Back to Journals » International Journal of Chronic Obstructive Pulmonary Disease » Volume 15
Influence of Comorbidities on the Survival of COPD Patients According to Phenotypes
Authors Ji Z , Hernández-Vázquez J , Domínguez-Zabaleta IM , Xia Z, Bellón-Cano JM , Gallo-González V , Ali-García I , Matesanz-Ruiz C, López-de-Andrés A, Jiménez-García R, Buendía-García MJ, Gómez-Sacristán, Girón-Matute WI, Puente-Maestu L , de Miguel-Díez J
Received 13 July 2020
Accepted for publication 22 September 2020
Published 30 October 2020 Volume 2020:15 Pages 2759—2767
DOI https://doi.org/10.2147/COPD.S270770
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 4
Editor who approved publication: Dr Richard Russell
Zichen Ji,1,2 Julio Hernández-Vázquez,3 Irene Milagros Domínguez-Zabaleta,1 Ziyi Xia,2 José María Bellón-Cano,4 Virginia Gallo-González,1 Ismael Ali-García,3 Carmen Matesanz-Ruiz,3 Ana López-de-Andrés,5 Rodrigo Jiménez-García,6 María Jesús Buendía-García,3 Ángela Gómez-Sacristán,7 Walther Iván Girón-Matute,1,2,7 Luis Puente-Maestu,1,2,7 Javier de Miguel-Díez1,2,7
1Pulmonology Service, Gregorio Marañón General University Hospital, Madrid, Spain; 2Faculty of Medicine, Complutense University of Madrid, Madrid, Spain; 3Pulmonology Section, Infanta Leonor University Hospital, Madrid, Spain; 4Research Support Service, Gregorio Marañón Health Research Institute, Madrid, Spain; 5Preventive Medicine and Public Health Teaching and Research Unit, Health Sciences Faculty, Rey Juan Carlos University, Alcorcón, Madrid, Spain; 6Public Health and Maternal and Child Health Department, Faculty of Medicine, Complutense University of Madrid, Madrid, Spain; 7Gregorio Marañón Health Research Institute, Madrid, Spain
Correspondence: Zichen Ji
Pulmonology Service, Gregorio Marañón University General Hospital, Madrid 28007, Spain
Tel +34 617 882 547
Email [email protected]
Background: Chronic obstructive pulmonary disease (COPD) usually occurs alongside other conditions. Few studies on comorbidities have taken into account the phenotypes of COPD patients. The objective of this study is to evaluate the prevalence of comorbidities included in the Charlson index and their influence on the survival of patients with COPD, taking phenotypes into account.
Methods: An observational study was conducted on a group of 273 patients who had COPD and underwent spirometry in the first half of 2011, with a median prospective follow-up period of 68.15 months. The survival of these patients was analyzed according to the presence of various comorbidities.
Results: Of the 273 patients, 93 (34.1%) died within the follow-up period. An increased presence of chronic ischemic heart disease (CIHD), chronic heart failure (CHF), chronic kidney disease (CKD), and malignancy was found in deceased patients. All of these conditions shorten the survival of COPD patients globally; however, when considering phenotypes, only CHF influences the exacerbator with chronic bronchitis phenotype, CKD influences the non-exacerbator phenotype, and malignancy influences the positive bronchodilator test (BDT) and exacerbator with chronic bronchitis phenotypes. In the multivariate model, advanced age (hazard ratio, HR: 1.05; p=0.001), CHF (HR: 1.74; p=0.030), and the presence of malignancy (HR: 1.78; p=0.010) were observed as independent mortality risk factors.
Conclusion: The survival is shorter in the presence of CIHD in overall COPD patients and also CHF, CKD, and malignancy for certain phenotypes. It is important to pay attention to these comorbidities in the comprehensive care of COPD patients.
