Back to Journals » Journal of Multidisciplinary Healthcare » Volume 15
Impact of Smoking on COVID-19 Symptoms in Non-Vaccinated Patients: A Matched Observational Study from Qatar
Authors Haider MZ , Al-Mannai A, Al-Sirhan S, Elsabagh A, Nasser N, Al-Quraishi N, Ouda A, Erradi K, Ashour AA , Gupta I, Abdulmajeed J, Al-Romaihi HE, Bansal D, Musa OAH, Abd Farag EAB, Al-Thani MHJ, Al Moustafa AE
Received 1 November 2021
Accepted for publication 10 December 2021
Published 17 March 2022 Volume 2022:15 Pages 531—540
DOI https://doi.org/10.2147/JMDH.S347130
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Scott Fraser
Mohammad Z Haider,1 Amna Al-Mannai,1 Sally Al-Sirhan,1 Ahmed Elsabagh,1 Nasser Nasser,1 Noora Al-Quraishi,1 Amr Ouda,1 Khadija Erradi,1 Anas A Ashour,2 Ishita Gupta,1,3 Jazeel Abdulmajeed,4 Hamad E Al-Romaihi,4 Devendra Bansal,4 Omran AH Musa,4 Elmoubasher Abu Baker Abd Farag,4 Mohammed HJ Al-Thani,4 Ala-Eddin Al Moustafa1,3,5
1College of Medicine, QU Health, Qatar University, Doha, Qatar; 2Department of Internal Medicine, Hamad Medical Corporation, Doha, Qatar; 3Biomedical and Pharmaceutical Research Unit, QU Health, Qatar University, Doha, Qatar; 4Ministry of Public Health, Doha, Qatar; 5Biomedical Research Centre, Qatar University, Doha, Qatar
Correspondence: Ala-Eddin Al Moustafa, College of Medicine, QU Health, Qatar University, PO Box 2713, Doha, Qatar, Tel +974 4403 7817, Fax +974 4403-3333, Email [email protected]
Purpose: Predisposition to acute illness from COVID-19 is suggested to correlate with cigarette smoking as it augments the risk of developing cardiovascular and respiratory illnesses, including infections. However, the effects of smoking on COVID-19 symptoms are not well described and controversial. In this study, we aim to explore the associations between smoking and COVID-19 symptoms.
Subjects and Methods: A cross-sectional study using the Ministry of Public Health (MoPH), Qatar database was administered to a Qatari population with confirmed COVID-19 disease who filled in pre-defined phone-call questionnaire between 27th February 2020 and 31st December 2020. We analyzed 11,701 non-vaccinated COVID-19 individuals (2952 smokers and 8749 non-smokers) with confirmed RT-PCR test results. The association of smoking and the presence of symptoms as well as patient characteristics was calculated using Pearson’s Chi-square and Fisher’s exact tests, adjusting for potential covariates.
Results: Compared with the non-smokers, symptomatic COVID-19 infection is significantly higher in smokers. In addition, we found fever as the most common symptom developed in COVID-19 patients followed by cough, headache, muscle ache, and sore throat. As compared to other symptoms, association of smoking with chills and abdominal pain was less evident (P < 0.05 and P < 0.001, respectively). However, both groups showed similar rates of developing cough.
Conclusion: In conclusion, smoking is associated with COVID-19 symptoms frequency in non-vaccinated patients; nevertheless, further investigations are necessary to understand the mechanism of this association which could generate new targets for the management of COVID-19 in smoker patients.
Keywords: COVID-19, smoking, frequency, Qatar, symptoms
Introduction
Coronavirus disease 2019 (COVID-19), is a severe respiratory illness that is highly infectious, caused by the novel severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2).1 It is evident that SARS-CoV-2 infection leads to multi-organ disease, with a high prevalence of acute respiratory distress syndrome (ARDS) and digestive tract complications.2 COVID-19 has a wide range of clinical symptoms ranging from asymptomatic to multi-organ dysfunction, and is classified into mild, moderate, severe, and critical based on the severity of its manifestation.3 The most common symptoms of COVID-19 include fever, cough, diarrhoea, and fatigue.3 Moreover, aged individuals as well as those who are immunocompromised, comorbid patients, and smokers are at a higher risk of casualty.4,5
Smoking is a known to trigger for respiratory tract inflammation, allergy, epithelial cells permeability, mucus development, and defective muco-ciliary clearance.6 Earlier studies reported that smoking practices elevate the risk of several bacterial and viral respiratory infections and correlates with worst outcomes in infected individuals.7–9 Smoking is a well- recognized risk factor for various respiratory and cardio-metabolic diseases, including chronic obstructive pulmonary disease (COPD) and bronchial asthma.10,11 In comparison to non-smokers, acute or chronic smokers can develop infection, tuberculosis, bacterial pneumonia, ARDS12–14 with severe complications14–16 and present with several co-morbidities, including emphysema, atherosclerosis, and immune dysregulation,17 thus, promoting the onset and progression of COVID-19. Nevertheless, the role of smoking in COVID-19 remains controversial and understated.14,18–21 As some studies demonstrated an association between smoking and COVID-19 severity,16,22 while others failed to report such a correlation.23,24 As such, a previous report across acute care hospitals and associated outpatient clinics found smoking as an independent risk factor for COVID-19 hospitalization,25 another meta-analysis described smoking as a risk factor for the development and progression of severe COVID-19.26,27 In contrast, a study by Farsalinos et al,20 found smoking prevalence relatively lower in COVID-19 patients.
