Back to Journals » ClinicoEconomics and Outcomes Research » Volume 12
Impact of Atypical Antipsychotics as Adjunctive Therapy on Psychiatric Cost and Utilization in Patients with Major Depressive Disorder
Authors Yan T, Greene M, Chang E , Houle CR, Tarbox MH, Broder MS
Received 20 September 2019
Accepted for publication 17 January 2020
Published 7 February 2020 Volume 2020:12 Pages 81—89
DOI https://doi.org/10.2147/CEOR.S231824
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Prof. Dr. Dean Smith
Tingjian Yan,1 Mallik Greene,2 Eunice Chang,1 Christy R Houle,3 Marian H Tarbox,1 Michael S Broder1
1Partnership for Health Analytic Research, LLC, Beverly Hills, CA 90212, USA; 2Otsuka Pharmaceutical Development & Commercialization, Inc, Princeton, NJ, 08540, USA; 3Lundbeck, Deerfield, IL 60015, USA
Correspondence: Tingjian Yan
Partnership for Health Analytic Research, LLC, 280 S. Beverly Dr., Suite 404, Beverly Hills, CA 90212, USA
Tel +1 310-858-9555
Email [email protected]
Introduction: Patients with major depressive disorder (MDD) incur high costs, despite established treatment options. Adding an atypical antipsychotic (AAP) to antidepressant therapy has shown to reduce depressive symptoms in MDD, but it remains unclear with which adjunctive AAP to initiate. As economic burden is one factor that can influence treatment selection, this study’s objective was to evaluate the impact of adjunctive AAP choice on psychiatric costs and healthcare utilization in MDD.
Materials and Methods: This retrospective cohort study analyzed de-identified data from: (1) IBM® MarketScan® Commercial (C), Medicare Supplemental (MS), and MarketScan Multi-State Medicaid (M) Databases, and (2) Optum® Clinformatics® Datamart. Adult MDD patients were included if they had: initiated adjunctive AAPs during study identification period (7/1/15-9/30/16 MarketScan C/MS, and Optum; 7/1/15-6/30/16 MarketScan M), and ≥ 12 months of continuous enrollment before (baseline) and after (follow-up) first treatment date. Models included generalized linear models (GLMs) for psychiatric costs (total inpatient and outpatient services, excluding outpatient pharmacy costs), and a two-part model (logistic regression for psychiatric hospitalizations, GLM for psychiatric hospitalization costs among hospitalized patients); models were adjusted for baseline characteristics.
Results: The final study sample consisted of 10,325 patients (7657 aripiprazole, 1219 brexpiprazole, 827 lurasidone, 622 quetiapine). Using brexpiprazole as reference, lurasidone and quetiapine users had $1662 and $3894 higher psychiatric costs, respectively. Psychiatric costs were not statistically significantly different between aripiprazole and brexpiprazole (p> 0.05). Quetiapine users had $15,159 (p< 0.001) higher psychiatric hospitalization costs among those hospitalized, and higher odds of psychiatric hospitalization [2.11 (1.46– 3.04); p< 0.001] compared to brexpiprazole users. No statistically significant differences observed in psychiatric hospitalization risk comparing aripiprazole and lurasidone with brexpiprazole (p> 0.05).
Conclusion: In MDD, brexpiprazole users had significantly lower psychiatric costs than lurasidone and quetiapine users, and significantly lower psychiatric hospitalization risk than quetiapine users. Adjunctive AAP choice may impact subsequent healthcare costs and utilization in MDD.
Keywords: atypical antipsychotics, adjunctive therapy, psychiatric cost, healthcare utilization, major depressive disorder
Introduction
Major depressive disorder (MDD) is one of the most common mental health disorders in the United States (US) and affects approximately 6.7% of all adults in the US.1 Of patients with MDD, 64% have severe impairment, defined as having severe problems with the ability to manage at home and work, and have and maintain relationships and a social life.1,2 MDD is associated with a high economic burden for patients, and those with more severe depression have higher direct and indirect costs and higher healthcare resource utilization.3 Despite the availability of a variety of treatment options, costs appear to be rising. There was a 21.5% increase in total healthcare costs from 2005 to 2010 (from $173.2 billion to $210.5 billion).4
One strategy recommended by clinical treatment guidelines for managing depression not resolved with initial antidepressant treatment is adding an atypical antipsychotic to antidepressant therapy.5 However, clinical trials have not directly compared the efficacy of individual atypical antipsychotics,5–8 making it unclear which adjunctive atypical antipsychotic (AAP) should be used first.
