Back to Journals » Journal of Multidisciplinary Healthcare » Volume 13
Indications and Clinical Profile of Neonatal Admissions: A Cross-Sectional Descriptive Analysis from a Single Academic Center in Jordan
Authors Khasawneh W , Sindiani A , Rawabdeh SA, Aleshawi A, Kanaan D
Received 14 August 2020
Accepted for publication 28 August 2020
Published 23 September 2020 Volume 2020:13 Pages 997—1006
DOI https://doi.org/10.2147/JMDH.S275267
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Scott Fraser
Wasim Khasawneh,1 Amer Sindiani,2 Saif Aldeen Rawabdeh,1 Abdelwahhab Aleshawi,3 Dana Kanaan1
1Department of Pediatrics and Neonatology, Faculty of Medicine, Jordan University of Science and Technology, Irbid, Jordan; 2Department of Obstetrics and Gynecology, Faculty of Medicine, Jordan University of Science and Technology, Irbid, Jordan; 3Department of Ophthalmology, Faculty of Medicine, Jordan University of Science and Technology, Irbid, Jordan
Correspondence: Wasim Khasawneh
Department of Pediatrics, Jordan University of Science and Technology, Irbid 22110, Jordan
Tel +962796041117
Fax +96227095777
Email [email protected]
Purpose: To review the indications and clinical profile of neonatal admissions at King Abdullah University Hospital in Jordan.
Materials and Methods: We conducted a cross-sectional review of all neonates admitted to the neonatal intensive care unit between September 2016 and September 2018. Collected data include demographic characteristics, indications for admission, morbidities and mortality, and discharge outcomes. Findings were reported among term and preterm infants.
Results: A total of 1444 infants were admitted during the study period of whom 1332 (92.2%) were inborn and 612 (42.4%) were term neonates. Of the 832 preterm infants, 545 were late preterm (34– 36 6/7 gestation) and 125 had very low birth weight (˂ 1500 grams); 925 (64%) were born by cesarean section. Respiratory failure of the newborn (41.2%) and prematurity (33.3%) were the main indications for admission among the whole cohort. Maternal prolonged premature rupture of membranes (PROM) was observed in nearly half the admissions of term infants. Hypoxic ischemic encephalopathy (3.2% vs 0.7%, p 0.01) and congenital anomalies (5% vs 1.2%, p 0.03) were more common in term infants. The rate of bronchopulmonary dysplasia was 39% among < 28-week and 28% among < 32-week premature infants. Sepsis was encountered in 59 infants. The overall mortality rate was 3.8%. Prematurity was the main predisposing factor for mortality (Adjusted OR: 9.9, 95% CI: 3.5, 27.6).
Conclusion: The majority of neonatal admissions at our institution are term and late preterm infants delivered by cesarean section. Prematurity, respiratory failure of the newborn, and suspected sepsis due to maternal PROM are the leading causes of admission. The mortality rate is within WHO target to achieve Sustainable Development Goal 3. Population-based studies are needed to make better conclusions that represent the whole Jordanian population. A revisit for the indications of cesarean deliveries may help to improve the neonatal outcomes.
Keywords: neonatal outcomes, Jordan, NICU admissions, neonatal mortality
Introduction
After birth, the smooth transition to the extra-uterine environment is essential to help the newborns survive the most critical period of life with good outcomes. The advance in neonatal care services over the past few decades has significantly improved the survival rate of newborns particularly premature neonates. As a result, neonatal admissions and the resulting neonatal morbidities are rising.1,2
Studies have shown that neonatal deaths are responsible for the majority of mortalities in children under the age of 5 years with a reported rate of 35% in the USA and up to 50% in certain low-income countries.3 The 2030 agenda for sustainable development by WHO calls for reduction in neonatal mortality rate to less than 12 per 1000 live births.4 In Jordan, death in the first 28 days of life has contributed to nearly 50% of all under 5-year mortalities in 2013.5 In the USA, the neonatal mortality rate (NMR) has dropped from more than 18 to less than 3 per 1000 live births in 2017.2 According to local Jordanian statistics, the NMR is about 15 per 1000 live births using gestational age (GA) of 20 weeks as the age of viability. This figure is reduced to nearly 10 per 1000 live births when using the WHO definition for the age of viability at 28-week gestation.6 Although variable factors contribute to neonatal mortality, the main reported causes are prematurity, congenital anomalies, infection, and birth depression.7 Accordingly, most of the neonatal deaths are potentially preventable by attaining adequate neonatal care at highly specialized centers.
