Back to Journals » Infection and Drug Resistance » Volume 13
In-vitro Antimicrobial Activity of Essential Oils and Spices Powder of some Medicinal Plants Against Bacillus Species Isolated from Raw and Processed Meat
Authors Hetta HF , Meshaal AK, Algammal AM , Yahia R , Makharita RR , Marraiki N, Shah MA , Hassan HAM , Batiha GES
Received 16 August 2020
Accepted for publication 11 November 2020
Published 4 December 2020 Volume 2020:13 Pages 4367—4378
DOI https://doi.org/10.2147/IDR.S277295
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Professor Suresh Antony
Helal F Hetta,1,2 Ahmed Kh Meshaal,3 Abdelazeem M Algammal,4 Ramadan Yahia,5 Rabab R Makharita,6,7 Najat Marraiki,8 Muhammad Ajmal Shah,9 Hebat-Allah M Hassan,2 Gaber El-Saber Batiha10
1Department of Internal Medicine, University of Cincinnati, College of Medicine, Cincinnati, OH 45267-0595, USA; 2Department of Medical Microbiology and Immunology, Faculty of Medicine, Assuit University, Assuit 71515, Egypt; 3Department of Botany and Microbiology, Faculty of Science, Al-Azhar University, Assiut 71524, Egypt; 4Department of Bacteriology, Immunology and Mycology, Faculty of Veterinary Medicine, Suez Canal University, Ismailia 41522, Egypt; 5Department of Microbiology and Immunology, Faculty of Pharmacy, Deraya University, Minia, Egypt; 6Biology Department, Faculty of Science and Arts Khulais, University of Jeddah, Jeddah, 21959, Saudi Arabia; 7Botany Department, Faculty of Science, Suez Canal University, Ismailia 41522, Egypt; 8Department of Botany and Microbiology, College of Science, King Saud University, Riyadh 11451, Saudi Arabia; 9Department of Pharmacognosy, Faculty of Pharmaceutical Sciences, Government College University, Faisalabad, Pakistan; 10Department of Pharmacology and Therapeutics, Faculty of Veterinary Medicines, Damanhour University, Damanhour 22511, Egypt
Correspondence: Helal F Hetta
Department of Medical Microbiology and Immunology, Faculty of Medicine, Assuit University, Assuit 71515, Egypt
Email [email protected]
Background and Aim: Bacillus species are widely distributed microorganisms in nature that are responsible for outbreaks of food poisoning and a common cause of food spoilage. This study aimed to isolate and identify foodborne Bacillus species from meat and to determine the antimicrobial activities of commercial essential oils and spices powder extracted from certain medicinal plants.
Methods: Sixty meat samples were collected in Assiut city and subdivided into raw meat and processed meat. Bacillus spp were isolated and identified according to their cultural characters, biochemical reactions, serological typing, and 16S rRNA gene sequencing. The antibacterial activity of essential oils and spices powder was measured by using well-diffusion and microbial count techniques.
Results: The prevalence of Bacillus spp. in the examined raw meat samples and processed meat samples was 13.34%, and 26.67%, respectively. There was a marked decrease in the total Bacillus species count after treatment of minced beef with essential oils and spices powder compared to the untreated one. Black seed oil was the most potent antibacterial essential oil among the tested oils present in this study.
Conclusion: Essential oils and spices powder of certain medicinal plants (cumin: Cuminum cyminum, black seeds: Nigella sativa, cloves: Syzygium aromaicum, cinnamon: Cinnamomum zeylanicum, and Marjoram: Origanum majorana) have a potential in vitro antimicrobial activity against Bacillus spp. Furthermore, Nigella sativa oil exhibited the most potent antibacterial activity against Bacillus spp.
Keywords: Bacillus spp., essential oils, processed meat, fresh meat, sequencing, antibacterial activity
Introduction
Meat is a daily part of the internationally human diet. It contains proteins (with all the essential amino acids), vitamins (A, B12, B6, D, and E), and minerals (iron and zinc), which are very important to human growth and well-being.1 Because of its nutrient content, meat is a highly perishable food since it represents good media for microorganism proliferation.1,2 According to the World Health Organization, 30% of the inhabitants in industrialized countries suffer every year from foodborne diseases3 with most of the cases attributed to the consumption of meat.4
Bacillus species are widely distributed microorganisms in nature. They have often been found to be responsible for outbreaks of food poisoning and are a common cause of food spoilage.5,6 Bacillus cereus is the most frequently isolated of naturally occurring bacilli. However, accurate identification for these bacterial species in many cases is difficult since they share many important morphological and biochemical properties.7
Different types of culture media such as MYP (mannitol–egg yolk–phenol red–polymyxin–agar) and PEMBA (polymyxin–pyruvate–egg yolk–mannitol–bromthymol blue-agar) were used for detection and the selective isolation of Bacillus spp. from food. The selectivity of these media is based on the hydrolysis of egg yolk lecithin and the absence of the use of mannitol by Bacillus spp. besides the presence of selective compounds like polymyxin.8
Several previous studies reported the emergence of multidrug-resistant bacterial pathogens that call for the need of natural herbal alternatives to the commonly used antimicrobial agents.9–14