In the absence of comparative trials, evidence from real-world retrospective studies can support clinical decision-making. While a few real-world studies compare different adjunctive AAPs in MDD, these studies have not incorporated all of the newly approved atypical antipsychotics,8,9 such as the serotonin-dopamine activity modulator brexpiprazole. Brexpiprazole was approved in 2015 as an adjunctive therapy for adults in MDD and has been shown to be an effective and well-tolerated treatment.10–12 In the only observational study that compared adjunctive brexpiprazole with lurasidone and quetiapine in MDD, there was no difference in psychiatric costs or hospitalization rates.13 However, this study used 2015–2016 claims data and only followed patients for 6 months. In the current study, we evaluated the impact of adjunctive antipsychotic choice on psychiatric costs and healthcare utilization in a 1-year follow-up time among a larger sample of patients with MDD, using more recent data from multiple large data sources to achieve maximal sample size.
Materials and Methods
Data Sources and Study Design
This retrospective cohort analysis used de-identified administrative claims data from the IBM® MarketScan® Commercial, Medicare Supplemental, and Multi-State Medicaid Databases (MarketScan is a trademark of IBM Corporation in the United States and other countries) as well as Optum® Clinformatics® Datamart. Both databases were analyzed separately, including separate data cleaning and study population cohort identification, but combined for the final analysis.
The MarketScan Commercial and Medicare Supplemental Databases represent health services of more than 43.6 million employees, dependents, and retirees in the United States with primary or Medicare supplemental coverage through privately insured fee-for-service, point-of-service, or capitated health plans. The databases include enrollment information and claims healthcare utilization information (eg, inpatient and outpatient services, and prescription drug claims). The MarketScan Multi-State Medicaid Database contains demographics and healthcare records of more than 44 million Medicaid enrollees from multiple states. Optum data covers over 10 years of patient experience and includes de-identified claims and clinical data from various health plans and healthcare providers for over 150 million people. All data were compliant with the Health Insurance Portability and Accountability Act (HIPAA) of 1996. As this study used de-identified administrative claims data, institutional review board approval was not required.
Sample Selection
Adult patients ≥18 years old with MDD were identified by the presence of at least one inpatient or two outpatient claims for MDD disorders (International Classification of Disease, 9th Revision, Clinical Modification [ICD-9-CM] codes: 296.2x, 296.3x;4,14 or 10th revision [ICD-10-CM] codes: F32.x-F32.5, F32.9, F33.0x-F33.4x) in any diagnosis field of a claim during the study period, which differed between databases: 07/01/2014 to 09/30/2017 for MarketScan Commercial and Medicare Supplemental; 07/01/2014 to 06/30/2017 for MarketScan Medicaid; and 07/01/2014 to 09/30/2017 for Optum (Figure 1).
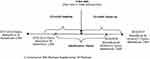 |
Figure 1 Study timeline. Abbreviations: C, commercial; MS, Medicare Supplemental; M, Medicaid. |
Adult patients with MDD who had at least 1 fill of aripiprazole, brexpiprazole, cariprazine, lurasidone, and quetiapine (150–300 mg per day) during the study identification (ID) period (07/01/2015 to 09/30/2016 for MarketScan Commercial and Medicare Supplemental and Optum; 07/01/2015 to 06/30/2016 for MarketScan Medicaid) were included in the analysis with a mutually exclusive cohort for each medication. Quetiapine use was limited to the labeled doses because of it being frequently prescribed off-label for insomnia. Aripiprazole, brexpiprazole, and quetiapine are currently approved by the FDA for treating MDD. Cariprazine and lurasidone were selected based on substantial evidence from a randomized clinical trial supporting use in patients with MDD.15–17 The study index date was defined as the start date of the first prescription fill of the adjunctive AAP (Figure 1). The adjunctive AAP used on the index date was the index therapy. Patients who had used the index therapy in the 12 months before the index date were excluded to ensure at least a 12-month clean period. Also, patients were required to have at least one pharmacy claim for an antidepressant in the 90 days before and the 90 days after the index date, as well as at least 30 days of antidepressant supply overlapping with the index therapy within 90 days after the index date.