Although, the effects of smoking on COVID-19 symptoms vary and are not well-described in literature, especially in Qatar, despite the high prevalence of smoking; we herein explored the correlation between smoking and COVID-19 symptoms in the population of Qatar. Our study pointed out that smoking is associated with symptomatic COVID-19 infection. The results of this study can help in understanding the correlation between smoking and COVID-19 symptoms, however, further studies are needed to investigate the mechanism behind this association.
Materials and Methods
Sample Collection
This is a matched cross-sectional study conducted using the Ministry of Public Health (MoPH), Qatar database. MoPH gathers de-identified information from all confirmed COVID-19 patients and stores the data in their electronic database. Between 27th February 2020 and 31st December 2020, a total of 105,745 patients were confirmed to have COVID-19 and were included in the database. Our study included all adult patients (≥18 years) with RT-PCR-confirmed COVID-19 infection. The study was approved by the MoPH institutional review board (IRB: ERC-826-3-2020) with waiver of informed consent and Qatar University IRB. The authors had no direct contact with any of the participants.
All patients filled a pre-defined phone-call based forms that included “yes/no” questions about smoking status, COVID-symptoms, and comorbidities. COVID-19 symptoms included in the questionnaire were fever, chills, cough, shortness of breath, headache, muscle ache, sore throat, diarrhoea, and abdominal pain. Smoking status was defined as either active smoker or non-smoker with no further classification based on the number of cigarettes. All non-smokers were considered with no previous history of smoking. Subjects who identified themselves as smokers were matched in a 1:3 ratio with non-smokers based on age, gender, nationality, and known comorbidities. A total of 2952 smokers were extracted from the database and were matched with 8749 non-smokers. None of these patients had received any dose of the COVID-19 vaccine.
Subjects were excluded if COVID-19 was confirmed by methods other than RT-PCR, had active or prior cancer, were immunocompromised, were receiving immunosuppressive drugs or had missing demographic data (age, gender, nationality, smoking status, and symptoms).
Statistical Analysis
Pearson’s Chi-square and Fisher’s exact tests were used to compare the presence of symptoms and the characteristics between the two groups. Continuous variables were expressed as mean ± standard deviation (SD) while categorical variables were presented by the count and percentage. All statistical analyses were carried out using Stata Statistical Software: Release 16 (College Station, TX: StataCorp LLC). Matching was performed using SPSS Fuzzy Case Control Matching extension. Two-tailed P-values of less than 0.05 was considered statistically significant.
Results
This study included 11,701 patients, of whom 2952 were smokers and 8749 were non-smokers. The two groups were matched based on age, gender, nationality, and comorbidities. Hence, both groups had similar characteristics. The mean age of the study population was 34.5 years with the vast majority being males (97.3%). The most common nationality in this study was Indian which comprises around 25% of the population, followed by Qatari, Nepalese, and Bangladeshi. Diabetes and cardiovascular diseases were the most frequent comorbidities in the study population, each affecting around 6% of the patients. The rest of the comorbidities were rare. Four subjects from the smokers group underwent surgeries previously which was significantly higher than the non-smokers group, where only two had prior surgeries (P < 0.05) (Table 1).
 |
Table 1 Baseline Characteristics of Study Participants Based on Their Smoking Status. Data are Presented as Mean ± Standard Deviation or as Number of Subjects (Percentage) |
As indicated in Table 2, our data revealed that a significantly higher proportion of smokers developed symptomatic COVID-19 infection (75.3%) compared to the non-smokers (62.8%; P < 0.001), indicating that smokers have a risk ratio of 1.2 (95% confidence interval: 1.17–1.23) to develop symptoms related to COVID-19 infection when compared to non-smokers (Figure 1).
 |
Table 2 The COVID-19 Symptoms Based on Patients’ Smoking Status. Data are Presented as Number of Subjects (Percentage) |
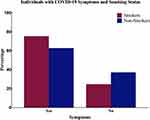 |
Figure 1 Bar chart showing the percentage of COVID-19 patients who experienced any COVID-19 related symptoms stratified based on their smoking status. |
On the other hand, and as demonstrated in Figure 2, the type of symptoms experienced in relation with smoking status show that fever is the most frequent symptom affecting 45% of confirmed COVID-19 cases, followed by cough, headache, muscle ache, and sore throat. Less frequent symptoms, present in less than 10% of patients, include chills, shortness of breath, diarrhoea, and abdominal pain. Smoking is associated with a significantly higher rate of developing all types of symptoms except cough, as both groups showed similar rates of developing cough (Table 2). On the other hand, the association of smoking with chills and abdominal pain was less evident when compared to the rest of the symptoms (P < 0.05 and P < 0.001; respectively) (Table 2).
