Back to Journals » International Journal of Nanomedicine » Volume 15
High UV-Vis-NIR Light-Induced Antibacterial Activity by Heterostructured TiO2-FeS2 Nanocomposites
Authors Mutalik C, Hsiao YC, Chang YH, Krisnawati DI, Alimansur M , Jazidie A, Nuh M, Chang CC, Wang DY, Kuo TR
Received 19 September 2020
Accepted for publication 28 October 2020
Published 12 November 2020 Volume 2020:15 Pages 8911—8920
DOI https://doi.org/10.2147/IJN.S282689
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Farooq A. Shiekh
Chinmaya Mutalik,1,* Yu-Cheng Hsiao,1,2,* Yi-Hsuan Chang,3 Dyah Ika Krisnawati,4 Moh Alimansur,4 Achmad Jazidie,5,6 Mohammad Nuh,7 Chia-Che Chang,8 Di-Yan Wang,8 Tsung-Rong Kuo1,3
1International PhD Program in Biomedical Engineering, College of Biomedical Engineering, Taipei Medical University, Taipei 11031, Taiwan; 2Graduate Institute of Biomedical Optomechatronics, College of Biomedical Engineering, Taipei Medical University, Taipei 11031, Taiwan; 3Graduate Institute of Nanomedicine and Medical Engineering, College of Biomedical Engineering, Taipei Medical University, Taipei 11031, Taiwan; 4Dharma Husada Nursing Academy, Kediri, East Java 64114, Indonesia; 5Department of Electrical Engineering, Institut Teknologi Sepuluh Nopember, Surabaya 60111, Indonesia; 6Universitas Nahdlatul Ulama Surabaya, Surabaya 60111, Indonesia; 7Department of Biomedical Engineering, Institut Teknologi Sepuluh Nopember, Surabaya 60111, Indonesia; 8Department of Chemistry, Tunghai University, Taichung 40704, Taiwan
*These authors contributed equally to this work
Correspondence: Di-Yan Wang
Department of Chemistry, Tunghai University, Taichung 40704, Taiwan
Tel +886-4-23590121
Email [email protected]
Tsung-Rong Kuo
Graduate Institute of Nanomedicine and Medical Engineering, College of Biomedical Engineering, Taipei Medical University, Taipei 11031, Taiwan
Tel +886-2-27361661
Email [email protected]
Purpose: Antibiotic resistance issues associated with microbial pathogenesis are considered to be one of the most serious current threats to health. Fortunately, TiO2, a photoactive semiconductor, was proven to have antibacterial activity and is being widely utilized. However, its use is limited to the short range of absorption wavelength.
Methods: In this work, heterostructured TiO2-FeS2 nanocomposites (NCs) were successfully prepared by a facile solution approach to enhance light-induced antibacterial activity over a broader absorption range.
Results: In TiO2-FeS2 NCs, FeS2 NPs, as light harvesters, can effectively increase light absorption from the visible (Vis) to near-infrared (NIR). Results of light-induced antibacterial activities indicated that TiO2-FeS2 NCs had better antibacterial activity than that of only TiO2 nanoparticles (NPs) or only FeS2 NPs. Reactive oxygen species (ROS) measurements also showed that TiO2-FeS2 NCs produced the highest relative ROS levels. Unlike TiO2 NPs, TiO2-FeS2 NCs, under light irradiation with a 515-nm filter, could absorb light wavelengths longer than 515 nm to generate ROS. In the mechanistic study, we found that TiO2 NPs in TiO2-FeS2 NCs could absorb ultraviolet (UV) light to generate photoinduced electrons and holes for ROS generation, including ⋅O2− and ⋅OH; FeS2 NPs efficiently harvested Vis to NIR light to generate photoinduced electrons, which then were transferred to TiO2 NPs to facilitate ROS generation.
Conclusion: TiO2-FeS2 NCs with superior light-induced antibacterial activity could be a promising antibacterial agent against bacterial infections.
