Back to Journals » Cancer Management and Research » Volume 12
Identification of Patients with Brain Metastases with Favorable Prognosis After Local and Distant Recurrence Following Stereotactic Radiosurgery
Authors Jiang X
Received 25 February 2020
Accepted for publication 13 May 2020
Published 3 June 2020 Volume 2020:12 Pages 4139—4149
DOI https://doi.org/10.2147/CMAR.S251285
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Sanjeev K. Srivastava
Xuechao Jiang
Department of Radiation Oncology, Binzhou Center Hospital Affiliated to Binzhou Medical College, Binzhou, Shandong, People’s Republic of China
Correspondence: Xuechao Jiang
Department of Radiation Oncology, Binzhou Center Hospital Affiliated to Binzhou Medical College, Binzhou, Shandong, People’s Republic of China
Tel/Fax +86 543-5325652
Email [email protected]
Purpose: This retrospective study aimed to determine the prognostic factors associated with overall survival after intracranial local and distant recurrence in patients undergoing stereotactic radiosurgery (SRS) for brain metastases.
Patients and Methods: Clinical characteristics and therapeutic parameters of 251 patients, who were treated with initial stereotactic radiosurgery for brain metastases and later experienced intracranial recurrence, were analyzed to identify prognostic factors of post-recurrence overall survival (PROS). A Cox proportional hazard model was applied for univariate and multivariate analyses.
Results: Among the 251 patients, the median post-recurrence overall survival was 8 months, and the six-month PROS rate was 60.2%. The interval from initial radiosurgery treatment to intracranial recurrence (hazard ratio [HR]:0.970), the number of brain recurrent tumors (HR:1.245), the number of extracranial metastatic organs (HR:1.183), recursive partition analysis (RPA) (HR:1.778), and Eastern Cooperative Oncology Group Performance Status (ECOG PS) (HR:2.442) were identified as independent prognostic factors. The patients who received local treatment for solitary brain recurrence achieved better survival (the median survival time after recurrence was 22 months). In patients without extracranial metastasis, the median post-recurrence overall survival of the local treatment group was longer than that in the whole brain radiation therapy (WBRT) group (P< 0.001) and the systemic therapy group (P< 0.001).
Conclusion: A shorter interval from initial stereotactic radiosurgery to recurrence, an increasing number of brain recurrences and extracranial metastatic organs, and poor RPA and ECOG PS values are associated with poor post-recurrence prognosis. When the number of brain recurrent tumors and extracranial metastatic organs was limited, local treatment including stereotactic radiosurgery, surgery or intensity-modulated radiation therapy (IMRT) improved the post-recurrence overall survival.
Keywords: stereotactic radiosurgery, brain metastases, prognostic factors, intracranial recurrence, salvage treatment
Introduction
In recent years, the overall survival of patients with brain metastases has been gradually prolonged, and intracranial local recurrence following stereotactic radiosurgery (SRS) has become an urgent clinical issue.1–4 Overall, 40–70% of new brain metastases occur during follow-up after radiosurgery treatment.5–8 Different types of salvage treatments include local treatment (SRS, surgery, intensity-modulated radiation therapy [IMRT], etc.), whole brain radiation therapy (WBRT), and systemic therapy (chemotherapy, targeted therapy, etc.). The effect of these salvage treatment modalities on patient survival after intracranial recurrence has not drawn much attention and has not been fully elucidated.
In the present study, we analyzed the patients who had undergone radiosurgery treatment for brain metastases and then experienced the first episode of recurrence. The intracranial recurrence patterns, salvage treatment modalities, and prognostic factors associated with survival were analyzed and reported to determine which patients could benefit from appropriate treatment modalities to achieve long-term survival.
Patients and Methods
Patient Cohort
We retrospectively analyzed the data of 604 patients with brain metastases who were treated with CyberKnife (Accuray, Sunnyvale, California) radiosurgery from September 2007 to July 2017 at Tianjin Medical University Cancer Institute and Hospital. All data collected until either death or the last follow-up were assessed. The patients were followed up for the first time 1 month after the initial CyberKnife radiosurgery, and every 3 months thereafter during the first year, and finally re-examined when necessary for a maximum of 6 months. 29 patients were lost to follow-up. In addition, 54 patients survived without intracranial recurrence, and 229 patients showed no brain recurrence before death. These patients received neither local treatment nor whole brain radiotherapy. Systemic treatments, including target therapy and chemotherapy, were provided to control extracranial diseases and the primary disease. At last, a total of 251 patients who developed brain recurrence(s) were included in the study. The present study was conducted in accordance with the institutional ethical guidelines and was approved by the Medical Ethics Committee of Tianjin Medical University Cancer institute and Hospital. In addition, this study was carried out according to the principles expressed in the Declaration of Helsinki. All patients had provided written informed consent for their data to be used for research.