Keywords: COPD, phenotypes, comorbidities, risk factors, mortality, survival
Introduction
Chronic obstructive pulmonary disease (COPD) is a widely prevalent condition with high morbidity and mortality rates.1 It often leads to comorbidities2–4 because the disease has systemic involvement and shares common risk factors with other pathologies. For this reason, a comorbidome has been described, where the associations of COPD with other diseases are represented.5
Numerous studies have analyzed the influence of certain diseases on COPD and vice versa, including ischemic heart disease,6 heart failure,7 cerebrovascular disease,8 diabetes mellitus (DM),9 and lung cancer.10
There are large cohort or population studies that have analyzed the set of comorbid conditions that COPD patients could suffer from. Gershon et al11 described the prevalence of comorbidities in COPD patients and proposed studies to determine the long-term influence of these conditions on the evolution of COPD. Although some similar studies exist, most of them have been carried out over a decade ago.12 For this reason, it is important to obtain updated the data to adapt to changes in the epidemiology of these diseases.
The classification of COPD patients into different phenotypes is a recommendation that has been added to clinical practice guidelines in recent years,13,14 since not all COPD patients clinically behave in the same way or present the same evolution.15 Therefore, it is also interesting to investigate the influence of these comorbid conditions on each COPD phenotype.
The objective of the present study was to evaluate the prevalence of comorbidities included in the Charlson index in patients with COPD, both overall and for each phenotype, and to also identify those comorbid conditions associated with higher mortality rates.
Patients and Methods
Design
We conducted an observational non-interventional study of a cohort of patients with established diagnoses of COPD. They were recruited in the stable phase, and a prospective follow-up was made, with the aim of avoiding any possible biases that may exist in a retrospective study.
The patients that participated in the research were over 40 years old and had a history of smoking, with a pack-year index equal to or greater than 10. They had a previous and confirmed diagnosis of COPD, established using a spirometry test, and they underwent scheduled spirometry tests between January 1, 2011 and June 30, 2011.
Those patients who had a forced expiratory volume in one second (FEV1) above 70% of the predicted value were excluded from the study. Further, neither patients with non-obstructive respiratory pathologies nor patients included in clinical trials were admitted.
Parameters
We collected the parameters during recruitment and at the end of the study. The recruitment period ran from January to June 2011, and the follow-up period ended in April 2017.
The demographic data of patients and their comorbidities were collected in the recruitment phase: age, gender, weight, height, body mass index (BMI), date of inclusion, FEV1, smoking history, COPD phenotype, pharmacological treatment of disease, respiratory therapies, and comorbidities included in the Charlson index, which has been widely used to evaluate the prognosis of patients, taking 22 comorbid conditions into account.
Mortality data were collected in the follow-up phase: the last follow-up date of surviving patients and the cause of death and death date of deceased patients.
Definition of Phenotypes
All patients included in the study were classified into one of the following four COPD phenotypes: non-exacerbator (NE), positive response to bronchodilator (PRTB), exacerbator with emphysema (EWE), and exacerbator with chronic bronchitis (EWCB).
The process followed to classify COPD patients into these phenotypes was as follows. First, patients with a positive response to the bronchodilator test, defined by an increment of FEV1 of at least 200 mL in absolute value and at least 12% in comparison to FEV1 without a bronchodilator, were considered to be PRTB phenotype. Third, patients who were not classified into either of these two phenotypes were considered as EWE or as EWCB phenotypes, according to the following criteria: if dyspnea was the patient’s main symptom and radiological emphysema or a decrease in the capacity of diffusion of carbon monoxide (DLCO) existed (which supports the diagnosis of emphysema), they were classified as EWE phenotype. If their predominant symptom was cough and expectoration for at least 3 months per year in the last two years, they were considered to be patients with EWCB phenotype.
This classification process did not exactly conform to the definition provided in the Spanish guidelines on COPD (GesEPOC).13 According to GesEPOC, a patient has asthma-COPD overlap (ACO) phenotype if they demonstrate a very positive response in the bronchodilator test, defined as an FEV1 increment greater than 400 mL in absolute value and 15% in relative value. Similar to other published studies,15 we think that this definition provides very high specificity but little sensitivity for this phenotype. Instead of the ACO phenotype, we use the PRTB phenotype.
