Back to Journals » Cancer Management and Research » Volume 11
HOXA10 deteriorates gastric cancer through activating JAK1/STAT3 signaling pathway
Authors Chen W, Wu G, Zhu Y, Zhang W, Zhang H, Zhou Y, Sun P
Received 12 January 2019
Accepted for publication 29 May 2019
Published 15 July 2019 Volume 2019:11 Pages 6625—6635
DOI https://doi.org/10.2147/CMAR.S201342
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Rituraj Purohit
Wenchao Chen,* Gang Wu,* Yuanzeng Zhu, Wei Zhang, Han Zhang, Yang Zhou, Peichun Sun
Department of Gastrointestinal Surgery, Henan Provincial People’s Hospital, People’s Hospital of Zhengzhou University, School of Clinical Medicine, Henan University, Zhengzhou, Henan 450003, People’s Republic of China
*These authors contributed equally to this work
Background: HOXA10 has been reported to be deregulated in many kinds of cancers including gastric cancer. But its role in gastric cancer progression is controversial. Therefore, the current study was performed to explore the role and mechanism of HOXA10 in gastric cancer.
Materials and methods: IHC and Western blotting assays were used to assess HOXA10 expression in gastric cancer tissues and cells. Lentivirus infection was used to alter HOXA10, STAT3 and JAK1 expression in gastric cancer NCI-N87 and MKN28 cells. MTT, cloning formation, flow cytometry and in vivo xenotransplantation experiments were carried out to assess cell proliferation, cloning formation, apoptosis and tumorigenesis.
Results: HOXA10 expression was obviously increased in gastric cancer tissues and cells when compared with the normal gastric tissue samples and cells. Upregulation of HOXA10 significantly enhanced cell proliferation, cloning formation and tumorigenesis abilities and reduced cell apoptosis in gastric cancer, and promoted the activation of JAK1/STAT3 signaling. In addition, we showed that the effects of HOXA10 on the promotion of cell viability and tumorigenesis and cell apoptosis repression were all weakened when JAK1 or STAT3 was downregulated.
Conclusion: This study demonstrates that HOXA10 functions as an oncogene in gastric cancer through activating JAK1/STAT3 signaling.
Keywords: HOXA10, JAK1/STAT3, viability, apoptosis, tumorigenesis, gastric cancer
Instruction
Gastric cancer is one of the most common malignant cancers with high prevalence in Asia, particularly in China, and it is also considered as one of the leading causes of cancer-related death worldwide.1 Although the development of new inhibitors or agents, such as DNA-binding small molecules,2 immunomodulatory natural product-like compound 1,3 the tumor-triggered drug released from calcium carbonate-encapsulated gold nanostars4 and the substituted quinazoline and quinoxaline derivatives5 have been identified to exert an anti-tumor role, the mechanism of them on cancer treatment may be complex. Nowadays, findings have demonstrated that the dysregulation of multiple genes is strongly implicated in carcinogenesis,6 enlightening that investigations of the molecular mechanisms of gastric cancer may be necessary for new drug development.
HOXA10 is a member of the HOX transcription factor family which is divided into four clusters (cluster A, B, C and D). Apparently, HOXA10 belongs to A cluster. Through regulating gene expression, morphogenesis, embryo viability and hematopoietic lineage commitment, HOXA10 plays important roles in carcinogenesis.7 In detail, HOXA10 has been reported to be frequently overexpressed in human acute myeloid leukemia,8 oral squamous cell carcinoma,9 ovarian clear cell adenocarcinoma10 and prostate cancer,11 and its high expression is closely related to an advanced clinical progress and poor prognosis. However, HOXA10’s role in gastric cancer is conflicting. For instance, Sentani et al12 reported that HOXA10 was positively expressed in about 30% of gastric cancer cases, and it was rarely expressed in the non-neoplastic gastric mucosa except intestinal metaplasia, and its expression was significantly negative correlated with the invasion depth and predicted a better prognosis of gastric cancer patients with the intestinal mucin phenotype. Nevertheless, work by Lim et al13 revealed that the high expression of HOXA10 predicted a poor prognosis in patients with gastric cancer. Therefore, further study should be carried out to explore the function of HOXA10 in gastric cancer.