Definition of Recurrence and Treatment Modalities for Recurrent Disease
Intracranial recurrence was diagnosed based on contrast enhanced magnetic resonance imaging (MRI) scans. Local recurrence was defined as the recurrence within a previously irradiated volume while distant recurrence was defined as the brain recurrence outside the previously irradiated volume. First, MRI scan and resonance spectroscopy were performed to distinguish local recurrence from brain radiation necrosis. Then, PET-CT was further carried out if a clear diagnosis could not be made. All patients were evaluated by radiation oncologists and physicists. The date of recurrence was defined as the date of diagnosis of intracranial recurrence. The overall survival after the diagnosis of recurrence was defined as post-recurrence overall survival (PROS). The salvage treatment modalities in this study included local treatment (SRS/surgery/IMRT), WBRT and systemic therapy. The choice of surgery, SRS, IMRT or WBRT was dependent on the patient’s general conditions, the number of brain metastases, and the size of each metastatic lesion. SRS was applied for 1–3 brain metastases no more than 3.5 cm in diameter, and dose selection was consistent with the Radiation Therapy Oncology Group (RTOG) 90–05 guidelines.9 Surgery was mainly applied for local recurrence, which was solitary, or for limited (1–3) distant recurrent tumors more than 3.5 cm in diameter. If the patient showed severe symptom along with surgical indications, resection was also conducted. In this study, resection was performed on 16 local recurrent tumors and 2 distant recurrent tumors. WBRT was generally performed for patients with more than 3 brain metastases. The regimen of the systemic therapy was selected according to the pathology of primary disease and patient conditions. Table 1 shows the clinical baseline characteristics of the patients with intracranial recurrence.
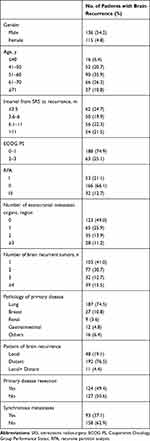 |
Table 1 Characteristics of 251 Patients with Brain Recurrence |
Statistical Analysis
The collected data included age, gender, date of initial stereotactic radiosurgery, pathology of the primary disease, date of diagnosis of intracranial recurrence, the number of brain recurrent tumors, the number of extracranial metastatic organs and follow-up information. The impact of the following factors on PROS was evaluated: age at recurrence, gender, pathology of the primary disease, the interval from initial SRS to recurrence, the number of brain recurrent tumors (0, 1, 2, 3, ≥4), the number of extracranial metastatic organs, Eastern Cooperative Oncology Group Performance Status (ECOG PS), RPA, the pattern of recurrence (local or distant), resection of the primary disease and synchronous brain metastasis. The Chi-square test was used to compare the difference of treatment methods of different groups. Survival curves were calculated using the Kaplan–Meier method and were compared using the Log-rank test. Significant variables of PROS obtained using the univariate analysis were subsequently tested with a multivariate analysis using the Cox proportional hazards model and the forward likelihood ratio method. SPSS software version 20.0 (IBM, Armonk, New York, USA) was used to carry out all statistical calculations. All statistical tests were 2-sided, and differences were considered statistically significant if the P value was less than 0.05.
Results
The median PROS of the 251 patients was 8 months, whereas the three-month post-recurrence overall survival rate was 76.0% and the six-month post-recurrence overall survival rate was 60.2%. The Kaplan-Meier estimates of PROS are shown in Figure 1A.