Cause of Death
In the group of deceased patients with a known cause of death, for both in deaths produced inside and outside the hospital, we were able to establish four primary causes of death: respiratory, cardiovascular, infectious, and malignancy. The respiratory causes included COPD exacerbation, respiratory failure, and respiratory acidosis. The cardiovascular causes included acute myocardial infarction, decompensation of heart failure, and pulmonary thromboembolism. The infectious causes included infections in any location. The malignancy causes included the complications arising from tumor progression without any other underlying causes. If several different causes of death were present, only the predominant one was considered.
Statistical Analysis
A histogram was used to check the normal distribution. The quantitative variables that had a normal distribution were expressed in terms of their mean and standard deviation (SD). The quantitative variables that did not follow a normal distribution were expressed in terms of their median and interquartile range (IQR). Qualitative variables were expressed as frequencies and percentages.
The analysis of variance test was used to compare quantitative variables with normal distribution, while the Kruskal–Wallis test was used to analyze the quantitative variables without normal distributions. Depending on the sample size, the chi-squared test or Fisher’s exact test were used to compare qualitative variables and proportions.
The univariate comparison of mortality based on the presence or absence of certain comorbid conditions, was performed using the long-rank test. The analysis of survival was graphically represented in Kaplan–Meier graphs.
A Cox regression was performed during multivariate analysis, with the aim of adjusting mortality to gender, age, phenotypes and pulmonary function with the presence of chronic ischemic heart disease (CIHD), chronic heart failure (CHF), chronic kidney disease (CKD), and malignancy. The proportionality of risks of the included variables was also checked. Further, the level of statistical significance considered for all comparisons was two-tailed p < 0.05.
The statistical program used for all analyses was SPSS version 26, except for the graphical representation and the survival analysis, where Stata version 15 was used.
Ethics and Informed Consent
Our study was approved by the Ethics Committee of Gregorio Marañón University General Hospital, with approval code HUIL-1507. Patients signed informed consent prior to taking part in the study.
This study was conducted in accordance with the Declaration of Helsinki.
Results
During the recruitment period, 273 patients met the eligibility criteria. All of them agreed to participate in the study and none withdrew early. Of all the patients included in this study, 243 (89%) of whom were male. The median of the follow-up period was 68.16 months (IQR 40.96–72.12). The mean age was 68 years (SD 10.62). The mean height was 1.63 m (SD 0.08) and the mean weight was 75.03 kg (SD 16.89); therefore, the mean BMI was 28.05 kg/m2 (SD 5.49). Regarding pulmonary function, the mean absolute value of FEV1 was 1211 mL (SD 417) and 48.64% predicted (SD 12.59). At inclusion, 92 patients (34%) were active smokers. In relation to COPD pharmacological treatment, 242 patients (88.6%) had chronic treatment with a long-acting adrenergic β-2‐agonist (LABA) at the time of recruitment, 254 patients (93%) were taking a long-acting muscarinic antagonist (LAMA), and 212 (77.7%) were receiving inhaled corticosteroids (ICS). Regarding oxygen and positive pressure therapies, continuous positive airway pressure (CPAP) was used by 31 patients (11.4%), 14 patients (5.1%) were treated with bi-level positive airway pressure (BiPAP) therapy, and long-term oxygen therapy (LTOT) was used in 91 patients (33.3%). Full descriptive data, overall and for each phenotype, are shown in Table 1.
 |
Table 1 Description and Comparison of General Characteristics of All Patients and According to Phenotypes |
The presence of the comorbidities included in the Charlson index—both overall and for each phenotype—is shown in Table 2. It also shows the scores for this index. A higher proportion of patients with CIHD were observed in the EWCB phenotype (p=0.000), a higher presence of dementia was found in the exacerbator with emphysema phenotype (p=0.017), and the only patient with cerebrovascular disease belonged to the exacerbator with emphysema phenotype (p=0.027).
 |
Table 2 Comorbidities in All Patients and According to Phenotypes |
Table 3 shows the presence of comorbidities included in the Charlson index as a function of survival or death during follow-up. A higher proportion of CIHD, CHF, CKD, and malignancy were found among deceased patients, a finding that was statistically significant (p=0.041, p=0.003, p=0.022 and p=0.004, respectively). In addition, the deceased patient group also had a higher Charlson comorbidity index score (p=0.000).