Patients were further required to have one diagnosis for MDD during the 12 months before (baseline period) or on the index date, and have continuous enrollment during baseline and the 12 months after the index date (follow-up period).
Patients were excluded if they had at least one diagnosis of schizophrenia, a schizoaffective disorder, schizophreniform, or bipolar disorder anytime during the study period. Due to incomplete data associated with the MarketScan Medicaid database, patients who were Medicare and Medicaid dual eligible, did not have pharmacy coverage or mental health coverage information, or had a capitated plan were also excluded. To prevent including potential duplicate records, patients with the same age, gender, region, insurance type, index date, and index medication found in both the MarketScan and Optum databases were randomly removed from one of the databases.
Study Measures
Baseline variables potentially related to illness severity were examined using data in the 1-year pre-index period. These included patient demographics (age, gender, and insurance); Charlson Comorbidity Index (CCI),18,19 number of Healthcare Cost and Utilization Project (HCUP) chronic conditions,20 and number of psychiatric (anxiety, bipolar disorder, depression, personality disorder, and substance abuse disorder) and non-psychiatric (obesity, type 2 diabetes mellitus, hyperlipidemia, and hypertension) comorbidities; medication use (non-index antipsychotics; psychiatric: antidepressants, anti-anxiety medications, sedatives or hypnotics, and mood stabilizers; non-psychiatric: anti-diabetic medications, lipid-lowering medications, and anti-hypertensive medications); and emergency department (ED) visits and hospitalizations.
The primary outcome measures of interest included psychiatric [with a primary diagnosis of any mental disorder (ICD-9-CM code: 290.xx-311.xx; ICD-10-CM code: F01.xx-F99.xx)] hospitalization and costs (total inpatient and outpatient service costs, as distinct from psychiatric pharmacy costs) during the follow-up period. Inpatient service costs were calculated as the sum of the inpatient claims (e.g., room and board, inpatient pharmacy, and hospitalization services); outpatient service costs were calculated as the sum of the outpatient claims (e.g., office and ED visits, and outpatient hospitalization; excluded psychiatric injectables). The secondary outcomes of interest included psychiatric pharmacy costs and medication adherence. Psychiatric pharmacy costs were calculated as the sum of the claims for psychiatric medications in oral or long-acting injectable (LAI) formulations from the pharmacy or outpatient setting. Medication adherence was measured by the proportion of days covered (PDC), in the follow-up period. PDC was calculated by taking the number of available days of index therapy and dividing by 365.21 Adherence was defined as PDC ≥0.80.
Statistical Analysis
Descriptive, unadjusted analyses were performed to assess differences among cohorts at baseline. Specifically, chi-squared tests were used for categorical variables and one-way analysis of variant or Kruskal–Wallis test was used for continuous variables, depending on the variable distributions.
To examine the association among AAPs and health outcomes, generalized linear models (GLMs) with Gamma distribution and log link function22 were performed for psychiatric costs, given that the distribution of the costs is right-skewed. A two-part model was conducted for psychiatric hospitalization costs, with logistic regression for psychiatric hospitalizations and a GLM for hospitalization costs among hospitalized patients. All models were adjusted for age group, gender, insurance type, CCI, MDD with psychosis/psychotic features, any psychiatric comorbidities, hyperlipidemia, hypertension, and any ED visits, hospitalizations, non-index antipsychotic use, psychiatric medication use, and non-psychiatric medication use at baseline. In the absence of a single acceptable measure of MDD severity available in administrative claims data, we used ED and hospital utilization, as well as non-index antipsychotic use as proxies for severity of MDD. Only statistically significant (p<0.05) covariates were presented in the final models.
All costs were adjusted to 2017 US dollars using the medical care component of the Consumer Price Index. All data transformation and statistical analyses were performed using SAS© version 9.4 (SAS Institute, Cary, North Carolina).
