Back to Journals » International Journal of General Medicine » Volume 16
Immune Fitness, Migraine, and Headache Complaints in Individuals with Self-Reported Impaired Wound Healing
Authors Balikji J, Mackus M, Garssen J , Hoogbergen MM, Verster JC
Received 19 March 2023
Accepted for publication 25 April 2023
Published 3 June 2023 Volume 2023:16 Pages 2245—2253
DOI https://doi.org/10.2147/IJGM.S413258
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Woon-Man Kung
Jessica Balikji,1 Marlou Mackus,1 Johan Garssen,1,2 Maarten M Hoogbergen,3 Joris C Verster1,4
1Division of Pharmacology, Utrecht Institute for Pharmaceutical Sciences, Utrecht University, Utrecht, 3584 CG, the Netherlands; 2Division of Plastic Surgery, Catharina Ziekenhuis, Eindhoven, 5623 EJ, the Netherlands; 3Global Centre of Excellence Immunology, Nutricia Danone Research, Utrecht, 3584 CT, the Netherlands; 4Centre for Human Psychopharmacology, Swinburne University, Melbourne, VIC, 3122, Australia
Correspondence: Joris C Verster, Tel +31 30 253 6909, Email [email protected]
Background: Having chronic wounds and impaired wound healing are associated with psychological distress. The current study aims to evaluate migraine and headache complaints in young adults with self-reported impaired wound healing.
Methods: A survey was conducted among N=1935 young adults (83.6% women), 18– 30 years old, living in the Netherlands. Wound healing status was verified, immune fitness was assessed using a single-item rating scale, and ID Migraine was completed. In addition, several questions were answered on past year’s headache experiences (including frequency, quantity, type, location, and severity).
Results: In both the control group (p < 0.001) and the IWH group (p = 0.002) immune fitness was significantly lower among those that reported headaches compared to those that reported no headaches. Individuals with self-reported impaired wound healing (IWH) scored significantly higher on the ID Migraine scale, and individuals of the IWH group scored significantly more often positive for migraine (ie, an ID Migraine score ≥ 2). They reported a younger age of onset of experiencing headaches, and significantly more often reported having a beating or pounding headache than the control group. Compared to the control group, the IWH group reported being significantly more limited in their daily activities compared to the control group.
Conclusion: Headaches and migraines are more frequently reported by individuals with self-reported impaired wound healing, and their reported immune fitness is significantly poorer compared to healthy controls. These headache and migraine complaints significantly limit them in their daily activities.
Keywords: headache, migraine, impaired wound healing, wound infection, slow healing wounds, chronic wounds, immune fitness
Introduction
A chronic wound is defined as an interruption in the continuity of the skin and integrity of the tissue that requires a prolonged time (>8 weeks) to heal, does not heal, or recurs.1 The most prevalent forms of chronic wounds (70–90%) are leg ulcers caused by vascular insufficiency,2,3 followed by foot diabetic ulceration.4,5 Chronic wounds are prevalent and constitute an underestimated public health problem: over 8 million Americans suffer from chronic wounds with or without infection, and the economic costs for chronic wound management have been estimated to range from $28 to $31 billion.6 Slow-healing wounds cause disability, decreased productivity, and loss of independence.7,8
The healing of a wound requires proper circulation, immune status, nutrition, and avoidance of negative mechanical forces. In healthy individuals, the wound healing process takes 3–14 days to complete and is classically divided into three overlapping stages: acute inflammation, proliferation, and granulation tissue formation, and tissue remodeling with wound contraction.9–11 During the inflammatory phase, hemostasis and inflammation occur. Neutrophils and macrophages appear on the wound surface to remove necrotic tissue, debris, and bacteria from the wound. A functioning immune system and an adequate release of growth factors are required for this phase of wound healing. In the proliferative phase, fibroblasts proliferate and produce a collagen matrix, and re-epithelization and angiogenesis occur. During the remodeling phase, fibroblasts reorganize the collagen matrix, and wound contraction occurs. This phase lasts until the granulation tissue is replaced by scar tissue. Wounds gain approximately 80% of their final strength in the first 3 weeks of normal wound healing.11 When any of the wound healing process components is compromised, healing may be delayed.