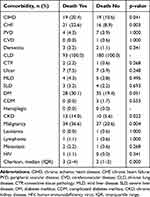 |
Table 3 Mortality According to Each Comorbidity and Charlson Index |
Figure 1 shows a graphical comparison of the survival rate between COPD patients without comorbidities and those suffering from CIHD, CHF, CKD or malignancy. It is clear that a greater proportion of COPD patients without comorbidities survived in the follow-up period in comparison to patients with comorbidities (Table 3). Figure 2 shows the same comparison, taking COPD phenotypes into account.
 |
Figure 1 Kaplan–Meier graph of survival according to the presence of chronic ischemic heart disease, chronic heart failure, chronic kidney disease and malignancy in overall COPD patients. |
 |
Figure 2 Kaplan–Meier graphs of survival according to the presence of chronic ischemic heart disease, chronic heart failure, chronic kidney disease and malignancy in each phenotype. |
Table 4 shows the numerical results for the survival, log-rank, and p-values of COPD patients as a function of COPD phenotypes and comorbid conditions. Comorbid conditions—such as CIHD, CHF, CKD, and malignancy—were shown to reduce the overall survival rates of COPD patients (p=0.012, p=0.000, p=0.050 and p=0.000, respectively). However, taking into account the phenotypes, CHF shortened the survival rates in the exacerbator with chronic bronchitis phenotype (p=0.031), CKD reduced survival rates in the non-exacerbator phenotype (p=0.031), and malignancy decreased survival rates in positive bronchodilator response phenotype (p=0.000) and exacerbator with chronic bronchitis phenotype (p=0.003).
 |
Table 4 Survival, Log-Rank and p-value in the Univariate Comparison of Survival According to Each Comorbidity and Phenotype |
On one hand, Table 5 shows that increased age, CHF, and malignancy are risk factors independently associated with mortality (p=0.001, p=0.030 and p=0.010, respectively). On the other hand, it is shown that a high FEV1 absolute value is a protective factor that is independently associated with mortality (p=0.014).
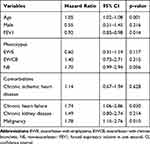 |
Table 5 Risk Factors Independently Associated with Mortality |
Table 6 shows the causes of death among patients with comorbid conditions, with a significant increase in mortality. In some cases where death occurred outside of the hospital, information could not be obtained regarding the cause of death. Using the information available regarding the cause of death, we only observed that patients who suffered from malignancies usually died owing to tumor progression (p=0.000).
 |
Table 6 Cause of Death in Patients Presenting Comorbidity |
Discussion
The main conclusion of our study is that there are some comorbid conditions—such as CIHD, CHF, CKD, and malignancy—that reduce the overall survival rates of COPD patients. If we take COPD phenotypes into account, these comorbidities do not affect all COPD patients in the same way: CHF shortens survival rates in the exacerbator with chronic bronchitis phenotype, CKD reduces survival in the non-exacerbator phenotype, and malignancy decreases survival in positive bronchodilator response and exacerbator with chronic bronchitis phenotypes. Two of these comorbidities (CHF and malignancy) and advanced age are risk factors independently associated with mortality. However, improved lung function, defined as a higher absolute value of FEV1, is a protective factor independently associated with mortality. Regarding the cause of death of deceased patients with comorbid conditions, it was only found that patients with malignancy died of this cause.
It is important to note that, in our study, only patients with COPD of at least moderate severity were considered. Therefore, patients with mild COPD have not been taken into account, since survival in these patients could depend more on other diseases than on COPD.
The results obtained in our study are similar to those published by Almagro et al,16 who also described cardiovascular diseases and CKD as risk factors independently associated with mortality in patients with COPD. The difference between our study and the Almagro study is that the latter investigates a cohort of COPD patients hospitalized for exacerbation, and our study focuses on a population of stable COPD patients. Therefore, our study takes patients with the non-exacerbator phenotype into account.
