Back to Journals » Diabetes, Metabolic Syndrome and Obesity » Volume 11
Influence of gastric bypass surgery on resting energy expenditure, body composition, physical activity, and thyroid hormones in morbidly obese patients
Authors Mirahmadian M, Hasani M, Taheri E, Qorbani M , Hosseini S
Received 23 April 2018
Accepted for publication 25 June 2018
Published 23 October 2018 Volume 2018:11 Pages 667—672
DOI https://doi.org/10.2147/DMSO.S172028
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Ming-Hui Zou
Mehri Mirahmadian,1 Motahareh Hasani,2 Ehsaneh Taheri,3 Mostafa Qorbani,4 Saeed Hosseini5
1Department of Nutrition and Biochemistry, School of Nutrition Sciences and Dietetics, Tehran University of Medical Sciences, 2School of Public Health, Iran University of Medical Sciences, 3Endocrinology and Metabolism Molecular and Cellular Sciences Institute, Tehran University of Medical Sciences, 4Noncommunicable Diseases Research Center, Endocrinology and Metabolism Population Sciences Institute, Tehran University of Medical Sciences, 5School of Nutrition, Tehran University of Medical Sciences, Tehran, Iran
Introduction: One way to lose weight is bariatric surgery. Various studies have shown that after Roux-en-Y gastric bypass (RYGB), resting energy expenditure (REE) decreased may be a result of changes in fat-free mass (FFM) and thyroid-stimulating hormone levels. The aim of our study was to assess changes in body composition, REE, and thyroid hormones in patients undergoing RYGB.
Methods: A total of 42 patients participated (21 undergoing RYGB and 21 age- and weight-matched subjects as controls) in our study. Weight, body-mass index, body composition, resting metabolic rate, physical activity, and thyroid hormones were measured at baseline in cases and 3 months after surgery in case and control groups.
Results: At 3 months after surgery, patients lost an average of 21.7±1.4 kg weight, and fat mass, FFM, and REE decreased significantly. REE was higher in cases compared to controls. T4, T3, and thyroid-stimulating hormone at 3 months after RYGB decreased, but the decrease was significant only in T4 compared to controls. FFM was higher and fat mass lower in cases compared to controls.
Conclusion: We detected a meaningful difference in REE before and after surgery, but we did not detect any meaningful difference in REE between controls and cases.
Keywords: gastric bypass surgery, resting energy expenditure, body composition
Introduction
Obesity is recognized as a major public-health problem that has reached epidemic proportions in both developed and developing countries. Obesity is an independent risk factor for developing other chronic diseases.1 Obesity is a considered a disease with multiple causes. In addition to genetic factors, unlimited access to high-calorie foods and industrial and urban lifestyles accompanied by less physical activity plays roles in overweight, obesity, and increasing numbers of people suffering from both.2,3 In the 2000s, the World Health Organization predicted that about 2.3 billion people would be suffering from overweight and 700 million individuals affected by obesity by 2015. The overall prevalence rate of obesity and over weight in Iran was reported as 42.8% of women in 2005.4
Bariatric surgery is recognized as a long-term treatment for morbid obesity.5 The number of bariatric surgery procedures performed in the US increased from about 200,000–220,000 cases in the period 2003–2008.6 Among all kinds of bariatric surgery, Roux-en-Y gastric bypass (RYGB) is the most common procedure in the US. RYGB is often done as a laparoscopic surgery. The size of the upper stomach is reduced to a small pouch in this operation. This pouch attaches directly to a part of the small intestine called the Roux limb. This reduces intake and absorption of food. Bypass surgery can reduce about 60% of excess body weight by restricting energy intake and decreasing absorption of nutrients.7
Obesity is considered a disease and a very important factor in being affected by chronic diseases, such as type 2 diabetes, hypertension, dyslipidemia, heart ischemia, osteoarthritis, and polycystic ovary syndrome, in addition to some cancers. These conditions and the increasing trend of obesity show the importance of the issue and examining treatment methods of obesity.2 Various studies have shown that after RYGB, resting energy expenditure (REE) decreases. A systematic overview of effect of RYGB on weight loss has shown that 31.3% of weight loss following the surgery is due to reduction in fat-free mass (FFM).8 In a study performed by de Moraes et al, it was shown that RYGB resulted in reductions in thyroid-stimulating hormone (TSH) levels. Reductions of TSH and FFM are considered factors resulting in reduction of REE after surgery.6 REE is defined as energy consumed through necessary body activities to provide for natural body functions and homeostasis, such as breathing, blood circulation, organic compound synthesis, and pumping ions through the membrane. REE is affected by different factors, such as size of body, body composition (fat mass [FM] and FFM), age, sex, hormonal status, and some other factors (caffeine, nicotine, alcohol, and fever). Considering the role of obesity in the creation of chronic diseases and its high prevalence, the authors decided to determine levels of REE changes and weight loss stemming from RYGB, in order to show that REE changes following bariatric surgery play a role in levels of weight loss resulting from these types of surgeries.