Previous research revealed that self-reported impaired wound healing in young adults was associated with poorer mood, attention deficits, reduced quality of life, and poorer immune fitness.12,13 The psychological distress of having chronic wounds was also shown to be associated with increased susceptibility to experiencing immune-related complaints14 and health issues such gastrointestinal complaints15 or poor sleep and increased levels of experiencing insomnia.16 Given these frequent comorbidities and their potential negative impact on both disease course and treatment compliance,6 it is important to further investigate these factors. The aim of the current article was therefore to investigate the possible relationship between impaired wound healing and migraine headaches.
Migraine is a common headache disorder, with a prevalence of 15% of the world’s population (~1 billion people) and affects women three times more often (~18%) than men (~6%).17,18 The pathophysiology of migraine constitutes the involvement of both vascular and neuronal mechanisms. The visual aura experienced by some patients with migraine arises from cortical spreading depression and the subsequent activation of perivascular nerve afferents. This leads to vasodilatation of and neurogenic inflammation of cranial vessels, which results in throbbing pain.19,20
Most vascular risk factors are related to lower levels of endothelial progenitor cells (EPCs) and endothelial dysfunction.21 EPCs are cell types that derive from bone marrow, circulate in peripheral blood, are capable of proliferation and differentiation into endothelial cells, and play an important role in angiogenesis (forming new blood vessels) in damaged tissues.22,23 Moreover, EPCs maintain the integrity and function of the vascular endothelium, being considered EPCs as a reflection of endothelial repair capacity.24 Furthermore, a loss in the number and function of EPC has also been found in patients with migraine.25 These values decrease even more during headache. Thus, a relationship between migraine and endothelial function has been suggested.26
Previous research has shown that EPCs may contribute to neovascularization during wound healing, limb ischemia,27–29 endothelization of vascular grafts,30,31 and atherosclerosis.32 One significant impairment of ischemic wounds is deficient tissue-level neovascularization.33 Neovascularization is essential for wound healing because it replaces damaged capillaries and re-establishes the supply of oxygen and nutrients. Literature has demonstrated that macro- and microangiopathy have been implicated in the pathogenesis of diabetic foot ulcers.34,35 Furthermore, reduced levels and impaired function of EPCs are found in diabetic patients.36,37 As a result, wound-healing mechanisms are compromised.36–41 Transplantation of EPC has demonstrated promising results in wound healing.42
Although the pathophysiology of migraine is not fully understood, calcitonin gene-related peptide (CGRP) plays a causative role in migraine. For example, increased CGRP plasma levels were shown during migraine attacks,43 and inhibition of CGRP release decreased both plasma levels of CGRP and the severity of migraine symptoms.43,44 Another study demonstrated that intra-venous provocation with CGRP induces migraine attacks in migraine patients.45 The role of CGRP in migraine is modulating nociception and maintaining neurogenic inflammation, which leads to pain sensitization. Despite its involvement in inflammatory processes,46–48 it has also been associated with wound healing processes.49 This is thought to be mediated through its ability to enhance keratinocyte proliferation,50 promote revascularization,51 and to reduce the expression of inflammatory mediators such as tumor necrosis factor-α (TNF-α) and macrophage infiltration.52
Immune fitness refers to the capacity of the body to respond to health challenges (such as infections) by activating an appropriate immune response, essential to maintain health, prevent and resolve disease, and improve quality of life.53 In the current study, immune fitness was assessed with a single-item rating scale ranging from 0 (very poor) to 10 (excellent).54–56
Given that EPCs and CGRP play a crucial role in migraine as well as in wound healing, the present study aimed to evaluate the association between migraine and impaired wound healing. As there are no biomarkers for immune fitness or headache, the study comprised an anonymous online survey, and all data were self-reported. It was hypothesized that migraine contributes to a higher incidence of impaired wound healing, which also results in poorer immune fitness.
Methods
Via Facebook advertisements in the fall of 2016, Dutch university students were recruited to complete an anonymous online survey on food and health. The cross-sectional survey was designed in SurveyMonkey and conducted in the Dutch language. Subjects could participate if they were students between the age of 18 to 30 years old. The study complied with the Declaration of Helsinki and was approved by the Psychology Ethics Committee of the University of Groningen (Approval code: 16072-O). Electronic informed consent was obtained from all subjects.