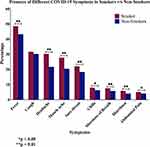 |
Figure 2 Bar chart showing the rate of developing known COVID-19 symptoms based on the patients’ smoking status. |
Discussion
In this investigation we explored for the first time the correlation between smoking and COVID-19 symptoms frequency in the population of Qatar. Our data revealed that current smoking is associated with a risk of developing symptoms suggestive of COVID-19, as well as a greater symptom burden, indicating an impact of smoking on COVID-19 disease frequency. Additionally, amongst patients positive for COVID-19, smokers had a higher symptom burden in comparison to non-smokers.
In our cohort, 97% of COVID-19 patients are males and 3% are females, which concurs with recent studies of COVID-19 infection.28–31 Furthermore, in this study, the estimated median age for patients is 34 years which is concordant with previous studies among COVID-19 patients in other countries32,33 as well as in Qatar and Saudi Arabia.29,30,34,35 As compared to other parts of the world, COVID-19 is found to affect younger populations in the Gulf Region.29,30,34 Such a trend is observed in our study, nevertheless, the age-groups effected with COVID-19 range from 15 to 87 years, indicating disease susceptibility across these ages. High infection and severity rates in adult patients are found to be associated with smoking, as seen in previous studies.36,37
Approximately 15% of COVID-19 patients investigated here exhibited various comorbidities as reported in previous studies.27,29,38 In this study, we found that both smokers and non-smokers had diabetes (6%), followed by cardiovascular disease (6%) and asthma (1%). This is in concordance with other reports which showed diabetes and/or hypertension as the most common comorbidities.27,29,32,34,39–41 However, our findings revealed that smoking had no significant association with diabetes and cardiovascular comorbidities. Along with cardiac and chronic respiratory diseases, lung and chronic kidney diseases are reported as risk factors associated with worse outcomes in COVID-19 patients.42,43
Similar to our data, various cohort studies revealed an association between smoking and COVID-19 disease comorbidity, incidence and mortality.14,16,22,27 In this context, smoking is known to damage the vascular endothelial,44 a characteristic feature in COVID-19 pathophysiology. A meta-analysis of 13 published Chinese studies pointed out older age (age > 65 years), male gender, and smoking were risk factors for disease progression in COVID-19 patients.45 On the other hand, a recent meta-analyses showed significant correlation between smoking and COVID-19 progression.27,46 Another meta-analysis based on 15 studies also reported concordant data where 22% of current and 46% of former smokers had severe COVID-19 complications.14 A recent study by Almazeedi et al47 was performed based on a cohort of 1096 COVID-19 patients in Kuwait and they reported that age > 50 years, smoking, elevated C-reactive protein (CRP) and prolactin levels as well as a quick sequential organ failure assessment (qSOFA) score greater than 0 as risk factors associated with COVID-19 severity. Likewise, a study on 10,713 COVID-19 patients from Brazil also reported smoking and pulmonary disease to augment the risk of COVID-19 severity.48 In addition to studies carried out in larger cohorts, studies in small cohorts also showed similar results.49,50 Recently, Archie et al51 postulated a plausible role of smoking on cerebrovascular and neurological dysfunction in COVID-19 patients. Recently, Jiménez-Ruiz11 performed a systematic review and meta-analysis and reported that smoking is a risk factor for a severe COVID-19 infection with a plausibility of developing a more critical condition. Similarly, another investigation showed a clear association between smoking and disease severity, with approximately 3% of deceased patients being smokers.52 Moreover, a meta-analysis that included 7 studies found that smokers had twice the risk of developing COVID-19 severity.53
Although, smoking may not essentially elevate the risk of developing COVID-19, the underlying mechanisms involving the biological and inflammatory signaling pathways induced by COVID-19 infection can be severe for smokers.21 Likewise, earlier research showed increased susceptibility to COVID-19 infection is attributed to the overexpression of one of the key SARS-CoV-2 receptors, angiotensin converting enzyme 2 (ACE2) receptor.4,17,54 Smoking increases susceptibility to COVID-19 infection via the triggering of peripheral nicotinic acetylcholine receptors (nAChRs) present in various organ systems.55 Nicotine disrupts homeostasis of the renin angiotensin system (RAS), and overexpresses the ACE/angiotensin (ANG)-II/ANG II type 1 receptor axis, resulting in cardiovascular and pulmonary diseases.55 Hence, nicotine can affect the nAChRs and enhance the SARS-CoV-2 host entry.56 Moreover, studies reported that smokers with diabetes, cardiovascular disease, COPD, asthma or cancer can enhance the expression pattern of ACE2; hence, they are more susceptible to develop COVID-19 infection and have worst prognosis.57,58 Diabetes can overexpress ACE2 expression via hyperinsulinemia along with other mechanisms.59 During diabetes, the function of Treg cells is compromised and expression of tumor necrosis factor-alpha (TNF-α) is reduced, further enhancing infections.59,60 On the other hand, cardiovascular patients are also prone to COVID-19 disease severity. In hypertensive patients, ACE2 modulators (ACE1 inhibitors, ANG blockers, and thiazolidinediones) are often prescribed to treat hypertension and can overexpress ACE2;39 a risk factor for the onset of COVID-19 disease. Likewise, the viral capsid of SARS-CoV-2 binds to the ACE2 surface and activates TMPRSS2, (Transmembrane serine protease 2, an androgen-responsive gene highly expressed in men), thus, increased TMPRSS2 expression in men increases their proneness to COVID-19 disease.61 Furthermore, it has been demonstrated that cigarette exposure can increase the expression pattern of ACE2 and TMPRSS2 in patients with COPD and idiopathic pulmonary fibrosis (IPF);17,62–66 in this context, it is important to highlight that COPD is reported as a major risk factor for COVID-19 infection.53,67 Smokers suffering from cancer are also found to be at a higher risk of developing COVID-19 infection due compromised immunity, thus providing an effectual environment for viral replication.68