Several molecular techniques that rely on DNA sequencing have been used to reveal the genetic relationship of bacillus strains.15 Enterobacterial repetitive intergenic consensus-PCR (ERIC-PCR) was used to screen for genetic relatedness. On the other hand, the application of pulsed-field gel electrophoresis (PFGE) has been proven to be useful for the discrimination and epidemiological characterization of B. cereus group strains.16
The 16S rRNA gene sequencing will continue to be the gold standard for identifying bacteria. The automation of this technique could enable it to be used routinely in clinical microbiology laboratories to replace the traditional phenotypic tests.17
Bacillus oceanisediminis H2T was first isolated from sediment of the South Sea in China18 and was found to be most closely related to Bacillus firmus19 and Bacillus infantis20 by 16S rRNA gene analysis. Recently, Bacillus oceanisediminis species is a rod-shaped aerobic bacterium, Gram-positive and spore-forming, and first isolated from marine sediment of the South Korean coast.21
The genus Brevibacillus was reported by Shida22 with the reclassification of 10 species of the genus Bacillus. A recent study reported that, 25 bacterial isolates were identified by 16S rRNA sequences analysis collected from public tap water in India, from the 25 isolates, 5 were of Bacillus (B. pumilus, B. flexus, B. megaterium, B. marisflavi and B. oceanosediminis).23
In a recent study, Fancello et al reported that 72 isolates were purified from olive samples collected in Italy and according to the 16S rDNA sequencing analysis, the bacterial isolates primarily fitting to Bacillus spp., Brevibacillus spp., Micrococcus spp., Pantoea spp., Kocuria spp., Staphylococcus spp., Lysinibacillus spp. and Lactobacillus spp. From them 17 isolates belonging to Bacillus spp. as follows: 11 strains as B. amyloliquefaciens, 5 strains as B. subtilis and 1 strain as B. megaterium. Also, 18 isolates belonging to Brevibacillus spp. as follows: 9 strains as Br. agri, 6 strains as Br. invocatus and 3 strains as Br. parabrevis.24 This study aimed to isolate and identify the foodborne Bacillus species by using conventional and molecular techniques and to determine the antimicrobial activities of commercial essential oils and spices powder extracted from certain medicinal plants (cumin: Cuminum cyminum, black seeds: Nigella sativa, cloves: Syzygium aromaicum, cinnamon: Cinnamomum zeylanicum, and Marjoram: Origanum majorana).
Materials and Methods
Sample Collection and Processing
Sixty meat samples (n = 60), including raw meat (n = 15), and processed meat (n = 45), were collected from butchery and markets in Assiut city, Egypt. The collected samples were transferred to the microbiology laboratory at the Faculty of Science (Al-Azhar University, Assiut Branch, Egypt) as soon as possible. The obtained samples were immediately cut and minced aseptically with a grinder through a 4 mm sterilized plate diameter (AC110V, China). All samples were kept at −20 °C for further analysis. The raw meat samples included beef, mutton meat, veal meat and chicken, while the processed meat samples included beef burgers, beef luncheon, ground beef, ground chicken, beef sausage, basterma, chicken luncheon and chicken burgers.
Isolation and Identification of Bacillus spp
Isolation of Bacillus spp
Three media were used for the isolation, and identification of Bacillus species; nutrient agar (Difco), Mannitol Egg Yolk Polymyxin agar (MEYP) (Difco, USA) (It depends on 1) selective inhibitory agent; polymyxin that inhibit the growth of other bacterial pathogens and contaminants. 2) Indicator systems; mannitol and phenol red, and egg yolk)25 and trypticase soy agar (Difco, USA) (support the growth of a wide variety of fastidious microorganisms).26 Twenty-five grams from each sample have been aseptically weighed and added to 225 mL of peptone water and overnight incubation at 37 °C in a shaker incubator. After 24 hours of enrichment, 0.1 mL of the broth was streaked on nutrient agar, MEYP agar, and trypticase soy agar plates and incubated at 37 °C for 24–48 hours. The characteristic colonies on MEYP agar are pink in color and were surrounded by a precipitate zone with the same color.
Phenotypic Identification of Bacillus spp
The obtained pure suspected colonies were identified according to their culture characters on nutrient agar, MEYP agar, and trypticase soy agar, morphological characters using Gram’s stain, and biochemical reactions including oxidase test, urease test, catalase test, indole production test, Voges–Proskauer test, citrate utilization test, and sugar fermentation pattern.27,28
Molecular Typing of Bacillus spp
A single pure colony of the isolate was taken and streaked on a nutrient agar plate and incubated overnight at 37 °C until a good colony morphology and size were obtained. DNA extraction was carried out according to the QIAamp DNA Mini Kit (Qiagen, USA). The 16S rRNA gene was amplified as described by Lane,29 using the universal primers 27F and 1492R. The PCR conditions were as follows: initial denaturation at 94 ºC for 5 min, then 30 cycles of denaturation at 94 ºC for 40 sec., annealing at 52 ºC for 30 sec., and extension at 72 ºC for 1 min., and final extension at 72 ºC for 7 min.30
The PCR amplicon was purified by the QIAquick gel extraction kit (Qiagen, USA) and sequenced using the same pair of primers. The sequence of the PCR amplicon was visualized and annotated using the Applied Biosystem Automated 3730XL DNA sequencer (Solgent Company, Daejeon, South Korea).