From a cardiovascular point of view, numerous studies have shown the negative effect of the coexistence of these diseases with COPD on patient survival.17–20 These diseases appear together very frequently, possibly due to common pathogenesis through tobacco use and systemic inflammation.21,22
Although there are only a few studies on the topic, it has been found that CKD worsens the prognosis of COPD,7 and vice versa,23 which aligns with the findings in our study. The pathophysiological mechanism by which this phenomenon occurs is not clear, but it seems that the malnutrition caused by CKD can cause COPD patients to lose exercise capacity faster. As a consequence, they have lower lung function, quality of life, and survival rates.24
The relationship between COPD and malignancies, particularly lung cancer, has been widely researched. The significant effect of lung cancer on COPD patients is well known.25–29 These results are consistent with those of the present study; malignancy is the only comorbid condition that has been found to be a significant cause of death. The mechanism of this association is influenced by not only tobacco use, but also by genetic and molecular mechanisms shared by COPD and malignancies.30
Some studies have found that dementia is a risk factor causing higher mortality;31,32 however, this was not corroborated by our findings. This could be attributed to the fact that this disease makes it difficult to perform spirometry tests, which is why few patients included in the study present this pathology.
COPD is a heterogeneous disease, for which different phenotypes have been defined over the years to better classify the disease. Several large-scale studies have shown that the existence of comorbidities differs according to phenotypes.33,34 The difference could account for the differing influence of each comorbidity on each phenotype found in this study. Future studies should focus on each of these comorbidities to better understand the role they play in the different COPD phenotypes.
On one hand, COPD causes systemic inflammation, which can affect other organs; on the other hand, COPD shares risk factors with other diseases.2 The comorbidities that have been identified as risk factors independently associated with mortality are possibly caused by a combination of both mechanisms.
The protective effect of elevated lung function found in our study matches evidence found in existing literature.13,14
Our study has certain strengths. First, in comparison to studies using retrospective population data—which take into account a wide variety of comorbidities—ours offers a prospective design. To this end, we obtained data on comorbidities present at the beginning of the study, not at the end of the follow-up period. This allowed us to understand the patient’s prognosis from first contact. This prospective design also facilitates the elimination of certain biases present in retrospective studies. Second, in contrast to studies that specifically investigate certain comorbidities, we analyzed all the comorbidities included in the Charlson index. Third, the analysis of comorbid conditions within each COPD phenotype is a novel approach to the topic.
However, our study also has some limitations. First, the comorbid conditions collected are limited to those present at the time of inclusion, without taking into account those that may appear during the follow-up period. Second, the sample size was not sufficient to include patients with low-prevalence comorbid conditions. Third, as this is a single-center study in Spain, the sample only represents the Spanish population. Therefore, it can be difficult to generalize the conclusions for the general population, where the presence of comorbidities may differ. Fourth, our cohort is predominantly male; this may not accurately reflect the presence of comorbidities in women. Finally, because our study is focusses on COPD phenotypes, the patients have not been grouped according to the ABCD GOLD classification. Future studies with specific designs at follow-up and with larger sample sizes are necessary to overcome these limitations.
Conclusions
Chronic ischemic heart disease, chronic heart failure, chronic kidney disease, and malignancy negatively affect the survival rates of patients with COPD. In daily clinical practice, comprehensive care for COPD patients—particularly taking these comorbidities into account—is important owing to their negative effects on COPD patients.
Funding
Menarini supported this study with an unconditional scholarship. This research is a part of the work funded by the Madrid Society of Pneumology and Thoracic Surgery (XVIII Awards Edition, 2018) and the Spanish Society of Pneumology and Thoracic Surgery (project no. 733 of the 2018 edition).
Disclosure
Julio Hernández-Vázquez reports grants from MENARINI, during the conduct of the study. Luis Puente-Maestu reports advisory for MSD, grants from ESTEVE, GSK, NOVARTIS, MENARINI, and personal fees from BOSTON-SCIENTIFIC and CHIESI, outside the submitted work. Javier de Miguel-Díez reports grants from SEPAR and NEUMOMADRID, during the conduct of the study. The authors declare no other potential conflicts of interest for this work.
