Back to Journals » OncoTargets and Therapy » Volume 13
Inhibition of HMGB1 Overcomes Resistance to Radiation and Chemotherapy in Nasopharyngeal Carcinoma
Authors Zhu X, Cong J, Lin Z, Sun J, Yang B, Li A
Received 19 November 2019
Accepted for publication 25 March 2020
Published 14 May 2020 Volume 2020:13 Pages 4189—4199
DOI https://doi.org/10.2147/OTT.S239243
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Sanjeev K. Srivastava
Xuewei Zhu,1 Jianan Cong,2 Zhang Lin,3 Jing Sun,4 Ben Yang,3 Aipeng Li5
1Department of Otolaryngology Head and Neck Surgery, China-Japan Union Hospital of Jilin University, Changchun, Jilin, People’s Republic of China; 2Department of Ophthalmology, Changchun City Central Hospital, Changchun, Jilin, People’s Republic of China; 3Department of Ophthalmology, China-Japan Union Hospital of Jilin University, Changchun, Jilin, People’s Republic of China; 4Department of Biochemistry and Molecular Biology, The George Washington University, Washington, DC, USA; 5Department of Ophthalmology, The First Hospital of Jilin University, Changchun, Jilin, People’s Republic of China
Correspondence: Ben Yang; Aipeng Li 126 Xiantai St, Changchun, Jilin, China
Email [email protected]; [email protected]
Objective: This study aimed to investigate the effect of high mobility group protein B1 (HMGB1) on chemoresistance and radioresistance in nasopharyngeal carcinoma (NPC).
Materials and Methods: HMGB1-knockout HK1 cell lines were generated using clustered regularly interspaced short palindromic repeats/CRISPR-associated protein 9 (CRISPR/Cas9) system. Western blotting was used to evaluate the protein expression level of HMGB1. DNA repair efficiency of non-homologous end joining (NHEJ) and homologous recombination (HR) was monitored through NHEJ and HR reporter assay. Cellular protein–protein interaction between HMGB1 and NHEJ apparatus was determined by immunoprecipitation. Direct protein–protein interaction was examined by affinity capture assay with purified protein. Protein-DNA binding was evaluated by chromatin fractionation assay. Cell viability assay was employed to measure cell sensitivity to ionizing radiation (IR) or cisplatin.
Results: HMGB1-knockout NPC cells showed significant decrease in NHEJ efficiency. HMGB1 immunoprecipitated NHEJ key factors in NPC cells and promoted DNA-binding activity of Ku70. Mutational analysis revealed that serine 155 of Ku70 was required for its direct interaction with HMGB1. HMGB1 was highly expressed in radio- and chemoresistant NPC cells. Deficiency of HMGB1 sensitized wild-type (WT) and resistant NPC cells to IR and cisplatin. Glycyrrhizin, which is HMGB1 inhibitor, impaired DNA binding of HMGB1 and exhibited excellent synergy with IR and cisplatin.
Conclusion: HMGB1 promotes NHEJ via interaction with Ku70 resulting in resistance to IR and cisplatin. Inhibition of HMGB1 by glycyrrhizin is a potential therapeutic regimen to treat cisplatin and IR resistant NPC patients.
