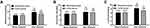Back to Journals » Drug Design, Development and Therapy » Volume 16
Influences of Bifid Triple Viable Capsules Plus Cetirizine on Gut Microbiota and Immune Function in Children with Eczema
Received 23 February 2022
Accepted for publication 9 July 2022
Published 3 August 2022 Volume 2022:16 Pages 2509—2515
DOI https://doi.org/10.2147/DDDT.S363702
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Prof. Dr. Tin Wui Wong
Zhenxing Su,1 Yaqin Kang2
1Dermatology, First Hospital of Shanxi Medical University, Taiyuan City, People’s Republic of China; 2Pediatrics, First Hospital of Shanxi Medical University, Taiyuan City, People’s Republic of China
Correspondence: Zhenxing Su, Dermatology, First Hospital of Shanxi Medical University, No. 85, Jiefang South Road, Yingze District, Taiyuan City, People’s Republic of China, Email [email protected]
Objective: Infantile eczema (IE), a common pediatric allergic skin disease caused by multiple inherent and external factors, is common in infants and young children, with skin lesions and itching as the main clinical manifestations. At present, its pathological mechanism has not been thoroughly clarified, but scholars believe that it is related to the joint action of various internal and external factors. This research aims to investigate the influences of bifid triple viable capsules plus cetirizine (CTZ) on gut microbiota (GMB) and immunity in children with eczema.
Methods: The complete clinical data of 162 cases of IE presented between July 2019 and July 2020 to the First Hospital of Shanxi Medical University were retrospectively analyzed. Children treated by CTZ alone were assigned to the control group (n = 81) and those by CTZ plus bifid triple viable capsules were included in the observation group (n = 81). Therapeutic efficacy, adverse reactions (ARs), disease recurrence, as well as changes in GMB, inflammatory factors (IFs) and immunoglobulins (Igs) were observed.
Results: The observation group was observed with a higher overall response rate and increased fecal Lactobacillus, Escherichia coli (E. coli) and Bifidobacterium counts after treatment versus the control group. After treatment, reduced IgG and IgM, as well as IFs, were found in both groups, with lower levels in the observation group. A lower incidence of ARs was determined in the observation group.
Conclusion: With high efficacy for the treatment of IE, bifid triple viable capsules plus CTZ can validly regulate the GMB of children, improve their immune function and clinical symptoms, and reduce the disease recurrence rate, which is worthy of clinical promotion.
Keywords: bifid triple viable capsules, cetirizine, eczema, gut microbiota, immune function
Introduction
Prenatal and early childhood environments, including smoke, airborne allergens, air pollution, various microorganisms, and environmental compounds, can lead to immune stress responses in children.1,2 Among them, environmental factors are closely associated with childhood eczema.3 Eczema is a chronic, recurrent and intensely itchy inflammatory skin disease that often occurs in skin folds, such as those on the elbows or behind the knees.4 The condition, which significantly affects approximately one-fifth of children, usually begins in infancy and sometimes continues into adulthood.5,6 The increasing incidence of eczema not only affects children’s health, but also brings a heavy financial, life and psychological burden to their caregivers.7
Cetirizine (CTZ) is a kind of histamine-1 (H1) receptor antagonist, which is often used in the treatment of allergies.8 Histamine (HA), a key mediator of allergic reactions, causes pruritus by acting upon H1, stimulates nasal mucosa secretion, shrinks bronchial and intestinal smooth muscles, and relaxes microvascular smooth muscles.9 First- and second-generation anti-HA drugs, both of which have similar pharmacological action and therapeutic use, can be distinguished as the second-generation anti-HA agents have fewer side effects due to their stronger selectivity for H1 peripheral receptors.10 As an oral agent, CTZ is the second generation of anti-HA drugs and was listed in the Italian market 30 years ago.11 CTZ is widely applied to children and teenagers to treat allergic rhinitis and chronic spontaneous urticaria.12 CTZ acts on these allergies by selectively inhibiting peripheral H1 receptors.13 However, after drug withdrawal, the allergy is easy to recur, which will induce itching again.14 Another drug, bifid triple viable capsules, is composed of Bifidobacterium, Lactobacillus and Enterococcus, which has been shown to be effective against diarrhea caused by GMB disturbances or enteritis and has been approved by the State Food and Drug Administration of China as a nonprescription drug.15 Pharmacological studies have shown that bifid triple viable, given orally, can alleviate intestinal inflammations via regulating beneficial and harmful flora like Lactobacillus and Escherichia coli.16–18 However, there is a shortage of research on the role of the two drugs in infantile eczema (IE). Accordingly, this research aims to determine the influences of the combined treatment of the two drugs through detecting related indexes such as gut microbiota (GMB) and immune function.