References
1. Alfageme I, de Lucas P, Ancochea J, et al. Nuevo estudio sobre la prevalencia de la EPOC en España: resumen del protocolo EPISCAN II, 10 años después de EPISCAN. Arch Bronconeumol. 2019;55(1):38–47. doi:10.1016/j.arbres.2018.05.011
2. de Miguel Díez J, García TG, Maestu LP. Comorbilidades de la EPOC. Arch Bronconeumol. 2010;46:20–25. doi:10.1016/S0300-2896(10)70058-2
3. De la Iglesia Martínez F, Serrano Arreba J. Enfermedad obstructiva crónica (EPOC) y comorbilidades. Galicia Clínica. 2012;73(Supl 1):30. doi:10.22546/17/310
4. Yin H, Yin S, Lin Q, Xu Y, Xu H, Liu T. Prevalence of comorbidities in chronic obstructive pulmonary disease patients. Medicine. 2017;96(19):e6836. doi:10.1097/MD.0000000000006836
5. Divo M, Cote C, de Torres JP, et al. Comorbidities and Risk of Mortality in Patients with Chronic Obstructive Pulmonary Disease. Am J Respir Crit Care Med. 2012;186(2):155–161.
6. Alqahtani F, Welle GA, Elsisy MF, et al. Incidence, Characteristics, and Outcomes of Acute Myocardial Infarction among Patients Admitted with Acute Exacerbation of Chronic Obstructive Lung Disease. COPD J Chronic Obstr Pulm Dis. 2015:15. doi:10.1080/15412555.2020.1757054.
7. de Miguel-díez J, Morgan JC, García J-G. The association between COPD and heart failure risk: a review. International Journal of Chronic Obstructive Pulmonary Disease. 2013;8:305–312. doi:10.2147/COPD.S31236
8. Corlateanu A, Covantev S, Mathioudakis AG, Botnaru V, Cazzola M, Siafakas N. Chronic Obstructive Pulmonary Disease and Stroke. COPD: Journal of Chronic Obstructive Pulmonary Disease. 2018;15(4):405–413. doi:10.1080/15412555.2018.1464551
9. Mannino DM, Thorn D, Swensen A, Holguin F. Prevalence and outcomes of diabetes, hypertension and cardiovascular disease in COPD. Eur Respir J. 2008;32(4):962–969. doi:10.1183/09031936.00012408
10. Lin H, Lu Y, Lin L, Meng K, Fan J. Does chronic obstructive pulmonary disease relate to poor prognosis in patients with lung cancer? Medicine. 2019;98(11):e14837. doi:10.1097/MD.0000000000014837
11. Gershon AS, Mecredy GC, Guan J, Victor JC, Goldstein R, To T. Quantifying comorbidity in individuals with COPD: a population study. Eur Respir J. 2015;45(1):51–59. doi:10.1183/09031936.00061414
12. Incalzi RA, Fuso L, Rosa MD, et al. Co-morbidity contributes to predict mortality of patients with chronic obstructive pulmonary disease. Eur Respir J. 1997;10(12):2794–2800. doi:10.1183/09031936.97.10122794
13. Miravitlles M, Soler-Cataluña JJ, Calle M, et al. Guía española de la enfermedad pulmonar obstructiva crónica (GesEPOC) 2017. Tratamiento farmacológico en fase estable. Arch Bronconeumol. 2017;53(6):324–335. doi:10.1016/j.arbres.2017.03.018
14. Global Initiative for Chronic Obstructive Lung Disease. Global strategy for the diagnosis, management and prevention of chronic obstructive pulmonary disease [Internet]. 2019https://goldcopd.org.
15. Hernández Vázquez J, Ali García I, Jiménez-García R, et al. COPD phenotypes: differences in survival. Int J Chron Obstruct Pulmon Dis. 2018;13:2245–2251.
16. Almagro P, Cabrera FJ, Diez J, et al. Comorbidities and Short-term Prognosis in Patients Hospitalized for Acute Exacerbation of COPD: the EPOC en Servicios de Medicina Interna (ESMI) Study. Chest. 2012;142(5):1126–1133. doi:10.1378/chest.11-2413
17. Neder JA, Rocha A, Alencar MCN, et al. Current challenges in managing comorbid heart failure and COPD. Expert Rev Cardiovasc Ther. 2018;16(9):653–673. doi:10.1080/14779072.2018.1510319
18. Struß N, Bauersachs J, Welte T, Hohlfeld JM. Linksherzfunktion bei COPD. Herz. 2019;44(6):477–482. doi:10.1007/s00059-019-4816-5
19. Morgan AD, Zakeri R, Quint JK. Defining the relationship between COPD and CVD: what are the implications for clinical practice? Ther Adv Respir Dis. 2018;12:1753465817750524.