Keywords: NPC, HMGB1, NHEJ, resistance, Ku70, glycyrrhizin
Introduction
Nasopharyngeal carcinoma (NPC) has a particularly high incidence in southern China, especially in the Cantonese region. The disease is relatively rare globally.1 The annual incidence is approximately 30 cases per 100,000 persons1 in Asia. While the incidence is less than 1 case per 100,000 persons in Europe and the US.2 Several risk factors, such as Epstein–Barr virus infection, host genetics, and environmental exposures, have been shown to be associated with the etiology of NPC.3–5 NPC is one of the most challenging head and neck squamous cell carcinomas because most patients are diagnosed at advanced stages.6 The front-line treatment for NPC is radiation therapy. Recently, intensity-modulated radiation therapy (IMRT) was incorporated in clinical radiotherapy to minimize the radiation doses.7 Radiation therapy is commonly given in combination with chemotherapeutic drugs for advanced NPC, such as cisplatin, carboplatin, and doxorubicin.8 Therefore, radioresistance and chemoresistance are the major obstacles for NPC treatment. Unfortunately, the mechanisms underlying radio- and chemoresistance for NPC remain unclear.9
High mobility group protein B1 (HMGB1), is a member of the high‐mobility group protein superfamily and is highly abundant in cells. The importance of HMGB1 is demonstrated by its detection in variety types of cancer in which upregulated expression of HMGB1 is associated with proliferation and metastasis.10–13 Recently, overexpression of HMGB1 has been found to be correlated with cell proliferation and poor prognosis in human NPC.14,15 HMGB1 participates in a variety of cellular processes, including chromatin structure and transcriptional regulation, differentiation, DNA repair, and inflammation.16 The well-established role of HMGB1 was described as a DNA-binding protein. It induces DNA bending and facilitates assembly of DNA-binding proteins.17–19 HMGB1 participates in nucleotide excision repair, base excision repair, mismatch repair, and non-homologous end-joining (NHEJ), indicating that HMGB1 might contribute to DNA repair-related radioresistance and chemoresistance.20
Both IR and cisplatin target DNA in cancer cells resulting in DNA double-strand breaks (DSB), which lead to cell death. NHEJ is one of the main DSB repair (DSBR) pathways in human, which does not require sister chromatid as the homologous template.21 Therefore, NHEJ can be incorporated throughout the whole cell cycle. While the other main DSBR pathway, homologous recombination (HR), repairs DSB accurately in a template-dependent manner.22 Core factors of NHEJ include Ku70/80 heterodimer, DNA-protein kinase catalytic subunit (DNA-PKcs), XLF, and XRCC4/Ligase IV complex.21 NHEJ also requires accessory factors for more efficient and accurate repair, such as DNA end processing factors and polymerase.23–27 HMGB1 has been implicated in NHEJ via stimulating ligation in vitro even without compatible DNA ends.28,29 NHEJ repairs DSB with incompatible ends, which are often generated by ionizing radiation.30 Therefore, we hypothesize that HMGB1 participates in NHEJ and radioresistance in human NPC cells. In this study, we observed that HMGB1 promotes efficient NHEJ via interaction with NHEJ core factor Ku70. The mutational analysis of Ku70 showed that serine 155 is essential for protein–protein interaction between Ku70 and HMGB1. Importantly, we found that HMGB1 deficiency or inhibition significantly sensitize both WT and resistant NPC cells to IR and cisplatin, suggesting targeting HMGB1 can potentially improve radio- or chemotherapy for NPC patients.
Materials and Methods
Cell Lines and Cell Cultures
HK1 (Hong Kong NPC AoE Cell Line Repository, a kindhearted gift from George Tsao of University of Hong Kong, the use of the cell line was approved by China-Japan Union Hospital of Jilin University.) Cells were cultured at 37°C in 5% CO2 atmosphere in RPMI-1640 medium (Gibco, NY, USA) with 10% fetal bovine serum for less than 2 months. HK1-IRR cells were generated by exposing HK1-WT cells to 1–4 Gy of IR once per week for 3 months. In detail, HK1-WT cells were exposed to 1 Gy of IR and allowed recover for a week. This procedure was repeated for another 2 weeks. The cells were subsequently exposed to 2 Gy, 3 Gy, and 4 Gy of IR for another 3 weeks/dose. Similarly, HK1-CR cells were generated by exposing HK1-WT cells to 5–10 µM of cisplatin for 3 months. In detail, HK1-WT cells were exposed to 5 µM of cisplatin for 2 days and released in drug-free medium for 5 days. This procedure was repeated for another 5 weeks. The cells were subsequently exposed to 10 µM of cisplatin for another 6 weeks.
Cell Viability Assay
Cells were seeded at 5x103 cells/well in 96-well plate and cultured for overnight to allow adherence. Cells were exposed to IR and recovered for 72 h or incubated with drug for 72 h before performing the cell viability assay. Cell viability was detected by using Sulforhodamine B (SRB) assay. Briefly, cells were fixed by 10% trichloroacetic acid at 4°C for 1 h. The plate was washed with water and air-dried at room temperature. Cells were stained by 0.02% SRB for 1 h at room temperature. Plates were washed for 3 times with 1% acetate acid and air-dried. Tris buffer (10 mM tris-HCl, pH 10.5) was used to extract SRB and the absorbance was measured at 510 nm by microplate reader (MTX Lab Systems).