Prognostic Factors for the PROS of 251 Patients
In the multivariate analysis, the interval from initial SRS to recurrence (P=0.008), the number of brain recurrent tumors (P=0.006), the number of extracranial metastatic organs (P=0.041), RPA (P<0.0001), and ECOG PS (P<0.00001) were shown to be independent prognostic factors of PROS. The results of univariate and multivariate analyses are shown in Table 2. The median PROS was calculated as 10.5 months, 9 months, 8 months, and 3.5 months for patients with 0, 1, 2, and ≥3 regions of extracranial metastases, respectively. The Kaplan-Meier estimates for PROS stratified by the number of extracranial metastatic organs are shown in Figure 1B. The median PROS calculated according to the number of brain recurrent tumors was 10.5 months in patients with 1 lesion, 9 months in patients with 2 lesions, 8 months in patients with 3 lesions and 3 months in patients with ≥4 lesions. Kaplan-Meier estimates for PROS stratified by the number of brain recurrent tumors are shown in Figure 1C. The median PROS was 11 months and 3 months for patients with PS 0–1, PS 2–3, respectively. The Kaplan-Meier estimates for PROS stratified by PS are shown in Figure 1D. The median PROS of patients with RPA I, II, and III was 17 months, 8.5 months and 3 months, respectively. The Kaplan-Meier estimates for PROS stratified by RPA are shown in Figure 1E. We determined median survival (Table 3) according to RPA level updated at initial SRS and at the time of recurrence. Initial RPA level correlated with survival duration after the first course of SRS (P<0.00001).
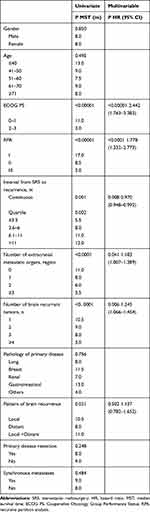 |
Table 2 Prognostic Factors for Post-Recurrence Overall Survival in 251 Patients |
The impact of pathology of the primary disease on PROS could not be demonstrated (P=0.756). In this study, 97 patients received local treatment including SRS or surgery or IMRT, and 40 patients received WBRT. The median PROS was 18.5 months in the local treatment group and 8 months in the WBRT group (Figure 2A). The median PROS was lower in the systemic therapy group (6 months) than that in the local treatment group (Figure 2B).
Factors Affecting the Interval from Initial SRS to Recurrence
The multivariate analysis identified the number of intracranial metastases at the time of initial SRS and age (P<0.001) as the prognostic factors for the interval from SRS to recurrence by multivariate analysis (Table 4). The presence of ≥2 brain metastases at initial SRS was associated with longer time interval to recurrence (P=0.01). As a continuous variable, increasing number of brain metastases at the time of initial SRS was also associated with shorter time interval (P=0.01).
Salvage Treatment for Patients with Single and Multiple Brain Recurrences
To evaluate the effect of the number of brain recurrent tumors on PROS, the difference in the effects of salvage treatment on patients with single tumor lesions and multiple tumor lesions was analyzed (Table 5). There were 103 patients with single tumor lesions and 148 patients with multiple tumor lesions. In the group of single brain metastasis, the number of patients treated with local treatment (SRS/surgery/IMRT) and WBRT was 59 (57.3%) and 38 (34.5%), respectively (P<0.00001). The patients with multiple brain recurrent tumors were more likely to be treated with WBRT than with a local treatment (P<0.003). After local treatment, the median PROS of the patients with solitary tumor was 22 months, and that of the patients with multiple recurrent tumors (≥3) was 12 months (P=0.039).
Salvage Treatment for Patients with or without Extracranial Metastases
A total of 123 (74.9%) patients showed no extracranial metastasis, and 128 patients showed 1 or more extracranial metastatic organs. There was no statistical difference in the application of various salvage treatments between the two groups. In this study, 51 (41.5%) patients showing no extracranial metastasis received local treatments, and the median PROS was longer than that of the patients treated with WBRT (P<0.001) and systemic therapy (P<0.001). Among patients with 1 region of extracranial metastasis, the PROS of those undergoing local treatment was similar to the PROS of those undergoing WBRT (P=0.428), but superior to the PROS of those treated with systemic therapy (P=0.028). Among patients with 2 or more regions of extracranial metastases, there was no statistical difference in the PROS between the local treatment group and the WBRT group (P=0.051), nor was there any statistical difference in the PROS between the local treatment group and the systemic therapy group (P=0.575).