Methods
This study was conducted on 21 patients aged 18–60 years with severe obesity scheduled to undergo RYGB surgery at the bariatric surgery center of a university hospital (Rasol Hospital) in Tehran during 2014–2015. All participants completed follow-up assessments in clinic visits. In accordance with US National Institutes of Health criteria for bariatric surgery, patients who underwent surgery had body-mass index BMI values >40 kg/m2.
A total of 21 controls were selected randomly from persons matched for age and sex and postoperative weight for modifying confounding effects. All participants filled out a written consent form and a demographic information form. Exclusion criteria were having any diagnosed diseases, including thyroid disorders, liver and kidney dysfunction, and history of type 1 and 2 diabetes. Also, patients who used drugs, including hormonal agents, nonsteroidal anti-inflammatory, corticosteroids, antipsychotics, and sibutramine, currently or in the 6 months preceding surgery were excluded from the study. Body composition, REE, physical activity level, and anthropometric and biochemical parameters were measured at 1 day before the surgery and at 3 months after surgery. The study protocol was approved by the medical ethics committee of Tehran University of Medical Sciences.
Anthropometry and physical activity assessment
Weight and height were measured using a Seca 725 scale (Seca, Hamburg, Germany) while subjects wore light clothes and no shoes in a standing posture. Accuracy of weight and height was within 0.1 kg and 0.5 cm, respectively. BMI was calculated as weight (kg) divided by height squared (m2). We used the International Physical Activity Questionnaire to estimate physical activity levels of participants.
Resting energy expenditure and body composition
REE was measured using a MetaCheck indirect calorimeter (model 7100; Korr Medical Technologies, Salt Lake City, Utah, USA), previously validated in adults. All measurements were performed while participants were sitting in a thermoneutral room after a 12-hour overnight fast and having refrained from caffeine, nicotine, or exercise for at least 4 hours. Consumption of caffeine, smoking, and physical activity were not allowed for at least 4 hours before measurements. The device measures oxygen consumption through breath-by-breath sampling over 15–20 minutes and then calculates REE and respiratory quotient. Body composition was measured by bioelectrical impedance analysis on a Tanita scale (BC-418MA; Tanita, Tokyo, Japan) with 200 kg maximum weight capacity and ability to measure 1%–75% body fat.
Blood sampling
For all participants, after overnight fasting, 10 mL venous blood was drawn from the antecubital vein of the right arm while patients were seated. Blood samples were collected in EDTA-containing tubes for plasma separation and tubes without anticoagulant for serum separation. All blood samples were transported to the laboratory of Shariati Hospital in Tehran and stored at −79°C until used. To measure serum levels of T3 (ng/mL) and T4 (µg/dL), radioimmunoassays were used, and for measurement of TSH (µIU/mL) immunoradiometric assays were applied.
Statistical analysis
SPSS version 16 (SPSS, Chicago, IL, USA) was used for all statistical analyses. Continuous data are presented as mean ± SD. Paired independent t-tests were used for comparisons between variables, including weight, BMI, and dietary intake components before and after the RYGB surgery.