Generation of HMGB1-Deficient HK1 Cell Line by Using CRISPR/Cas9
Cas9 along with HMGB1 guide RNA plasmid was constructed by ligating oligonucleotide duplexes, which targets exon1 of HMGB1, into BbsI cut pX330-U6-Chimeric_BB-CBh-hSpCas9 (Addgene #42230). The plasmid was transfected into HK1-WT cells and selected by using puromycin. Cells were harvested and seeded in 96-well plate at 100 cells/mL and incubated for 14 days. Induvial clones were screened for HMGB1 expression.
HR and NHEJ Reporter Assay
A total of 10 μg of HR or NHEJ reporter plasmid was linearized by restriction enzyme NheI in 50 μL reaction for 6 h in a 37°C water bath. Linearized DNA were separated from non-linearized DNA by using gel extraction kit (QIAGEN). One microgram of linearized plasmid was transfected into HK1 cells by using Lipofectamine 2000 (ThermoFisher) according to manufacturer’s protocol. The linear DNA will be integrated into the chromosomal DNA in HK1 cells through HR. The plasmid contains geneticin-resistant gene. Therefore, cells with successfully integrated reporter (stable transfected cells) were selected by 1 mg/mL geneticin for 14 days. Stable transfected cells were seeded at 3×105 cells/mL in a 6-well plate. The plasmid contains GFP gene that is disrupted by insertion flanked by I-SceI restriction sites. Thus, the insertion was removed and one DSB was generated by transfecting 2 µg of I-SceI plasmid in stable transfected cells and incubating for 48 h. Successful DSBR will result in restoration of GFP expression that can be used to quantify DSBR efficiency. The green fluorescent intensity was measured by plate reader (MTX Lab Systems).
Ionizing Radiation
A total of 1×106 cells were resuspended in 10 mL medium and exposed to ionizing radiation by Gammator 50 137Cs source irradiator. IR-treated cells were then seeded for further cell viability assay.
Affinity Capture Assay
Fifty micrograms of GST-HMGB1 was bound to 50 µL of glutathione agarose in 300 µL binding buffer (50 mM tris-HCl pH 7.4, 100 mM KOAc, 1 mM EDTA, 1 mM DTT, 1x protease inhibitor) at 4°C with gentle mix for 1 h. The agarose was collected by centrifugation at 4°C, 1000×g for 1 min and washed three times with 1 mL binding buffer. The agarose was incubated with 50 µg of His-Ku70 or His-Ku70-S155A in 500 µL binding buffer at 4°C with gentle mix for 3 h. The agarose resin was washed three times with 1 mL binding buffer and the agarose was retained. Agarose-bound protein was eluted by using 50 µL of elution buffer (50 mM Tris-HCl, pH 8.0, 10 mM glutathione) for 15 min at 4°C. The supernatant was collected by centrifugation at 1000×g for 1 min. Agarose-bound protein was detected by using SDS-PAGE and visualized by using Western blot assay.
Chromatin Fractionation Assay
A total of 3 × 106 cells were harvested and washed with ice-cold PBS, and extracted by using CSK buffer (100 mM NaCl, 300 mM sucrose, 3 mM MgCl2, 0.7% Triton X-100, 10 mM PIPES, pH 7.0) supplemented with 1x protease inhibitor, 0.3 mg/mL RNase A at 4°C for 10 min. Cells were then washed 3 times in ice-cold PBS and harvested in SDS loading buffer for analysis by SDS-PAGE and followed with Western Blotting assay.31
siRNA Interference and Transfection
ON-TARGETplus HMGB1 siRNA was purchased from Dharmacon (L-018981-00-005). siHMGB1 was transfected into HK1 cells using Lipofectamine RNAiMAX (Thermo Fisher Scientific) according to the manufacturer’s instructions. siGL2 was incorporated as negative control.
For transfection, HMGB1 plasmid (Addgene #31609) was transfected into HK1 cells using Lipofectamine 3000 (Thermo Fisher Scientific) according to the manufacturer’s instructions. For complementation of HMGB1, transfection was performed 24 h after siRNA interference.
Statistical Analysis
Graphpad Prism 7 (Graphpad Software Inc., USA) was used for all statistical analysis. Results shown are the mean ± SD from 3 independent experiments. Student’s t-test was used to analyze differences between groups. P < 0.05 was considered statistically significant.