Results
Sample Selection
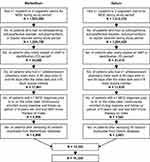
Of the 2,845,172 identified patients with MDD during the study period (1,532,862 MarketScan; 1,312,310 Optum), 51,466 newly started brexpiprazole, quetiapine (150–300 mg per day), lurasidone, aripiprazole, or cariprazine during the ID period; of those patients, 50,817 were on a single adjunctive AAP on the index date. 29,440 patients had at least one antidepressant pharmacy claim each in 90 days before and 90 days after the index date, and at least 15 days of supply overlapping with the first prescription of the index therapy days’ supply. After excluding those without at least one MDD diagnosis before or on the index date, continuous enrollment during baseline and follow-up, or monotherapy, and who were <18 years of age, 10,501 patients remained. Potential duplicates were removed from each database; cariprazine initiators were further excluded due to the small sample size. The final study sample consisted of 10,325 patients with MDD: 7657 aripiprazole, 1219 brexpiprazole, 827 lurasidone, and 622 quetiapine (Figure 2).
Baseline Characteristics
The mean (SD) age of the overall study population was 48.0 (15.8) years. The majority of the MDD patients were female (70.3% overall) and had commercial insurance (77.0%) (Table 1).
 |
Table 1 Baseline Characteristics |
Baseline characteristics (age, gender, insurance, comorbid illness, ER visits, and hospitalization rates) differed across adjunctive AAP users. Compared with other AAP users with MDD, brexpiprazole users were statistically significantly older but had lower ER visits and hospitalization rates at baseline (p<0.001 for all comparisons). Medication use at baseline was common and also differed among AAP users (p<0.001 for all comparisons) (Table 1).
Medication Adherence, Costs, and Healthcare Utilization in the 12-Month Follow-Up
Adherence differed across the adjunctive AAP cohorts (p<0.001), but the overall unadjusted medication adherence was low in this population (%PDC≥0.80=26.8%). Quetiapine users had the highest unadjusted medication adherence (29.6%), and lurasidone users had the lowest (20.7%).
Unadjusted psychiatric costs (excluding pharmacy costs) during the follow-up period were lowest for brexpiprazole with mean (SD) of $3371 (10,708) compared with $3871 (13,937) for aripiprazole, $6224 (20,656) for lurasidone, and $10,670 ($37,689) for quetiapine. These differences were primarily driven by brexpiprazole users having the lowest mean (SD) hospitalization costs [$763 (6317)] compared with the other adjunctive AAP users. While brexpiprazole had the highest psychiatric pharmacy costs [$8961 (6104)] among the various AAP users, lurasidone users had the highest total psychiatric costs [$14,196 (21,541)] (Table 2).
 |
Table 2 Unadjusted Psychiatric Costs (Adjusted to Y2017 Dollar) During the 12-Month Follow-Up |
After controlling for baseline differences, the adjusted psychiatric costs (excluding pharmacy costs) differed significantly across the cohorts (p<0.001) (Table 3). When compared with brexpiprazole users, the psychiatric costs were significantly higher in lurasidone users ($1662; p<0.001) and quetiapine users ($3894; p<0.001), but did not differ between aripiprazole and brexpiprazole users ($91; p=0.563). The adjusted psychiatric pharmacy costs were significantly higher in brexpiprazole users compared to the other AAPs (Table 3). Among hospitalized patients, the adjusted psychiatric hospitalization costs were $15,159 (p<0.001) higher in quetiapine users than in brexpiprazole users (Table 3). The odds of psychiatric hospitalization were also significantly higher in quetiapine users than in brexpiprazole users [2.11 (1.46–3.04); p<0.001]. No statistically significant differences were observed in risk of hospitalization comparing aripiprazole and lurasidone users with brexpiprazole users (p>0.05). Across AAPs, the lowest psychiatric hospitalization rates were found in brexpiprazole users (4.3%, p<0.001).
 |
Table 3 Adjusteda Estimates of Psychiatric Healthcare Utilization and Costs |
Discussion
In this large real-world study using data from multiple nationally representative claims databases, we found that adjunctive brexpiprazole was associated with $2170 and $5898 lower psychiatric costs than lurasidone and quetiapine, respectively. Brexpiprazole users had a 4.3% lower hospitalization rate than quetiapine users (4.3% vs 8.6%), and hospitalization costs were $642 lower in brexpiprazole than in quetiapine users. Differences between brexpiprazole and aripiprazole were not statistically significant. The proportion of patients who were adherent to the index AAP was low (%PDC≥0.80=26.8%).