Janus kinase (JAK)/signal transducer and activator of transcription (STAT) signaling plays crucial role in the regulation of cell proliferation, survival, apoptosis and differentiation.14 Once JAKs are activated, phosphorylated STAT proteins translocate into the nucleus and promotes the activation of target genes including oncogenes and thereby accelerate carcinogenesis.15,16 In gastric cancer, Xiao et al17 recently reported that microRNA-340 promoted the progression of gastric cancer partially depending on the activation of JAK/STAT signaling. Similarly, Chen et al18 revealed that the inhibition of JAK/STAT signaling pathway was strongly implicated in omphalia lapidescens protein-mediated repression of gastric cancer. However, the effect of HOXA10 on the activation of JAK/STAT signaling remains unclear.
The present study was performed with two main objectives, one is to explore the effects of HOXA10 in gastric cancer progression, and the other is to probe whether HOXA10 promotes/inhibits gastric cancer progression via regulating the JAK/STAT signaling.
Materials and methods
Ethic statement
Protocols involving human or animals were, respectively, performed in accordance with the Helsinki Declaration or the NIH Guide for the Care and Use of Laboratory Animals and were approved by the ethical committee of Henan Provincial People’s Hospital.
Tissue samples
Twenty cases of gastric cancer tumor tissue specimens were obtained from patients with primary gastric adenocarcinoma who underwent curative gastrectomy as the primary treatment in our hospital between 2010 and 2015. All tissues were stored at −80°C until further analysis. All patients had signed informed consent before samples collection.
Immunohistochemistry (IHC)
Paraffin-embedded gastric cancer tissues and para-carcinoma tissues with 4 μm in thickness were subjected to IHC. In brief, the tissue sections were deparaffinized and immersed in 3% H2O2 to remove the endogenous peroxidase activity. After antigen retrieval and milk blocking, the sections were incubated with anti-HOXA10 antibody at 1:100 dilution (No. ab191470, Abcam, MA, USA). Then, the sections were incubated with the corresponding secondary antibody (Zhongshanqinqiao, Beijing, China) and counterstained with Chromogen 3,3ʹ-diaminobenzidine tetrachloride (DAB) (Serva, Heidelberg, Germany) at 1:5000 dilution. Cell nucleus was dyed with Harris hematoxylin.
Cell culture
Human normal gastric epithelial cell line GES 1 and gastric cancer cell lines MKN28, SGC-7901 and NCI-N87 were purchased from BeNa Culture Collection (Beijing, China). For cell culture, GES 1 and MKN28 cells were maintained in Dulbecco’s modified Eagle medium (DMEM) with high glucose (Thermo Fisher Scientific, MA, USA), supplemented with 10% fetal bovine serum (FBS; Thermo Fisher Scientific). NCI-N87 and SGC-7901 cells were cultured in RPMI-1640 medium (Thermo Fisher Scientific) with 10% FBS supplement.
Lentivirus obtainment and stable transfection cell line establishment
The lentivirus vector with neomycin resistance used to upregulate HOXA10 expression in gastric cancer cells, referred to OE-HOXA10, and the short hair RNAs (shRNAs) with puromycin resistance used to downregulate HOXA10 (sh-HOXA10) and JAK1 (sh-JAK1), and their negative control (NC) were all synthesized by GenePharma (Shanghai, China).
To establish stable transfection cell lines, 7 μg/mL puromycin or 7 μg/mL puromycin +100 μg/mL G418 were added into the culture medium of NCI-N87 cells transfected with OE-HOXA10 or OE-HOXA10+ sh-JAK1 for 2 weeks, respectively. Then, the survival cells were seeded into 6-well plate to amplify for cloning formation, immunofluorescence staining and xenotransplantation assays.
Real-time quantitative PCR (qPCR) assay
Total RNA was extracted from cells with an RNeasy Mini Kit (Qiagen, Valencia, CA). After quantification, a total of 1 μg RNA was reversely transcripted into cDNA with a First Strand cDNA Synthesis Kit (CWBIO, Jiangsu, China). Then, the mRNA quantitation of HOXA10/JAK1 in gastric cancer cells was determined by qPCR assay with SYBR Green regent (Thermo Fisher Scientific) on an ABI PRISM 7700 Sequence Detection System (Applied Biosystems, Foster City, CA, USA). GAPDH was used as an internal reference to normalize HOXA10/JAK1 expression. Primer sequences are listed as follows:
HOXA10: sense- 5ʹ-AAGGTGAAAACGCAGCCAAC-3ʹ,
HOXA10: antisense-5ʹ-CTAATCTCTAGGCGCCGCTC-3ʹ;
JAK1: sense-5ʹ-GCATCGAGCGCACAAAGTTA-3ʹ,
JAK1: antisense-5ʹ-ACCAGTAGGGTTGAGGGACA-3ʹ;
STAT3: sense-5ʹ-GGAGAAACAGGATGGCCCAA-3ʹ,
STAT3: antisense-5ʹ-AGGCGTGATTCTTCCCACAG-3ʹ;
GAPDH: sense-5ʹ-CCACTAGGCGCTCACTGTTCT-3ʹ,
GAPDH: antisense-5ʹ-GCATCGCCCCACTTGATTTT-3ʹ.