Admission to the neonatal intensive care unit (NICU) carries significant social and financial burdens on the involved families and healthcare facilities.8 In the US, the NICU admission rate has increased significantly by nearly 25% in the period 2007 to 2012.1 More surprisingly, term babies contributed the most to the increased overall NICU admission rates. This observation could have been explained by the increase in Cesarean section (CS) procedures and the increased elective deliveries at early-term gestation (37–38.6 weeks).1
To improve neonatal survival with better overall outcomes and less severe morbidities, early identification of the risk factors is paramount so that appropriate interventions can be directed towards the most prevalent and treatable neonatal illnesses. To achieve this goal, it is important to study the pattern of neonatal admissions. Most reports that examined neonatal outcomes following NICU admission have focused on certain subgroups like premature infants or infants with very low birth weight (VLBW, less than 1500 grams).9–11 Most US hospitals are engaged in national networks, like the Vermont Oxford Network to study VLBW infants and focus on long-standing research and quality improvement projects by comparing the data from different institutions on a regular basis.12 To better compare the outcomes at different NICU set-ups, the level of neonatal care units has been classified into 4 major levels with level-4 units offering the most specialized services including surgical interventions.13
In Jordan, neonatal care services are offered through academic, military, governmental, and private health institutions. With the advance in technology and the increasing availability of resources over the past decade, there has been a steady improvement in the level of newborn care across the nation. Currently, there are ten-level 3 to 4 NICUs capable of accommodating a total of nearly 400 sick neonates in the country. In addition, level 2 units that care for less premature and less sick infants are distributed throughout major Jordanian cities.14 Besides geographic location, insurance policy coverage is the main factor that determines where newborns that require specialized care are admitted. Despite the widespread neonatal units of different levels, no population-based data had been reported about NICU admission and outcomes. Most published data about neonatal outcomes in Jordan studied the patterns and causes of mortality,6,15 and only limited research has been conducted about single-center NICU admission outcomes.16 In 2015, the epidemiologic patterns of the NICU admissions at Al-Bashir hospital, a public institution that has the largest NICU in the country, were studied. The authors concluded that late preterm gestation and CS deliveries were the main indications for NICU admission. They also reported a mortality rate of 8.7% among the admitted neonates in their center with a median gestational age of 30 weeks at time of death.16 Otherwise, there is a paucity of data and research about neonatal outcomes from Jordan and the Middle East. Thus far, there has been no reported data about the prevalence and patterns of NICU admissions at any of the academic referral centers in the country. The purpose of this study is to assess the prevalence of NICU admission and evaluate the indications and clinical profile of admissions at our level-3 NICU at King Abdullah University Hospital (KAUH) in North of Jordan. The patterns and outcomes are reported among term and preterm infants.
Materials and Methods
A retrospective single-center cross-sectional descriptive analysis was conducted to evaluate the indications and clinical profile of all NICU admissions at KAUH in the period between September 2016 and September 2018. KAUH is the only academic hospital in North of Jordan. It is the main referral center in the region that provides access to healthcare services for nearly two million. The annual number of deliveries is around 3500. In addition to a 25-bed level one nursery that cares for healthy late preterm and term infants, our NICU is level- 3 unit that is managed by three neonatology consultants and more than 25 pediatric residents. This 18-year-old, 30-incubator unit accommodates all inborn neonates that need specialized care. In addition, 5–10% of the admissions are transferred from local public and private hospitals. With the exception of pediatric cardiothoracic surgery, all pediatric and surgical subspecialties are available for consults in our unit.
A list for all newborns who were admitted to the NICU during the study period was obtained from the hospital electronic database. All NICU admissions, including inborn and outborn infants, were included in this review. Excluded from the analysis were all stillbirths and all infants with complex congenital heart disease who were transferred to an outside institution within the first 72 hours. Data were extracted from the electronic medical records after a comprehensive review of the admission notes, daily progress notes, consultation notes, discharge summaries, problem lists, medication sheets, and laboratory and radiologic investigations. Data collection was conducted by well-trained pediatric residents and interns using pretested variables and documented in an excel spreadsheet. Demographic characteristics, indications for NICU admission, morbidities, and outcomes were reported as documented by the admission physician.
Data were abstracted about gestational age (GA), birth weight, gender, maternal age, mode of delivery, source of admission, reason for admission, respiratory support including invasive mechanical ventilation or continuous positive airway pressure (CPAP), type of intravenous access, feeding regimen and complications, laboratory investigations, blood product transfusion, sepsis, antibiotic therapy, and results of standard screening studies per protocols such as head ultrasound and ROP screening exam. Also collected are data about disposition including mortality, length of stay as well as the weight and corrected gestational age at discharge.