Results
HMGB1 Contributes to Efficient NHEJ in Human NPC Cells
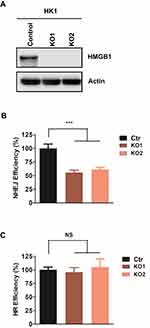
Although HMGB1 stimulating ligase IV activity in vitro has been reported28, whether HMGB1 regulates NHEJ efficacy in vivo has not yet been demonstrated. To investigate the role of HMGB1 in NHEJ in human NPC cells, we used CRISPR/cas9 to generate HMGB1-knockout HK1 (KO) cells. We randomly selected two clones and we observed that both KO1 and KO2 lack detectable HMGB1 protein expression (Figure 1A). We next used a robust NHEJ reporter assay to examine NHEJ efficiency in HMGB1-KO cells. We found that HMGB1 deficiency resulted in 45% and 37% decrease of NHEJ efficiency in HK-KO1 and HK-KO2, respectively, as compared to that in HK1-WT cells (p<0.001) (Figure 1B). NHEJ and HR are two major DSBR pathways that often collaborate or compete in eukaryotic cells.32 Since HMGB1 affects NHEJ efficiency in NPC cells, we then evaluated HR efficiency using HR reporter assay. As shown in Figure 1C, HMGB1 deficiency did not significantly alter HR efficiency versus WT cells. These results suggest that HMGB1 maintains NHEJ efficiency but not potentially impacts pathway choice between NHEJ and HR.
HMGB1 Interacts with Ku70 and Promotes DNA-Binding Activity of Ku70 in NHEJ
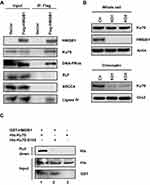
To elucidate how HMGB1 affects NHEJ, we first used immunoprecipitation assay to determine whether HMGB1 interacts with NHEJ core factors. As shown in Figure 2A, we detected that HMGB1 forms protein–protein interactions with Ku70, DNA-PKcs, and Ligase IV in HK1 cells. Since the most well-known function of HMGB1 is bending DNA structure by DNA binding, we hypothesized that HMGB1 regulates DNA binding of NHEJ core factors. Using chromatin fractionation assay, we found that Ku70–DNA interaction significantly decreased in HMGB1-deficient HK1 cell (Figure 2B), indicating that HMGB1 participates in NHEJ by facilitating Ku70-DNA binding.
Since interaction between HMGB1 and Ku70 in Figure 2A could be mediated by DNA or other proteins, we further determined whether HMGB1 directly interacts with Ku70 in vitro using purified GST-tagged-HMGB1 (GST-HMGB1) and HIS-tagged-Ku70 (HIS-Ku70). GST-HMGB1 binds to glutathione agarose that is used to exclude non-GST-tagged protein or non-HMGB1-binding protein. Using this affinity capture assay, we found that HIS-Ku70 was retained by GST-HMGB1 on glutathione agarose (Figure 2C, lane 1), demonstrating HMGB1 and Ku70 have direct protein–protein interaction. Fell et al showed that serine 155, which is located in vWA domain of Ku70, can be phosphorylated after IR-induced DNA double-strand breaks.33 Interestingly we found that Ku70-S155A failed to interact with HMGB1 in vitro (Figure 2C, lane 2), suggesting HMGB1–Ku70 interaction could involve in DNA damage response generated by IR.
HMGB1 Deficiency Sensitizes NPC Cells to IR
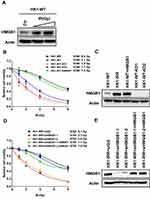
Given that NHEJ is the major pathway for re-joining incompatible DNA ends, which are often a result from IR, we hypothesized that HMGB1 expression is affected in response to IR. We treated HK1-WT cells with 0–4 Gy of IR, recovered the cells for 48 h and evaluated HMGB1 protein expression using Western Blotting. As shown in Figure 3A, HMGB1 expression was significantly induced by IR, suggesting HMGB1 participates in radioresistance in NPC cells.