Western blotting assay
To detect the protein expression of HOXA10, JAK1, JAK2, JAK3, Tyk2, STAT3, STAT5B, phospho-STAT3 (p-STAT3), p-JAK1, p-JAK2, p-JAK3, p-Tyk2 and GAPDH, Western blotting assay was performed. The extraction and isolation of total protein from tissues and cells were carried out with the help of RIPA lysis buffer (Beyotime, Jiangsu, China). After incubation with RIPA lysis buffer on ice for 30 mins, the mixture was centrifuged for 20 mins at 4°C at a speed of 1,500 g, and the supernatant was collected. After being quantified with BCA kit (Thermo Fisher Scientific) and degenerated at 100°C for 10 mins, equal amounts of protein from each sample were loaded onto 10% SDS-PAGE, followed by immunoblotting using the indicated primary antibodies overnight at 4°C. Subsequently, the membranes were incubated with secondary antibodies for 1 hr at room temperature. The blot bands were visualized with an enhanced chemiluminescence regent (ECL; Millipore, Billerica, MA, USA) and detected by the gel imaging instrument (Eberhardzell, Germany). And ImageJ software was used to quantify protein level. Primary antibodies used in this experiment are as follows: HOXA10 (1:1,000 dilution; No. ab191470; Abcam), JAK1 (1:1,000 dilution; No. 3332, Cell Signaling Technology, CA, USA), p-JAK1 (1:1,000 dilution; No. 3331, Cell Signaling Technology), JAK2 (1:1,000 dilution; No. 3230, Cell Signaling Technology), p-JAK2 (1:1,000 dilution; No. 3771, Cell Signaling Technology), JAK3 (1:1,000 dilution; No. 3775, Cell Signaling Technology), p-JAK3 (1:1,000 dilution; No. 5031, Cell Signaling Technology), Tyk2 (1:1,000 dilution; No. 9312, Cell Signaling Technology), p-Tyk2 (1:1,000 dilution; No. 9321, Cell Signaling Technology), STAT3 (1:1,000 dilution; No. 9139, Cell Signaling Technology), p-STAT3 (1:1,000 dilution; No. 4113, Cell Signaling Technology), STAT5B (1:1,000 dilution; No. 34,662, Cell Signaling Technology), p-STAT5B (1:1,000 dilution; No. ab52211, Abcam) and GAPDH (1:5,000 dilution; No. TA-08, Zhongshanjinqiao).
MTT assay
Cell proliferation was assessed by MTT assay (Solarbio, Beijing, China) according to the manufactory’s instructions. In detail, 3×103 gastric cancer cells were seeded into 96‑well plates with 200 μL growth medium. Then, the lentivirus vectors were added into cell culture medium which was replaced with fresh complete medium every 2 days. After 1, 2, 3, 4 or 5 days of lentivirus infection, 20 μg of MTT solution was added into cell culture medium and cultured for further 4 hrs at 37°C. Next, 150 μL dimethyl sulfoxide (DMSO) was added to each well and incubated for 10 mins at 37°C The absorbance used to assess cell viability was measured at 570 nm.
Cloning formation assay
Stable transfection cell lines (control, OE-HOXA10 and sh-HOXA10 in both MKN28 and NCI-N87 cells) in logarithmic growth phase were trypsinized and collected. Then, 500 cells from each group were seeded onto a 6-well culture plate and cultured at 37°C for 14 days, with the medium being replaced every 2 days. Then, the culture was terminated and the cells were fixed with 4% paraformaldehyde for 15 mins at room temperature, followed by being stained with 1% crystal violet regent (Solarbio). After being washed with PBS for 5 times, the colony numbers were counted.