Subjects indicated whether or not they had experienced wound infections or slow-healing wounds during the past year. If they answered ‘yes’ to either of these two questions they were allocated to the impaired wound healing (IWH) group. The other subjects served as a control group. A single-item rating assessed immune fitness on an 11-point scale that ranged from 0 (very poor) to 10 (excellent).53,54 The test–retest reliability of the scale is 0.85 to 0.89,55,56 and its outcome has been significantly related to various mental and physical health constructs54,57–60 and quality of life.54 ID Migraine was completed to evaluate migraine complaints.61 The ID Migraine consists of three questions, which can be answered with yes (score 1) or no (score 0). The sum score of the three questions is computed. An overall ID Migraine score of ≥2 implies a positive screen for having a migraine (sensitivity of 81%, specificity of 75%).61
Subjects were asked whether or not they had experienced a headache during the past year (yes/no answering format). If they answered affirmative, they completed a series of questions related to their headache. The questions were developed by investigators (M.M. and J.C.V.) to gain more insight into headache complaints. First, the age of onset of experiencing headaches was recorded. Second, it was assessed how often they experience hangovers per month. A third question asked whether or not they had family members with headache complaints (yes/no answering format). A fourth question concerned the location of the headache (left, right, or both left and right). Question 5 (yes/no answering format) assessed the type of headache pain. Subjects could choose (multiple answers possible) between (1) beating, pounding, (2) drilling, (3) stabbing, (4) tension headache (like a tight band around the head), (5) as if a knife is stabbed in the head or eye, and (6) continuously present, uninterrupted. Question 6 (yes/no answering format) concerned the starting time of the headache. As starting time, subjects could choose (multiple answers possible) between (1) I wake up with a headache, (2) during the day, (3) during the night, (4) only on the weekend, (5) before or during menstruation (females only), and (6) around ovulation (females only). Finally, question 7 asked whether subjects could predict the onset of their headache. The answering possibilities to choose from were (1) no, (2) yes, on the same day, (3) yes, 1 day before, (4) yes, 2 days before, and (5) yes, more than 2 days before.
Statistical analyses were conducted with SPSS (IBM Corp. Released in 2013. IBM SPSS Statistics for Windows, Version 29.0. Armonk, NY, USA: IBM Corp.). In case of missing data, subjects were omitted from the corresponding analysis. Data from the IWH group and control group were compared with the Independent-Samples Kruskal–Wallis test. Percentual data were compared with Chi-Squared tests. Differences between groups were considered statistically significant if p < 0.05 (2-sided). Spearman correlations were computed between immune fitness and the overall ID Migraine score, and between immune fitness and the monthly frequency of having headaches. Correlations were considered significant if p < 0.05 (2-sided).
Results
Data from n=1935 subjects (83.6% women) was used for the analysis. A total of 82.0% of them reported having had headaches during the past year. The demographics of the participants are summarized in Table 1. Immune fitness was significantly lower in the IWH group than in the control group (p < 0.001). Other differences between the IWH group and the control group were not significant.
 |
Table 1 Demographics of the IWH Group and the Control Group |
Demographics based on headache status are summarized in Table 2. In both the control group (p < 0.001) and the IWH group (p = 0.002) immune fitness was significantly lower among those that reported headaches compared to those that reported no headaches. Other differences between those with or without past-year headaches were not significant.
 |
Table 2 Demographics According to Headache Status |
For those who reported past year headaches, the characteristics of their headaches are summarized in Table 3. The age of onset of experiencing headaches was significantly younger in the IWH group than in the control group. With regard to the type of headache, the IWH group significantly more often reported having a beating or pounding headache than the control group. No other significant differences were found between the groups regarding the location, type, and starting time of the headache. The control group could significantly more frequently predict the onset of the headache 1 day before its start, whereas the IWH group significantly more frequently could predict the onset of the headache 2 days before its start.