The BLAST tool from the National Center of Biotechnology Information (NCBI) website has been used to analyze the sequence obtained. Phylogenetic analysis of sequence was constructed using the software MEGA 6.0.31
Assessment of Antibacterial Activity of Some Medicinal Plants
Plant Samples Collection and Preparation
Five plant samples including cumin (Cuminumcyminum), black seeds (Nigella sativa), cloves (Syzygium aromaicum), cinnamon (Cinnamomum zeylanicum), and marjoram (Origanum Majorana) have been used in the treatment of beef meat samples. The powder samples were collected from the local market in Assiut city. The plant volatile oil samples were collected from National Research Center—Unit of oils extraction.
Treatment of Minced Beef Meat Samples with Both the Spices Powder and Essential Oils of the Plant Samples Separately for the Bacterial Count
Minced beef samples were mixed in a sterile mixer with spices powder (0.5% and 1% of minced beef meat as weight/weight), and the essential oils (0.25% and 0.5% of minced beef meat as volume/weight). Three groups were made from the samples; the first group was treated with either 0.5% or 1% spices, the second group was treated with either 0.25% or 0.5% essential oils, and the third group was kept as a control group. Each sample was packed in polyethylene bags and stored at 4 °C ± 1, and the bacterial count was done at intervals of 0, 3, 6, 9, 12, and 15 days.
Antimicrobial Activity of Essential Oils and Spices Powder of the Plant Samples Using the Well-Diffusion Assay
The antibacterial activity of essential oils and spices powder was examined in a well-diffusion assay, by adding the essential oils or the powder extract into the well of agar plates inoculated with the isolated strain that was confirmed to be Bacillus species based on culture characteristics, biochemical reactions, and 16S rRNA gene sequence analysis.32
The agar plates were prepared as follows: a pure culture of Bacillus spp. was grown in nutrient broth at 37 °C for 18–24 hours in shaker incubator until the final concentration was 108 CFU/mL. Twenty millilitres of plain nutrient agar was poured into each sterile petri dish. A 6 mm well punched in the solid agar plates via a sterile cork borer. Each plate was surface inoculated by 0.2 mL of the culture of Bacillus spp. broth in triplicates.
Serial concentrations of each tested essential oil were made (% w/v) in dimethylsulfoxide (DMSO) (10% aqueous) solvent as follows: 50 and 100 mg/mL and sterilized by filtration by passing through a 0.22-μm membrane filter. DMSO without the essential oil was used as a control. For spices powder, 100 g of the powder was dissolved in 500 mL ethyl alcohol34 and left for 72 hours at room temperature on a shaker,33 then the extract was dried by rotary evaporator with a vacuum at 25 °C. The residues obtained were weighed and reconstituted by ethyl alcohol and sterilized by filtration by passing through a 0.22-μm membrane filter. The concentrations made from each residue were as follows: 50 and 100 mg/mL. Fifty microlitres of each tested sample was pipette to the wells of the inoculated agar plates aseptically.35–37 The plates were incubated at 35 °C for 24 hours. After incubation, the inhibitory zones were measured in millimeters.
Statistical Analysis
GraphPad Prism program version 8.0.1 (244) (San Diego, CA, USA) was used for statistical analysis. P-values less than 0.05 were considered significant. Kruskal–Wallis test was used when groups compared to the control group to analyze the results for statistically significant differences.
Results
Isolation and Identification of Bacillus spp
Conventional Methods (Cultural and Biochemical Methods)
Only 14 isolates out of 297 presumptive isolated colonies have been characterized as Bacillus species among all the examined samples. The details are as follows: Bacillus spp. have been detected in 2 out of 6 fresh chicken meat samples (33.34%), 3 out of 12 beef luncheon samples (25.0%), 5 out of 12 ground beef samples (41.67%), only 1 out of 6 basterma samples(16.67%), and 3 out of 3 beef burger samples (100.0%), while Bacillus spp. has not been detected in fresh veal meat, fresh mutton meat and chicken luncheon samples (Table 1).
 |
Table 1 Occurrence of Bacillus spp. in Samples Collected During the Three Periods |
Regarding the phenotypic characteristics; Bacillus oceanisediminis is a Gram-positive, rod-shaped, aerobic, motile, catalase-positive, and endospore-forming microorganism. The optimal growth temperature was 37 °C; the optimal pH was 7.0. Also, according to the results obtained from the API 50 CHB/E medium, the characteristics strongly confirmed that the isolate belongs to the genus Bacillus. Concerning the phenotypic characteristics of Brevibacillus invocatus; it is a Gram-positive, motile, rod-shaped, strictly aerobic microorganism. The growth at 30 °C is initially slow, with a more rapid increase in growth rate following 24 hours’ incubation; after 3–4 days, the slightly umbonate colonies are 1–8 mm in diameter, with slightly irregular margins. Colonies are brownish yellow, some with a single whitish concentric zone at the margin, and they are butyrous and have silky surfaces; the centers are opaque and the edges are translucent. Brevibacillus invocatus is catalase-positive. Nitrate reduction-negative. Casein, gelatin, starch, and urea are not hydrolyzed and indole is not produced) (Table 2).