Flow cytometry assay
Annexin V (FITC) Apoptosis Detection kit (BD Biosciences, San Diego, CA, USA) was recruited to test cell apoptosis. After 48 hrs of lentivirus infection, NCI-N87 and MKN28 cells were collected with 0.25% of EDTA‑free trypsin (Thermo Fisher Scientific) and washed with PBS for one time. Subsequently, the cells were incubated with 100 μL of 1X binding buffer solution containing 5 μL of Annexin V and 5 μL PI solution for 15 mins in the dark. Then, the cells were washed with 1X of binding buffer for three times and resuspended with 500 μL of 1X binding buffer. Cell apoptosis was detected with a Beckman FC500 flow cytometer (Beckman Coulter, Inc., Brea, California, USA) and analyzed by FlowJo 7.6 software.
Immunofluorescence staining
Immunofluorescence staining was used to detect the effect of HOXA10 on the subcellular location of STAT3 protein. Briefly, sh-HOXA10 or OE-HOXA10 stably transfected NCI-N87 and MKN28 cells were grown onto small glass coverslips in 24-well plate at 10% confluence. After 24 hrs of culture, the cells were washed with PBS and fixed with 4% paraformaldehyde for 15 mins at room temperature, followed by being permeabilized with 0.25% Triton X-100 for 10 mins and blocked with 5% goat serum diluted in PBS for 1 hr at room temperature. Next, the cells were probed with anti-STAT3 antibody (1:100 dilution; No. 9139, Cell Signaling Technology) overnight at 4°C, and subsequently incubated with IgG AF488 (Thermo Fisher Scientific) in the dark for 1 hr at room temperature. Cell nucleus was dyed with DAPI at 1:10,000 dilution. After being covered with anti-fade mounting regent (Solarbio, Beijing, China), the subcellular location of STAT3 was evaluated under a laser scanning microscope (Leica Microsystems CMS, USA).
Xenotransplantation assay
Four-week-old male athymic BALB/C nude mice purchased from Weitonglihua. Co., Ltd (Beijing, China) were used to detect the effects of HOXA10/JAK1 on cell tumorigenesis. In brief, 5×106 NCI-N87 cells with OE-HOXA10, OE-HOXA10+sh-JAK1, OE-HOXA10+sh-STAT3 or negative vector stable transfection were resuspended in 200 μL PBS and were then injected into the armpit area of nude mice, with 5 mice in each group. Twenty-eight days of post-transplantation, mice were sacrificed and the tumors were collected for weighing.
Statistical analysis
Every experiment in this study was performed ≥3times. Data were indicated as mean±standard deviation (SD). Statistical analyses were carried out by SPSS23.0 software with one-way ANOVA followed by the Tukey’s test for multiple groups and Student’s t-test for two groups. P<0.05 was considered as statistically significant difference.
Results
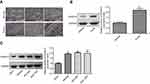
HOXA10 is overexpressed in gastric cancer tissues and cells
To further explore the effects of HOXA10 in the progression of gastric cancer, we first assessed its expression patterns in gastric cancer tissues and adjacent normal tissues using IHC and Western blotting assays. Compared with the normal para-carcinogenesis gastric tissues, HOXA10 protein expression was significantly increased in gastric cancer tissues (Figure 1A–B). To further determine its different expression profiles, we also compared its expression in normal gastric cell line GES 1 and gastric cancer cell lines, including NCI-C87, MKN28 and SGC-7901. Consistently with gastric cancer tissues, HOXA10 protein levels were obviously elevated in gastric cancer cells as compared to that in GES1 cells (Figure 1C). These findings confirmed that HOXA10 was highly expressed in gastric cancer.
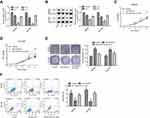
Overexpression of HOXA10 promotes cell viability and inhibits cell apoptosis in gastric cancer
Then, we explored HOXA10 roles in the progression of gastric cancer via carrying out gain-/loss-of-function assays. Transfection with the lentiviral vector OE-HOXA10 significantly increased the expression of HOXA10, and the lentiviral vector sh-HOXA10 RNA-1 used to downregulate HOXA10 showed the highest knockdown efficiency among the 3 shRNAs at both mRNA (Figure 2A) and protein levels (Figure 2B), in both MKN28 and NCI-N87 cells. Compared with OE-NC group, cells in OE-HOXA10 groups showed higher proliferation ability (Figure 2C–D), cloning formation ability (Figure 2E) and lower apoptosis population (Figure 2F), whereas sh-HOXA10 caused the opposite results. These results demonstrated that HOXA10 overexpression accelerated the progression of gastric cancer.