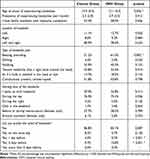 |
Table 3 Characteristics of the Reported Headaches |
Typical migraine complaints are summarized in Table 4. Compared to the control group, the IWH group reported being significantly more frequently bothered by light when having a headache and was significantly more limited in their daily activities compared to the control group. Compared to the control group, overall migraine scores of the IWH group were significantly higher, and individuals of the IWH group scored significantly more often positive for migraine (ie, an ID Migraine score ≥2).
 |
Table 4 ID Migraine Scores |
The correlations between immune fitness and headaches are shown in Figure 1. A significant and negative correlation was found between immune fitness and the overall ID Migraine score (r = −0.203, p < 0.001), and a significant and negative correlation was found between immune fitness and the monthly frequency of having headaches (r = −0.205, p < 0.001).
Discussion
This study demonstrated significant associations between self-reported impaired wound healing, migraine, and immune fitness. The analysis revealed that compared to the control group, headache and migraine were experienced significantly more often by individuals with impaired wound healing. In both the control group and the IWH group, immune fitness was significantly lower among those that reported headaches compared to those that reported no headaches.
Previous studies have shown lower levels of EPC and higher CGRP counts in patients with migraine. Induced inflammation by persistent stimulation of endothelium by CGRP could lead to a progressive decrease of EPC levels as occurs with other chronic diseases.62 This effect might appear with greater intensity during migraine pain attacks, according to the increased plasma levels of CGRP found during headache. However, data on migraine in chronic wound patients is lacking. A literature search revealed only one case study63 about a migraine patient with possible CGRP receptor antibody-related skin wound healing impairment as a systemic side effect of CGRP. This finding supports the notion that migraine patients undergoing CGRP block therapy should be more intensively monitored for impaired wound healing.
A strength of this study is its large sample size. There are, however, several limitations that must be mentioned. First, the convenience sample is not nationally representative. In line with Dutch university demographics, females were overrepresented in the sample. It is unclear to what extent the results obtained in this sample of young adults are representative of other age groups. Also, their health status was self-reported and not confirmed by a formal diagnosis. In general, younger people have better immune fitness compared to older people.64 Given this, the effects observed for the current sample may be more pronounced in older individuals. Future studies in formally diagnosed patients and controls should verify the current findings. It is then important to also collect data on possible diseases and comorbidities related to impaired wound healing (eg, diabetes). Second, because the assessments were self-reported and retrospective, recall bias may have influenced reporting. The self-reports were not confirmed by a physician. Hence, the fact that individuals may have different perceptions of the concepts of wound infection and slow healing wounds (which were not further explained in the survey) may have caused bias. Future studies applying a longitudinal design, including confirmation of assessments by a physician, could minimize this. Third, whereas the ID-Migraine scale is recognized as a valid and reliable screening instrument for migraine,61,65 it must be acknowledged that there are other, more elaborate, questionnaires to assess migraine. Instead of using these, the researchers developed a series of questions to evaluate the nature of experienced headaches. Although the questions are very straightforward, no formal validation study was conducted for these questions. Fourth, immune fitness was assessed via a single-item scale, and this reflects the personal opinion of the individual.53 Future studies could also include assessments of biomarkers of systemic inflammation (eg, blood cytokine concentrations) to further investigate the role of the immune system in the relationship between impaired wound healing and headache. Finally, lifestyle factors, such as nutrition or physical activity, were not considered in the current study. It is important to investigate their role in future studies, as they may play an essential role in both wound healing and headache.66–68
The study has clear implications. Headache and migraine were significantly more frequently reported by individuals with self-reported impaired wound healing, and their reported immune fitness is significantly poorer compared to healthy controls. The associated pain of having wounds is not limited to the location of the wound and may also comprise headache. This implies that it is important to verify headache and migraine complaints in individuals with self-reported impaired wound healing, and if present, to adequately treat these complaints. In addition, it is important to monitor lifestyle factors and their impact on immune fitness, as improving immune fitness may have a direct, positive effect on both wound healing and headache complaints.
In conclusion, headaches and migraine are more frequently reported by individuals with self-reported impaired wound healing, and their reported immune fitness is significantly poorer compared to healthy controls. These headache and migraine complaints significantly limit them in their daily activities. To improve future wound care, an interdisciplinary approach should take into account the increased susceptibility for migraine and headache of individuals with impaired wound healing.
Data Sharing Statement
Data and questionnaire are available from the corresponding author upon reasonable request.
Institutional Review Board Statement
The study complied with the Declaration of Helsinki and ethics approval was obtained from the University of Groningen Psychology Ethics Committee (Approval code: 16072-O).