System-based neonatal outcomes of interest reported in this analysis include intraventricular hemorrhage (IVH), periventricular leukomalacia (PVL), retinopathy of prematurity (ROP), respiratory distress syndrome (RDS), bronchopulmonary dysplasia (BPD), pneumothorax, pulmonary hemorrhage, pulmonary hypertension, necrotizing enterocolitis (NEC), feeding intolerance, sepsis, blood product transfusion, neonatal jaundice, and mortality. In our unit, definitions of neonatal pathologies and outcomes are based on clinical presentation and supportive laboratory investigations utilizing the standard international criteria as applicable.
The term prematurity was used to describe live-born neonates delivered before completed 37 weeks of gestation. Birth weight was classified using the WHO weight classification system and categorized into low birth weight (LBW: birth weight less than 2500 grams), very low birth weight (VLBW: birth weight less than 1500 grams) and extremely low birth weight (ELBW: birth weight less than 1000 grams).17
Grades of IVH were reported using Papile’s classification as follows: germinal matrix bleeding (grade 1), intraventricular bleeding without dilatation (grade 2), intraventricular bleeding with dilatation (grade 3), and intraparenchymal extension or periventricular infarction (grade 4).18 In our analysis, severe IVH refers to grade 3 or 4.
For ROP, we utilize the International classification of retinopathy of prematurity in assessing the stage of ROP as follows: demarcation line (Stage 1), visible ridge (Stage 2), ridge blood vessels (Stage 3), subtotal retinal detachment (Stage 4), and total retinal detachment (Stage 5).19 In our analysis, high-stage ROP refers to stage 3 or more.
RDS diagnosis was given to any preterm or occasionally term babies with signs of respiratory distress and typical CXR finding of hyaline membrane disease including reticulgranular shadows, air bronchogram, or white-out lung fields. BPD was defined according to the new BPD definition of oxygen dependence at 28 days of life and classified according to the oxygen requirement at 36-week corrected gestational age or at discharge into mild BPD (room air), moderate (fractionated inspired oxygen of 21–30%) and severe (fractionated inspired oxygen of more than 30%).20
Pneumothorax diagnosis was established with typical radiological findings or with positive transillumination sign. Pulmonary hypertension refers to typical echocardiographic findings using the calculated tricuspid regurgitation (TR) jet as a marker.21 Bell’s classification system was applied for the diagnosis of NEC, and we included definitive cases of typical clinical presentation associated with pneumatosis intestinalis or portal venous gas (stage two), or pneumoperitoneum (stage 3).22
Sepsis was defined as bacterial isolation from blood or cerebrospinal fluid (CSF) samples. Early-onset sepsis (EOS) refers to any case of sepsis in the first 72 hours of life while late-onset sepsis (LOS) used for cases diagnosed after 72 hours.23 The isolated bacteria were identified and reported as well. In the case of coagulase-negative staphylococcus (CONS) growth, the diagnosis of sepsis was attained with two positive cultures 24 hours apart, or with one positive culture associated with abnormal white blood cell count and differential and/or C-reactive protein (CRP).24
Birth depression refers to neonates born flaccid with respiratory depression that requires resuscitation beyond the routine newborn care steps. Hypoxic ischemic encephalopathy (HIE) diagnosis was established according to the criteria set by the American academy of pediatrics (AAP) and American college of obstetrics and gynecology (ACOG) in the presence of peripartum event associated with an APGAR score of 5 or less at 5 minutes of age, or significant metabolic acidosis with PH of 7.2 or less and/or base deficit of at least 16 in the context of clinical encephalopathy according to Sarnat definition and staging.25,26
Regarding cases of hyperbilirubinemia, diagnosis and management were according to the AAP guidelines.27 We reported cases of ABO incompatibility, Rh isoimmunization, duration of phototherapy, IVIG administration, and exchange transfusion procedures.
For certain variables and outcomes, we calculated the total number of days. This was applied for the total antibiotic days, invasive mechanical ventilation, CPAP, and phototherapy. We reviewed data that pertain to term and preterm infants using completed 37-week gestation at birth as the marker of differentiation and reported their outcomes accordingly.
Statistical analyses were performed using IBM SPSS Statistics Software version 21 (IBM Corp., Armonk, NY, USA). Data were presented as frequency distributions and percentages for categorical variables, and mean ± standard deviation for continuous variables. Data was tested at a significance level of 0.05. Pearson χ2 test was used to investigate the significance of association between categorical variables, while Student’s t-test and ANOVA were applied to examine the significance level for continuous normally distributed variables. The normality of the distribution of data was tested using the Kolmogorov–Smirnov test. If a significant (P<0.05) relationship was found, then a posthoc residual analysis for categorical variables and a Fisher’s least significant difference test for continuous variables were applied to determine the exact significance between groups for each variable.