 |
Table 2 Biochemical Reaction Results of Selected Strains |
Molecular Identification of Bacillus spp. Using 16S rRNA Gene Sequencing
Two isolates (AKM5 & AKM6) from those, which identified to be Bacillus species, were chosen to be additionally confirmed via 16S rRNA gene sequence analysis using the BLASTn tool of the National Center of Biotechnology Information (NCBI). The isolate AKM5 was related to Bacillus oceanisedimins and the isolate AKM6 was related to Brevibacillus invocatus. Our sequences were submitted to the GenBank database with the accession numbers KX863513 for AKM5 isolate and accession number KX863514 for AKM6 isolate (Table 3).
 |
Table 3 Sequence Analysis of the 16S rDNA Gene by BLASTn Tool |
A phylogenetic tree based on the assessment of the 16S rRNA sequence with reference strains was created. The phylogenetic analysis was made with 1,500 bp and 2000bp sequences for Bacillus using the software MEGA 6 (Figure 1).
 |
Figure 1 Phylogenetic dendrogram based on 16S rRNA gene sequences showing the site of strain AKM5 & AKM6 between members of diverse genus species. The evolutionary history was inferred using the UPGMA method.59 The optimal tree with the sum of branch length = 0.13321378 is shown. The tree is drawn to scale, with branch lengths in the same units as those of the evolutionary distances used to infer the phylogenetic tree. The evolutionary distances were computed using the Kimura 2-parameter method60 and are in the units of the number of base substitutions per site. The analysis involved 21 nucleotide sequences. All positions containing gaps and missing data were eliminated. There were a total of 258 positions in the final dataset. Evolutionary analyses were conducted in MEGA6.61 |
Assessment of Antibacterial Activity of Some Medicinal Plants
Treatment of Minced Beef with Plant Samples for the Microbial Count
The results show that the treatment of minced beef with a level of 0.50% essential oil has more antibacterial activity than 0.25% essential oils of the same plant on Bacillus species and the same matter for spices powder. The differences between the control group and each treated minced beef sample after 15 days’ storage were statistically significant, except for samples treated with 0.25% essential oil of marjoram it was non-significant. Generally, the essential oils have better antibacterial activity than spices powder of the same plants. The details of the results were as follows:
Changes in Bacillus species Log10 count (CFU/g) in minced beef samples treated with essential oils during storage at 4±1°C up to 15 days:
Bacillus spp. was counted in different minced samples contain essential oils of cumin, black seeds, cloves, cinnamon, and marjoram under levels of 0.25% and 0.50% and control sample during storage at 4±1°C up to 15 days. The results revealed that Bacillus spp. counts increased in the control sample; thus, its counts at zero time were (3.333 log10 CFU/g) and reached to (3.439 log10 CFU/g) at the end of storage periods. However, for the treated samples, after 12 days of the refrigerated storage, the microbial count reached zero except samples treated with marjoram oil 0.25%, it reached zero count after 15 days of storage. After 15 days of storage, the difference between the control group and the samples treated with 0.25% essential oil of cumin, black seeds, cloves and cinnamon was statistically significant (p = 0.0138, 0.0011, 0.0052 and 0.0088, respectively). Moreover, after 15 days of storage, the difference between the control group and all samples treated with 0.5% essential oil of cumin, black seeds, cloves, cinnamon and marjoram was statistically significant (p = 0.0010, 0.0001, 0.0013, 0.0037 and 0.0047, respectively). However, the difference was non-significant between the control group and the samples treated with 0.25 essential oil of marjoram (p = 0.1412) after 15 days of storage (Table 4) (Figure 2).
 |
Table 4 Changes in Bacillus spp. Log10 Count (CFU/g) of Minced Samples Treated with Essential Oils During Storage at 4±1°C Up to 15 Days |
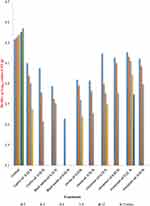 |
Figure 2 Changes in Bacillus spp. Log10 counts (CFU/g) of treated minced samples with essential oils during storage at 6±1°C for up to 15 days. |
Changes in Bacillus species Log10 counts (CFU/g) in minced beef samples treated with spices powder throughout storage at 4±1°C up to 15 days:
Bacillus spp. was counted in different minced samples contain spices powder of cumin, black seeds, cloves, cinnamon, and marjoram under levels of 0.5% and 1.0% and control sample during storage at 4±1°C up to 15 days. The results revealed that Bacillus spp. count increased in the control sample; thus, its counts at zero time were (3.333 log10 CFU/g) and reached to (3.439 log10 CFU/g) at the end of storage periods. Concerning the treated samples, the microbial count reached zero after 12 days of the refrigerated storage, except samples treated with cinnamon and marjoram spices powder under the level of 0.5%, which reached zero count after 15 days of storage. After 15 days of storage the difference between the control group and the samples treated with 0.5% spices powder of cumin, black seeds, cloves, cinnamon and marjoram was statistically significant (p = 0.0229, 0.0082, 0.0136, 0.0114 and 0.0184, respectively). Furthermore, the difference between the control group and the samples treated with 1% spices powder of cumin, black seeds, cloves, cinnamon and marjoram was statistically significant (p = 0.0029, <0.0001, 0.0014, 0.0044 and 0.0049) after 15 days of storage (Table 5) (Figure 3).