Methods
General Data
Data sources: One hundred and sixty-two cases of IE presented between July 2019 and July 2020 to the First Hospital of Shanxi Medical University and met the inclusion criteria were enrolled. According to different treatment therapies, 81 cases were assigned to the control group and 81 to the observation group. The basic data (gender, month age and average course of disease) showed no significant difference (P > 0.05), indicating comparability of the two groups. The Ethics Committee of the First Hospital of Shanxi Medical University approved this study, and the families of the children all provided informed consent. This study complies with the Declaration of Helsinki.
Inclusion criteria: (1) Age: 4 months to 3 years old; (2) Diagnosed of eczema (non-exudative type); (3) No drug allergies nor probiotics taken half a month before treatment; (4) Normal intelligence level and mental state. Exclusion criteria: (1) Immune, infectious and congenital diseases; (2) Other skin diseases; (3) Viral and fungal infections; (4) Heart, liver and kidney dysfunction.
Methods
CTZ was given to patients in the control group. First, children were given routine treatment to prevent allergens, and Erfukang lotion diluted at 1:5 was applied externally, followed by the external application of Dr. Yu Body Lotion, 3 times a day. After that, they were given CTZ drops of different doses according to different age groups. For children aged 6 months to 1 year, CTZ was given 5 drops/time, twice a day. Children over 1 year old were administered with CTZ at 7 drops/time, twice daily. The treatment lasted for 30 days. On this basis, children were also treated with bifid triple viable capsules, per os, 1 tablet/time, 2 times per day for children of any age. Also, children were treated for 30 days. Children in the control group received the same treatment except for bifid triple viable capsules.
Test Results
Detection of GMB in Two Groups
GMB was detected before and 3 days after treatment. Children’s feces were collected and the contents of GMB such as Lactobacillus, Bifidobacterium and Escherichia coli were measured. The specific steps were as follows: 1g of fresh feces was put into a container, and mixed evenly with phosphate buffer (PBS), followed by centrifugation, sedimentation and water rinsing. Quantitative PCR was used to detect the proportion of bacteria in the samples. Fresh feces samples were pretreated and fecal bacterial DNA was extracted according to the instructions of the fecal bacterial genomic DNA Extraction Kit (ThermoFisher). Three real-time PCR reactions were performed for each diluted cDNA on StepOne Plus Real-time PCR System instrument (Thermo Fischer Scientific). The conditions were denaturation at 95°C for 5 min, 95°C for 15s, 50°C for 1 min, 72°C for 45s and 87°C for 5s, for 40 cycles.
Determination of Inflammatory Factors (IFs) and Immunoglobulins (Igs)
Blood samples were obtained from all children before and 3 days after treatment. Enzyme-linked immunosorbent assay (ELISA) kits (Dakewe Biotech), able to detect concentrations as low as 10 pg/mL of IL-4 and 3.9 pg/mL of IL-10, were responsible for serum IL-4 and IL-10 quantification, following the supplier’s instructions. IgG and IgM were determined using available ELISA by ELISA ImmunoWELL kits (Ben-Bio, San Diego, CA).
Adverse Reaction (AR) Rate
The incidence of ARs of the two groups was detected and compared, and the main indicators were dry mouth, diarrhea and lethargy. ARs with a cut-off below 2% were not listed.
Overall Response Rate (ORR) and Recurrence Rate in Two Groups
The ORR 1 month after treatment was tested, and the criteria were as follows: Markedly effective: A ≥ 70% skin lesion resolution in children; Effective: A ≥ 30% skin lesion resolution in children; Ineffective: A < 30% resolution or even an increase in skin lesions. ORR = number of cases of (markedly effective+effective)/total number of children × 100%. In addition, the recurrence rate after treatment was measured.