Adding an AAP to antidepressant therapy has shown to be efficacious in reducing depressive symptoms in patients with MDD.6,10,23,24 However, the majority of previous studies comparing the economic impact of selecting one AAP over another used older data.9,25 A recent cost-effectiveness model estimated that brexpiprazole had $866 and $737 lower medical costs compared to quetiapine 150 mg/day and quetiapine 300 mg/day; however, the model’s cost inputs were derived from clinical trial data and limited by follow-up time and lack of head-to-head comparisons of treatments.26 A prior real-world study comparing brexpiprazole with lurasidone and quetiapine during the 6 months following the initiation of these drugs reported no significant differences in psychiatric costs.13 A recent study comparing brexpiprazole and extended-release quetiapine reported a medical cost difference that was slightly less than what our study identified ($2124 vs $3894); however, this study examined all-cause costs and only included the extended-release form of quetiapine.27 Also consistent with our study, quetiapine has previously been shown to have higher medical costs and risk of hospitalization when compared to aripiprazole.9
The current study contributes to the existing literature in several ways. First, the number of patients we were able to examine was much higher than commonly enrolled in clinical trials of adjunctive AAPs for the treatment of MDD.28 Second, unlike the clinical trials which involve in a very restricted study population, this study included a more generalizable and diverse sample of patients with a variety of insurance types. Third, as patients may not respond to AAPs right away,25,29 the follow-up period of 12 months in our study allowed for a longer observation period. Fourth, this was the first study to compare brexpiprazole and aripiprazole. The lack of statistically significant differences between these drugs may be explained by their pharmacologic similarities.
This study has several limitations. First, MDD diagnosis was identified from health insurance claims, which are designed for reimbursement purposes and can contain errors. Second, the study design required patients to have antidepressant therapy before and after initiating an adjunctive AAP, but we could not determine whether antidepressant therapy had been optimized. Information such as symptom relief, an indicator of treatment effectiveness and one of the most significant outcomes for patients with depression,30 could not be measured. We could not address selection bias beyond controlling for observables. However, the large sample size and the inclusion of reliable measures of cost and utilization make health insurance claims data a valuable source of information. Lastly, we did not use propensity score (PS) matching to make the groups more comparable. While PS matching may have theoretical advantages over covariate-adjusted regression (CAR), it is not necessarily superior in a real-world setting. A recent study comparing PS methods with CAR found that matching tended to give less precise estimates in some cases.31 Additionally, our study included multiple treatment groups. Many commonly available PS methods for multiple treatments may lead to biased estimates.32
Conclusions
In this large real-world study, adding adjunctive brexpiprazole to antidepressant therapy was associated with lower psychiatric costs in patients with MDD compared to treatment with lurasidone and quetiapine. Use of adjunctive brexpiprazole was also associated with lower psychiatric hospitalization risk compared to quetiapine users. There are no statistically significant differences were found between brexpiprazole and aripiprazole drug groups. These findings suggest that choice of adjunctive AAP may impact subsequent healthcare costs and utilization in patients with MDD.
Abbreviations
95% CI, 95% confidence interval; AAP, atypical antipsychotic; CAR, covariate adjusted regression; CCI, Charlson comorbidity index; ED, emergency department; GLM, generalized linear model; HCUP, Healthcare Cost and Utilization Project; HIPAA, Health Insurance Portability and Accountability Act; ID, identification; IRR, incidence rate ratio; LAI, long-acting injectable; MDD, major depressive disorder; OR, odds ratio; PDC, proportion of days covered; PS, propensity score; US, United States.
Author Contributions
All authors made substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data; took part in drafting the article or revising it critically for important intellectual content; gave final approval of the version to be published; and agree to be accountable for all aspects of the work.
Funding
This work was supported by Otsuka Pharmaceutical Development & Commercialization, Inc. and Lundbeck.
Disclosure
Dr Tingjian Yan, Dr Eunice Chang, Ms Marian H Tarbox, and Dr Michael S Broder are employees of Partnership for Health Analytic Research, LLC, which was paid by Otsuka Pharmaceutical Development & Commercialization, Inc. to conduct this research. They report other paid works from AstraZeneca, Janssen Pharmaceuticals, and Sage Therapeutics, outside the submitted work. Dr Christy R Houle is an employee of Lundbeck. Dr Mallik Greene was an employee of Otsuka Pharmaceutical Development & Commercialization, Inc, at the time this research was conducted. The authors report no other conflicts of interest in this work.