Results
A total of 1444 infants were admitted to the NICU during the study period. Of the admitted neonates, 1332 were born at our institution representing a cumulative admission rate of 19% (out of 7020 total deliveries). Nine hundred and twenty-five (64%) were delivered by CS. Fifty-three percent were males. Term neonates accounted for 42.4% (612) of all neonatal admissions. Of the 832 preterm neonates admitted during the study period, nearly two-thirds (545) were born at late preterm gestation. Among the admitted cohort, LBW and VLBW infants constituted 39.3% (568) and 8.7% (125). Table 1
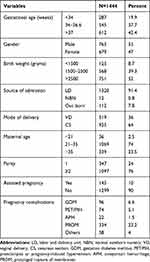 |
Table 1 Neonatal and Maternal Characteristics |
The indications for NICU admissions are reported in Table 2. Besides prematurity (33.3%), the main reason for admission among the whole cohort was respiratory failure secondary to RDS or TTN (41.2%). On the other hand, maternal PROM (49%), hyperbilirubinemia (25%), and birth depression (12.7%) were the main factors for admission among term infants. Congenital anomalies were seen in 40 cases (2.8%).
 |
Table 2 Indications for NICU Admission |
Table 3 summarizes the clinical outcomes. There are significant differences in the morbidities and complications observed during the NICU stay between preterm and term infants. Admitted term infants were more likely to have HIE (3.2% vs 0.7%, p 0.01) and congenital anomalies (5% vs 1.2%, p 0.03). Among premature neonates, 7 (˂1%) developed grade 3–4 IVH and 9 (˂1%) had ROP of stage 3 or higher. Of the premature babies with RDS, the majority (70%) were treated with respiratory support only, while 30% required surfactant administration. Of those who received surfactant, the majority (152/244) received one dose only, while 92/244 required 2 or 3 doses. The total number of premature infants who required invasive mechanical ventilation during the hospital stay was 178 (21.4%) for a median duration of 1.5 days compared with 500 (60%) who required CPAP only for a median of 5.3 days. Pneumothorax and pulmonary hemorrhage affected 20 (2.4%) and 9 (1.1%) neonates, respectively. Thirty-eight premature neonates (4.6%) were diagnosed with BPD, of whom 50% had a moderate or severe degree. More specifically, the rate of BPD was reported among <28-week and <32-week premature infants at 39% and 28%, respectively.
 |
Table 3 Complications and Morbidities of Neonatal Admissions |
Culture-proven sepsis was diagnosed in 59 infants. Of those, 45 were premature and 14 were term neonates. The main isolated pathogens in EOS were Streptococcus agalataciae and Klebsiella pneumoniae, while coagulase-negative Staphylococcus and ESBL positive Klebsiella pneumoniae contributed to the majority of LOS cases.
In our unit, the majority of admitted infants (86%) received formula feeding and nearly 20% received supportive partial parenteral nutrition in the form of Dextrose and 10% amino acid solutions. Although feeding intolerance was commonly encountered during NICU admission, the proportion of premature neonates who developed confirmed NEC was 1.4% (22) of whom only three babies required surgical intervention with partial bowel resection and creation of ileostomy. Less than 10% received packed red blood cell transfusion once during their NICU stay and 5% required transfusion on two or more occasions. IVIG was prescribed as an adjunctive therapy for hyperbilirubinemia due to autoimmune hemolysis in 30 cases (5%), and double volume exchange transfusion was performed in 9 cases (1.4%).
The overall mortality rate among our NICU admissions was found to be 3.8%. Higher mortality rate was observed in premature infants compared with term infants (51 cases or 6.1% vs 4 cases or 0.7%, p 0.001). The mean length of stay among premature infants was 12.5 days ± 15.9 compared to 5.3 days ± 9.6 among term infants (p 0.005). Table 4
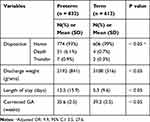 |
Table 4 Discharge Outcomes |
Discussion
Among all deliveries at our institution, this single-center retrospective analysis reported a neonatal admission rate of less than 20%. Overall, the main indications for admission include prematurity and respiratory failure related to RDS and TTN, while maternal PROM, hyperbilirubinemia, and HIE contributed the most to NICU admission among term infants. Also, this study highlighted the difference in the epidemiology and clinical outcomes between term and preterm infants.