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study.
Author Contributions
All authors made a significant contribution to the work reported, whether that is in the conception, study design, execution, acquisition of data, analysis and interpretation, or in all these areas; took part in drafting, revising, or critically reviewing the article; gave final approval of the version to be published; have agreed on the journal to which the article has been submitted; and agree to be accountable for all aspects of the work.
Funding
This research received no external funding.
Disclosure
J.G. is a part-time employee of Nutricia Research and has received research grants from Nutricia Research Foundation, Top Institute Pharma, Top Institute Food and Nutrition, GSK, STW, NWO, Friesland Campina, CCC, Raak-Pro, and EU. Over the past 3 years, J.C.V. has acted as a consultant for Eisai, KNMP, Red Bull, Sen-Jam Pharmaceutical, and Toast! The other authors have no potential conflicts of interest to disclose.
References
1. Wysocki AB. Wound fluids and the pathogenesis of chronic wounds. J Wound Ostomy Continence Nurs. 1996;23:283–290. doi:10.1016/s1071-5754(96)90047-9
2. Leung PC. Diabetic foot ulcers: a comprehensive review. Surgeon. 2007;5:219–231. doi:10.1016/S1479-666X(07)80007-2
3. Tam M, Moschella SL. Vascular skin ulcers of limbs. Cardiol Clin. 1991;9:555–563. doi:10.1016/S0733-8651(18)30293-5
4. Sumpio BE, Lee T, Blume PA. Vascular evaluation and arterial reconstruction of the diabetic foot. Clin Podiatr Med Surg. 2003;20:689–708. doi:10.1016/S0891-8422(03)00088-0
5. Wu SC, Driver VR, Wrobel JS, Armstrong DG. Foot ulcers in the diabetic patient: prevention and treatment. Vasc Health Risk Manag. 2007;3:65–76.
6. Sen CK. Human wounds and its burden: an updated compendium of estimates. Adv Wound Caref. 2019;8:39–48. doi:10.1089/wound.2019.0946
7. Rathur HM, Boulton AJ. The diabetic foot. Clin Dermatol. 2007;25:109–120. doi:10.1016/j.clindermatol.2006.09.015
8. Sweitzer SM, Fann SA, Borg TK, Baynes JW, Yost MJ. What is the future of diabetic wound care? Diabetes Educ. 2006;32:197–210. doi:10.1177/0145721706286897
9. Baum CL, Arpey CJ. Normal cutaneous wound healing: clinical correlation with cellular and molecular events. Dermatol Surg. 2005;31:674–686. doi:10.1097/00042728-200506000-00011
10. Goldman R. Growth factors and chronic wound healing: past, present, and future. Adv Skin Wound Care. 2004;17:24–35. doi:10.1097/00129334-200401000-00012
11. Singer AJ, Clark RA. Cutaneous wound healing. N Engl J Med. 1999;341:738–746. doi:10.1056/NEJM199909023411006
12. Balikji J, Hoogbergen MM, Garssen J, Verster JC. Mental resilience, mood, and quality of life in young adults with self-reported impaired wound healing. Int J Environ Res Public Health. 2022;19:2542. doi:10.3390/ijerph19052542
13. Balikji J, Hoogbergen MM, Garssen J, Verster JC. Inattention, impulsivity, and hyperactivity pose individuals with impaired wound healing at increased risk for accidents and injury. Brain Sci. 2022;12:961. doi:10.3390/brainsci12080961
14. Balikji J, Hoogbergen MM, Garssen J, Verster JC. Self-reported impaired wound healing in young adults and their susceptibility to experiencing immune-related complaints. J Clin Med. 2022;11:980. doi:10.3390/jcm11040980
15. Balikji J, Garssen J, Hoogbergen MM, Verster JC. The association of irritable bowel complaints and perceived immune fitness among individuals that report impaired wound healing: supportive evidence for the gut-brain-skin axis. Gastroenterol Insights. 2021;12:423–432. doi:10.3390/gastroent12040040