On the contrary, while several studies reported an association between disease severity and smoking,16,22 few investigations found no difference or a negative correlation between smoking and COVID-19 severity.23,24 Moreover, a recent review reported that current smokers are at a comparatively lower risk of developing COVID-19 as compared to never smokers, however, these findings aim to analyse certain factors that could make the interpretation of these results more complex.19 Hence, it is necessary to conduct interventional studies in the general population and in patients post-COVID-19 disease. In addition to investigations analysing the correlation between smoking and COVID-19 severity, reports on behavioural changes in tobacco consumption during the pandemic found varying data. While, in USA there was an increase by 41% in tobacco consumption during the onset of COVID-19,69 in Italy, smokers had considered quitting.70 However, the same study found that former smokers and never smokers wanted to start smoking.70 In China and England as well, there was an increase in smoking among smokers and relapsed smoking habits among former smokers.71,72 As of currently, both epidemiological/clinical evidence and in-silico findings indicate that COVID-19 infection is a nAChR disease that could be prevented and may be controlled by nicotine.56,73 Smoking was suggested to exert a protective measure as its nicotine acts as a nAChR agonist and competes with the SARS-COV-2 for the binding site, hence leading to a decline in accessible viral adhesion sites.74 Nevertheless, the plausibility of nicotine displaying protective effect against COVID-19 can be partially masked by smoking-related toxicity. In addition, abrupt cessation of nicotine consumption when smokers are hospitalized need further validation in laboratory studies using nicotine-based pharmaceutical products. Considering the limited and contradicting past investigations available on this topic, we hypothesized a strong association of smoking with the risk of developing respiratory diseases. However, coughing, which is the major respiratory depression symptom was reported in both smokers and non-smokers nearly equally, with only a non-statistically significant difference of 1.4% more for smokers. Moreover, significant differences were reported in other symptoms such as fever, muscle ache, abdominal pain, diarrhoea, headaches, and shortness of breath, all of which are also symptoms of nicotinic overstimulation of the cholinergic system75 which is activated by the nicotine in cigarettes and triggers COVID-19 disease.76,77
Although, our present study included a large sample size and points out a strong link between smoking and symptoms development among COVID-19 patients, there are several factors limiting the interpretation of our investigation. The present results remain limited by the available information. In particular, detailed data on possible sources of exposure to smoking are lacking. The data was self-reported by patients via phone-call based forms. The patients reported their symptoms through this form. Additionally, there was no classification based on the number of packs smoked per day. Thus, heavy smokers and light smokers were grouped together. Moreover, patients who reported as non-smokers are assumed to have no history of smoking and thus, we believe that further studies are needed to investigative the consequences and underlying mechanisms of smoking and its association with COVID-19 infection and its frequency in patients. In addition to cigarette smoking, effects of waterpipe and electronic cigarette use on COVID-19 transmission, clinical progression and frequency of COVID-19 should be further studied. Lastly, the association between COPD frequency and COVID-19 should be also analysed.
Conclusion
In conclusion, our present study demonstrates an association between smoking and COVID-19 symptoms frequency in non-vaccinated patients. While we believe that further studies in a larger cohort are required to clearly determine the association between smoking and COVID-19 symptoms which can help in controlling COVID-19 induced morbidity and mortality especially in smoker patients. Nevertheless, and based on our present study it is evident that discontinuing of smoking practices should be contemplated as a part of the strategies to address COVID-19 infection management in the international arena, as smoking increases both the likelihood of symptomatic disease and the frequency of disease.
Ethics Approval and Consent to Participate
Patient consent was waived as this was a matched cross-sectional study conducted using the Ministry of Public Health (MoPH), Qatar database. The study was conducted according to the guidelines of the Declaration of Helsinki, and approved by the Institutional Review Board (or Ethics Committee) of Ministry of Public Health (IRB: ERC-826-3-2020).
Acknowledgments
We would like to thank Qatar’s Ministry of Public Health for providing the data used in this study. Also, we would like to thank the Department of Population Medicine at College of Medicine, Qatar University for their support provided throughout the study.