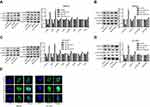
Overexpression of HOXA10 promotes the activation of JAK1/STAT3 signaling in gastric cancer
Next, we explored whether JAK/STAT signaling was involved in HOXA10-induced gastric cancer progression. Western blotting results showed that upregulation of HOXA10 with OE-HOXA10 transient transfection significantly enhanced the expressions of JAK1 and p-JAK1, with JAK2, JAK3, Tyk2 and their phosphorylation levels showed no obvious change, and downregulation of HOXA10 caused the opposite results (Figure 3A and C). In addition, HOXA10 overexpression significantly increased the phosphorylated level of STAT3, with no notable change in the expression levels of STAT3, STAT5B and p-STAT5B in both MKN28 (Figure 3B) and NCI-N87 cells (Figure 3D), and sh-HOXA10 treatment showed the opposite results (Figure 3B and D). Additionally, HOXA10 upregulation significantly enhanced the nuclear accumulation of STAT3 protein, and knockdown of HOXA10 obviously reduced the nuclear accumulation of STAT3 protein (Figure 3E). These discoveries demonstrated that HOXA10 promoted the activation of JAK1/STAT3 signaling in gastric cancer.
Downregulation of either JAK1 or STAT3 abolishes the effects of HOXA10 on the promotion of gastric cancer cells transformation to malignant phenotype
Next, we explored whether HOXA10 promoted gastric cancer progression through activating JAK1/STAT3 pathway. Sh-JAK1 was used to repress JAK1/STAT3 pathway. Among the three shRNAs, sh-2 targeting human JAK1 gene showed the best knockdown efficiency at both mRNA and protein levels (Figure 4A–B), which was chosen for further study. Compared with cells in OE-HOXA10 group, cells in OE-HOXA10+sh-JAK1 group displayed a reduced proliferation (Figure 4C–D) and cloning formation (Figure 4E) abilities and an increased apoptosis rate (Figure 4F), in both MKN28 and NCI-N87 cells.
In addition, we explored the role of STAT3 in HOXA10-mediated gastric cancer progression. The results showed that sh-2-STAT3 significantly reduced the mRNA and protein expression of STAT3 in both MKN28 and NCI-N87 cell lines (Figure 5A–B). Sh-STAT3 transfection significantly reduced the expression of STAT3 and p-STAT3 in MKN28 (Figure 5C) and NCI-N87 cells (Figure 5D). Moreover, downregulation of STAT3 significantly blunted HOXA10 roles in cell proliferation (Figure 5E–F) and clone formation ability (Figure 5G) promotion and cell apoptosis inhibition (Figure 5H) in gastric cancer MKN28 and NCI-N87 cells.
HOXA10 promotes in vivo tumor formation through activating JAK1/STAT3 signaling pathway
Moreover, we also explored the effects of HOXA10/JAK1/STAT3 axis in the tumorigenesis of gastric cancer NCI-N87 cells. The results demonstrated that overexpression of HOXA10 apparently enhanced the tumorigenesis of NCI-N87 cells, whereas this effect was significantly weakened when JAK1 or STAT3 was downregulated (Figure 6). The above results suggested that HOXA10 promoted the progression of gastric cancer via activating JAK1/STAT3 signaling.
Discussion
With the aim of further exploring the molecular mechanisms underlying gastric cancer progression, we probed the role and mechanisms of HOXA10 in gastric cancer. The original results of Western blotting and IHC demonstrated that HOXA10 was upregulated in gastric cancer tissues and cells. Importantly, we reveal that HOXA10 functions as an oncogene in the occurrence and development of gastric cancer through activating JAK1/STAT3 signaling.
Up to now, the function of HOXA10 in carcinogenesis is controversial, as it serves as oncogene in some malignancies and tumor suppressor in others. For example, overexpression of HOXA10 was identified to promote the progression of prostate cancer,11 pancreatic cancer,19 ovarian cancer,20 nasopharyngeal carcinoma21 and neck squamous cell carcinoma.22 However, HOXA10 was reported to be downregulated and inhibited the progression of endometrial cancer23 and breast cancer.24 In gastric cancer, works by Shao,25 Sentani12 and Lim13 all showed that HOXA10 was over-expressed in gastric cancer, but their reports on HOXA10 function were different. For instance, Works by Shao et al25 revealed that HOXA10 together with microRNA-196b-5p (miR-196b-5p) was over-expressed in gastric cancer, and TFF1 reduced the expression of HOXA10 and miR-196b-5p in gastric cancer, leading to the inhibition of cell proliferation and invasion in gastric cancer AGS cells, which indirectly suggests that HOXA10 functions as an oncogene in gastric cancer. Similarly, Han et al26 found that HOXA10 upregulation significantly enhanced the proliferation, migration and invasion of gastric cancer SGC7901 and GGC823 cells. Lim et al13 revealed that HOXA10 high expression predicted a poor prognosis in patients with gastric cancer. However, Sentani et al12 found that knockdown of HOXA10 obviously increased the viability and motility of gastric cancer MKN45 and MKN74 cells. In the present study, through gain-/loss-of-function assays, we demonstrated that upregulation of HOXA10 in gastric cancer NCI-N87 and MKN28 cells significantly enhanced cell proliferation, cloning formation and tumorigenesis abilities, and reduced cell apoptosis, suggesting that HOXC10 functions as an oncogene in gastric cancer, which was consistent with the works by Han et al26 and Shao et al.25 I think that the different cell contents may be responsible for these inconsistent results; hence, more researches should be done to further clarify HOXA10 role in gastric cancer.