Results
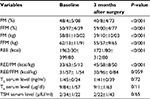
A total of 42 persons participated in this study: 21 subjects who underwent RYGB surgery and 21 subjects as a control group. All participants were female. Follow-up was 100% at 3 months after surgery. The mean age of subjects in the surgery and control groups was 34.33±8.55 and 35.11±7.90 years, respectively. The mean of weight in participants was 121/9±19/4 kg and BMI 46/1±6/4 kg/m2 before surgery (Table 1). Three months after RYGB surgery, mean weight and BMI had decreased to 94.8±18.0 kg and 35.8±5.47 kg/m2, respectively. Mean total weight loss and change in BMI at 3-month follow-up were statistically significant (P<0.001 for both). Table 2 shows the demographic and anthropometric characteristics of morbidly obese patients before and after surgery. After surgery, mean FM, FFM, and REE decreased compared to baseline. The average reduction in REE was 239.5±87 kcal at 3 months after surgery. On the other hand, REE:FM ratio (t=1.33, P<0.001) had increased significantly, but REE:FFM ratio (t=0.78, P=0.059) had decreased at 3 months after surgery compared to baseline. T4, T3, and TSH at 3 months after RYGB had decreased, but not statistically significant.
  | Table 1 Comparison of BMI and Weight average in baseline and 3 months after surgery Notes: Data are expressed as mean±SD. P<0.05 is statistically significant. |
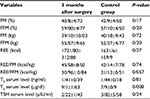
Table 3 shows the demographic and anthropometric characteristics of obese patients at 3 months postsurgery and controls. Three months after surgery in obese patients, FM was lower and FFM higher compared to controls. Also, REE was higher in patients 3 months after surgery compared to controls. On the other hand, REE:FM ratio (P=0.74) was higher but REE:FFM ratio (P=0.057) lower at 3 months after surgery compared to controls. At 3 months after surgery, serum levels of T3 (t=024, P=0.81) and T4 (t=2.81, P=0.008) were higher and TSH (t=1.19, P=0.24) lower in cases compared to controls. Only T4 serum levels showed statistically significant differences. Levels of physical activity are shown in percentiles 25, 50, and 75 in Table 4. There were no significant statistical differences between patients’ physical activity before vs after surgery. On the other hand, the number of patients with mild or moderate activity was significantly higher than the control group (Table 5). Also, the number of subjects in the control group with severe activity was significantly higher than obese patients after surgery (P<0.001).
  | Table 4 Comparison of physical activity level in baseline and 3 months after surgery Note: Wilcoxon Signed Rank Test*. |
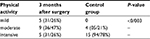  | Table 5 Comparison of physical activity level in case group and control groups Note: Pearson χ2. |
Discussion
This study aimed to evaluate the impact of RYGB on REE. The effect of weight-loss surgery was investigated via age, weight, BMI, REE, body composition, serum level of thyroid hormones, and level of physical activity in the preoperative period and at 3 months after surgery in the case group, and these variables in the case group 3 months after surgery were compared with the control group. Our results showed that 3 months after surgery, mean weight loss was 27.1±1.4 kg and BMI loss 10.3±0.7 kg/m2 compared to presurgery. Mean weight and BMI loss were 35.6.1±2.4 kg and 12.9±0.7 kg/m2, respectively, in Tamboli et al10 and 33.4.1±7.6 kg,12.9±2.3 kg/m2 in Carrasco et al9 at 6 months after surgery. Our findings were consistent with other studies.9,10 However, mean follow-up in other studies was at least 6 months, while ours was 3 months.
In our study, resting metabolic rate significantly reduced by about 240.5±87 kcal at 3 months after surgery compared to prior to surgery, while this value was 396±157 kcal in Carrasco et al9 and about 440 kcal in Carey et al11 at 6 months after surgery. It is possible that the short duration of our study was the cause of the lower reduction compared to other research. In terms of body composition, FM and FFM had significantly decreased by about 19.71±0.1 kg and 6.56±2.26 kg 3 months after surgery compared to prior.
Carrasco et al found that FM and FFM decreased by 20.6±6.9 and 7.8±4.8 kg in 6 months after RYGB surgery. With an average reduction of 3.25±3.63 kcal, the REE:FFM ratio decreased significantly from 33.4±4.1 to 30.1±2.6 kcal/kg.9 Das et al found that REE reduced after weight loss in bariatric surgery due to FFM reduction,12 due to body FFM including skeletal muscles, organs, bones, and visceral tissue. Skeletal muscles and organs make lean body mass, which underpins FFM metabolic activity and plays a major role in determining REE.12
The results of other similar studies have shown that FM and FFM kg decrease. FM and FFM percentages decrease too. Though the body FM percentage in our study decreased similarly to other studies, the FFM percentage increased. This contradiction can be explained by FFM percentage being measured and compared in our study at 3 months after surgery and before surgery, while it has been longer in similar studies. On the other hand, in our study there was more FM reduction compared to FFM reduction, as the increase in FFM percentage represented only mathematical calculations of weight loss with further reduction in FM and less reduction in FFM. Moreover, the results showed no significant difference in terms of data (REE, FM, FFM, REE/FM, REE/FFM) between cases and controls. It can be concluded that body composition is the main component that determines REE and REE loss following bariatric surgery, due to reduction in FFM.
