Back to Journals » Drug Design, Development and Therapy » Volume 8
Influence of caffeine and hyaluronic acid on collagen biosynthesis in human skin fibroblasts
Authors Donejko M, Przylipiak A , Rysiak E, Głuszuk K, Surażyński A
Received 22 June 2014
Accepted for publication 12 August 2014
Published 15 October 2014 Volume 2014:8 Pages 1923—1928
DOI https://doi.org/10.2147/DDDT.S69791
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Professor Shu-Feng Zhou
Magdalena Donejko,1 Andrzej Przylipiak,1 Edyta Rysiak,2 Katarzyna Głuszuk,2 Arkadiusz Surażyński2
1Department of Esthetic Medicine, 2Department of Medicinal Chemistry, Faculty of Pharmacy, Medical University of Białystok, Białystok, Poland
Aim: The aim of this study was to evaluate the effect of caffeine on collagen biosynthesis in human skin fibroblasts and the influence of hyaluronic acid (HA) on this process.
Materials and methods: Collagen, [3H]-thymidine incorporation, and prolidase activity were measured in confluent human skin fibroblast cultures that had been treated with 1, 2, and 5 mM caffeine and with caffeine and 500 µg/mL HA. Western immunoblot analysis was performed to evaluate expression of ß1-integrin receptor, insulin-like growth factor receptor phospho-Akt protein and mitogen-activated protein kinase (phospho-extracellular signal-regulated kinase).
Results: Caffeine inhibited collagen biosynthesis in a dose-dependent manner. The mechanism of this process was found at the level of prolidase activity. Caffeine significantly inhibited the enzyme activity. The addition of HA had no effect on collagen biosynthesis or prolidase activity in fibroblasts incubated with caffeine. Caffeine also had an inhibitory effect on DNA biosynthesis. HA, however, did not have any significant effect on this process. The inhibition of the expression of ß1-integrin and insulin-like growth factor receptor in fibroblasts incubated with the caffeine indicates a possible mechanism of inhibition of collagen biosynthesis.
Conclusion: Caffeine reduces collagen synthesis in human cultured skin fibroblasts. HA did not have any significant protective effect on this process. This is the first study to our knowledge that reports caffeine-induced inhibition of collagen synthesis in human skin fibroblasts.
Keywords: collagen, caffeine, hyaluronic acid, fibroblast
Introduction
Caffeine (methyltheobromine) is an alkaloid and a widespread ingredient of several pharmacy drugs. Caffeine is also present in soft drinks, energy drinks, and coffee infusions. Mean daily consumption of this substance oscillates between 3 and 7 mg/kg body mass. Cytochrome P450 is responsible for metabolizing this alkaloid. Caffeine exhibits anti-inflammatory activity through the inhibition of tumor necrosis factor synthesis and through increasing the internal cell concentration of cyclic adenosine monophosphate and has also displayed antioxidant and radioprotective properties.1 It has been shown that caffeine has an influence on cell migration and proliferation.2 However, regardless of concentration, caffeine disrupts the progression of the cell cycle, which leads to apoptosis.3,4 Hence, one of the adverse effects of caffeine use is the inhibition of the wound healing process.5
Collagen biosynthesis plays an important role in wound healing. Collagen, a dominant protein in the body, is the main element of the extracellular matrix. This protein affects the structure maintenance (elasticity and durability) of many organs, including skin, bones, cartilage, and tendons.6 The amount of collagen in a cell is the resultant of its processes of synthesis and degradation. The influence of caffeine on these processes has not so far been explained.
A substance proven to possess a protective effect on collagen synthesis is hyaluronic acid (HA).7,8 HA belongs to the glycosaminoglycan family of polysaccharides and is composed of a D-glucuronic acid connected via glycosidic bonds with a particle of N-acetylglucosamine.9,10 In the body of animals, hyaluronan is most abundant in the extracellular matrix, where, along with chondroitin sulfate, heparan sulfate, and scleroproteins, it becomes its main carbohydrate component. Hyaluronan forms a layer that determines the adhesion, migration, and shape of the cell around the exterior of the cell.7 Half of all HA in the body is contained in the skin. Preparations or implants containing HA accelerate the healing process in injuries to the retina, in diabetic foot, in bone and mucus membrane injuries, and in venous ulcers.