16. Balikji J, Garssen J, Hoogbergen MM, Roth T, Verster JC. Insomnia complaints and perceived immune fitness in students with and without self-reported impaired wound healing. Medicina. 2022;58:1049. doi:10.3390/medicina58081049
17. Lipton RB, Stewart WF, Diamond S, Diamond ML, Reed M. Prevalence and burden of migraine in the United States: data from the American migraine study II. Headache. 2001;41:646–657. doi:10.1046/j.1526-4610.2001.041007646.x
18. Breslau N, Rasmussen BK. The impact of migraine: epidemiology, risk factors, and co-morbidities. Neurology. 2001;56:S4–S12. doi:10.1212/WNL.56.suppl_1.S4
19. Goadsby PJ. Recent advances in understanding migraine mechanisms, molecules and therapeutics. Trends Mol Med. 2007;13:39–44. doi:10.1016/j.molmed.2006.11.005
20. Silberstein SD. Migraine pathophysiology and its clinical implications. Cephalalgia. 2004;24(suppl 2):2–7. doi:10.1111/j.1468-2982.2004.00892.x
21. Hill JM, Zalos G, Halcox JP, et al. Circulating endothelial progenitor cells, vascular function, and cardiovascular risk. N Engl J Med. 2003;348:593–600. doi:10.1056/NEJMoa022287
22. Asahara T, Murohara T, Sullivan A, et al. Isolation of putative progenitor endothelial cells for angiogenesis. Science. 1997;275:964–967. doi:10.1126/science.275.5302.964
23. Rosenzweig A. Endothelial progenitor cells. New Engl J Med. 2003;348:581–582. doi:10.1056/NEJMp020175
24. George J, Shmilovich H, Deutsch V, Miller H, Keren G, Roth A. Comparative analysis of methods for assessment of circulating endothelial progenitor cells. Tissue Eng. 2006;12:331–335. doi:10.1089/ten.2006.12.331
25. Lee ST, Chu K, Jung KH, et al. Decreased number and function of endothelial progenitor cells in patients with migraine. Neurology. 2008;70:1510–1517. doi:10.1212/01.wnl.0000294329.93565.94
26. Bigal ME, Kurth T, Hu H, Santanello N, Lipton RB. Migraine and cardiovascular disease: possible mechanisms of interaction. Neurology. 2009;72:1864–1871. doi:10.1212/WNL.0b013e3181a71220
27. Kalka C, Masuda H, Takahashi T, et al. Transplantation of ex vivo expanded endothelial progenitor cells for therapeutic neovascularization. Proc Natl Acad Sci USA. 2000;97:3422–3427. doi:10.1073/pnas.97.7.3422
28. Majka SM, Jackson KA, Kienstra KA, Majesky MW, Goodell MA, Hirschi KK. Distinct progenitor populations in skeletal muscle are bone marrow-derived and exhibit different cell fates during vascular regeneration. J Clin Invest. 2003;111:71–79. doi:10.1172/JCI16157
29. Takahashi T, Kalka C, Masuda H, et al. Ischemia- and cytokine-induced mobilization of bone marrow-derived endothelial progenitor cells for neovascularization. Nat Med. 1999;5:434–438. doi:10.1038/7434
30. Kaushal S, Amiel GE, Guleserian KJ, et al. Functional small-diameter neovessels created using endothelial progenitor cells expanded ex vivo. Nat Med. 2001;7:1035–1040. doi:10.1038/nm0901-1035
31. Shi Q, Rafii S, Wu MH, et al. Evidence for circulating bone marrow-derived endothelial cells. Blood. 1998;92:362–367. doi:10.1182/blood.V92.2.362
32. Sata M, Saiura A, Kunisato A, et al. Hematopoietic stem cells differentiate into vascular cells that participate in the pathogenesis of atherosclerosis. Nat Med. 2002;8:403–409. doi:10.1038/nm0402-403
33. Kim HJ, Jang SY, Park JI, et al. Vascular endothelial growth factor-induced angiogenic gene therapy in patients with peripheral artery disease. Exp Mol Med. 2004;36:336–344. doi:10.1038/emm.2004.44
34. La Fontaine J, Harkless LB, Davis CE, Allen MA, Shireman PK. Current concepts in diabetic microvascular dysfunction. J Am Podiatr Med Assoc. 2006;96:245–252. doi:10.7547/0960245