Keywords: antibacterial agent, antibacterial mechanism, reactive oxygen species, light harvester, light-induced antibacterial activity
Introduction
The vigor and resistance of bacterial pestilences are growing day-by-day, and microbial infections are on the rise, creating serious hazards to human health worldwide. Antibiotic-resistant pathogenic infections can soon increase the mortality rate to millions each year.1–5 A photoactive semiconductor, titanium dioxide (TiO2), with economically feasible and biocompatible properties has shown unique antibacterial activity and has been in use for the past few decades.6–8 TiO2 was shown to be an effective antibacterial agent under ultraviolet (UV) light irradiation, by generating reactive oxygen species (ROS) which can cause irreparable damage to the cell envelope of microbes.9–11 The effective generation of radicals such as H2O2, ⋅OH, and ⋅O2− by TiO2 is attributable to its reactivity to light, and it seems to be more efficient when doped or conjugated with other transition elements, noble metals, polymers, carbon, nitrogen, sulfur, or boron to form TiO2 nanocomposites (NCs). TiO2 NCs showed superior antibacterial activities to multidrug-resistant bacteria because of their ability to efficiently absorb light and generation ROS.12–20 The efficient light absorption of TiO2 NCs can be ascribed to contracted bandgaps of TiO2 due to the conjugation of inorganic and organic materials to TiO2.21–32
To extend the absorption range, the NCs of iron disulfide (FeS2) nanoparticles (NPs) conjugated with TiO2 NPs (FeS2-TiO2 NCs) were synthesized. Recently, FeS2-TiO2 NCs were explored as a novel photocatalyst to harvest the light in the region from UV to near-infrared (NIR) for application in energy storage and conversion.33 FeS2 NPs with a small bandgap (~0.95 eV) have been utilized as efficient light harvesters to enhance the absorption from UV to NIR.34 FeS2-TiO2 NCs also exhibited high photoelectric cell (PEC) performance for reducing CO2 to methanol because the bandgap energy of FeS2-TiO2 NCs is narrowed to 1.70 eV for significant enhancement of the photocatalytic performance with visible (Vis) light irradiation.35 Furthermore, TiO2-FeS2 NCs with photocatalytic applications in the UV-Vis-NIR region have shown great potential to be TiO2-based photocatalysts in practical applications for highly active photocatalytic hydrogen evolution.36 However, antibacterial applications with the use of photoactive, biocompatible, and low-cost TiO2-FeS2 NCs are still limited. The most pressing need is to combat wide bacterial infections in fields such as medicine, food, and water quality with the light-activated antibacterial agent of TiO2-FeS2 NCs.
In this work, light-driven catalysts of TiO2-FeS2 NCs were synthesized by a hydrothermal process and annealing method. Characterization studies were carried out to validate the structure and optics of TiO2-FeS2 NCs using scanning electron microscopy (SEM), transmission electron microscopy (TEM), powder x-ray diffraction (XRD), Raman spectroscopy, and UV-Vis-NIR spectrophotometry. Furthermore, antibacterial activities of TiO2-FeS2 NCs were investigated against Escherichia coli (E. coli) under illumination of simulated AM1.5 sunlight and in the dark. To study light-harvesting properties of FeS2 NPs, the antibacterial efficiencies of TiO2-FeS2 NCs were also examined against E. coli under light irradiation with a 515-nm longpass filter. Moreover, to investigate details of the antibacterial mechanism, ROS generation of TiO2-FeS2 NCs against E. coli was measured before and after light irradiation.
Materials and Methods
Chemicals
Titanium dioxide (P25) (Acros), iron(III) nitrate nonahydrate (Fe((NO3))3) (Acros), sulfur (Acros), thioacetamide (TAA) (Acros), N,N-dimethylformamide (DMF) (JT/Macron), ampicillin (Bioshop), glycerol (Honeywell), lysogeny broth (LB) broth miller (Bioshop), LB agar miller (Bioshop), agar-A (Biobasic), 2′,7′-dichlorofluorescin diacetate (DCFH-DA) (Sigma-Aldrich), and Hoechst 33342 (Biotium) were commercially acquired.
Preparation of TiO2-FeS2 NCs Onto Carbon Fiber Paper (CFP)
Fe((NO3))3·9H2O (8 mmol), thioacetamide (100 mmol), and TiO2 (8 mmol, P25) were dissolved in 10 mL DMF and then transferred to a Teflon container. The Teflon container was fixed in an autoclave reactor and placed in a hot-air oven (JOV-40) at 180°C for 18 h. After cooling to room temperature, the solution was centrifuged (Heraeus multifuge X1R, Thermo Scientific) at 4000 rpm for 10 min and washed with ethanol to remove excess organic residues. Afterward, the primary product of FeS2-NCs was collected and dried in the oven. A mixture of the primary product of FeS2-NCs (0.1 g) and sulfur powder (0.3 g) was placed in a furnace (Thermofisher Lindberg Blue M) at 500°C for 1 h to obtain the final product of TiO2-FeS2 NCs. For antibacterial tests, TiO2 NPs (0.4 mg), FeS2 NPs (0.4 mg), and TiO2-FeS2 NCs (0.8 mg) were, respectively, drop-cast onto CFP (CeTech) with dimensions of 1 x 2 cm.