 |
Table 5 Changes in Bacillus spp. Log10 Count (CFU/g) of Minced Samples Treated with Spices Powders During Storage at 4±1°C Up to 15 Days |
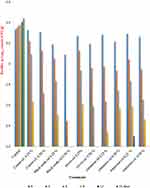 |
Figure 3 Changes in Bacillus spp. Log10 counts (CFU/g) of treated minced samples with spices powder throughout storage at 6±1°C for up to 15 days. |
Antimicrobial Activity of Essential Oils and Spices Powder Using the Well-Diffusion Assay Technique
The growth inhibition zones were measured are listed in Table 6. The black seeds had the highest antibacterial activity followed by marjoram then cloves then cumin and finally the least antibacterial activity for cinnamon. Generally, the antibacterial activity of essential oils is stronger than the spices powder for the same plant (Table 6).
 |
Table 6 Antibacterial Activities of Some Essential Oils and Spices Powder Against Bacillus spp. |
Discussion
The uncontrolled widespread use of antibiotics in veterinary and health sectors as well as the bacterial antibiotic-resistant genes are incriminated in the development of such multidrug-resistant strains.38–41 The emergence of multidrug-resistant bacteria call for the need of natural herbal alternatives to the commonly used antimicrobial agents.42–51 The foodborne diseases resulting from the consumption of Gram-positive and Gram-negative bacteria-contaminated food have been of vital concern to public health. Good hygiene practices, adequate preservative techniques for processed foods and the use of antimicrobial substances such as antibiotics are the various control and treatment methods in developed countries.52
Many studies stated that food analysis is a useful tool for identifying bacterial species including Bacillus spp. which cause foodborne diseases.53,54 Our results showed that 14 isolates were identified as Bacillus spp. by cultural, biochemical, and serological characteristics. Bacillus spp. was not recognized in fresh veal meat samples, 2 of 6 fresh chicken meat samples (33.34%), not recognized in fresh mutton meat, 3 of 12 beef luncheon (25.0%), not recognized in chicken luncheon, 5 of 12 ground beef (41.67%), only 1 of 6 basterma (16.67%), and 3 of 3 beef burger (100.0%). These results assure the fact that Bacillus spp. are associated with food from animal sources either fresh or processed and not specifically with beef.
In light of our results, we found that the highest occurrence of Bacillus spp. is in beef burger samples, which may indicate that the prevalence of Bacillus spp. is higher in processed meat than in fresh meet. According to 16S rRNA gene sequence analysis, the two identified isolates are Bacillus oceanisedimins and Brevibacillus invocatus; thus to our knowledge, this is the first report isolating both Bacillus oceanisedimins and Brevibacillus invocatus from both fresh and processed meat samples, and more investigations are required to identify their roles in meat samples.
The results of the treatment of minced beef with essential oils of cumin, black seeds, cloves, cinnamon, and marjoram under levels 0.25% and 0.50% during storage showed that statistically significant decrease in Bacillus spp. counts in comparison to control samples during storage except for essential oil of marjoram under levels 0.25%, accordingly we recommend the use of marjoram essential oil not less than 1% (volume/weight). Furthermore, there was an increase in Bacillus spp. count in the control sample. Moreover, after storage for 6 days Bacillus spp. disappeared in minced beef samples contained black seeds essential oils with level 0.50%. After 15 days of the refrigerated storage, there were no any Bacillus spp. in all studied samples except the sample contained marjoram oil at 0.25% and 0.50% and the control.
The disappearance of Bacillus spp. in samples treated with essential oils of black seeds, cloves, cumin, and cinnamon meanwhile increased the Bacillus spp. count in a control sample, after 15 days of storage, indicates that the disappearance of Bacillus spp. in treated samples may be attributed to the antimicrobial activity of these essential oils. One of the most important findings of the current study was the reduced effectiveness of the major components of essential oil compared with whole oil mixtures. Many reports of the antibacterial activity of essential oil components have tested their efficacy against vegetative bacterial cells.55–57
The results of antimicrobial activity of essential oils and spices powder using Well-diffusion assays showed that the black seeds essential oil had the strongest antibacterial activity (biggest inhibition zone), while the Cumin oil had the least antibacterial activity (smallest inhibition zone) among the essential oils present in this study. The results also showed that the Marjoram spices power has the strongest antibacterial activity among spices powder tested in this study, while Cumin spices powder had the least antibacterial activity among the spices powder tested in this study. Therefore, it is obvious that the cumin plant had the least antibacterial among plants present in this study. These results are in agreement with Özcan et al, which reported that essential oils of some spices may be used as antimicrobial agents to prevent the spoilage of food products.58
Study limitation and future recommendations: The small sample size is the main limitation of this study. As part of the antimicrobial activity of essential oils, it is better to evaluate the minimum inhibitory concentration (MIC) and the minimum bactericidal concentration (MBC).
Conclusion
Bacillus species are commonly associated with food from animal sources that is considered a public health threat. The highest prevalence of Bacillus spp. was observed in processed meat, especially beef burger. Essential oils and spices powder of certain medicinal plants (black seeds: Nigella sativa, cloves: Syzygium aromaicum, cinnamon: Cinnamomum zeylanicum, and Marjoram: Origanum majorana) have a potential in vitro antimicrobial activity against Bacillus spp. Furthermore, Nigella sativa oil exhibited the most potent antibacterial activity against Bacillus spp., while Cumin (Cuminum cyminum) showed the least antibacterial activity.