The widespread use of caffeine in food products, often in very high concentrations, and its permeability into cells creates a need to monitor its effects on cellular processes.11,12 The aim of this study was the assessment and recognition of the impairing mechanism of caffeine on the production of collagen in human skin fibroblasts and the evaluation of the role of HA in this process.
Materials and methods
Cell culture
Human skin fibroblasts were purchased from the American Type Culture Collection (Rockville, MD, USA). The cells were maintained in Dulbecco’s modified Eagle medium supplemented with 10% fetal bovine serum (HyClone Laboratories, Logan, UT, USA), 2 mM glutamine, 50 U/mL penicillin, and 50 μg/mL streptomycin at 37°C in a 5% CO2 incubator.
Collagen production
The method described by Peterkofsky and Diegelmann was used to determine the production of collagen.13 Radioactive precursor 5[3H]-proline (5 μCi/mL, 28 Ci/mmol) was labeled on confluent cultures of fibroblasts cultured in growth medium with varying concentrations of caffeine and HA 500 μg/mL for 24 hours and then in digested proteins with purified Clostridium histolyticum collagenase.
[3H]-thymidine incorporation
The effect of the studied substances on fibroblast [3H]-thymidine incorporation was examined. In 24-well tissue culture dishes, cells were plated at 1×105 cells/well, using 1 mL growth medium in each well. Forty-eight hours later (1.6±0.1×105 cells/well), various concentrations of caffeine with or without 500 μg/mL hyaluronan were added for 24 hours at 37°C to the culture wells. Thereafter, 0.5 μCi [3H]-thymidine (6.7 Ci/mmol) was added to the wells and cultures were incubated at 37°C for 4 hours. Subsequently, the cells were rinsed three times with 1 mL 0.05 M Tris-HCl and twice with 5% trichloroacetic acid. This was followed by cell lysis in 1 mL 0.1 M NaOH containing 1% sodium dodecyl sulfate. Next, 4 mL scintillation liquid was added and radioactivity was measured in scintillation counter.
In general, [3H]-thymidine incorporation is considered to express cell proliferation.
Determination of prolidase activity
The method of Myara et al was used to determine the activity of prolidase,14 with colorimetric determination of proline applying Chinard’s reagent. Centrifugation at 200× g for 15 minutes was done after harvesting the cells. Discarding the supernatant and suspension of the cell pellet were the next steps of the procedure. We used 1 mL 50 mM 4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid (HEPES) at pH 7.8 to suspend the cells and sonicated them for 3×10 seconds at 0°C. After that, we centrifuged specimens (12,000× g, 30 minutes) at 4°C. We used the supernatant for protein determination (Bradford method) and to perform prolidase activity assays. To activate the prolidase incubation with Mn(II) the following is necessary: 100 μL cell extract supernatant to mix with 100 μL 50 mM HEPES at pH 7.8 under the presence of MnCl2 to achieve a final concentration of 1 mM in the mixture. Next was incubation for 24 hours at 37°C. After that, the prolidase reaction was initiated as follows: We added 100 μL of the activated mixture to 100 μL of 94 mM glycyl-proline for a final concentration of 47 mM. Then the samples were incubated for 1 hour at 37°C. To terminate the reaction after that, 1 mL of 0.45 M trichloroacetic acid was added. To parallel blank tubes, trichloroacetic acid was added at time “zero.” After centrifugation of specimens at 10,000× g for 15 minutes, the released proline was determined as follows: we added 0.5 mL trichloroacetic acid supernatant to 2 mL of a 1:1 mixture of glacial acetic acid and Chinard’s reagent (25 g of ninhydrin dissolved at 70°C in 600 mL glacial acetic acid and 400 mL 6 M orthophosphoric acid) and incubated for 10 minutes at 90°C. We used colormetric absorbance at 515 nm to determine the amount of released proline and reported them as nanomoles per minute per milligram of protein.