Statistical Methods
SPSS19.0 (Chinese version, SPSS Inc.) was utilized for statistical analysis of comprehensive data. A Chi-squared test was used for the comparison of counting data (n[%]) between groups. The paired t test was used for the comparison of measurement data (Mean±SD) within groups while independent sample t test for between-group comparisons. The difference was significant when P < 0.05.
Results
General Information
The general data (gender, average month age, average course of disease, average weight, etc.) showed no evident differences between groups (P > 0.05). Table 1
 |
Table 1 Children’s General Information |
Fluorescence Quantitative Sample Analysis of Two Groups
Bifidobacterium, Escherichia coli and Lactobacillus counts showed non-significant differences between groups prior to treatment (P > 0.05), while they increased after three days of treatment in both groups, especially in the observation group (P < 0.05). Figure 1
The Levels of IFs
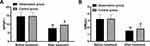
The levels of IFs differed insignificantly before treatment (P > 0.05). After three days of treatment, IL-4 and IL-10 levels decreased in both groups, with more obvious alleviation in inflammation in the observation group (P < 0.05). Figure 2
The Levels of IgG and IgM
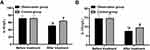
Similarly, no obvious inter-group differences were determined in Ig G and Ig M prior to treatment (P > 0.05). Reduced Ig G and Ig M were found in research group and control group after three days of treatment, with more obvious improvements in the observation group (P < 0.05). Figure 3
The Incidence of ARs in Two Groups
The occurrence of ARs in both the research group and control group was compared and the results showed a significantly lower incidence in the observation group (P < 0.05). Table 2
 |
Table 2 Incidence of Adverse Events |
The Overall Response Rate in Two Groups
The inter-group comparison of ORR within 1 month after treatment revealed a statistically higher ORR in the observation group (P < 0.05). Table 3
 |
Table 3 Total Effective Rate |
The Recurrence Rate in Two Groups
The recurrence rate was lower in the observation group than in the control group (P < 0.05). Table 4
 |
Table 4 Total Recurrence Rate |
Discussion
As an inflammation of the skin, eczema is a non-infectious chronic skin inflammation characterized by itching, as well as red rashes in most cases.19 This type of eczema/dermatitis is very common in childhood, affecting up to 25% of children worldwide.20 Therefore, there is a lot of research on the treatment of the disease. Based on the results obtained in this study, we are here to discuss the influences of bifid triple viable capsules plus CTZ therapy.
First of all, GMB detection results showed better GMB recovery in the observation group using bifid triple viable capsules plus CTZ therapy compared with the control group using CTZ alone. Probiotics in the intestine can modulate GMB, enhance gastrointestinal function, promote nutrient absorption, boost immunity and minimize bacterial growth.21 Meanwhile, probiotics can stimulate regulatory T cells in children with eczema and suppress Th2 reaction, thus adjusting GMB and improving eczema symptoms.22,23 Probiotic flora abnormalities, on the other hand, may lead to abnormal T cell reaction and the development of acute eczema.24,25 Combining this with the results of this study, it can be seen that the joint treatment did improve the GMB of the children due to the use of bifidum triple viable bacteria on the basis of CTZ.
In terms of the relief of inflammation, better improvement in the observation group despite reduced IFs in both groups after treatment. A study on rhinitis shows that CTZ tablets have a favorable effect on inflammation and remarkable anti-allergic properties.26,27 Both groups of patients took CTZ tablets, so the inflammatory reactions of both groups were well alleviated. However, a pharmacological study reported that in the model of enteritis in mice, oral administration of bifid triple viable capsules can reduce intestinal inflammation and down-regulate pro-IFs.15,16 Therefore, combined with the results of this study, the IFs of children treated with the combination of the two drugs will be better improved.