Funding
This research received no external funding. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Van Doremalen N, Bushmaker T, Morris DH, et al. Aerosol and surface stability of SARS-CoV-2 as compared with SARS-CoV-1. New Engl J Med. 2020;382(16):1564–1567. doi:10.1056/NEJMc2004973
2. Cascella M, Rajnik M, Cuomo A, Dulebohn SC, Di Napoli R. Features, evaluation and treatment coronavirus (COVID-19). In: Statpearls [Internet]. StatPearls Publishing; 2020.
3. Wang Y, Wang Y, Chen Y, Qin QJ. Unique epidemiological and clinical features of the emerging 2019 novel coronavirus pneumonia (COVID‐19) implicate special control measures. J Med Virol. 2020;92(6):568–576. doi:10.1002/jmv.25748
4. Brake SJ, Barnsley K, Lu W, McAlinden KD, Eapen MS, Sohal SS. Smoking upregulates angiotensin-converting enzyme-2 receptor: a potential adhesion site for novel coronavirus SARS-CoV-2 (Covid-19). J Clin Med. 2020;9(3):841. doi:10.3390/jcm9030841
5. Hassan SA, Sheikh FN, Jamal S, Ezeh JK, Akhtar AJC. Coronavirus (COVID-19): a review of clinical features, diagnosis, and treatment. Cureus. 2020;12(3):e7355. doi:10.7759/cureus.7355
6. Strzelak A, Ratajczak A, Adamiec A, Feleszko W. Tobacco smoke induces and alters immune responses in the lung triggering inflammation, allergy, asthma and other lung diseases: a mechanistic review. Int J Environ Res Public Health. 2018;15(5):1033. doi:10.3390/ijerph15051033
7. Bagaitkar J, Demuth DR, Scott DA. Tobacco use increases susceptibility to bacterial infection. Tob Induc Dis. 2008;4(1):12. doi:10.1186/1617-9625-4-12
8. Zambon JJ, Grossi SG, Machtei EE, Ho AW, Dunford R, Genco RJ. Cigarette smoking increases the risk for subgingival infection with periodontal pathogens. J Periodontol. 1996;67(10 Suppl):1050–1054. doi:10.1902/jop.1996.67.10s.1050
9. Arcavi L, Benowitz NL. Cigarette smoking and infection. Arch Intern Med. 2004;164(20):2206–2216. doi:10.1001/archinte.164.20.2206
10. Eapen MS, Sharma P, Moodley YP, Hansbro PM, Sohal SS. Dysfunctional immunity and microbial adhesion molecules in smoking-induced pneumonia. Am J Respir Crit Care Med. 2019;199(2):250–251. doi:10.1164/rccm.201808-1553LE
11. Jiménez-Ruiz CA, López-Padilla D, Alonso-Arroyo A, Aleixandre-Benavent R, Solano-Reina S, de Granda-orive JI. COVID-19 and smoking: a systematic review and meta-analysis of the evidence. Arch Bronconeumol. 2021;57 Suppl 1:21–34. doi:10.1016/j.arbres.2020.06.024
12. Han L, Ran J, Mak Y-W, et al. Smoking and influenza-associated morbidity and mortality: a systematic review and meta-analysis. Epidemiology. 2019;30(3):405–417. doi:10.1097/EDE.0000000000000984
13. Lawrence H, Hunter A, Murray R, Lim W, McKeever T. Cigarette smoking and the occurrence of influenza–systematic review. J Infect. 2019;79(5):401–406. doi:10.1016/j.jinf.2019.08.014
14. Alqahtani JS, Oyelade T, Aldhahir AM, et al. Prevalence, severity and mortality associated with COPD and smoking in patients with COVID-19: a rapid systematic review and meta-analysis. PLoS One. 2020;15(5):e0233147. doi:10.1371/journal.pone.0233147
15. Zhang -J-J, Dong X, Cao -Y-Y, et al. Clinical characteristics of 140 patients infected with SARS‐CoV‐2 in Wuhan, China. Allergy. 2020;75(7):1730–1741. doi:10.1111/all.14238
16. Guan W-J, Ni Z-Y, Hu Y, et al. Clinical characteristics of coronavirus disease 2019 in China. N Engl J Med. 2020;382(18):1708–1720. doi:10.1056/NEJMoa2002032
17. Smith JC, Sausville EL, Girish V, et al. Cigarette smoke exposure and inflammatory signaling increase the expression of the SARS-CoV-2 receptor ACE2 in the respiratory tract. Dev Cell. 2020;53(5):514–529.e513. doi:10.1016/j.devcel.2020.05.012
18. Vardavas CI, Nikitara K. COVID-19 and smoking: a systematic review of the evidence. Tob Induc Dis. 2020;18:20. doi:10.18332/tid/119324
19. Simons D, Shahab L, Brown J, Perski O. The association of smoking status with SARS-CoV-2 infection, hospitalization and mortality from COVID-19: a living rapid evidence review with Bayesian meta-analyses (version 7). Addiction. 2021;116(6):1319–1368. doi:10.1111/add.15276
20. Farsalinos K, Angelopoulou A, Alexandris N, Poulas K. COVID-19 and the nicotinic cholinergic system. Eur Respir J. 2020;56(1):2001589. doi:10.1183/13993003.01589-2020
21. Leung JM, Sin DD. Smoking, ACE-2, and COVID-19: ongoing controversies. Eur Respir J. 2020;56:2001759. doi:10.1183/13993003.01759-2020
22. Liu W, Tao ZW, Wang L, et al. Analysis of factors associated with disease outcomes in hospitalized patients with 2019 novel coronavirus disease. Chin Med J. 2020;133(9):1032–1038. doi:10.1097/cm9.0000000000000775
23. Chen X, Tong J, Xiang J, Hu J. Retrospective study on the epidemiological characteristics of 139 patients with novel coronavirus pneumonia on the effects of severity. Chongqing Med. 2020;3:1–9.