To examine the role of HMGB1 in radio-sensitivity, we measured cell viability of HMGB1-deficient NPC cells. Compared to HK1-WT cells, HMGB1 deficiency significantly increased cell sensitivity to IR (Figure 3B). To exclude the possibility that improved sensitivity to IR in HMGB1-deficient cells could be masked by compensatory changes, we overexpressed HMGB1 in HK1-WT cells (Figure 3C). As expected, overexpression of HMGB1 rescued IR resistance to similar level to IR-resistant HK1 cells (HK1-IRR) (Figure 3B). Notably, HMGB1 expression level in HK1-IRR cells was higher than in HK1-WT cells. In addition, we also overexpressed HMGB1 in HK1-WT-KO cells and observed similar result that HMGB1 restored IR resistance in HMGB1-deficient cells (Supplementary Figure 1A and C). These findings suggest that HMGB1 participates in IR resistance in NPC. To directly test whether HMGB1 contributes to IR resistance, we monitored cell viability in HMGB1-knockdown HK1-IRR cells. Improved sensitivity was confirmed in cell viability assay in which IR sensitivity in HMGB1-knockdown HK1-IRR cells was 3-fold higher than in control HK1-IRR cells (Figure 3D and E). Complementation of HMGB1 in HMGB1-knockdown HK1-IRR cells rescued IR resistance (Figure 3D) furthermore confirmed that HMGB1 deficiency indeed sensitizes NPC cells to IR.
HMGB1 Deficiency Sensitizes NPC Cells to Cisplatin
Cisplatin is commonly used as chemotherapy strategy following radiation treatment in NPC. We then investigated whether HMGB1 related to cell survival in response to cisplatin treatment. Similar to the response to IR, HMGB1 expression level also increased with cisplatin treatment in HK1 cells (Figure 4A). Importantly, HMGB1 deficiency sensitized both HK1-WT and HK1-cisplatin resistant (HK1-CR) cells to cisplatin (Figure 4B-E, Supplementary Figure 1B and C), indicating HMGB1 contributes to chemoresistance in human NPC.
Glycyrrhizin Inhibits DNA Binding of HMGB1 and Sensitizes NPC Cells to IR and Cisplatin
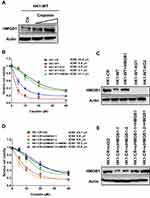
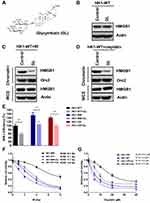
Natural compound glycyrrhizin (GL) (Figure 5A), which is the major constituent of glycyrrhiza glabra (liquorice) root, has been demonstrated to directly bind HMGB1.34 Importantly, GL binds to the DNA-binding concave of HMGB135 and disrupts HMGB1–DNA interaction.34 Since HMGB1 participates in NHEJ and contributes to radio- and chemoresistance in NPC, we next examined the effect of GL on HMGB1 in NPC cells. Although 1 µM of GL did not induce HMGB1 degradation (Figure 5B), it impeded HMGB1 chromatin binding stimulated by IR (Figure 5C) or cisplatin (Figure 5D) in HK1 cells, leading us to question whether GL compromise NHEJ in NPC cells. Indeed, we found that GL significantly inhibited NHEJ efficiency in HK1-WT, HK1-IRR and HK1-CR cells (Figure 5E). Therefore, we hypothesized that inhibition of HMGB1 by GL sensitizes NPC cells to IR and cisplatin. We incubated NPC cells with 1 µM of GL 24 h prior to IR or cisplatin treatment. As shown in Figure 5F and G, GL sensitized both WT and resistant NPC cells to IR and cisplatin, suggesting GL is a potential leading compound to overcome radio- and chemoresistance in NPC.
Discussion
In this study, we elucidated that HMGB1 contributes to NHEJ efficiency by inducing Ku70–DNA interaction. We also observed that HMGB1 physically interacts with Ku70 and the mutational analysis demonstrated that serine 155 of Ku70 is necessary for Ku70–HMGB1 interaction. Importantly, we found that both IR and cisplatin treatment can induce HMGB1 expression in NPC cells and HMGB1 knockout significantly sensitizes NPC cells to IR and cisplatin. We also revealed that glycyrrhizin inhibits HMGB1 DNA-binding activity and generates excellent synergy with IR and cisplatin.