References
1. The National Institute of Mental Health. Major depression. mental health information - statistics. [Published February 2019]. Available from: https://www.nimh.nih.gov/health/statistics/major-depression.shtml#part_155029.
2. Bose J, Hedden SL, Lipari RN, Park-Lee E. Key Substance Use and Mental Health Indicators in the United States: Results from the 2017 National Survey on Drug Use and Health. Rockville (MD): Center for Behavioral Health Statistics and Quality, Substance Abuse and Mental Health Services Administration; 2018. Available from: https://www.samhsa.gov/data/.
3. Chow W, Doane MJ, Sheehan J, Alphs L, Le H. Economic burden among patients with major depressive disorder: an analysis of healthcare resource use, work productivity, and direct and indirect costs by depression severity. Am J Manag Care. 2019;16:e188–e196.
4. Greenberg PE, Fournier -A-A, Sisitsky T, Pike CT, Kessler RC. The economic burden of adults with major depressive disorder in the United States (2005 and 2010). J Clin Psychiatry. 2015;76(2):155–162. doi:10.4088/JCP.14m09298
5. Patkar AA, Pae C-U. Atypical antipsychotic augmentation strategies in the context of guideline-based care for the treatment of major depressive disorder. CNS Drugs. 2013;27(S1):29–37. doi:10.1007/s40263-012-0031-0
6. Wen XJ, Wang LM, Liu ZL, Huang A, Liu YY, Hu JY. Meta-analysis on the efficacy and tolerability of the augmentation of antidepressants with atypical antipsychotics in patients with major depressive disorder. Braz J Med Biol Res. 2014;47(7):605–616. doi:10.1590/1414-431X20143672
7. Philip NS, Carpenter LL, Tyrka AR, Price LH. Augmentation of antidepressants with atypical antipsychotics: a review of the current literature. J Psychiatr Pract. 2008;14(1):34–44. doi:10.1097/01.pra.0000308493.93003.92
8. McIntyre RS, Weiller E. Real-world determinants of adjunctive antipsychotic prescribing for patients with major depressive disorder and inadequate response to antidepressants: a case review study. Adv Ther. 2015;32(5):429–444. doi:10.1007/s12325-015-0207-3
9. Nadkarni A, Kalsekar Y, Hebden F. Medical costs and utilization in patients with depression treated with adjunctive atypical antipsychotic therapy. Clin Outcomes Res. 2013;5:49–57. doi:10.2147/CEOR.S36526
10. Eaves S, Rey JA. Brexpiprazole (Rexulti): a new monotherapy for schizophrenia and adjunctive therapy for major depressive disorder. P T Peer-Rev J Formul Manag. 2016;41(7):418–422.
11. Al Shirawi MI, Edgar NE, Kennedy SH. Brexpiprazole in the treatment of major depressive disorder. Clin Med Insights Ther. 2017;9:1179559X1773180. doi:10.1177/1179559X17731801
12. Fava M, Okame T, Matsushima Y, Perry P, Weiller E, Baker RA. Switching from inadequate adjunctive or combination treatment options to brexpiprazole adjunctive to antidepressant: an open-label study on the effects on depressive symptoms and cognitive and physical functioning. Int J Neuropsychopharmacol. 2016;20(1):22–30. doi:10.1093/ijnp/pyw087
13. Broder MS, Greene M, Yan T, Chang E, Hartry A, Yermilov I. Medication adherence, health care utilization, and costs in patients with major depressive disorder initiating adjunctive atypical antipsychotic treatment. Clin Ther. 2019;41:221–232. doi:10.1016/j.clinthera.2018.12.005
14. Gauthier G, Guérin A, Zhdanava M, et al. Treatment patterns, healthcare resource utilization, and costs following first-line antidepressant treatment in major depressive disorder: a retrospective US claims database analysis. BMC Psychiatry. 2017;17(1):222. doi:10.1186/s12888-017-1385-0
15. Suppes T, Silva R, Cucchiaro J, et al. Lurasidone for the treatment of major depressive disorder with mixed features: a randomized, double-blind, placebo-controlled study. Am J Psychiatry. 2016;173(4):400–407. doi:10.1176/appi.ajp.2015.15060770
16. Fava M, Durgam S, Earley W, et al. Efficacy of adjunctive low-dose cariprazine in major depressive disorder: a randomized, double-blind, placebo-controlled trial. Int Clin Psychopharmacol. 2018;33(6):312–321. doi:10.1097/YIC.0000000000000235
17. Durgam S, Earley W, Guo H, et al. Efficacy and safety of adjunctive cariprazine in inadequate responders to antidepressants: a randomized, double-blind, placebo-controlled study in adult patients with major depressive disorder. J Clin Psychiatry. 2016;77(03):371–378. doi:10.4088/JCP.15m10070
18. Charlson ME, Pompei P, Ales KL, MacKenzie CR. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis. 1987;40(5):373–383. doi:10.1016/0021-9681(87)90171-8
19. Deyo RA, Cherkin DC, Ciol MA. Adapting a clinical comorbidity index for use with ICD-9-CM administrative databases. J Clin Epidemiol. 1992;45(6):613–619. doi:10.1016/0895-4356(92)90133-8
20. Agency for Healthcare Research and Quality. HCUP chronic condition indicator. healthcare cost and utilization project (HCUP). [Published June 2015]. Available from: www.hcup-us.ahrq.gov/toolssoftware/chronic/chronic.jsp.