35. Ngo BT, Hayes KD, DiMiao DJ, Srinivasan SK, Huerter CJ, Rendell MS. Manifestations of cutaneous diabetic microangiopathy. Am J Clin Dermatol. 2005;6:225–237. doi:10.2165/00128071-200506040-00003
36. Tepper OM, Galiano RD, Capla JM, et al. Human endothelial progenitor cells from type II diabetics exhibit impaired proliferation, adhesion, and incorporation into vascular structures. Circulation. 2002;106:2781–2786. doi:10.1161/01.CIR.0000039526.42991.93
37. Loomans CJ, de Koning EJ, Staal FJ, et al. Endothelial progenitor cell dysfunction: a novel concept in the pathogenesis of vascular complications of type 1 diabetes. Diabetes. 2004;53:195–199. doi:10.2337/diabetes.53.1.195
38. Fadini GP, Miorin M, Facco M, et al. Circulating endothelial progenitor cells are reduced in peripheral vascular complications of type 2 diabetes mellitus. J Am Coll Cardiol. 2005;45:1449–1457. doi:10.1016/j.jacc.2004.11.067
39. Keswani SG, Katz AB, Lim FY, et al. Adenoviral-mediated gene transfer of PDGF-B enhances wound healing in type I and type II diabetic wounds. Wound Repair Regen. 2004;12:497–504. doi:10.1111/j.1067-1927.2004.12501.x
40. Loomans CJ, De Koning EJ, Staal FJ, Rabelink TJ, Zonneveld AJ. Endothelial progenitor cell dysfunction in type 1 diabetes: another consequence of oxidative stress? Antioxid Redox Signal. 2005;7:1468–1475. doi:10.1089/ars.2005.7.1468
41. Vasa M, Fichtlscherer S, Aicher A, et al. Number and migratory activity of circulating endothelial progenitor cells inversely correlate with risk factors for coronary artery disease. Circ Res. 2001;89:E1–E7. doi:10.1161/hh1301.093953
42. Suh W, Kim KL, Kim JM, et al. Transplantation of endothelial progenitor cells accelerates dermal wound healing with increased recruitment of monocytes/macrophages and neovascularization. Stem Cells. 2005;23:1571–1578. doi:10.1634/stemcells.2004-0340
43. Goadsby PJ, Edvinsson L, Ekman R. Vasoactive peptide release in the extracerebral circulation of humans during migraine headache. Ann Neurol. 1990;28:183–187. doi:10.1002/ana.410280213
44. Edvinsson L, Haanes KA, Warfvinge K, Krause DN. CGRP as the target of new migraine therapies—successful translation from bench to clinic. Nat Rev Neurol. 2018;14:338–350. doi:10.1038/s41582-018-0003-1
45. Lassen LH, Ashina M, Christiansen I, et al. Nitric oxide synthase inhibition: a new principle in the treatment of migraine attacks. Cephalalgia. 1998;18:27–32. doi:10.1046/j.1468-2982.1998.1801027.x
46. Salmona M, Damaj M, Marubio LM, et al. Altered neuroadaptation in opiate dependence and neurogenic inflammatory nociception in alpha CGRP-deficient mice. Nat Neurosci. 2001;4:357–358. doi:10.1038/86001
47. Zhang L, Hoff AO, Wimalawansa SJ, et al. Arthritic calcitonin/α calcitonin gene-related peptide knockout mice have reduced nociceptive hypersensitivity. Pain. 2001;89:265–273. doi:10.1016/S0304-3959(00)00378-X
48. Benschop RJ, Collins EC, Darling RJ, et al. Development of a novel antibody to calcitonin gene-related peptide for the treatment of osteoarthritis-related pain. Osteoarthr Cartil. 2014;22:578–585. doi:10.1016/j.joca.2014.01.009
49. Khalil Z, Helme R. Sensory peptides as Neuromodulators of wound healing in aged rats. J Gerontol Ser a Biol Sci Med Sci. 1996;51A:B354–B361. doi:10.1093/gerona/51A.5.B354
50. Roggenkamp D, Köpnick S, Stäb F, et al. Epidermal nerve fibers modulate Keratinocyte growth via Neuropeptide signaling in an innervated skin model. J Invest Dermatol. 2013;133:1620–1628. doi:10.1038/jid.2012.464