Although delivery at term gestation is considered to be low risk, mature neonates with normal birth weight might exhibit certain illnesses that require escalation of care and admission to the NICU. In our study, term infants accounted for 42% of the total NICU admissions during the study period. This finding is consistent with a population-based report of infants from 38 US states which showed nearly half of all NICU admissions to be delivered at more than 37-week gestation.1 Maternal PROM was among the main factors associated with admission of our studied term neonates. The same finding was reported by Al-Wassia who studied the risk factors for admission of term infants at a tertiary Saudi teaching hospital.28 Moreover, nearly half the admitted term infants in our unit were delivered by CS which was electively performed at early-term gestation (37–38 6/7 weeks) in the majority of cases. Among those, more than half required respiratory support in the form of invasive mechanical ventilation or CPAP for a median of 3 days. A similar concern was raised by Burgess et al who reported respiratory problems requiring NICU admission in almost half of the 36 to 41-week infants included in their analysis.29 This highlights the importance of adhering to the ACOG and RCOG recommendations for avoiding elective CS deliveries at early-term gestation to minimize these potentially preventable complications.30
The proportion of preterm admission in our unit is 58%. This is relatively higher than what is reported from other low-income countries.31 Of notice, two-thirds of the preterm infants in our cohort were born at late preterm gestation. The increasing rate of late preterm delivery is consistent with other reports.32,33 This might be explained by the increase in assisted reproductive technology resulting in multiple gestation and increasing CS delivery rate. Thirty-nine percent of the admitted neonates in our study belong to the LBW category and this is comparable with studies from other low-income countries.34,35
One of the major causes of morbidity and mortality among premature infants is RDS and BPD. In our cohort, although half of the premature babies were diagnosed with RDS, the majority required CPAP respiratory support and only 21% required invasive ventilation for a mean of 1.5 days. Simultaneously, surfactant administration was required in 30%, the majority required one dose only. The improving rate of antenatal care attendance, the increasing use of antenatal steroids, and the availability of non-invasive ventilation and CPAP are known factors to reduce the RDS severity.36 Fortunately, less than 5% of the premature infants included in our study developed BPD and only 6 infants had severe BPD. The wide variation in the reported incidence of BPD globally, ranging from 5% to 75%, might be attributed to the heterogeneity of the studied neonates and the difference in management practices and in disease definition.37 To better understand the trend in BPD and to get a more fair comparison between different centers, BPD rate should be reported according to specific GA-stratified subgroups. In our cohort, the incidence was 39% among extremely premature (GA < 28 weeks) and 28% among very premature (GA <32 weeks). Our findings are consistent with population-based cohorts from Canada and the USA,38,39 and better than reports from the region. For instance, the incidence of BPD among < 32-week premature Saudi infants was recently reported by Al-Shehri et al to be 45.9% with nearly half of the cases categorized as severe BPD.40
The cumulative incidence of sepsis in our cohort was 4.4%. Of the confirmed cases, three-fourths affected premature infants and this is likely explained by the relatively immunocompromised status of prematurity, the use of umbilical catheters and other central lines, and the longer duration of hospitalization. EOS contributed to 22% of all cases with Group B Streptococcus (GBS) and Klebsiella pneumoniae being the most isolated pathogens. On the other hand, coagulase-negative Staphylococcus and Klebsiella pneumoniae were the most common bacterial causes of LOS. The incidence and epidemiologic trend of sepsis in our cohort is comparable to other reports that showed GBS to remain the leading cause of EOS, and CONS and gram-negative bacteria to be the main causes of LOS.41 Maternal PROM was reported in 24%. It is not clear if these cases are overestimated since not all cases of suspected PROM were confirmed by PH or microscopic testing. Although maternal PROM was a major factor for neonatal admission in our cohort, the rate of culture-proven sepsis among this subgroup was less than 1%.
The other potential complications of prematurity included in our analysis showed that severe IVH and high-stage ROP affected less than 2% among the whole premature group. However, among the most susceptible subgroup; ie, the ˂ 32-week premature infants, the incidence was 8% and 7%, respectively. The low rates can be explained by the relatively low number of extremely premature and VLBW infants in our cohort. In addition, the short duration of invasive and non-invasive ventilation with strict control of oxygen supplement could have protected against ROP. The outcome of ROP was recently studied in a single-center military hospital in Jordan and the incidence was found to be 17%.42 In Saudi Arabia, Waheeb et al reported that ≥ stage 3 ROP affected 15.2% of ˂ 32-week premature infants in two tertiary hospitals in Jeddah.43
In the present study, the overall mortality rate was 3.8% (55 out of 1444) among all admissions representing 8 deaths per 1000 live births (55 out of 7020). The main cause of mortality was extreme prematurity (30/55). Our finding is in line with the recommendations to reduce perinatal and neonatal mortality in order to achieve the Sustainable Development Goal number 3 which targets a NMR of less than 12 per 1000 live births by 2030.4 The average global NMR is 18/1000 live births with a wide variation ranging from 2.3 to 30 with low rates in North America and West Europe and high rates in West Africa.2 The relatively low rate reported in our study is less than the average NMR of 12.6/1000 live births in the Middle East region and is significantly better than what was reported 5 years ago from Al Bashir hospital, the main public hospital in Jordan. In their analysis, they reported a mortality rate of 8.9% among their NICU admissions. Prematurity and low birth weight were the main associated factors with death in their analysis. At a local national level as determined by the Jordan Perinatal and Neonatal Study, the NMR has been 14.9 per 1000 live births.44 In Jordan and most other countries, prematurity remains the leading cause of death in the neonatal period and among children under the age of 5 years.45
The average duration of hospitalization in our cohort is reasonably low. This could have been explained by the fact that 80% of our admitted neonates are term and late preterm infants. The financial burden of NICU stay is one of the major obstacles that face the involved families and healthcare providers.8 Our policy is to discharge infants home as soon as they are stable enough given that parents receive comprehensive counselling and outpatient follow up appointments are scheduled within a short period of time after discharge.