Prognostic Factors for the PROS of Patients with Lung Cancer
Since lung cancer was the main primary disease (74.5%), the prognostic factors for the post-recurrence overall survival of patients with lung cancer were analyzed (Table 6). The median PROS of patients with lung cancer was 8 months. There were 138 cases of non-small cell lung cancer, 21 cases of small cell lung cancer, and 28 cases of unknown pathology. In the univariate analysis, the interval from initial SRS to recurrence, the number of extracranial metastatic organs, the number of brain recurrent tumors, the pattern of recurrence, RPA and ECOG PS were identified as the significant prognostic factors for PROS. In the multivariate analysis, the interval from initial SRS to recurrence (P=0.01), the number of extracranial metastatic organs (P=0.006), the number of brain recurrent tumors (P=0.001), RPA (P<0.001), and ECOG PS (P<0.00001) were identified as significant prognostic factors for PROS. Excluding 28 patients with unknown pathology, the median PROS of patients with non-small cell lung cancer (9 months) was longer than that of patients with small cell lung cancer (5.5 months) (P=0.048).
 |
Table 3 Median Overall Survival in Months After Initial SRS and After Brain Recurrence |
 |
Table 4 Multivariate Cox Proportional Hazards Model for Brain Recurrence After Initial SRS |
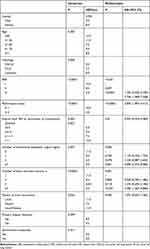 |
Table 6 Prognostic Factors for Post-Recurrence Overall Survival in Patients with Lung Cancer |
Follow-Up
In this study, 87 of the 251 patients (35.1%) suffered neurologic death. 67 (27.5%) patients had stable intracranial lesions and died of systemic causes, and 46 patients (19.1%) died of both uncontrolled cerebral metastases and extracranial progression. The cause of death in 18 patients was undetermined.
Among the 251 patients, 48 patients (19.5%) only showed local recurrences, 192 patients (76.5%) only showed distant recurrences, and 11 patients (4.4%) showed both. The 7 patients with local recurrent tumors were treated with repeat radiosurgery treatment. By the last follow-up, 2 patients survived, 1 died of uncontrolled intracranial tumor and 3 died of extracranial metastasis. No brain radiation necrosis occurred in 6 of the 7 patients, while the condition of the remaining 1 patient was unknown because the patient was lost to follow-up.
Discussion
Previous studies reported that surgery or SRS for oligometastatic diseases helped some patients to achieve long-term survival10–13 due to the following 2 reasons: First, oligometastatic lesions have particular biological characteristics; second, intensive local treatment can prolong disease-free survival.14,15 For patients with brain recurrences following the initial SRS, the salvage local treatment may still promote survival.16 In this study, the number of brain recurrent tumors and the number of extracranial metastatic organs were identified as independent prognostic factors for PROS. The patients with solitary brain recurrence were more likely to receive a local treatment, while the patients with multiple brain recurrent tumors were more likely to receive WBRT. The local treatment could prolong the PROS of patients without extracranial metastasis. In the group of multiple (≥2 regions) extracranial metastatic organs, the patients receiving the local treatment achieved no survival advantage compared with those receiving WBRT. It is suggested that the state of oligometastasis and the local treatment of some patients lead to a better survival. By reducing the disease burden, local treatments may inhibit the immunosuppressive or proangiogenic mechanisms that leading to micrometastatic progression.15 Therefore, if patients have a smaller number of intracranial and extracranial tumor lesions when the intracranial recurrence occurs, local treatments appear to be more reasonable. Otherwise, WBRT or even supportive care alone may be more reasonable given the poor life expectancy of the patients. To apply effective local treatments for brain metastasis, a follow-up system should be established to detect brain recurrence and extracranial metastasis before their progression.
This study showed that PROS was improved with the prolongation of the interval from Cyberknife radiosurgery to intracranial recurrence. The brain metastasis velocity (BMV) score was recently proposed by Farris et al17 and then externally validated18 which showed that a lower BMV score was associated with an improved overall survival following a distant brain failure. This result indicated that the smaller number of new intracranial metastases and a longer intracranial disease-free interval are favorable for survival after intracranial recurrence, which is consistent with the results of this research. In addition, it was shown that the presence of ≥2 initial brain metastases was correlated with a higher BMV, and a higher BMV at the time of the first failure was associated with an increased number of brain metastases at the time of the second failure. These results can help clinicians to decide whether to recommend additional SRS or salvage WBRT. A larger number of intracranial metastases at the time of initial SRS, which was associated with a shorter interval from initial SRS to recurrence in this study, was reported to be a poor prognostic factor for recurrence.19,20 Some studies have reported the number of intracranial tumors was an important predictor of distant brain metastases after SRS.21,22 Patients with multiple brain metastases should be followed up systematically and regularly so that intracranial recurrence can be found in a timely fashion.