21. Nau D. Proportion of Days Covered (PDC) as a Preferred Method of Measuring Medication Adherence. Springfield (VA): Pharmacy Quality Alliance; 2012. Available from: http://www.pqaaliance.org/files/PDCvsMPRfinal.pdf.
22. Deb P, Norton EC. Modeling health care expenditures and use. Annu Rev Public Health. 2018;39(1):489–505. doi:10.1146/annurev-publhealth-040617-013517
23. Waite R. Use of second generation antipsychotics for treatment-resistant major depressive disorder. Ment Health Clin. 2014;4(5):246–256. doi:10.9740/mhc.n207192
24. Spielmans GI, Berman MI, Linardatos E, Rosenlicht NZ, Perry A, Tsai AC. adjunctive atypical antipsychotic treatment for major depressive disorder: a meta-analysis of depression, quality of life, and safety outcomes. Hay PJ, ed. PLoS Med. 2013;10(3):e1001403. doi:10.1371/journal.pmed.1001403
25. Halpern R, Nadkarni A, Kalsekar I, et al. Medical costs and hospitalizations among patients with depression treated with adjunctive atypical antipsychotic therapy: an analysis of health insurance claims data. Ann Pharmacother. 2013;47(7–8):933–945. doi:10.1345/aph.1R622
26. Sussman M, Yu J, Kamat SA, et al. Cost-effectiveness of brexpiprazole adjunctive treatment for major depressive disorder. J Affect Disord. 2017;207:54–62. doi:10.1016/j.jad.2016.09.006
27. Seetasith A, Greene M, Hartry A, Burudpakdee C. Real-world economic outcomes of brexpiprazole and extended-release quetiapine adjunctive use in major depressive disorder. Clin Outcomes Res. 2019;11:741–755. doi:10.2147/CEOR.S220007
28. Komossa K, Depping AM, Gaudchau A, Kissling W, Leucht S. Second-generation antipsychotics for major depressive disorder and dysthymia. In: The Cochrane Collaboration editor, Cochrane Database of Systematic Reviews. Chichester (UK): John Wiley & Sons, Ltd; 2010. doi:10.1002/14651858.CD008121.pub2
29. Seetasith A, Greene M, Hartry A, Burudpakdee C. Changes in healthcare resource use and costs associated with early versus delayed initiation of atypical antipsychotic adjunctive treatment in major depressive disorder. J Med Econ. 2018;21(9):888–901. doi:10.1080/13696998.2018.1484373
30. Halfin A. Depression: the benefits of early and appropriate treatment. Am J Manag Care. 2007;13:S92–S97.
31. Elze MC, Gregson J, Baber U, et al. Comparison of propensity score methods and covariate adjustment. J Am Coll Cardiol. 2017;69(3):345–357. doi:10.1016/j.jacc.2016.10.060
32. Lopez MJ, Gutman R. Estimation of causal effects with multiple treatments: a review and new ideas. Stat Sci. 2017;32(3):432–454. doi:10.1214/17-STS612
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.