If patients are selected according to appropriate research criteria and surgery is successful, metabolic and physiological changes in the body in relation to REE are equivalent to someone who naturally has the same weight and body composition without any intervention. Faria et al investigated the importance of REE in weight return in patients undergoing bariatric surgery. In this study, 36 patients undergoing RYGB were divided into two groups. The first group maintained their weight, while in the second group patients had weight gain at least >2 kg after 2 years. Faria et al found that patients with higher FM and lower REE percentage after surgery were much more likely to return to their previous weight.13
The results of our study and similar others suggest that a low metabolic rate can be a factor in regaining weight after RYGB surgery. Therefore, resting metabolic rate in patients undergoing bariatric surgery is important in weight loss and weight return. In our study, T3 and TSH levels had decreased and T4 levels increased at 3 months after surgery compared to prior. However, these changes were not statistically significant.
In another study conducted by Raftopoulos et al,14 among 224 patients who received RYGB surgery between 1999 and 2002, 23 (10.3%) had hypothyroidism. Rates of improved thyroid function and unchanged thyroid function were evaluated, and it was found that at 17-month follow-up ten of 23 (43.5%) had improved thyroid function. The drug treatment was completely eliminated in two patients and the need for thyroxine treatment decreased among the remaining eight.14,15 Our study results were consistent with other similar studies.16,17 Many theories have been suggested about the impact of obesity on thyroid function. Hypothyroidism affects the metabolism of carbohydrates and lipids by reduction in REE and has a role in body-weight increase and metabolic side effects. On the other hand, elevation of TSH serum may be caused by obesity,an increase of adipose tissue and adipocytokins stimulate the pituitary gland. After weight-loss surgery, weight reduction improves thyroid hormones, which may result in the secretion of adipocytokines in relation to obesity. One of the adipocytokines is leptin. Leptin plays a role in regulating TRH mRNA gene expression in the hypothalamic periventricular nucleus.18 Leptin is secreted by lipid cells, and its serum level is proportional to the amount of FM. As a result, changed leptin levels may have a role in thyroid-hormone levels due to weight loss.19
In the present study, there was no statistically significant difference in physical activity 3 months after surgery compared to before surgery. Physical activity in percentile 25 improved of mild level to moderate level. Boan et al studied patients after RYBG surgery through questionnaires, and found that their physical activity had improved at 6 months after surgery.20 The sensitivity of patients in regard to weight is the most important factor that can increase the amount of physical activity in patients undergoing bariatric surgery.
Limitations
There were several limitations to this study, such as small sample and short follow-up. In addition, this was a cross-sectional study, such that serum levels of T3, T4, and TSH were measured only once. Future studies should include long-term follow-up after surgery and larger samples to determine the effect of RYGB surgery on the amount of REE. In addition, to our knowledge, no previous study has evaluated all of our variables (body composition, REE, physical activity, and thyroid hormones) before and after RYGB surgery.
Conclusion
Our results clearly demonstrated that the resting metabolic rate of the body and influencing factors, such as FM and FFM, had decreased at 3 months after RYGB surgery. The basal metabolic rate in the case group at 3 months after RYGB was at a lower level than cases with the same weight. In regard to the importance and role of REE in weight loss, it can be concluded that lower REE levels after surgery might be a factor in weight returning among patients who undergo bariatric surgery. However, if patients are not selected according to the correct criteria for surgery, such as those who have low REE from the beginning, there is a lower chance of weight loss after surgery, In our study, 3 months seemed to be insufficient to evaluate REE and compare it with the control group. Surgical side effects and physiological and anatomical adjustments, eg, lower levels of TSH and FFM, are factors that may affect REE. Therefore, the quality of weight loss should be considered among patients who undergo bariatric surgery.
Acknowledgment
This study was supported by Tehran University of Medical Sciences (grant EC_00299).
Disclosure
The authors report no conflicts of interest in this work.