The RPA was developed with the goal of predicting the survival of patients with newly diagnosed brain metastases,23 so as guide treatment decisions. Local therapy may be a more attractive treatment option for patients with a longer expected survival. It was found in this study that the prognostic value of RPA in the patient remained high for the multivariate analysis carried out at the time recurrence after initial radiosurgery treatment. As such, an updated RPA level may be a useful tool in selecting patients most likely benefited from local treatments.
This study also had some limitations. Firstly, the pathological heterogeneity produced some deficiencies in the survival analysis. Previous studies have shown that the correlation between survival and the pathology of the primary disease in patients with brain metastases following SRS was controversial,24–30 and the results of comparison among patients with different histopathological results were inconclusive.6,25,27,28 There is no relevant literature reporting the effect of histopathology on the PROS of patients undergoing the initial radiosurgery treatment. According to the results of this study, there was no correlation between the pathology of primary disease and PROS. Since a number of studies have reported the effect of histopathology on the survival of patients with lung cancer,2,19,31,32 the factors related to the PROS in patients with lung cancer were also analyzed in this study. The post-recurrence overall survival of patients with small cell lung cancer was worse than that of patients with non-small cell lung cancer (P=0.048). These results suggested that the effect of histopathology on the survival of patients with lung cancer was also worthy of attention after salvage treatment.
Secondly, only the number of brain metastases was examined in this study, although the size of brain tumor was also reported to be a prognostic factor for patients with brain metastases. Thus, further improvements are necessary for future studies to include more potential prognostic factors.
Thirdly, the follow-up of patients was not performed systematically after the initial stereotactic radiosurgery. Therefore, it remains unknown whether systematic follow-up may promote the early detection of recurrent lesions suitable for local treatments. Because the choice of treatment modalities for recurrent disease was not prospective, the survival evaluation in this study for the treatment of patients with multiple metastases may be biased.
Additionally, several ongoing trials will further our understanding in specific populations. The study (NCT00922974) is a randomized Phase III study evaluating how well image-guided radiosurgery or stereotactic body radiation therapy works and compares it to external-beam radiation therapy in treating patients with localized spine metastasis. NCT00811655 is a randomized phase II study evaluating 6-month local control utilizing pre-operative SRS followed by surgical resection for patients with 1–4 brain metastases.33 The trial (NCT01345539) is a phase II study evaluating feasibility of radiosurgery for all metastatic sites for patients presenting with oligometastatic disease. NCT02898727 is a Phase II study evaluating the treatment of local therapy for the treatment of brain metastases from HER2 positive breast cancer. Together, these efforts and others with intensive follow-up after initial radiosurgery will increase the chance of detection of oligo-recurrence or subclinical recurrence(s).
Conclusion
The interval from the initial stereotactic radiosurgery to recurrence, the number of brain recurrent tumors, the number of extracranial metastatic organs, RPA and ECOG PS status are independent prognostic factors for post-recurrence overall survival. For patients with fewer brain recurrences and extracranial metastases, a local treatment (SRS/surgery/IMRT) can prolong the post-recurrence survival after the initial radiosurgery treatment.
Ethics and Consent
The present study was conducted in accordance with the institutional ethical guidelines and was approved by the Medical Ethics Committee of Tianjin Medical University Cancer Institute and Hospital. Also, this study was carried out according to the principles expressed in the Declaration of Helsinki. All patients had provided written informed consent for their data to be used for research.
Acknowledgments
I thank all the patients who trusted me and all the physicians and staff who helped with this study.
Disclosure
The author declares no potential conflicts of interest in the study.