20. Onishi K. Total management of chronic obstructive pulmonary disease (COPD) as an independent risk factor for cardiovascular disease. J Cardiol. 2017;70(2):128–134.
21. Cavaillès A, Brinchault-Rabin G, Dixmier A, et al. Comorbidities of COPD. Eur Respir Rev off J Eur Respir Soc. 2013;22(130):454–475. doi:10.1183/09059180.00008612
22. Kirkham PA, Barnes PJ. Oxidative Stress in COPD. CHEST. 2013;144(1):266–273. doi:10.1378/chest.12-2664
23. Fedeli U, De Giorgi A, Gennaro N, et al. Lung and kidney: a dangerous liaison? A population-based cohort study in COPD patients in Italy. <![CDATA[International Journal of Chronic Obstructive Pulmonary Disease]]>. 2017;12:443–450. doi:10.2147/COPD.S119390
24. Trudzinski FC, Alqudrah M, Omlor A, et al. Consequences of chronic kidney disease in chronic obstructive pulmonary disease. Respiratory Research. 2019;20(1):151. doi:10.1186/s12931-019-1107-x
25. Lim JU, Yeo CD, Rhee CK, et al. <p>Comparison of clinical characteristics and overall survival between spirometrically diagnosed chronic obstructive pulmonary disease (COPD) and non-COPD never-smoking stage I-IV non-small cell lung cancer patients. <![CDATA[International Journal of Chronic Obstructive Pulmonary Disease]]>. 2019;14:929–938. doi:10.2147/COPD.S190244
26. Gao Y, Guan W, Liu Q, et al. Impact of COPD and emphysema on survival of patients with lung cancer: A meta-analysis of observational studies. Respirology. 2016;21(2):269–279. doi:10.1111/resp.12661
27. Wang W, Dou S, Dong W, et al. Impact of COPD on prognosis of lung cancer: from a perspective on disease heterogeneity. <![CDATA[International Journal of Chronic Obstructive Pulmonary Disease]]>. 2018;13:3767–3776. doi:10.2147/COPD.S168048
28. Yi Y-S, Ban WH, Sohng K-Y. Effect of COPD on symptoms, quality of life and prognosis in patients with advanced non-small cell lung cancer. BMC Cancer. 2018;18(1):1053. doi:10.1186/s12885-018-4976-3
29. Media AS, Persson M, Tajhizi N, Weinreich UM. Chronic obstructive pulmonary disease and comorbidities’ influence on mortality in non-small cell lung cancer patients. Acta Oncol Stockh Swed. 2019;58(8):1102–1106. doi:10.1080/0284186X.2019.1612942
30. Seijo LM, Zulueta JJ. Understanding the Links Between Lung Cancer, COPD, and Emphysema: A Key to More Effective Treatment and Screening. Oncol Williston Park N. 2017;31(2):93–102.
31. García-Sanz M-T, Cánive-Gómez J-C, Senín-Rial L, et al. One-year and long-term mortality in patients hospitalized for chronic obstructive pulmonary disease. J Thorac Dis. 2017;9(3):636–645. doi:10.21037/jtd.2017.03.34
32. Xie F, Xie L. <p>COPD and the risk of mild cognitive impairment and dementia: a cohort study based on the Chinese Longitudinal Health Longevity Survey. <![CDATA[International Journal of Chronic Obstructive Pulmonary Disease]]>. 2019;14:403–408. doi:10.2147/COPD.S194277
33. Garcia-Aymerich J, Gómez FP, Benet M, et al. Identification and prospective validation of clinically relevant chronic obstructive pulmonary disease (COPD) subtypes. Thorax. 2011;66(5):430–437. doi:10.1136/thx.2010.154484
34. Agusti A, Calverley PM, Celli B, et al. Characterisation of COPD heterogeneity in the ECLIPSE cohort. Respir Res. 2010;11(1):122.
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