This study is not without limitations. The main limitation was the retrospective nature of this review in which the collected data relied on the documentation of the treating teams in the electronic medical charts. This might contribute to an underestimate or overestimate of certain diagnosis like the incidence of RDS and hyperbilirubinemia. In addition, being a single-center analysis over a relatively short period of time, conclusions about the generalizability of our findings cannot be accurately made to represent the whole Jordanian population. Another limitation is that this study involves a descriptive analysis of the neonatal outcomes among all NICU admissions including preterm and full-term infants, and so, the reported significant differences in their outcomes are expected, given the different pathophysiology in both groups. Our future research is intended to analyze the main predisposing factors for the most serious outcomes among each subgroup separately.
Conclusions
In conclusion, this comprehensive analysis of neonatal admission and outcomes represents the first study from an academic institution and the second overall report from Jordan. Prematurity and respiratory failure of the newborn related to RDS and TTN are the main indications for NICU admission. The overall outcome is very reassuring and comparable to most centers in high-income countries. The mortality rate at our center falls within the WHO target range to reduce the under-5 mortality rate. To improve the outcome furthermore, quality improvement projects should be implemented to study the increasing elective operative CS procedures and late preterm deliveries. Population-based studies are needed to make better conclusions that represent the whole Jordanian population. Creating a national database for all newborns and NICU admissions to report and compare mortality and morbidities is encouraged considering the difference in healthcare quality and availability of resources.
Abbreviations
NICU, neonatal intensive care unit; PROM, prolonged rupture of membranes; VLBW, very low birth weight; WHO, World Health Organization; AAP, American Academy of Pediatrics; ACOG, American College of Obstetrics and Gynecology; RCOG, Royal College of Obstetrics and Gynecology; IVH, intraventricular hemorrhage; PVL, periventricular leukomalacia; ROP, retinopathy of prematurity; RDS, respiratory distress syndrome; BPD, brobhopulmonary dysplasia; NEC, necrotizing enterocolitis; NMR, neonatal mortality rate; GA, gestational age; CS, cesarean section; CPAP, continuous positive airway pressure; EOS, Early-onset sepsis; LOS, Late-onset sepsis; TTN, transient tachypnea of newborn.
Data Sharing Statement
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Ethics Approval and Informed Consent
An Institutional Review Board (IRB) approval was obtained from Jordan University of Science and Technology (IRB number 388-2017). Also, administrative approval was obtained from KAUH to review the electronic medical records. Parental consent was waived as this study involves chart review and analysis was performed on de-identified data. Patient data privacy and confidentiality are maintained as this study was conducted in compliance with the ethical standards per Helsinki declaration.
Acknowledgments
The authors would like to extend deep thanks to all healthcare workers at the neonatal ICU at KAUH for their great efforts taking care of our precious neonates.
Author Contributions
All authors made substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data; took part in drafting the article or revising it critically for important intellectual content; agreed to submit to the current journal; gave final approval of the version to be published; and agree to be accountable for all aspects of the work.
All authors have approved the submitted version and have agreed to be accountable for all its contents.
Disclosure
All authors declare that they have no competing interests.
References
1. Harrison W, Goodman D. Epidemiologic trends in neonatal intensive care, 2007–2012. JAMA Pediatr. 2015;169(9):855–862. doi:10.1001/jamapediatrics.2015.1305
2. Hug L, Alexander M, You D, Alkema L. National, regional, and global levels and trends in neonatal mortality between 1990 and 2017, with scenario-based projections to 2030: a systematic analysis. Lancet Glob Health. 2019;7(6):e710–e720. doi:10.1016/S2214-109X(19)30163-9
3. National and regional under-5 mortality rate by economic status for low-income and middle-income countries: a systematic assessment. UNICEF DATA. Available from: https://data.unicef.org/resources/u5mr-by-economic-status/.