Acknowledgments
The authors extend their appreciation to the researchers supporting project number (RSP-2020/201), King Saud University, Riyadh, Saudi Arabia.
Disclosure
The authors report no conflicts of interest for this work.
References
1. Zerabruk K, Retta N, Muleta D, Tefera AT. Assessment of microbiological safety and quality of minced meat and meat contact surfaces in selected butcher shops of Addis Ababa, Ethiopia. J Food Qual. 2019;2019.
2. Holck A, Axelsson L, McLeod A, Rode TM, Heir EJ. Health and safety considerations of fermented sausages. J Food Qual. 2017;2017.
3. Mouafo HT, Baomog A, Adjele JJ, Sokamte AT, Mbawala A, Ndjouenkeu R. Microbial profile of fresh beef sold in the markets of ngaoundéré, cameroon, and antiadhesive activity of a biosurfactant against selected bacterial pathogens. J Food Qual. 2020;2020.
4. Salifou C, Boko K, Ahounou G, et al. Diversité de la microflore initiale de la viande et sécurité sanitaire des consommateurs. Int J Biol Chem Sci. 2013;7(3):1351–1369.
5. Goepfert J, Spira W, Elshafie HJ, Technology F. Bacillus cereus: food poisoning organism. A review. J Milk Food Technol. 1972;35(4):213–227.
6. Camele I, Elshafie HS, Caputo L, Sakr SH. Bacillus mojavensis: biofilm formation and biochemical investigation of its bioactive metabolites. J Biol Res/Bollettino della Società Italiana di Biologia Sperimentale. 2019;92:1.
7. Kwon G-H, Lee H-A, Park J-Y, et al. Development of a RAPD-PCR method for identification of Bacillus species isolated from Cheonggukjang. Int J Food Microbiol. 2009;129(3):282–287.
8. Hendriksen NB, Hansen B. Diagnostic properties of three conventional selective plating media for selection of Bacillus cereus, B. thuringiensis and B. weihenstephanensis. Folia Microbiologica. 2011;56(6):535–539.
9. Algammal AM, El-Kholy AW, Riad EM, et al. Genes encoding the virulence and the antimicrobial resistance in enterotoxigenic and shiga-toxigenic E. coli isolated from diarrheic calves. Toxins. 2020;12:6. doi:10.3390/toxins12060383
10. Algammal AM, El-Sayed ME, Youssef FM, et al. Prevalence, the antibiogram and the frequency of virulence genes of the most predominant bacterial pathogens incriminated in calf pneumonia. AMB Express. 2020;10(1):99. doi:10.1186/s13568-020-01037-z
11. Algammal AM, Enany ME, El-Tarabili RM, Ghobashy MOI, Helmy YA. Prevalence, antimicrobial resistance profiles, virulence and enterotoxins-determinant genes of MRSA isolated from subclinical bovine mastitis in Egypt. Pathogens (Basel, Switzerland). 2020;9:5.
12. Algammal AM, Mohamed MF, Tawfiek BA, Hozzein WN, El Kazzaz WM, Mabrok M. Molecular typing, antibiogram and PCR-RFLP based detection of aeromonas hydrophila complex isolated from oreochromis niloticus. Pathogens (Basel, Switzerland). 2020;9:3.
13. Enany ME, Algammal AM, Nasef SA, et al. The occurrence of the multidrug resistance (MDR) and the prevalence of virulence genes and QACs resistance genes in E. coli isolated from environmental and avian sources. AMB Express. 2019;9(1):192. doi:10.1186/s13568-019-0920-4
14. El-Sayed M, Algammal A, Abouel-Atta M, Mabrok M, Emam A. Pathogenicity, genetic typing, and antibiotic sensitivity of Vibrio alginolyticus isolated from Oreochromis niloticus and Tilapia zillii. Rev Med Vet. 2019;170:80–86.
15. Koeck J-L, Njanpop-Lafourcade B-M, Cade S, et al. Evaluation and selection of tandem repeat loci for Streptococcus pneumoniae MLVA strain typing. 2005;5(1):66.
16. Gdoura-Ben Amor M, Siala M, Zayani M, et al. Isolation, identification, prevalence, and genetic diversity of Bacillus cereus group bacteria from different foodstuffs in Tunisia. Front Microbiol. 2018;9:447.
17. Woo PC, Cheung EY, Leung K-W, Yuen K-Y. Identification by 16S ribosomal RNA gene sequencing of an Enterobacteriaceae species with ambiguous biochemical profile from a renal transplant recipient. Diagn Microbiol Infect Dis. 2001;39(2):85–93. doi:10.1016/S0732-8893(01)00206-1
18. Zhang J, Wang J, Fang C, et al. Bacillus oceanisediminis sp. nov., isolated from marine sediment. Int J Syst Evol Microbiol. 2010;60(12):2924–2929.
19. Manzo N, D’Apuzzo E, Coutinho PM, Cutting SM, Henrissat B, Ricca E. Carbohydrate-active enzymes from pigmented Bacilli: a genomic approach to assess carbohydrate utilization and degradation. Int J Syst Evol Microbiol. 2011;11(1):198.