24. Huang C, Wang Y, Li X, et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet. 2020;395(10223):497–506. doi:10.1016/s0140-6736(20)30183-5
25. Killerby ME, Link-Gelles R, Haight SC, et al. Characteristics associated with hospitalization among patients with COVID-19 — metropolitan Atlanta, Georgia, March–April 2020. MMWR Morb Mortal Wkly Rep. 2020;69(25):790–794. doi:10.15585/mmwr.mm6925e1
26. Karanasos A, Aznaouridis K, Latsios G, et al. Impact of smoking status on disease severity and mortality of hospitalized patients with COVID-19 infection: a systematic review and meta-analysis. Nicotine Tob Res. 2020;22(9):1657–1659. doi:10.1093/ntr/ntaa107
27. Zhang H, Ma S, Han T, et al. Association of smoking history with severe and critical outcomes in COVID-19 patients: a systemic review and meta-analysis. Eur J Integr Med. 2021;43:101313. doi:10.1016/j.eujim.2021.101313
28. Omar SM, Musa IR, Salah SE, Elnur MM, Al-Wutayd O, Adam I. High mortality rate in adult COVID-19 inpatients in Eastern Sudan: a Retrospective Study. J Multidiscip Healthc. 2020;13:1887–1893. doi:10.2147/jmdh.S283900
29. Shaikh F, Aldhafferi N, Buker A, et al. Comorbidities and risk factors for severe outcomes in COVID-19 patients in Saudi Arabia: a Retrospective Cohort Study. J Multidiscip Healthc. 2021;14:2169–2183. doi:10.2147/JMDH.S317884
30. Al Kuwari HM, Abdul Rahim HF, Abu-Raddad LJ, et al. Epidemiological investigation of the first 5685 cases of SARS-CoV-2 infection in Qatar, 28 February–18 April 2020. BMJ Open. 2020;10(10):e040428. doi:10.1136/bmjopen-2020-040428
31. Argenziano MG, Bruce SL, Slater CL, et al. Characterization and clinical course of 1000 patients with coronavirus disease 2019 in New York: retrospective case series. BMJ. 2020;369:m1996. doi:10.1136/bmj.m1996
32. Feng Y, Ling Y, Bai T, et al. COVID-19 with different severities: a multicenter study of clinical features. Am J Respir Crit Care Med. 2020;201(11):1380–1388. doi:10.1164/rccm.202002-0445OC
33. Liu K, Chen Y, Lin R, Han K. Clinical features of COVID-19 in elderly patients: a comparison with young and middle-aged patients. J Infect. 2020;80(6):e14–e18. doi:10.1016/j.jinf.2020.03.005
34. Alsofayan YM, Althunayyan SM, Khan AA, Hakawi AM, Assiri AM. Clinical characteristics of COVID-19 in Saudi Arabia: a national retrospective study. J Infect Public Health. 2020;13(7):920–925. doi:10.1016/j.jiph.2020.05.026
35. Alyami MH, Naser AY, Orabi MAA, Alwafi H, Alyami HS. Epidemiology of COVID-19 in the Kingdom of Saudi Arabia: an Ecological Study. Front Public Health. 2020;8(506). doi:10.3389/fpubh.2020.00506
36. Nikolich-Zugich J, Knox KS, Rios CT, Natt B, Bhattacharya D, Fain MJ. SARS-CoV-2 and COVID-19 in older adults: what we may expect regarding pathogenesis, immune responses, and outcomes. GeroScience. 2020;42(2):505–514. doi:10.1007/s11357-020-00186-0
37. Joly BS, Siguret V, Veyradier A. Understanding pathophysiology of hemostasis disorders in critically ill patients with COVID-19. Intensive Care Med. 2020;46(8):1603–1606. doi:10.1007/s00134-020-06088-1
38. AlJishi JM, Alhajjaj AH, Alkhabbaz FL, et al. Clinical characteristics of asymptomatic and symptomatic COVID-19 patients in the Eastern Province of Saudi Arabia. J Infect Public Health. 2021;14(1):6–11. doi:10.1016/j.jiph.2020.11.002
39. Zhou F, Yu T, Du R, et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet. 2020;395(10229):1054–1062. doi:10.1016/s0140-6736(20)30566-3
40. Baradaran A, Ebrahimzadeh MH, Baradaran A, Kachooei AR. Prevalence of comorbidities in COVID-19 patients: a systematic review and meta-analysis. Arch Bone Jt Surg. 2020;8(Suppl1):247–255. doi:10.22038/abjs.2020.47754.2346
41. Khamis F, Al-Zakwani I, Al Naamani H, et al. Clinical characteristics and outcomes of the first 63 adult patients hospitalized with COVID-19: an experience from Oman. J Infect Public Health. 2020;13(7):906–913. doi:10.1016/j.jiph.2020.06.002