References
1. Baschnagel AM, Meyer KD, Chen PY, et al. Tumor volume as a predictor of survival and local control in patients with brain metastases treated with gamma knife surgery. J Neurosurg. 2013;119(5):1139–1144. doi:10.3171/2013.7.JNS13431
2. Cho KR, Lee MH, Kong DS, et al. Outcome of gamma knife radiosurgery for metastatic brain tumors derived from non-small cell lung cancer. J Neurooncol. 2015;125(2):331–338. doi:10.1007/s11060-015-1915-x
3. Jensen CA, Chan MD, McCoy TP, et al. Cavity-directed radiosurgery as adjuvant therapy after resection of a brain metastasis. J Neurosurg. 2011;114(6):1585–1591. doi:10.3171/2010.11.JNS10939
4. Lucas JT
5. Hanssens P, Karlsson B, Yeo TT, Chou N, Beute G. Detection of brain micrometastases by high-resolution stereotactic magnetic resonance imaging and its impact on the timing of and risk for distant recurrences. J Neurosurg. 2011;115(3):499–504. doi:10.3171/2011.4.JNS101832
6. Yamamoto M, Serizawa T, Shuto T, et al. Stereotactic radiosurgery for patients with multiple brain metastases (JLGK0901): a multi-institutional prospective observational study. Lancet Oncol. 2014;15(4):387–395. doi:10.1016/S1470-2045(14)70061-0
7. Chang EL, Wefel JS, Hess KR, et al. Neurocognition in patients with brain metastases treated with radiosurgery or radiosurgery plus whole-brain irradiation: a randomised controlled trial. Lancet Oncol. 2009;10(11):1037–1044. doi:10.1016/S1470-2045(09)70263-3
8. Kocher M, Soffietti R, Abacioglu U, et al. Adjuvant whole-brain radiotherapy versus observation after radiosurgery or surgical resection of one to three cerebral metastases: results of the EORTC 22952–26001 study. J Clin Oncol. 2011;29(2):134–141. doi:10.1200/JCO.2010.30.1655
9. Shaw E, Scott C, Souhami L, et al. Single dose radiosurgical treatment of recurrent previously irradiated primary brain tumors and brain metastases: final report of RTOG protocol 90-05. Int J Radiat Oncol Biol Phys. 2000;47(2):291–298. doi:10.1016/S0360-3016(99)00507-6
10. Collen C, Christian N, Schallier D, et al. Phase II study of stereotactic body radiotherapy to primary tumor and metastatic locations in oligometastatic nonsmall-cell lung cancer patients. Ann Oncol. 2014;25(10):1954–1959. doi:10.1093/annonc/mdu370
11. Kawabe T, Phi JH, Yamamoto M, Kim DG, Barfod BE, Urakawa Y. Treatment of brain metastasis from lung cancer. Prog Neurol Surg. 2012;25:148–155.
12. Flannery TW, Suntharalingam M, Regine WF, et al. Long-term survival in patients with synchronous, solitary brain metastasis from non-small-cell lung cancer treated with radiosurgery. Int J Radiat Oncol Biol Phys. 2008;72(1):19–23. doi:10.1016/j.ijrobp.2007.12.031
13. Ashworth A, Rodrigues G, Boldt G, Palma D. Is there an oligometastatic state in non-small cell lung cancer? A systematic review of the literature. Lung Cancer. 2013;82(2):197–203. doi:10.1016/j.lungcan.2013.07.026
14. Gomez DR, Blumenschein GR, Lee JJ, et al. Local consolidative therapy versus maintenance therapy or observation for patients with oligometastatic non-small-cell lung cancer without progression after first-line systemic therapy: a multicentre, randomised, controlled, Phase 2 study. Lancet Oncol. 2016;17(12):1672–1682. doi:10.1016/S1470-2045(16)30532-0
15. Gomez DR, Tang C, Zhang J, et al. Local consolidative therapy vs. maintenance therapy or observation for patients with oligometastatic non-small-cell lung cancer: long-term results of a multi-institutional, Phase II, randomized study. J Clin Oncol. 2019;37(18):1558–1565. doi:10.1200/JCO.19.00201
16. Hafez N, Gettinger S. Oligometastatic disease and local therapies: a medical oncology perspective. Cancer J. 2020;26(2):144–148. doi:10.1097/PPO.0000000000000439
17. Farris M, McTyre ER, Cramer CK, et al. Brain metastasis velocity: a novel prognostic metric predictive of overall survival and freedom from whole-brain radiation therapy after distant brain failure following upfront radiosurgery alone. Int J Radiat Oncol Biol Phys. 2017;98(1):131–141. doi:10.1016/j.ijrobp.2017.01.201
18. McTyre ER, Soike MH, Farris M, et al. Multi-institutional validation of brain metastasis velocity, a recently defined predictor of outcomes following stereotactic radiosurgery. Radiat Oncol. 2020;142:168–174.