20. Ko KS, Oh WS, Lee MY, et al. Bacillus infantis sp. nov. and Bacillus idriensis sp. nov., isolated from a patient with neonatal sepsis. Int J Syst Evol Microbiol. 2006;56(11):2541–2544.
21. Lee Y-J, Lee S-J, Jeong H, et al. Draft genome sequence of Bacillus oceanisediminis 2691. Int J Syst Evol Microbiol.2012;194(22):6351–6352.
22. Shida O, Takagi H, Kadowaki K, Komagata K, Microbiology E. Proposal for Two New Genera, Brevibacillus gen. nov. and Aneurinibacillus gen. nov.. Int J Syst Evol Microbiol.1996;46(4):939–946.
23. Kaur R, Singh D, Kesavan AK, Kaur R. Molecular characterization and antimicrobial susceptibility of bacterial isolates present in tap water of public toilets. Int Health. 2020;12(5):472–483. doi:10.1093/inthealth/ihz074
24. Fancello F, Multineddu C, Santona M, et al. Bacterial biodiversity of extra virgin olive oils and their potential biotechnological exploitation. Microorganisms. 2020;8(1):97. doi:10.3390/microorganisms8010097
25. Mossel D, Koopman M, Jongerius E. Enumeration of Bacillus cereus in foods. Appl Microbiol. 1967;15(3):650–653. doi:10.1128/AEM.15.3.650-653.1967
26. Rosenfeld D, Wolff DB, Atlas D. General probability-matched relations between radar reflectivity and rain rate. J Appl Meteorol. 1993;32(1):50–72. doi:10.1175/1520-0450(1993)032<0050:GPMRBR>2.0.CO;2
27. Cowan ST, Steel KJ. Cowan and Steel’s Manual for the Identification of Medical Bacteria. Cambridge university press; 2004.
28. El-Hadedy D, El-Nour SA. Identification of Staphylococcus aureus and Escherichia coli isolated from Egyptian food by conventional and molecular methods. J Genetic Eng Biotechnol. 2012;10(1):129–135. doi:10.1016/j.jgeb.2012.01.004
29. Lane D. 16S/23S rRNA sequencing. In: Nucleic Acid Techniques in Bacterial Systematics. 1991:115–175.
30. Yahia R, Hanora A, Fahmy N, Aly KA. Quorum sensing signal production by sponge-associated bacteria isolated from the Red Sea, Egypt. Afr J Biotechnol. 2017;16(32):1688–1698. doi:10.5897/AJB2017.16078
31. Tamura K, Stecher G, Peterson D, Filipski A, Kumar S. MEGA6: molecular evolutionary genetics analysis version 6.0. Mol Biol Evol. 2013;30(12):2725–2729. doi:10.1093/molbev/mst197
32. Elshafie HS, Caputo L, De Martino L, et al. Biological investigations of essential oils extracted from three Juniperus species and evaluation of their antimicrobial, antioxidant and cytotoxic activities. Journal of Applied Microbiology. 2020;129(5):1261–1271. doi:10.1111/jam.14723
33. Rajasekar A, Wilkinson S, Sekar R, Bridge J, Medina-Roldan E, Moy CKS. Biomineralisation performance of bacteria isolated from a landfill in China. Can J Microbiol. 2018;64(12):945–953. doi:10.1139/cjm-2018-0254
34. Alam S, Ahmad R, Pranaw K, Mishra P, Khare SK. Asparaginase conjugated magnetic nanoparticles used for reducing acrylamide formation in food model system. Bioresour Technol. 2018;269:121–126. doi:10.1016/j.biortech.2018.08.095
35. Adiguzel A, Ozer H, KiliC H, CetiN B. Screening of antimicrobial activity of essential oil and methanol extract of Satureja hortensis on foodborne bacteria and fungi. Czech J Food Sci. 2007;25(2):81. doi:10.17221/753-CJFS
36. De M, De AK, Banerjee AB. Screening of spices for antimicrobial activity. J Spices Aromatic Crops. 1999;8(2):135–144.