Western blot analysis
For Western blot analysis, slab polyacrylamide-SDS/polyacrylamide gel electrophoresis (SDS/PAGE) was applied, as described by Laemmli.15 Equal amounts of total cellular protein (20 μg) were submitted to the electrophoresis on SDS-PAGE gels. Samples were mixed with Laemmli sample buffer containing 2.5% SDS (with reducing agent). After SDS-PAGE, the gels were stored for 5 minutes in 25 mM Tris, 0.2 M glycine in 20% (v/v) methanol to equilibrate. The samples were transferred to 0.2 μm pore-sized nitrocellulose at 100 mA for 1 hour by applying a LKB 2117 Multiphor II electrophoresis unit. The nitrocellulose was incubated with monoclonal anti-phospho-mitogen activated protein kinase (MAPK) antibody (extracellular signal-regulated kinase [ERK]1/ERK2) at a ratio of 1:1,000, monoclonal anti-phospho-AKT antibody at a ratio of 1:1,000, and polyclonal anti-β-actin antibody at ratio of 1:500 in 5% dried milk in Tris-buffered saline with Tween 20 (TBS-T) (20 mmol/L Tris-HCl buffer at pH 7.4, containing 150 mmol/L NaCl and 0.05% Tween 20) for 1 hour. To perform the analysis phospho-MAPK (ERK1/ERK2) and phospho-AKT, anti-mouse immunoglobulin G (whole-molecule) alkaline phosphatase conjugate was added at ratio of 1:5,000 in TBS-T. The second antibody-alkaline phosphatase conjugated, anti-rabbit immunoglobulin G (whole-molecule), was added at ratio of 1:5,000 in TBS-T, to analyze β-actin, the second antibody-alkaline phosphatase conjugated, and was incubated for 30 minutes under slow shaking. Next, nitrocellulose was washed with TBS-T (5×5 minutes) and submitted to 5-bromo-4-chloro-3-indolyl phosphate/nitro blue tetrazolium liquid substrate reagent.
Statistical methods
The mean values for at least three assays ± standard deviation (± SD) were calculated in all appropriate experiments. The statistical analysis was performed applying double-sided, unpaired Student’s t-test. SPSS version 16 was used, and P<0.05 was acknowledged as statistically significant.
Results
To assess the activity of caffeine on collagen synthesis in human skin fibroblasts, cells were incubated for 24 hours in 1, 2, and 5 mM caffeine as well as in caffeine with HA at a concentration of 500 μg/mL. In control cultures, collagen biosynthesis was intensive. It has been shown that caffeine significantly affects collagen synthesis (Figure 1). Exposure of the fibroblasts to different concentrations of the alkaloid led to the inhibition of the newly synthesized collagen by 48.04% (2.38% ± SD; n=3), 72.90% (3.60% ± SD; n=3), and 92.12% (3.81% ± SD; n=3), respectively, when compared with the control value. The addition of HA did not have a protective effect on the biosynthesis of collagen in cultures incubated with caffeine. The results presented provide evidence that caffeine has an inhibitory effect on collagen synthesis in confluent human skin fibroblasts, regardless of the presence of HA.
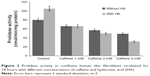
The enzyme responsible for recycling proline for collagen resynthesis is prolidase. Its activity was assessed in cultures of human skin fibroblasts in conditions established in the current experiment. Figure 2 presents the results. The activity of prolidase in control cultures is expressed by the value of 280 nmol proline obtained from a synthetic substrate in 1 minute, based on a milligram of protein extract of the cell homogenate. The addition of caffeine to cell cultures causes prolidase inhibition, respectively, at 17.50% (7.70% ± SD; n=3) in cultures incubated with caffeine at a concentration of 1 mM and at 28.75% (10.37% ± SD; n=3) and 38.75% (1.80% ± SD; n=3) in cultures incubated with caffeine at concentrations of 2 and 5 mM. The addition of HA did not have a significant effect on prolidase activity in cultures incubated with caffeine.
The influence of caffeine on prolidase activity may constitute a mechanism of the inhibitory effect of this alkaloid on collagen biosynthesis.
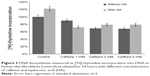
Research conducted so far states that caffeine has an inhibitory effect on cell division. For this reason, the influence of caffeine on DNA biosynthesis was also assessed. The dpm values of [3H]-thymidine incorporated in the DNA of these cells were assumed to constitute 100% (Figure 3). It has been ascertained that caffeine in a dose-dependent manner inhibits the biosynthesis of DNA. At the highest concentration, DNA biosynthesis was inhibited by 32%±0.98%. HA did not have any significant protective effect in counteracting the inhibition of DNA biosynthesis caused by the increased concentrations of caffeine.
The β1-integrin receptor plays a key role in the regulation of collagen biosynthesis, in DNA biosynthesis, and in prolidase activity. The expression of the integrin receptor has been assessed in cultures incubated with increasing concentrations of caffeine and in cultures incubated with HA and caffeine. The inhibition of the expression of the receptor in cultures incubated with the substances being studied indicates an inhibitory effect of caffeine that is directly proportional to the increase of its concentration. HA does not influence the level of expression of the receptor in cultures incubated with caffeine (Figure 4A).