4. WHO SDG 3. Ensure healthy lives and promote wellbeing for all at all ages. Available from: https://www.who.int/sdg/targets/en/.
5. Jordan (JOR). Demographics, health & infant mortality. UNICEF DATA. Available from: https://data.unicef.org/country/jor/.
6. Batieha AM, Khader YS, Berdzuli N, et al. Level, causes and risk factors of neonatal mortality, in Jordan: results of a national prospective study. Matern Child Health J. 2016;20(5):1061–1071.
7. Sankar MJ, Natarajan CK, Das RR, Agarwal R, Chandrasekaran A, Paul VK. When do newborns die? A systematic review of timing of overall and cause-specific neonatal deaths in developing countries. J Perinatol. 2016;36(S1):S1–S11.
8. Johnston KM, Gooch K, Korol E, et al. The economic burden of prematurity in Canada. BMC Pediatr. 2014;14(1).
9. Koller-Smith LIM, Shah P, Ye XY, et al. Comparing very low birth weight versus very low gestation cohort methods for outcome analysis of high risk preterm infants. BMC Pediatr. 2017;17(1).
10. Tsai L-Y, Chen Y-L, Tsou K-I, Mu S-C. Taiwan premature infant developmental collaborative study group. The impact of small-for-gestational-age on neonatal outcome among very-low-birth-weight infants. Pediatr Neonatol. 2015;56(2):101–107. doi:10.1016/j.pedneo.2014.07.007
11. Persson M, Shah PS, Rusconi F, et al. Association of maternal diabetes with neonatal outcomes of very preterm and very low-birth-weight infants an international cohort study. JAMA Pediatr. 2018;172(9):867–875. doi:10.1001/jamapediatrics.2018.1811
12. Horbar JD, Soll RF, Edwards WH. The Vermont Oxford Network: a community of practice. Clin Perinatol. 2010;37(1):29–47. doi:10.1016/j.clp.2010.01.003
13. Barfield WD, Papile LA, Baley JE, et al. Levels of neonatal care. Pediatrics. 2012;130(3):587–597.
14. Khader Y, Alyahya M, Al-sheyab N, Shattnawi KK. Evaluation of maternal and newborn health services in Jordan. J Multidiscip Healthc. 2018;11–439.
15. Obeidat N, Khader Y, Batieha A, Abdel Razeq N, Al-Sheyab N, Khassawneh M. Neonatal mortality in Jordan: secondary analysis of Jordan Population and Family Health Survey (JPFHS) data. J Matern Neonatal Med. 2019;32(2):217–224. doi:10.1080/14767058.2017.1377174
16. Sivasubramaniam P, Quinn CE, Blevins M, et al. Neonatal outcomes of infants admitted to a large government hospital in Amman, Jordan. Glob J Health Sci. 2015;7(4):217–234. doi:10.5539/gjhs.v7n4p217
17. Martin JA, Hamilton BE, Ventura SJ, Osterman MJK, Wilson E, Mathews TJ. Final data for 2010. Natl Vital Stat Rep. 2012;61(1):1–104.
18. Papile LA, Burstein J, Burstein R, Koffler H. Incidence and evolution of subependymal and intraventricular hemorrhage: a study of infants with birth weights less than 1500 gm. J Pediatr. 1978;92(4):529–534. doi:10.1016/S0022-3476(78)80282-0
19. Quinn GE. The international classification of retinopathy of prematurity revisited: an international committee for the classification of retinopathy of prematurity. Arch Ophthalmol. 2005;123(7):991–999.
20. Ehrenkranz RA, Walsh MC, Vohr BR, et al. Validation of the National Institutes of Health consensus definition of bronchopulmonary dysplasia. Pediatrics. 2005;116(6):1353–1360. doi:10.1542/peds.2005-0249
21. Walther FJ, Benders MJ, Leighton JO. Early changes in the neonatal circulatory transition. J Pediatr. 1993;123(4):625–632. doi:10.1016/S0022-3476(05)80966-7
22. Bell MJ, Ternberg JL, Feigin RD, et al. Neonatal necrotizing enterocolitis. Therapeutic decisions based upon clinical staging. Ann Surg. 1978;187(1):1–7.
23. Puopolo KM, Benitz WE, Zaoutis TE. Management of neonates born at ≥35 0/7 weeks’ gestation with suspected or proven early-onset bacterial sepsis. Pediatrics. 2018;142(6):1098–4275.