42. Khan A, Althunayyan S, Alsofayan Y, et al. Risk factors associated with worse outcomes in COVID-19: a retrospective study in Saudi Arabia. East Mediterr Health J. 2020;26(11):1371–1380. doi:10.26719/emhj.20.130
43. Abohamr SI, Abazid RM, Aldossari MA, et al. Clinical characteristics and in-hospital mortality of COVID-19 adult patients in Saudi Arabia. Saudi Med J. 2020;41(11):1217–1226. doi:10.15537/smj.2020.11.25495
44. Messner B, Bernhard D. Smoking and cardiovascular disease: mechanisms of endothelial dysfunction and early atherogenesis. Arterioscler Thromb Vasc Biol. 2014;34(3):509–515. doi:10.1161/atvbaha.113.300156
45. Zheng Z, Peng F, Xu B, et al. Risk factors of critical & mortal COVID-19 cases: a systematic literature review and meta-analysis. J Infect. 2020;81(2):e16–e25. doi:10.1016/j.jinf.2020.04.021
46. Patanavanich R, Glantz SA. Smoking is associated with COVID-19 progression: a meta-analysis. Nicotine Tob Res. 2020;22(9):1653–1656. doi:10.1093/ntr/ntaa082
47. Almazeedi S, Al-Youha S, Jamal MH, et al. Characteristics, risk factors and outcomes among the first consecutive 1096 patients diagnosed with COVID-19 in Kuwait. EClinicalMedicine. 2020;24:100448. doi:10.1016/j.eclinm.2020.100448
48. Soares RCM, Mattos LR, Raposo LM. Risk factors for hospitalization and mortality due to COVID-19 in Espírito Santo State, Brazil. Am J Trop Med Hyg. 2020;103(3):1184–1190. doi:10.4269/ajtmh.20-0483
49. Zhao Z, Chen A, Hou W, et al. Prediction model and risk scores of ICU admission and mortality in COVID-19. PLoS One. 2020;15(7):e0236618. doi:10.1371/journal.pone.0236618
50. Mohsin FM, Tonmon TT, Nahrin R, et al. Association between smoking and COVID-19 severity: evidence from Bangladesh. J Multidiscip Healthc. 2021;14:1923–1933. doi:10.2147/JMDH.S317603
51. Archie SR, Cucullo L. Cerebrovascular and neurological dysfunction under the threat of COVID-19: is there a comorbid role for smoking and vaping? Int J Mol Sci. 2020;21(11):3916. doi:10.3390/ijms21113916
52. Ujjan ID, Devrajani BR, Ghanghro AA, Shah SZA. The clinical and demographical profile of Coronavirus illness: the tale of Tablighi Jamaat and Zaireen in Quarantine/Isolation center at Sukkur and Hyderabad. Pak J Med Sci. 2020;36(Covid19–s4):S12–s16. doi:10.12669/pjms.36.COVID19-S4.2829
53. Zhao Q, Meng M, Kumar R, et al. The impact of COPD and smoking history on the severity of COVID-19: a systemic review and meta-analysis. J Med Virol. 2020;92(10):1915–1921. doi:10.1002/jmv.25889
54. Leung JM, Yang CX, Tam A, et al. ACE-2 expression in the small airway epithelia of smokers and COPD patients: implications for COVID-19. Eur Respir J. 2020;55(5):2000688. doi:10.1183/13993003.00688-2020
55. Oakes JM, Fuchs RM, Gardner JD, Lazartigues E, Yue X. Nicotine and the renin-angiotensin system. Am J Physiol Regul Integr Comp Physiol. 2018;315(5):R895–r906. doi:10.1152/ajpregu.00099.2018
56. Changeux JP, Amoura Z, Rey FA, Miyara M. A nicotinic hypothesis for Covid-19 with preventive and therapeutic implications. C R Biol. 2020;343(1):33–39. doi:10.5802/crbiol.8
57. Yilin Z, Yandong N, Faguang JJB. Role of angiotensin-converting enzyme (ACE) and ACE2 in a rat model of smoke inhalation induced acute respiratory distress syndrome. Burns. 2015;41(7):1468–1477. doi:10.1016/j.burns.2015.04.010
58. Radzikowska U, Ding M, Tan G, et al. Distribution of ACE2, CD147, CD26, and other SARS-CoV-2 associated molecules in tissues and immune cells in health and in asthma, COPD, obesity, hypertension, and COVID-19 risk factors. Allergy. 2020;75(11):2829–2845. doi:10.1111/all.14429
59. Li B, Yang J, Zhao F, et al. Prevalence and impact of cardiovascular metabolic diseases on COVID-19 in China. Clin Res Cardiol. 2020;109(5):531–538. doi:10.1007/s00392-020-01626-9