In addition, Ig G and Ig M levels of both groups decreased after treatment, while the two indexes of the observation group with combined treatment improved more obviously. Bifid triple viable capsules have been shown to not only effectively reduce various IFs and reduce inflammatory responses, but also effectively enhance CD4+ and reduce CD8+ cells in the human body.17,18 Another drug, CTZ, has the effect of improving immune cells, and minimizing cytokine storms caused by the immune system.8 Therefore, although the immunity of both groups was improved, the intestinal immune response of children in the observation group was better improved after treatment due to the combined use of drugs, which consequently produces various cytokines and antibodies, contributing to effectively enhanced local and systemic immune functions. As a result, the observation group has better treatment outcomes and a smaller chance of recurrence after treatment.
This study still shows some margins of improvement. The design of the study was not comprehensive enough to determine some other indicators that could reflect the effectiveness of the treatment, such as the level of T cells which is also related to immunologic function. Besides, we failed to investigate the patients’ family members’ satisfaction with the treatment and children’s compliance during the treatment process. Finally, the follow-up time was too short to determine the long-term therapeutic effect. In the future research, we will not only test and compare more indicators, but also continue to address these deficiencies to continuously improve the treatment methods.
Conclusion
To sum up, with remarkable curative effects in the treatment of IE, bifid triple viable capsules plus CTZ can significantly regulate the GMB of children, improve their immune function and clinical symptoms, and reduce disease recurrence, which deserves clinical expansion.
Data Sharing Statement
The labeled dataset used to support the findings of this study is available from the corresponding author upon request.
Funding
This study did not receive any funding in any form.
Disclosure
The authors declare no competing interests.
References
1. Wesemann DR, Nagler CR. The microbiome, timing, and barrier function in the context of allergic disease. Immunity. 2016;44(4):728–738. doi:10.1016/j.immuni.2016.02.002
2. Mims JW. Asthma: definitions and pathophysiology. Int Forum Allergy Rhinol. 2015;5(S1):S2–S6. doi:10.1002/alr.21609
3. Jackson-Browne MS, Henderson N, Patti M, Spanier A, Braun JM. The impact of early-life exposure to antimicrobials on asthma and eczema risk in children. Current Environ Health Rep. 2019;6(4):214–224. doi:10.1007/s40572-019-00256-2
4. George SM, Karanovic S, Harrison DA, et al. Interventions to reduce Staphylococcus aureus in the management of eczema. Cochrane Database Syst Rev. 2019. doi:10.1002/14651858.CD003871.pub3
5. Odhiambo JA, Williams HC, Clayton TO, Robertson CF, Asher MI. Global variations in prevalence of eczema symptoms in children from ISAAC Phase Three. J Allergy Clin Immunol. 2009;124(6):1251–1258. e1223. doi:10.1016/j.jaci.2009.10.009
6. Abuabara K, Yu A, Okhovat JP, Allen I, Langan SM. The prevalence of atopic dermatitis beyond childhood: a systematic review and meta‐analysis of longitudinal studies. Allergy. 2018;73(3):696–704. doi:10.1111/all.13320
7. Kyu HH, Abate D, Abate KH, et al. Global, regional, and national disability-adjusted life-years (DALYs) for 359 diseases and injuries and healthy life expectancy (HALE) for 195 countries and territories, 1990–2017: a systematic analysis for the Global Burden of Disease Study 2017. Lancet. 2018;392(10159):1859–1922.
8. Hogan RB, Hogan Iii RB, Cannon T, et al. Dual-histamine receptor blockade with cetirizine-famotidine reduces pulmonary symptoms in COVID-19 patients. Pulm Pharmacol Ther. 2020;63:101942. doi:10.1016/j.pupt.2020.101942
9. Parisi GF, Licari A, Papale M, et al. Antihistamines: ABC for the pediatricians. Pediatr Allergy Immunol. 2020;31:34–36. doi:10.1111/pai.13152
10. Tatarkiewicz J, Rzodkiewicz P, Żochowska M, Staniszewska A, Bujalska-Zadrożny M. New antihistamines–perspectives in the treatment of some allergic and inflammatory disorders. Arch Med Sci. 2019;15(2):537. doi:10.5114/aoms.2017.68534
11. Corsico AG, Leonardi S, Licari A, et al. Focus on the cetirizine use in clinical practice: a reappraisal 30 years later. Multidiscip Respir Med. 2019;14(1):1–7. doi:10.1186/s40248-018-0159-y
12. Parisi GF, Leonardi S, Ciprandi G, et al. Cetirizine use in childhood: an update of a friendly 30-year drug. Clin Mol Allergy. 2020;18(1):1–6. doi:10.1186/s12948-020-00118-5
13. Blaiss MS, Bernstein JA, Kessler A, et al. The role of cetirizine in the changing landscape of iv antihistamines: a narrative review. Adv Ther. 2021;39:1–15.