37. Mostafa AA, Al-Askar AA, Almaary KS, Dawoud TM, Sholkamy EN, Bakri MM. Antimicrobial activity of some plant extracts against bacterial strains causing food poisoning diseases. Saudi J Biol Sci. 2018;25(2):361–366. doi:10.1016/j.sjbs.2017.02.004
38. Algammal AM, Hetta HF, Elkelish A, et al. Methicillin-Resistant Staphylococcus aureus (MRSA): one health perspective approach to the bacterium epidemiology, virulence factors, antibiotic-resistance, and zoonotic impact. Infect Drug Resist. 2020;13:3255–3265. doi:10.2147/IDR.S272733
39. Algammal AM, Mabrok M, Sivaramasamy E, et al. Emerging MDR-Pseudomonas aeruginosa in fish commonly harbor oprL and toxA virulence genes and bla(TEM), bla(CTX-M), and tetA antibiotic-resistance genes. Sci Rep. 2020;10(1):15961. doi:10.1038/s41598-020-72264-4
40. Rangel-López L, Zaragoza-Bastida A, Valladares-Carranza B, et al. In vitro antibacterial potential of salix babylonica extract against bacteria that affect oncorhynchus mykiss and oreochromis spp. Animals. 2020;10:8. doi:10.3390/ani10081340
41. Makharita RR, Hetta HF, Abdelaziz MH, Hagagy FI, Ahmed AA, Algammal AM. Antibiogram and genetic characterization of carbapenem-resistant gram-negative pathogens incriminated in healthcare-associated infections. Infect Drug Resist. 2020;Volume 13(13):3991–4002. doi:10.2147/IDR.S276975
42. Saleh E, Morshdy AE, El-Manakhly E, et al. Effects of olive leaf extracts as natural preservative on retailed poultry meat quality. Foods. 2020;9(8):1017. doi:10.3390/foods9081017
43. Abd El-Baky RM, Farhan SM, Ibrahim RA, Mahran KM, Hetta HF. Antimicrobial resistance pattern and molecular epidemiology of ESBL and MBL producing Acinetobacter baumannii isolated from hospitals in Minia, Egypt. Alexandria J Med. 2020;56(1):4–13. doi:10.1080/20905068.2019.1707350
44. Abd El-Baky RM, Masoud SM, Mohamed DS, et al. Prevalence and some possible mechanisms of colistin resistance among multidrug-resistant and extensively drug-resistant Pseudomonas aeruginosa. Infect Drug Resist. 2020;13:323–332. doi:10.2147/IDR.S238811
45. Abd El-Baky RM, Sandle T, John J, Gea A-R, Hetta HF. A novel mechanism of action of ketoconazole: inhibition of the NorA efflux pump system and biofilm formation in multidrug-resistant Staphylococcus aureus. Infect Drug Resist. 2019;12:1703–1718. doi:10.2147/IDR.S201124
46. Abd Ellah NH, Abdel-Aleem JA, Abdo MN, Abou-Ghadir OF, Zahran KM, Hetta HF. Efficacy of ketoconazole gel-flakes in treatment of vaginal candidiasis: formulation, in vitro and clinical evaluation. Int J Pharm. 2019;567:118472. doi:10.1016/j.ijpharm.2019.118472
47. Abo-Shama UH, El-Gendy H, Mousa WS, et al. Synergistic and antagonistic effects of metal nanoparticles in combination with antibiotics against some reference strains of pathogenic microorganisms. Infect Drug Resist. 2020;13:351–362. doi:10.2147/IDR.S234425
48. Ahmed S, Ahmed S, Mohamed W, et al. Nosocomial vancomycin and methicillin resistant staphylococcal infections in intensive care units in Assiut University Hospitals. Egypt J Med Microbiol. 2011;20:2.
49. Al-Kadmy IMS, Ibrahim SA, Al-Saryi N, Aziz SN, Besinis A, Hetta HF. Prevalence of genes involved in colistin resistance in Acinetobacter baumannii: first report from Iraq. Microbial Drug Resist. 2020;26(6):616–622. doi:10.1089/mdr.2019.0243
50. El-Kazzaz W, Metwally L, Yahia R, Al-Harbi N, El-Taher A, Hetta HF. Antibiogram, prevalence of OXA carbapenemase encoding genes, and RAPD-genotyping of multidrug-resistant acinetobacter baumannii incriminated in hidden community-acquired infections. Antibiotics (Basel, Switzerland). 2020;9:9.
51. El-Mokhtar MA, Hetta HF. Ambulance vehicles as a source of multidrug-resistant infections: a multicenter study in Assiut City, Egypt. Infect Drug Resist. 2018;11:587. doi:10.2147/IDR.S151783
52. Aboaba O, Smith S, Olude F. Antibacterial effect of edible plant extract on Escherichia coli 0157: H7. Pak J Nutri. 2006;5(4):325–327.
53. El-Helow ERJFML. Identification and molecular characterization of a novel Bacillus strain capable of degrading Tween-80. FEMS Microbiol Letters. 2001;196(2):119–122.
54. Whittaker P, Fry FS, Curtis SK, et al. Use of fatty acid profiles to identify food-borne bacterial pathogens and aerobic endospore-forming bacilli. J Agr Food Chem. 2005;53(9):3735–3742.
55. Bakkali F, Averbeck S, Averbeck D, Idaomar MJF. Toxicology c. Biological effects of essential oils–a review. Food Chem Toxicol. 2008;46(2):446–475.
56. Cox SD, Mann CM, Markham JL, Gustafson JE, Warmington JR, Wyllie SGJM. Determining the antimicrobial actions of tea tree oil. Molecules. 2001;6(2):87–91.
57. Dorman H, Deans S. Antimicrobial agents from plants: antibacterial activity of plant volatile oils. J Appl Microbiol. 2000;88(2):308–316.
58. Özcan MM, Saǧdıç O, Özkan G. Inhibitory effects of spice essential oils on the growth of Bacillus species. J Med Food. 2006;9(3):418–421.
59. Sokal RR. Numerical Taxonomy. San Francisco, CA: Freeman; 1973.
60. Kimura M. A simple method for estimating evolutionary rates of base substitutions through comparative studies of nucleotide sequences. J Mol Evol. 1980;16(2):111–120.
61. Tamura K, Stecher G, Peterson D, Filipski A, Kumar S. MEGA6: molecular evolutionary genetics analysis version 6.0.. Mol Biol Evol. 2013;30:2725–2729.
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