24. Wynn JL, Wong HR, Shanley TP, Bizzarro MJ, Saiman L, Polin RA. Time for a neonatal-specific consensus definition for sepsis. Pediatr Crit Care Med. 2014;15(6):523–528.
25. Sarnat HB, Sarnat MS. Neonatal encephalopathy following fetal distress: a clinical and electroencephalographic study. Arch Neurol. 1976;33(10):696–705.
26. Millichap JG. Neonatal hypoxic-ischemic encephalopathy: predicting the outcome. AAP Gd Rounds. 2006;16(5):52.
27. Hyperbilirubinemia S. Management of hyperbilirubinemia in the newborn infant 35 or more weeks of gestation. Pediatrics. 2004;114(1):297–316.
28. Al-Wassia H, Saber M. Admission of term infants to the neonatal intensive care unit in a Saudi tertiary teaching hospital: cumulative incidence and risk factors. Ann Saudi Med. 2017;37(6):420–424.
29. Burgess APH, Katz J, Pessolano J, Ponterio J, Moretti M, Lakhi NA. Determination of antepartum and intrapartum risk factors associated with neonatal intensive care unit admission. J Perinat Med. 2016;44(5):589–596.
30. Khasawneh W, Obeidat N, Yusef D, Alsulaiman JW. The impact of cesarean section on neonatal outcomes at a university-based tertiary hospital in Jordan. BMC Pregnancy Childbirth. 2020;20(1):335.
31. Taha Z, Ali Hassan A, Wikkeling-Scott L, Papandreou D. Factors associated with preterm birth and low birth weight in Abu Dhabi, the United Arab Emirates. Int J Environ Res Public Health. 2020;17(4):1382.
32. Raju TNK, Higgins RD, Stark AR, Leveno KJ. Optimizing care and outcome for late-preterm (near-term) infants: a summary of the workshop sponsored by the National Institute of Child Health and Human Development. Pediatrics. 2006;118:1207–1214.
33. Kamath BD, Todd JK, Glazner JE, Lezotte D, Lynch AM. Neonatal outcomes after elective cesarean delivery. Obstet Gynecol. 2009;113(6):1231–1238.
34. Ali SR, Ahmed S, Lohana H. Disease patterns and outcomes of neonatal admissions at a secondary care hospital in Pakistan. Sultan Qaboos Univ Med J. 2013;13(3):418–421.
35. Santas G, Santas F. Trends of caesarean section rates in Turkey. J Obstet Gynaecol. 2018;38(5):658–662.
36. Course C, Chakraborty M. Management of respiratory distress syndrome in preterm infants in wales: a full audit cycle of a quality improvement project. Sci Rep. 2020;10(1):1–7.
37. Siffel C, Kistler KD, Lewis JFM, Sarda SP. Global incidence of bronchopulmonary dysplasia among extremely preterm infants: a systematic literature review. J Matern Neonatal Med. 2019;1–11.
38. Zysman-Colman Z, Tremblay GM, Bandeali S, Landry JS. Bronchopulmonary dysplasia – trends over three decades. Paediatr Child Health. 2013;18(2):86.
39. Lapcharoensap W, Gage SC, Kan P, et al. Hospital variation and risk factors for bronchopulmonary dysplasia in a population-based cohort. JAMA Pediatr. 2015;169(2):e143676.
40. Alshehri M, Alshehri M, Algossadi R, Alshehri M. Predictors of bronchopulmonary dysplasia in Saudi preterm infants. Ann Neonatol J. 2020.
41. Braye K, Foureur M, de Waal K, Jones M, Putt E, Ferguson J. Group B streptococcal screening, intrapartum antibiotic prophylaxis, and neonatal early-onset infection rates in an Australian local health district: 2006–2016. PLoS One. 2019;14(4).
42. Khraisat H, Khatatbeh A, Al-Essa M. Retinopathy of prematurity: are we screening enough babies in Jordan? JRMS. 2018;25(1):74–78.
43. Waheeb S, Alshehri K. Incidence of retinopathy of prematurity at two tertiary centers in Jeddah, Saudi Arabia. Saudi J Ophthalmol. 2016;30(2):109–112.
44. Khader Y, Al-sheyab N, Alyahya M, Batieha A. Registration, documentation, and auditing of stillbirths and neonatal deaths in Jordan from healthcare professionals’ perspectives: reality, challenges and suggestions. J Matern Neonatal Med. 2018;1–11.
45. Smith AC, Mutangiri W, Fox R, Crofts JF. Millennium development goal 4: reducing perinatal and neonatal mortality in low-resource settings. Obstet Gynaecol. 2014;16(1):1–5.
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