HMGB1 binds to and bends damaged DNA. However, the biological consequence of this role remains unknown. Our group for the first time found that HMGB1 deficiency impairs Ku70–DNA binding, which could result from failed exposure of damaged DNA. Therefore, HMGB1 may contribute to NHEJ by providing accessible substrates to NHEJ apparatus. Since Ku-DNA binding has been demonstrated as the initiation step of NHEJ, HMGB1 could serve at early stage of NHEJ or DNA damage response. We believe the role of HMGB1 is not limited to NHEJ because modulating chromatin structure occurs in almost all DNA repair pathways. Lange et al observed that HMGB1 enhances the repair of UVC and psoralen ICL-induced DNA damage.36 Thus, effects of HMGB1 on DNA repair remain to be explored.
HMGB1 plays important roles as both chromatin structural protein and cytokine. Therefore, it is reasonable that HMGB1 participates in DNA repair and inflammation.16,17,37 Our future directions include examining HMGB1 expression in NPC patients with allergic rhinitis (AR) history, which has been positively correlated with NPC.38 Secreted HMGB1 may improve tumor cell survival and invasion because of its multifunction, such as transcriptional regulation, V(D)J recombination, chromatin structure, and inflammation. The correlation between AR and NPC could be mediated by HMGB1 expression. The regulatory mechanism of HMGB1 in AR or NPC remains an interesting area to study.
NHEJ has been demonstrated to be correlated with chemoresistance or radioresistance in a variety of cancer types, such as colorectal cancer, glioma, pancreatic cancer, and hepatocellular carcinoma.39–42 Therefore, HMGB1, which contributes to NHEJ efficiency in NPC, should be positively related to radioresistance or chemoresistance. Our study demonstrated that HMGB1 contributes to therapeutic resistance mechanisms possibly due to its role in NHEJ. These results provide evidence supporting the hypothesis that targeting HMGB1 in human NPC may significantly sensitize the NPC patients to radio- and chemotherapy.
Conclusions
This study demonstrated that HMGB1 contributes to efficient NHEJ by interacting with Ku70. Knockout or inhibition of HMGB1 significantly sensitizes NPC cells to radio- and chemotherapy.
Abbreviations
HMGB1, high mobility group protein B1; NPC, nasopharyngeal carcinoma; IR, ionizing radiation; IMRT, intensity-modulated radiation therapy; DSBR, DNA double-strand break repair; NHEJ, non-homologous end joining; HR, homologous recombination; WT, wild type.
Disclosure
The authors declare that they have no competing interests.
References
1. Cao SM, Simons MJ, Qian CN. The prevalence and prevention of nasopharyngeal carcinoma in China. Chin J Cancer. 2011;30(2):114–119. doi:10.5732/cjc.010.10377
2. Yu MC, Yuan JM. Epidemiology of nasopharyngeal carcinoma. Semin Cancer Biol. 2002;12(6):421–429. doi:10.1016/S1044579X02000858
3. Andersson-Anvret M, Forsby N, Klein G, Henle W. Relationship between the Epstein-Barr virus and undifferentiated nasopharyngeal carcinoma, correlated nucleic acid hybridization and histopathological examination. Int J Cancer. 1977;20(4):486–494. doi:10.1002/ijc.2910200403
4. Lin DC, Meng X, Hazawa M, et al. The genomic landscape of nasopharyngeal carcinoma. Nat Genet. 2014;46(8):866–871. doi:10.1038/ng.3006
5. Ward MH, Pan WH, Cheng YJ, et al. Dietary exposure to nitrite and nitrosamines and risk of nasopharyngeal carcinoma in Taiwan. Int J Cancer. 2000;86(5):603–609. doi:10.1002/(SICI)1097-0215(20000601)86:5<603::AID-IJC1>3.0.CO;2-H
6. Licitra L, Bernier J, Cvitkovic E, et al. Cancer of the nasopharynx. Crit Rev Oncol Hematol. 2003;45(2):199–213. doi:10.1016/S1040-8428(01)00210-4
7. Lee AW, Lin JC, Ng WT. Current management of nasopharyngeal cancer. Semin Radiat Oncol. 2012;22(3):233–244. doi:10.1016/j.semradonc.2012.03.008
8. Paiar F, Di Cataldo V, Zei G, et al. Role of chemotherapy in nasopharyngeal carcinoma. Oncol Rev. 2012;6(1):e1. doi:10.4081/oncol.2012.e1
9. Wang J, Shi M, Hsia Y, et al. Failure patterns and survival in patients with nasopharyngeal carcinoma treated with intensity modulated radiation in Northwest China, a pilot study. Rad Oncol. 2012;7:2. doi:10.1186/1748-717X-7-2
10. Brezniceanu ML, Volp K, Bosser S, et al. HMGB1 inhibits cell death in yeast and mammalian cells and is abundantly expressed in human breast carcinoma. FASEB J. 2003;17(10):1295–1297. doi:10.1096/fj.02-0621fje
11. Kawahara N, Tanaka T, Yokomizo A, et al. Enhanced coexpression of thioredoxin and high mobility group protein 1 genes in human hepatocellular carcinoma and the possible association with decreased sensitivity to cisplatin. Cancer Res. 1996;56(23):5330–5333.