19. Kuremsky JG, Urbanic JJ, Petty WJ, et al. Tumor histology predicts patterns of failure and survival in patients with brain metastases from lung cancer treated with gamma knife radiosurgery. Neurosurgery. 2013;73(4):
20. Chung SY, Chang JH, Kim HR, Cho BC, Lee CG, Suh CO. Optimal dose and volume for postoperative radiotherapy in brain oligometastases from lung cancer: a retrospective study. Radiat Oncol J. 2017;35(2):153–162. doi:10.3857/roj.2017.00094
21. Ayala-Peacock DN, Peiffer AM, Lucas JT, et al. A nomogram for predicting distant brain failure in patients treated with gamma knife stereotactic radiosurgery without whole brain radiotherapy. Neuro-Oncology. 2014;16(9):1283–1288. doi:10.1093/neuonc/nou018
22. Rodrigues G, Warner A, Zindler J, Slotman B, Lagerwaard F. A clinical nomogram and recursive partitioning analysis to determine the risk of regional failure after radiosurgery alone for brain metastases. Radiat Oncol. 2014;111(1):52–58. doi:10.1016/j.radonc.2013.11.015
23. Gaspar L, Scott C, Rotman M, et al. Recursive partitioning analysis (RPA) of prognostic factors in three Radiation Therapy Oncology Group (RTOG) brain metastases trials. Int J Radiat Oncol Biol Phys. 1997;37(4):745–751. doi:10.1016/S0360-3016(96)00619-0
24. Kotecha R, Damico N, Miller JA, et al. Three or more courses of stereotactic radiosurgery for patients with multiply recurrent brain metastases. Neurosurgery. 2017;80(6):871–879. doi:10.1093/neuros/nyw147
25. Lima LC, Sharim J, Levin-Epstein R, et al. Hypofractionated stereotactic radiosurgery and radiotherapy to large resection cavity of metastatic brain tumors. World Neurosurg. 2017;97:571–579. doi:10.1016/j.wneu.2016.10.076
26. Rades D, Huttenlocher S, Dziggel L, et al. A new tool to predict survival after radiosurgery alone for newly diagnosed cerebral metastases. Asian Pac J Cancer Prev. 2015;16(7):2967–2970. doi:10.7314/APJCP.2015.16.7.2967
27. Gonda DD, Kim TE, Goetsch SJ, et al. Prognostic factors for stereotactic radiosurgery-treated patients with cerebral metastasis: implications on randomised control trial design and inter-institutional collaboration. Eur J Cancer. 2014;50(6):1148–1158. doi:10.1016/j.ejca.2014.01.001
28. Zindler JD, Rodrigues G, Haasbeek CJ, et al. The clinical utility of prognostic scoring systems in patients with brain metastases treated with radiosurgery. Radiat Oncol. 2013;106(3):370–374. doi:10.1016/j.radonc.2013.01.015
29. Andrews DW, Scott CB, Sperduto PW, et al. Whole brain radiation therapy with or without stereotactic radiosurgery boost for patients with one to three brain metastases: phase III results of the RTOG 9508 randomised trial. Lancet. 2004;363(9422):1665–1672. doi:10.1016/S0140-6736(04)16250-8
30. Pontoriero A, Conti A, Iatì G, et al. Prognostic factors in patients treated with stereotactic image-guided robotic radiosurgery for brain metastases: a single-center retrospective analysis of 223 patients. Neurosurg Rev. 2016;39(3):495–504. doi:10.1007/s10143-016-0718-7
31. Aydemir F, Tufan K, Cekinmez M, et al. Prognostic impact of histologic subtype in non-small cell lung cancer patients treated with gamma knife radiosurgery: retrospective analysis of 104 patients. Turk Neurosurg. 2017;27(1):14–21. doi:10.5137/1019-5149.JTN.14831-15.1
32. Kaul D, Angelidis A, Budach V, Ghadjar P, Kufeld M, Badakhshi H. Prognostic indices in stereotactic radiotherapy of brain metastases of non-small cell lung cancer. Radiat Oncol. 2015;10:244. doi:10.1186/s13014-015-0550-1
33. Huff WX, Agrawal N, Shapiro S, et al. Efficacy of pre-operative stereotactic radiosurgery followed by surgical resection and correlative radiobiological analysis for patients with 1-4 brain metastases: study protocol for a phase II trial. Radiat Oncol. 2018;13(1):252. doi:10.1186/s13014-018-1178-8
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.