It is reported that JAK/STAT signaling is abnormally activated in gastric carcinoma, which then regulates the expression of genes that involve in cell proliferation, apoptosis, migration and invasion.27,28 Accumulated evidence has verified that the hyperactivation of JAK/STAT signaling tends to protect cell from apoptosis and promote cell proliferation in gastric cancer.29,30 AG490, a JAK family-selective inhibitor, was identified to not only suppress the activation of JAK1/STAT3 in H pylori-infected gastric epithelial cells31 but also inhibit the activity of the LEF/β-catenin promoter32 and the downstream genes of β-catenin signaling, such as c-Myc,33 indicating that JAK1/STAT3 repression may mediate the inactivation of β-catenin. Work by Park et al.34 revealed that Wnt might induce the activation of STAT3. Taken together, these findings suggest that there might be a positive crosstalk between JAK/STAT and β-catenin pathways. As HOXA10 was reported to activate β-catenin signaling,35 we assumed that HOXA10 might activate JAK/STAT signaling. To make out this hypothesis, we performed Western blotting to assess the expression of JAK family (JAK1-3 and Tyk2) and results showed that HOXA10 upregulation only increased JAK1 expression and increased its phosphorylation. In addition, we observed that HOXA10 could significantly enhance the expression of p-STAT3 and promote STAT3 nuclear accumulation, suggesting that JAK1/STAT3 might be involved in HOXA10-mediated acceleration of gastric cancer progression. However, the crosstalk between HOXA10, JAK/STAT and β-catenin is not investigated in the present study, and we intend to explore it in our further study.
In mechanism, consistently with our speculate, we found that the downregulation of JAK1 significantly abolished the effects of HOXA10 on the promotion of cell viability and tumorigenesis and the decrease of cell apoptosis, which indicates that HOXA10 promotes the progression of gastric cancer through activating JAK1/STAT3 pathway. And this mechanism was further verified by the compensation experiment, in which we silenced STAT3 and overexpressed HOXA10, and the results showed that HOXA10-mediated STAT3 activation and cell viability enhancement, as well as cell apoptosis repression, were significantly reversed when STAT3 was downregulated in both MKN28 and NCI-N87 cells, which further illustrated that HOXA10 promoted the progression and gastric cancer through activating JAK1/STAT3 signaling. Previous study also explored the mechanism underlying HOXA10 in gastric cancer. For example, Shao et al25 revealed that TFF1 induced the DNA methylation at HOXA10 promoter reduced HOXA10 expression, which then reduced miR-196b-5p expression and repressed the progression of gastric cancer. Han et al26 used ingenuity pathway analysis and Western blotting assay to explore the interaction between HOXA10 and CD44, and preliminarily confirmed that HOXA10 interacted with CD44 via MSN (Moesin), which then accelerated the progression of gastric cancer.
In conclusion, the present study demonstrates that HOXA10 is highly expressed in gastric cancer, and it serves as an oncogene in gastric cancer progression through activating JAK1/STAT3 pathway. Collectively, our study further clarifies the role and mechanism of HOXA10 in gastric cancer, which might provide new means for gastric cancer treatment.