References
Calle EE, Rodriguez C, Walker-Thurmond K, Thun MJ. Overweight, obesity, and mortality from cancer in a prospectively studied cohort of U.S. adults. N Engl J Med. 2003;348(17):1625–1638. | ||
Flegal KM, Carroll MD, Kit BK, Ogden CL. Prevalence of obesity and trends in the distribution of body mass index among US adults, 1999-2010. JAMA. 2012;307(5):491–497. | ||
Nguyen DM, El-Serag HB. The epidemiology of obesity. Gastroenterol Clin North Am. 2010;39(1):1–7. | ||
Janghorbani M, Amini M, Willett WC, et al. First nationwide survey of prevalence of overweight, underweight, and abdominal obesity in Iranian adults. Obesity. 2007;15(11):2797–2808. | ||
Wadden TA, Butryn ML, Byrne KJ. Efficacy of lifestyle modification for long-term weight control. Obes Res. 2004;12 Suppl:151S–162S. | ||
Belle SH, Berk PD, Courcoulas AP, et al. Safety and efficacy of bariatric surgery: longitudinal assessment of bariatric surgery. Surg Obes Relat Dis. 2007;3(2):116–126. | ||
Buchwald H, Estok R, Fahrbach K, et al. Weight and type 2 diabetes after bariatric surgery: systematic review and meta-analysis. Am J Med. 2009;122(3):248–256. | ||
Chaston TB, Dixon JB, O’Brien PE. Changes in fat-free mass during significant weight loss: a systematic review. Int J Obes. 2007;31(5):743–750. | ||
Carrasco F, Papapietro K, Csendes A, et al. Changes in resting energy expenditure and body composition after weight loss following Roux-en-Y gastric bypass. Obes Surg. 2007;17(5):608–616. | ||
Tamboli RA, Hossain HA, Marks PA, et al. Body composition and energy metabolism following Roux-en-Y gastric bypass surgery. Obesity. 2010;18(9):1718–1724. | ||
Carey DG, Pliego GJ, Raymond RL. Body composition and metabolic changes following bariatric surgery: effects on fat mass, lean mass and basal metabolic rate: six months to one-year follow-up. Obes Surg. 2006;16(12):1602–1608. | ||
Das SK, Roberts SB, Mccrory MA, et al. Long-term changes in energy expenditure and body composition after massive weight loss induced by gastric bypass surgery. Am J Clin Nutr. 2003;78(1):22–30. | ||
Faria SL, Kelly EO, Faria OP, Ito MK. Snack-eating patients experience lesser weight loss after Roux-en-Y gastric bypass surgery. Obes Surg. 2009;19(9):1293–1296. | ||
Raftopoulos Y, Gagné DJ, Papasavas P, et al. Improvement of hypothyroidism after laparoscopic Roux-en-Y gastric bypass for morbid obesity. Obes Surg. 2004;14(4):509–513. | ||
de Moraes CM, Mancini MC, de Melo ME, et al. Prevalence of subclinical hypothyroidism in a morbidly obese population and improvement after weight loss induced by Roux-en-Y gastric bypass. Obes Surg. 2005;15(9):1287–1291. | ||
Fazylov R, Soto E, Cohen S, Merola S. Laparoscopic Roux-en-Y gastric bypass surgery on morbidly obese patients with hypothyroidism. Obes Surg. 2008;18(6):644–647. | ||
Chikunguwo S, Brethauer S, Nirujogi V, et al. Influence of obesity and surgical weight loss on thyroid hormone levels. Surg Obes Relat Dis. 2007;3(6):631–635. | ||
de Moraes CM, Mancini MC, de Melo ME, et al. Prevalence of subclinical hypothyroidism in a morbidly obese population and improvement after weight loss induced by Roux-en-Y gastric bypass. Obes Surg. 2005;15(9):1287–1291. | ||
Müller MJ, Bosy-Westphal A, Kutzner D, Heller M. Metabolically active components of fat-free mass and resting energy expenditure in humans: recent lessons from imaging technologies. Obes Rev. 2002;3(2):113–122. | ||
Boan J, Kolotkin RL, Westman EC, Mcmahon RL, Grant JP. Binge eating, quality of life and physical activity improve after Roux-en-Y gastric bypass for morbid obesity. Obes Surg. 2004;14(3):341–348. |
 © 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.