It is known that insulin-like growth factor 1 (IGF-1)-dependent effects include activation of the expression of the collagen gene, prolidase activity, and cellular proliferation. In this study, we observed a similar effect as in integrin receptors. In this condition, the inhibition of the expression of the IGF-1 receptor was proportional to the increase of caffeine concentration. HA also does not influence the level of expression of this receptor (Figure 4B).
The signal transmitted by the stimulated IGF-1 receptor and the β1-integrin receptor leads to the activation of MAPK and AKT,16,17 as can be seen in Figure 4C. In fibroblasts treated with caffeine, the expression of the MAPK kinases (phospho-ERK1/2) decreased in a dose-dependent manner when compared with control cells. The addition of HA to cells treated with caffeine had no effect on the decrease in the expression of phospho-ERK1/2. Caffeine added to cultured fibroblasts had no effect on the expression of phospho-AKT kinases and β-actin (Figure 4D).
Discussion
Our research confirms our earlier observations that caffeine may have an adverse effect on the wound healing process, as well as on the aging process, of the human skin.18 This is connected to its influence on collagen biosynthesis. The presented research results show that the exposure of human skin fibroblasts to caffeine has a directly proportional effect to the concentration of this substance and causes the reduction of newly synthesized collagen by the cell.
The rising popularity of energy drinks containing caffeine, as well as the popularity of the substance itself, warrants the importance of ascertaining the influence of this substance on the elements of the extracellular matrix.
The research performed shows that the mechanism responsible for the impairment of collagen in human skin fibroblasts treated with the studied alkaloid may be the inhibition of prolidase activity. On the one hand, this enzyme is responsible for proline scavenging for collagen resynthesis,12 but on the other hand, it also plays a key role in the regulation of numerous transcription factors (hypoxia-inducible factor 1-alpha, transforming growth factor beta, and nuclear factor kappa-light-chain-enhancer of activated B cells) responsible for processes connected with wound healing, inflammation, or angiogenesis.19 Both the process of collagen biosynthesis and the process regulating prolidase activity are controlled through signals generated by β-integrin receptors and IGF-1 receptors. This research has shown that caffeine impairs the expression of both of these receptors, which is directly relative to the applied dosage and the resulting proportional impairment of MAP kinases related to these receptors (ERK1/2). The impairment of the cellular signal generated by the above-mentioned receptors might be a mechanism of impairment of collagen biosynthesis caused by the presence of caffeine. This is the first report about caffeine-induced inhibition of collagen synthesis in human skin fibroblasts.
The signal generated by integrin receptors and IGF-1 receptors is a key element in the processes of cell proliferation and division. We have been able to show that caffeine, in a dose-dependent manner, causes significant impairment of DNA biosynthesis, and the mechanism of this process may explain disruptions in the signal transfer from these receptors, especially those disruptions in the expression of kinases of the MAPK group, which are important to this process.
The literature confirms that HA has a favorable effect on the regenerative processes connected with the impairment of collagen synthesis.20,21 In this case, however, the addition of HA did not reverse any changes in collagen that were caused by the caffeine. HA did not exert a protective effect on the disadvantageous influence of caffeine on individual regulatory elements of collagen biosynthesis. In contrast, caffeine may also lead to the impairment of HA effectiveness as a substance that has a protective influence on the level of collagen present in tissue during the processes of wound healing or counteraction of human skin aging.
Exposure of embryonic fibroblasts or forebrain neurons to energy drinks leads to the retardation of regenerative processes, especially in the process of wound healing.5 In contrast, the elucidation of the molecular mechanism of the impairment of collagen synthesis by caffeine may explain the protective effect of caffeinated drinks on the induction of liver cirrhosis.22 The explanation of the molecular mechanism of the impairment of collagen biosynthesis by caffeine may create a drug target in the pharmacotherapy of this process.
Disclosure
MD is a fellow in the project Studies, Research, Commercialization: A Support Programme of UMB Doctoral Students, submeasure 8.2.1 Human Capital Operational Programme, cofinanced from the European Union under the European Social Fund. This work was supported in part by grants from the Medical University of Białystok (123-30848F and 133-30508F).