14. Chung AH, La Grenade L, Harinstein LM. Pruritus after discontinuation of cetirizine. Ther Adv Drug Saf. 2019;10:2042098619859996. doi:10.1177/2042098619859996
15. Chen MY, Qiu ZW, Tang HM, et al. Efficacy and safety of bifid triple viable plus aminosalicylic acid for the treatment of ulcerative colitis: a systematic review and meta-analysis. Medicine. 2019;98(47). doi:10.1097/MD.0000000000017955
16. Shi C-Z, Chen H-Q, Liang Y, et al. Combined probiotic bacteria promotes intestinal epithelial barrier function in interleukin-10-gene-deficient mice. World J Gastroenterol. 2014;20(16):4636. doi:10.3748/wjg.v20.i16.4636
17. Zhao H-M, Huang X-Y, Zuo Z-Q, et al. Probiotics increase T regulatory cells and reduce severity of experimental colitis in mice. World j Gastroenterol. 2013;19(5):742. doi:10.3748/wjg.v19.i5.742
18. Song H, Wang W, Shen B, et al. Pretreatment with probiotic Bifico ameliorates colitis‐associated cancer in mice: transcriptome and gut flora profiling. Cancer Sci. 2018;109(3):666–677. doi:10.1111/cas.13497
19. Boyle RJ, Bath‐Hextall FJ, Leonardi‐Bee J, Murrell DF, Tang ML. Probiotics for treating eczema. Cochrane Database Syst Rev. 2008. doi:10.1002/14651858.CD006135.pub2
20. Woo YR, Han Y, Lee JH, et al. Real‐world prevalence and burden of genital eczema in atopic dermatitis: a multicenter questionnaire‐based study. J Dermatol. 2021;48(5):625–632. doi:10.1111/1346-8138.15817
21. Vandenplas Y, De Greef E, Devreker T, Veereman-Wauters G, Hauser B. Probiotics and prebiotics in infants and children. Curr Infect Dis Rep. 2013;15(3):251–262. doi:10.1007/s11908-013-0334-4
22. Vitaliti G, Pavone P, Guglielmo F, Spataro G, Falsaperla R. The immunomodulatory effect of probiotics beyond atopy: an update. J Asthma. 2014;51(3):320–332. doi:10.3109/02770903.2013.862259
23. Sun M, Luo J, Liu H, Xi Y, Lin Q. Can mixed strains of lactobacillus and bifidobacterium reduce eczema in infants under three years of age? A meta-analysis. Nutrients. 2021;13(5):1461. doi:10.3390/nu13051461
24. Song H, Yoo Y, Hwang J, Na Y-C, Kim HS. Faecalibacterium prausnitzii subspecies–level dysbiosis in the human gut microbiome underlying atopic dermatitis. J Allergy Clin Immunol. 2016;137(3):852–860. doi:10.1016/j.jaci.2015.08.021
25. Mobasheri A, Batt M. An update on the pathophysiology of osteoarthritis. Ann Phys Rehabil Med. 2016;59(5–6):333–339. doi:10.1016/j.rehab.2016.07.004
26. Kim Y-E, Son MJ, Jung SY, Kwon O, Lee J-H, Lee D-H. Socheongryong-tang for improving nasal symptoms associated with allergic rhinitis: a study protocol for a randomized, open-label, cetirizine controlled, clinical trial. Medicine. 2018;97(34):e11812.
27. Benazzo M, Leonardi S, Corsico A, et al. Cetirizine modifies quality of life and symptoms in children with seasonal allergic rhinitis: a pilot study. Acta Bio Medica. 2021;92(1):e2021003.
 © 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.