12. Poser I, Golob M, Buettner R, Bosserhoff AK. Upregulation of HMG1 leads to melanoma inhibitory activity expression in malignant melanoma cells and contributes to their malignancy phenotype. Mol Cell Biol. 2003;23(8):2991–2998. doi:10.1128/MCB.23.8.2991-2998.2003
13. Volp K, Brezniceanu ML, Bosser S, et al. Increased expression of high mobility group box 1 (HMGB1) is associated with an elevated level of the antiapoptotic c-IAP2 protein in human colon carcinomas. Gut. 2006;55(2):234–242. doi:10.1136/gut.2004.062729
14. Zhu X, Sun L, Wang Y. High mobility group box 1 (HMGB1) is upregulated by the Epstein-Barr virus infection and promotes the proliferation of human nasopharyngeal carcinoma cells. Acta Otolaryngol. 2016;136(1):87–94. doi:10.3109/00016489.2015.1082192
15. Wu D, Ding Y, Wang S, Zhang Q, Liu L. Increased expression of high mobility group box 1 (HMGB1) is associated with progression and poor prognosis in human nasopharyngeal carcinoma. J Pathol. 2008;216(2):167–175. doi:10.1002/path.2391
16. Czura CJ, Wang H, Tracey KJ. Dual roles for HMGB1, DNA binding and cytokine. J Endotoxin Res. 2001;7(4):315–321. doi:10.1177/09680519010070041401
17. Andersson U, Erlandsson-Harris H, Yang H, Tracey KJ. HMGB1 as a DNA-binding cytokine. J Leukoc Biol. 2002;72(6):1084–1091.
18. Schroter H, Bode J. The binding sites for large and small high-mobility-group (HMG) proteins studies on HMG-nucleosome interactions in vitro. Eur J Biochem. 1982;127(2):429–436.
19. Nightingale K, Dimitrov S, Reeves R, Wolffe AP. Evidence for a shared structural role for HMG1 and linker histones B4 and H1 in organizing chromatin. EMBO J. 1996;15(3):548–561. doi:10.1002/j.1460-2075.1996.tb00387.x
20. Lange SS, Vasquez KM. HMGB1, the jack-of-all-trades protein is a master DNA repair mechanic. Mol Carcinog. 2009;48(7):571–580. doi:10.1002/mc.20544
21. Lieber MR, Ma Y, Pannicke U, Schwarz K. Mechanism and regulation of human non-homologous DNA end-joining. Nat Rev Mol Cell Biol. 2003;4(9):712–720. doi:10.1038/nrm1202
22. Valerie K, Povirk LF. Regulation and mechanisms of mammalian double-strand break repair. Oncogene. 2003;22(37):5792–5812. doi:10.1038/sj.onc.1206679
23. Ma Y, Pannicke U, Schwarz K, Lieber MR. Hairpin opening and overhang processing by an Artemis/DNA-dependent protein kinase complex in nonhomologous end joining and V(D)J recombination. Cell. 2002;108(6):781–794. doi:10.1016/S0092-8674(02)00671-2
24. Li J, Summerlin M, Nitiss KC, Nitiss JL, Hanakahi LA. TDP1 is required for efficient non-homologous end joining in human cells. DNA Repair (Amst). 2017;60:40–49. doi:10.1016/j.dnarep.2017.10.003
25. Heo J, Li J, Summerlin M, et al. TDP1 promotes assembly of non-homologous end joining protein complexes on DNA. DNA Repair (Amst). 2015;30:28–37. doi:10.1016/j.dnarep.2015.03.003
26. Biehs R, Steinlage M, Barton O, et al. DNA double-strand break resection occurs during non-homologous end joining in G1 but is distinct from resection during homologous recombination. Mol Cell. 2017;65(4):671–684 e675. doi:10.1016/j.molcel.2016.12.016