51. Mishima T, Ito Y, Hosono K, et al. Calcitonin gene-related peptide facilitates revascularization during hindlimb ischemia in mice. Am J Physiol Circ Physiol. 2011;300:H431–H439. doi:10.1152/ajpheart.00466.2010
52. Zhang X, Zhuang J, Wu H, et al. Inhibitory effects of Calcitonin gene-related peptides on experimental vein graft disease. Ann Thorac Surg. 2010;90:117–123. doi:10.1016/j.athoracsur.2010.03.063
53. Verster JC, Kraneveld AD, Garssen J. The assessment of immune fitness. J Clin Med. 2023;12:22. doi:10.3390/jcm12010022
54. Van Schrojenstein Lantman M, Otten LS, Mackus M, et al. Mental resilience, perceived immune functioning, and health. J Multidiscip Healthc. 2017;10:107–112. doi:10.2147/JMDH.S130432
55. Wilod Versprille LJ, van de Loo AJ, Mackus M, et al. Development and validation of the Immune Status Questionnaire (ISQ). Int J Environ Res Public Health. 2019;16:4743. doi:10.3390/ijerph16234743
56. Verster JC, Mulder KEW, Hendriksen PA, et al. Test-retest reliability of single-item assessments of immune fitness, mood and quality of life. Heliyon. 2023;9:e15280.
57. Verster JC, Anogeianaki A, Kruisselbrink LD, Alford C, Stock A-K. Relationship of alcohol hangover and physical endurance performance: walking the Samaria Gorge. J Clin Med. 2020;9:E114. doi:10.3390/jcm9010114
58. Verster JC, Arnoldy L, van de Loo AJ, Kraneveld AD, Garssen J, Scholey A. The impact of having a holiday or work in Fiji on perceived immune fitness. Tour Hosp. 2021;2:95–112. doi:10.3390/tourhosp2010006
59. Kiani P, Balikji J, Kraneveld AD, Garssen J, Bruce G, Verster JC. Pandemic preparedness: the importance of adequate immune fitness. J Clin Med. 2022;11:2442. doi:10.3390/jcm11092442
60. Kiani P, Mulder KEW, Balikji J, Kraneveld AD, Garssen J, Verster JC. Pandemic preparedness: maintaining adequate immune fitness by attaining a normal, healthy bodyweight. J Clin Med. 2022;11:3933. doi:10.3390/jcm11143933
61. Lipton RB, Dodick D, Sadovski R, et al. A self-administered screener for migraine in primary care. The ID Migraine™ validation study. Neurology. 2003;61:375–382. doi:10.1212/01.WNL.0000078940.53438.83
62. Mikirova NA, Jackson JA, Hunninghake R, et al. Circulating endothelial progenitor cells: a new approach to anti- aging medicine? J Transl Med. 2009;7:106. doi:10.1186/1479-5876-7-106
63. Wurthmann S, Nägel S, Hadaschik E, et al. Impaired wound healing in a migraine patient as a possible side effect of calcitonin gene-related peptide receptor antibody treatment: a case report. Cephalalgia. 2020;40:1255–1260. doi:10.1177/0333102420933571
64. Pinti M, Appay V, Campisi J, et al. Aging of the immune system: focus on inflammation and vaccination. Eur J Immunol. 2016;46:2286–2301. doi:10.1002/eji.201546178
65. Cousins G, Hijazze S, Van de Laar FA, Fahey T. Diagnostic accuracy of the ID Migraine: a systematic review and meta-analysis. Headache. 2011;51:1140–1148. doi:10.1111/j.1526-4610.2011.01916.x
66. Russell L. The importance of patients’ nutritional status in wound healing. Br J Nurs. 2001;10:S42–S49. doi:10.12968/bjon.2001.10.Sup1.5336
67. Emery CF, Kiecolt-Glaser JK, Glaser R, Malarkey WB, Frid DJ. Exercise accelerates wound healing among healthy older adults: a preliminary investigation. J Gerontol a Biol Sci Med Sci. 2005;60:1432–1436. doi:10.1093/gerona/60.11.1432
68. Seng EK, Martin PR, Houle TT. Lifestyle factors and migraine. Lancet Neurol. 2022;21:911–921. doi:10.1016/S1474-4422(22)00211-3
 © 2023 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2023 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.