Acknowledgments
This study was supported by the funding from the Basic and frontier Technology Research Project of Henan Province (No. 122300410057 and No. 162300410119), Medical Science and Technology Project of Henan Province (No. 201602203), Natural Science Foundation of Henan Province (No. 162300410292) and the launch fund of Academician Workstation of Henan Province for Non-coding Nucleic Acid Transformation Research.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Torre LA, Bray F, Siegel RL, Ferlay J. Lortet-Tieulent J and Jemal A. Global cancer statistics, 2012. CA Cancer J Clin. 2015;65:87–108. doi:10.3322/caac.21262
2. Leung CH, Chan DS, Ma VP, Ma DL. DNA-binding small molecules as inhibitors of transcription factors. Med Res Rev. 2013;33:823–846. doi:10.1002/med.21266
3. Zhong Z, Liu LJ, Dong ZQ, et al. Structure-based discovery of an immunomodulatory inhibitor of TLR1-TLR2 heterodimerization from a natural product-like database. Chem Commun (Camb). 2015;51:11178–11181. doi:10.1039/c5cc02728d
4. Liu Y, Zhi X, Yang M, et al. Tumor-triggered drug release from calcium carbonate-encapsulated gold nanostars for near-infrared photodynamic/photothermal combination antitumor therapy. Theranostics. 2017;7:1650–1662. doi:10.7150/thno.17602
5. Noolvi MN, Patel HM, Bhardwaj V, Chauhan A. Synthesis and in vitro antitumor activity of substituted quinazoline and quinoxaline derivatives: search for anticancer agent. Eur J Med Chem. 2011;46:2327–2346. doi:10.1016/j.ejmech.2011.03.015
6. Katoh H, Ishikawa S. Genomic pathobiology of gastric carcinoma. Pathol Int. 2017;67:63–71. doi:10.1111/pin.12493
7. Cheng W, Jiang Y, Liu C, Shen O, Tang W, Wang X. Identification of aberrant promoter hypomethylation of HOXA10 in ovarian cancer. J Cancer Res Clin Oncol. 2010;136:1221–1227. doi:10.1007/s00432-010-0772-4
8. Shah CA, Wang H, Bei L, Platanias LC, Eklund EA. HoxA10 regulates transcription of the gene encoding transforming growth factor beta2 (TGFbeta2) in myeloid cells. J Biol Chem. 2011;286:3161–3176. doi:10.1074/jbc.M110.183251
9. Yamatoji M, Kasamatsu A, Yamano Y, et al. State of homeobox A10 expression as a putative prognostic marker for oral squamous cell carcinoma. Oncol Rep. 2010;23:61–67.
10. Li B, Jin H, Yu Y, et al. HOXA10 is overexpressed in human ovarian clear cell adenocarcinoma and correlates with poor survival. Int J Gynecol Cancer. 2009;19:1347–1352. doi:10.1111/IGC.0b013e3181a83f1d
11. Li B, Cao X, Weng C, et al. HoxA10 induces proliferation in human prostate carcinoma PC-3 cell line. Cell Biochem Biophys. 2014;70:1363–1368. doi:10.1007/s12013-014-0065-7
12. Sentani K, Oue N, Naito Y, et al. Upregulation of HOXA10 in gastric cancer with the intestinal mucin phenotype: reduction during tumor progression and favorable prognosis. Carcinogenesis. 2012;33:1081–1088. doi:10.1093/carcin/bgs121
13. Lim JY, Yoon SO, Seol SY, et al. Overexpression of miR-196b and HOXA10 characterize a poor-prognosis gastric cancer subtype. World J Gastroenterol. 2013;19:7078–7088. doi:10.3748/wjg.v19.i41.7078
14. Huang Y, Cen LP, Choy KW, et al. JAK/STAT pathway mediates retinal ganglion cell survival after acute ocular hypertension but not under normal conditions. Exp Eye Res. 2007;85:684–695. doi:10.1016/j.exer.2007.08.003
15. Spano JP, Milano G, Rixe C, Fagard R. JAK/STAT signalling pathway in colorectal cancer: a new biological target with therapeutic implications. Eur J Cancer. 2006;42:2668–2670. doi:10.1016/j.ejca.2006.07.006
16. Mullen M, Gonzalez-Perez RR. Leptin-induced JAK/STAT signaling and cancer growth. Vaccines (Basel). 2016;4. doi:10.3390/vaccines4030026
17. Xiao C, Hong H, Yu H, et al. MiR-340 affects gastric cancer cell proliferation, cycle, and apoptosis through regulating SOCS3/JAK-STAT signaling pathway. Immunopharmacol Immunotoxicol. 2018;40:278–283. doi:10.1080/08923973.2018.1455208
18. Chen L, Lu Z, Yang Y, Du L, Zhou X, Chen Y. Effects of purified omphalia lapidescens protein on metastasis, cell cycle, apoptosis and the JAK-STAT signaling pathway in SGC-7901 human gastric cells. Oncol Lett. 2018;15:4161–4170. doi:10.3892/ol.2018.7830
19. Cui XP, Qin CK, Zhang ZH, et al. HOXA10 promotes cell invasion and MMP-3 expression via TGFbeta2-mediated activation of the p38 MAPK pathway in pancreatic cancer cells. Dig Dis Sci. 2014;59:1442–1451. doi:10.1007/s10620-014-3033-6
20. Liu J, Jiang Y, Wan Y, Zhou S, Thapa S, Cheng W. MicroRNA665 suppresses the growth and migration of ovarian cancer cells by targeting HOXA10. Mol Med Rep. 2018;18:2661–2668. doi:10.3892/mmr.2018.9252
21. Shen ZH, Zhao KM, Du T. HOXA10 promotes nasopharyngeal carcinoma cell proliferation and invasion via inducing the expression of ZIC2. Eur Rev Med Pharmacol Sci. 2017;21:945–952.