60. Sardu C, D’Onofrio N, Balestrieri ML, et al. Outcomes in patients with hyperglycemia affected by COVID-19: can we do more on glycemic control? Diabetes Care. 2020;43(7):1408–1415. doi:10.2337/dc20-0723
61. Asselta R, Paraboschi EM, Mantovani A, Duga S. ACE2 and TMPRSS2 variants and expression as candidates to sex and country differences in COVID-19 severity in Italy. Aging. 2020;12(11):10087–10098. doi:10.18632/aging.103415
62. Saheb Sharif-Askari N, Saheb Sharif-Askari F, Alabed M, et al. Airways expression of SARS-CoV-2 receptor, ACE2, and TMPRSS2 is lower in children than adults and increases with smoking and COPD. Mol Ther - Methods Clin Dev. 2020;18:1–6. doi:10.1016/j.omtm.2020.05.013
63. Cruz T, López-Giraldo A, Noell G, et al. Multi-level immune response network in mild-moderate Chronic Obstructive Pulmonary Disease (COPD). Respir Res. 2019;20(1):152. doi:10.1186/s12931-019-1105-z
64. Kim WJ, Lim JH, Lee JS, Lee S-D, Kim JH, Oh Y-M. Comprehensive analysis of transcriptome sequencing data in the lung tissues of COPD subjects. Int J Genomics. 2015;2015:206937. doi:10.1155/2015/206937
65. McDonough JE, Ahangari F, Li Q, et al. Transcriptional regulatory model of fibrosis progression in the human lung. JCI Insight. 2019;4(22):e131597. doi:10.1172/jci.insight.131597
66. Pardo A, Gibson K, Cisneros J, et al. Up-regulation and profibrotic role of osteopontin in human idiopathic pulmonary fibrosis. PLoS Med. 2005;2(9):e251. doi:10.1371/journal.pmed.0020251
67. Lippi G, Henry BM. Chronic obstructive pulmonary disease is associated with severe coronavirus disease 2019 (COVID-19). Respir Med. 2020;167:105941. doi:10.1016/j.rmed.2020.105941
68. Chen N, Zhou M, Dong X, et al. Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a descriptive study. Lancet. 2020;395(10223):507–513. doi:10.1016/S0140-6736(20)30211-7
69. Kowitt SD, Cornacchione Ross J, Jarman KL, et al. Tobacco quit intentions and behaviors among cigar smokers in the United States in response to COVID-19. Int J Environ Res Public Health. 2020;17(15):5368. doi:10.3390/ijerph17155368
70. Caponnetto P, Inguscio L, Saitta C, Maglia M, Benfatto F, Polosa R. Smoking behavior and psychological dynamics during COVID-19 social distancing and stay-at-home policies: a survey. Health Psychol Res. 2020;8(1):9124. doi:10.4081/hpr.2020.9124
71. Sun Y, Li Y, Bao Y, et al. Brief report: increased addictive internet and substance use behavior during the COVID-19 pandemic in China. Am J Addict. 2020;29(4):268–270. doi:10.1111/ajad.13066
72. Patwardhan P. COVID-19: risk of increase in smoking rates among England’s 6 million smokers and relapse among England’s 11 million ex-smokers. BJGP Open. 2020;4(2):bjgpopen20X101067. doi:10.3399/bjgpopen20X101067
73. Reddy RK, Charles WN, Sklavounos A, Dutt A, Seed PT, Khajuria A. The effect of smoking on COVID-19 severity: a systematic review and meta-analysis. J Med Virol. 2021;93(2):1045–1056. doi:10.1002/jmv.26389
74. Farsalinos K, Barbouni A, Poulas K, Polosa R, Caponnetto P, Niaura R. Current smoking, former smoking, and adverse outcome among hospitalized COVID-19 patients: a systematic review and meta-analysis. Ther Adv Chronic Dis. 2020;11:2040622320935765. doi:10.1177/2040622320935765
75. Smith EW, Smith KA, Maibach HI, Andersson PO, Cleary G, Wilson D. The local side effects of transdermally absorbed nicotine. Skin Pharmacol. 1992;5(2):69–76. doi:10.1159/000211021
76. Berlin I, Thomas D, Le Faou A-L, Cornuz J. COVID-19 and smoking. Nicotine Tob Res. 2020;22(9):1650–1652. doi:10.1093/ntr/ntaa059
77. Alexandris N, Lagoumintzis G, Chasapis CT, et al. Nicotinic cholinergic system and COVID-19: in silico evaluation of nicotinic acetylcholine receptor agonists as potential therapeutic interventions. Toxicol Rep. 2021;8:73–83. doi:10.1016/j.toxrep.2020.12.013
 © 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