27. Ma Y, Lu H, Tippin B, et al. A biochemically defined system for mammalian nonhomologous DNA end joining. Mol Cell. 2004;16(5):701–713. doi:10.1016/j.molcel.2004.11.017
28. Yamanaka S, Katayama E, Yoshioka K, Nagaki S, Yoshida M, Teraoka H. Nucleosome linker proteins HMGB1 and histone H1 differentially enhance DNA ligation reactions. Biochem Biophys Res Commun. 2002;292(1):268–273. doi:10.1006/bbrc.2002.6647
29. Hiom K, Gellert M. Assembly of a 12/23 paired signal complex, a critical control point in V(D)J recombination. Mol Cell. 1998;1(7):1011–1019. doi:10.1016/S1097-2765(00)80101-X
30. Burma S, Chen BP, Chen DJ. Role of non-homologous end joining (NHEJ) in maintaining genomic integrity. DNA Repair (Amst). 2006;5(9–10):1042–1048. doi:10.1016/j.dnarep.2006.05.026
31. Sun J, Fu X, Wang Y, et al. Erianin inhibits the proliferation of T47D cells by inhibiting cell cycles, inducing apoptosis and suppressing migration. Am J Transl Res. 2016;8(7):3077–3086.
32. Kass EM, Jasin M. Collaboration and competition between DNA double-strand break repair pathways. FEBS Lett. 2010;584(17):3703–3708. doi:10.1016/j.febslet.2010.07.057
33. Fell VL, Schild-Poulter C. Ku regulates signaling to DNA damage response pathways through the Ku70 von Willebrand A domain. Mol Cell Biol. 2012;32(1):76–87. doi:10.1128/MCB.05661-11
34. Sakamoto R, Okano M, Takena H, Ohtsuki K. Inhibitory effect of glycyrrhizin on the phosphorylation and DNA-binding abilities of high mobility group proteins 1 and 2 in vitro. Biol Pharm Bull. 2001;24(8):906–911. doi:10.1248/bpb.24.906
35. Yamaguchi H, Kidachi Y, Kamiie K, Noshita T, Umetsu H. Structural insight into the ligand-receptor interaction between glycyrrhetinic acid (GA) and the high-mobility group protein B1 (HMGB1)-DNA complex. Bioinformation. 2012;8(23):1147–1153. doi:10.6026/97320630081147
36. Lange SS, Mitchell DL, Vasquez KM. High mobility group protein B1 enhances DNA repair and chromatin modification after DNA damage. Proc Natl Acad Sci U S A. 2008;105(30):10320–10325. doi:10.1073/pnas.0803181105
37. Gardella S, Andrei C, Ferrera D, et al. The nuclear protein HMGB1 is secreted by monocytes via a non-classical, vesicle-mediated secretory pathway. EMBO Rep. 2002;3(10):995–1001. doi:10.1093/embo-reports/kvf198
38. Chung SD, Wu CS, Lin HC, Hung SH. Association between allergic rhinitis and nasopharyngeal carcinoma, a population-based study. Laryngoscope. 2014;124(8):1744–1749. doi:10.1002/lary.24532
39. Chen X, Li WF, Wu X, et al. Dicer regulates non-homologous end joining and is associated with chemosensitivity in colon cancer patients. Carcinogenesis. 2017;38(9):873–882. doi:10.1093/carcin/bgx059
40. Sishc BJ, Davis AJ. The role of the core non-homologous end joining factors in carcinogenesis and cancer. Cancers. 2017;9:7.
41. Yang S, Wang XQ. XLF-mediated NHEJ activity in hepatocellular carcinoma therapy resistance. BMC Cancer. 2017;17(1):344.
42. Li YH, Wang X, Pan Y, Lee DH, Chowdhury D, Kimmelman AC. Inhibition of non-homologous end joining repair impairs pancreatic cancer growth and enhances radiation response. PLoS One. 2012;7(6):e39588. doi:10.1371/journal.pone.0039588
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.