22. Guo LM, Ding GF, Xu W, et al. MiR-135a-5p represses proliferation of HNSCC by targeting HOXA10. Cancer Biol Ther. 2018;19:1–28. doi:10.1080/15384047.2018.1450112
23. Zhang L, Wan Y, Jiang Y, et al. Upregulation HOXA10 homeobox gene in endometrial cancer: role in cell cycle regulation. Med Oncol. 2014;31:52. doi:10.1007/s12032-014-0374-0
24. Chen Y, Zhang J, Wang H, et al. miRNA-135a promotes breast cancer cell migration and invasion by targeting HOXA10. BMC Cancer. 2012;12:111. doi:10.1186/1471-2407-12-111
25. Shao L, Chen Z, Peng D, et al. Methylation of the HOXA10 promoter directs miR-196b-5p-dependent cell proliferation and invasion of gastric cancer cells. Mol Cancer Res. 2018;16:696–706. doi:10.1158/1541-7786.MCR-17-0655
26. Han Y, Lu S, Wen YG, et al. Overexpression of HOXA10 promotes gastric cancer cells proliferation and HOXA10(+)/CD44(+) is potential prognostic biomarker for gastric cancer. Eur J Cell Biol. 2015;94:642–652. doi:10.1016/j.ejcb.2015.08.004
27. Liu B, Lu Y, Li J, Liu Y, Liu J, Wang W. Leukemia inhibitory factor promotes tumor growth and metastasis in human osteosarcoma via activating STAT3. Apmis. 2015;123:837–846. doi:10.1111/apm.12427
28. Yeh JE, Frank DA. STAT3-interacting proteins as modulators of transcription factor function: implications to targeted cancer therapy. ChemMedChem. 2016;11:795–801. doi:10.1002/cmdc.201500482
29. Khanna P, Chua PJ, Bay BH, Baeg GH. The JAK/STAT signaling cascade in gastric carcinoma (Review). Int J Oncol. 2015;47:1617–1626. doi:10.3892/ijo.2015.3160
30. Yu RX, Hu XM, Xu SQ, Jiang ZJ, Yang W. Effects of fucoxanthin on proliferation and apoptosis in human gastric adenocarcinoma MGC-803 cells via JAK/STAT signal pathway. Eur J Pharmacol. 2011;657:10–19. doi:10.1016/j.ejphar.2010.12.006
31. Zhang Y, Turkson J, Carter-Su C, et al. Activation of Stat3 in v-Src-transformed fibroblasts requires cooperation of Jak1 kinase activity. J Biol Chem. 2000;275:24935–24944. doi:10.1074/jbc.M002383200
32. Liu YC, Lai WC, Chuang KA, et al. Blockade of JAK2 activity suppressed accumulation of beta-catenin in leukemic cells. J Cell Biochem. 2010;111:402–411. doi:10.1002/jcb.22714
33. Dang CV. c-Myc target genes involved in cell growth, apoptosis, and metabolism. Mol Cell Biol. 1999;19:1–11. doi:10.1128/mcb.19.1.1
34. Park B, Lim JW, Kim H. Lycopene treatment inhibits activation of Jak1/Stat3 and Wnt/beta-catenin signaling and attenuates hyperproliferation in gastric epithelial cells. Nutr Res. 2018. doi:10.1016/j.nutres.2018.07.010
35. Shah CA, Bei L, Wang H, Platanias LC, Eklund EA. HoxA10 protein regulates transcription of gene encoding fibroblast growth factor 2 (FGF2) in myeloid cells. J Biol Chem. 2012;287:18230–18248. doi:10.1074/jbc.M111.328401
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.