References
Azam S, Hadi N, Khan NU, Hadi SM. Antioxidant and prooxidant properties of caffeine, theobromine and xanthine. Med Sci Monit. 2003; 9(9):BR325–BR330. | ||
Merighi S, Benini A, Mirandola P, et al. Caffeine Inhibits Adenosine-Induced Accumulation of Hypoxia-Inducible Factor-1α, Vascular Endothelial Growth Factor, and Interleukin-8 Expression in Hypoxic Human Colon Cancer Cells. Mol Pharmacol. 2007;72:395–406. | ||
Bode AM, Dong Z. The enigmatic effects of caffeine in cell cycle and cancer. Cancer Lett. 2007;247(1):26–39. | ||
Jafari M, Rabbani A. Studies on the mechanism of caffeine action in alveolar macrophages: caffeine elevates cyclic adenosine monophosphate level and prostaglandin synthesis. Metabolism. 2004;53(6):687–692. | ||
Doyle W, Shide E, Thapa S, Chandrasekaran V. The effects of energy beverages on cultured cells. Food Chem Toxicol. 2012;50(10): 3759–3768. | ||
Fratzl P, editor. Collagen: Structure and Mechanics. New York: Springer; 2008. | ||
Evanko SP, Tammi MI, Tammi RH, Wight TN. Hyaluronan-dependent pericellular matrix. Adv Drug Deliv Rev. 2007;59(13):1351–1365. | ||
Karna E, Miltyk W, Surazyński A, Pałka JA. Protective effect of hyaluronic acid on interleukin-1-induced deregulation of beta1-integrin and insulin-like growth factor-I receptor signaling and collagen biosynthesis in cultured human chondrocytes. Mol Cell Biochem. 2008; 308(1–2):57–64. | ||
Girish KS, Kemparaju K. The magic glue hyaluronan and its eraser hyaluronidase: a biological overview. Life Sci. 2007;80(21): 1921–1943. | ||
Schaefer L, Schaefer RM. Proteoglycans: from structural compounds to signaling molecules. Cell Tissue Res. 2010;339(1):237–246. | ||
Barone JJ, Roberts HR. Caffeine consumption. Food Chem Toxicol. 1996;34(1):119–129. | ||
O’Neill SC, Donoso P, Eisner DA. The role of [Ca2+]i and [Ca2+] sensitization in the caffeine contracture of rat myocytes: measurement of [Ca2+]i and [caffeine]i. J Physiol. 1990;425:55–70. | ||
Peterkofsky B, Diegelmann R. Use of a mixture of proteinase-free collagenases for the specific assay of radioactive collagen in the presence of other proteins. Biochemistry. 1971;10(6):988–994. | ||
Myara I, Charpentier C, Lemonnier A. Optimal conditions for prolidase assay by proline colorimetric determination: application to iminodipeptiduria. Clin Chim Acta. 1982;125(2):193–205. | ||
Laemmli UK. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970;227(5259):680–685. | ||
Parsons CJ, Takashima M, Rippe RA. Molecular mechanisms of hepatic fibrogenesis. J Gastroenterol Hepatol. 2007;22(suppl 1):S79–S84. | ||
Zhang YE. Non-Smad pathways in TGF-beta signaling. Cell Res. 2009; 19(1):128–139. | ||
Salsas-Escat R, Stultz CM. The molecular mechanics of collagen degradation: implications for human disease. Exp Mech. 2009; 49(1):65–77. | ||
Surazynski A, Miltyk W, Czarnomysy R, Grabowska J, Palka J. Hyaluronic acid abrogates nitric oxide-dependent stimulation of collagen degradation in cultured human chondrocytes. Pharmacol Res. 2009;60(1):46–49. | ||
Karna E, Palka JA. Phosphoenolpyruvate-dependent inhibition of collagen biosynthesis, alpha2beta1 integrin and IGF-I receptor signaling in cultured fibroblasts. Mol Cell Biochem. 2008;315(1–2):61–67. | ||
Surazynski A, Miltyk W, Palka J, Phang JM. Prolidase-dependent regulation of collagen biosynthesis. Amino Acids. 2008;35(4):731–738. | ||
Furtado KS, Polletini J, Dias MC, Rodrigues MAM, Barbisan LF. Prevention of rat liver fibrosis and carcinogenesis by coffee and caffeine. Food Chem Toxicol. 2014;64:20–26. |
 © 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.