Structural and Optical Characterizations
SEM (JEOL JSM-7800F) and TEM (Hitachi HT-7700) were carried out to characterize the structures of the materials used in the work such as TiO2 NPs, FeS2 NPs, and TiO2-FeS2 NCs. To prepare TEM samples, TiO2 NPs, FeS2 NPs, and TiO2-FeS2 NCs were dispersed in ethanol and then drop-cast onto a copper mesh. After being air-dried, the copper mesh with the materials was used for TEM characterization. XRD studies were carried out using A Rigaku Miniflex 600 with Cu Kα radiation generated at 30 mA and 30 KeV. Scans for XRD were operated from 20° to 70°. Raman data were measured using an Olympus objective Plan N lens at 16 mW. SEM images, TEM images, XRD data, and surface-enhanced Raman spectroscopic (SERS) measurements were used to validate the structural properties of the TiO2 NPs, FeS2 NPs and TiO2-FeS2 NCs. A UV-Vis-NIR absorption spectrometer (Jasco V-770) was used to detect and validate the optical properties of TiO2 NPs, FeS2 NPs, and TiO2-FeS2 NCs.
Antibacterial Activity Test
To culture E. coli, LB medium was prepared by mixing 5 g of LB broth (Miller) and 1000 mL of sterilized water. Escherichia coli (150 µL) was cultured in medium containing of 3 mL of LB medium and 300 µL ampicillin (100 µg/mL) in a shaker at 160 rpm under 37°C for 3 h. The colony-forming unit (CFU) value of the E. coli solution was evaluated by the optical density (OD) at a wavelength of 600 nm (OD600). For E. coli, OD600 of 1.0 is calculated to be 8 × 108 CFU/mL. In this study, a solution of E. coli with an OD600 of 0.1 was utilized for the antibacterial test. For the light-induced antibacterial test, a solar simulator (Enlitech) was used to simulate AM1.5 sunlight. TiO2 NPs, FeS2 NPs, and TiO2-FeS2 NCs drop-cast onto CFP were immersed in E. coli solutions and then TiO2 NPs, FeS2 NPs and TiO2-FeS2 NCs were irradiated for 3 min with simulated AM1.5 sunlight. After light irradiation, E. coli solutions were cultured in a shaker at 160 rpm and 37°C. During culture, the OD600 values of E. coli solutions were measured every 30 min. Bacterial growth curves were used to evaluate the antibacterial activities of TiO2 NPs and TiO2-FeS2 NCs under light irradiation.
Analysis of ROS Generation
ROS generation was measured and validated using a 2ʹ,7ʹ-dichlorodihydrofluorescein diacetate (H2DCFDA) assay. For the H2DCFDA assay, H2DCFDA can be oxidized by ROS into 2ʹ,7ʹ-dichlorofluorescein (DCF). DCF is highly fluorescent and can be measured by fluorescence spectroscopy with excitation/emission at 488/525 nm. The fluorescence intensity of DCF revealed the total amount of ROS production. Furthermore, the dye of Hoechst 33,342 was applied to measure the total amount of bacteria. The fluorescence intensity of Hoechst 33,342 with excitation/emission at 350/461 nm wavelengths showed the total amount of bacteria. For the analysis of ROS generation, in brief, 10 µM of DCFH-DA and 1 µg/mL of Hoechst 33,342 were separately added to E. coli culture medium in a 96-well plate. Afterward, E. coli solutions were incubated at 37°C and 200 rpm for 20 min. After incubation, E. coli solutions were centrifuged at 104 g for 2 min, and the supernatants were removed. Precipitates were suspended in sterilized water. The washing procedures were repeated once. The fluorescence intensities of DCF (excitation/emission wavelength at 488/525 nm) and Hoechst 33342 (excitation/emission wavelength at 350/461 nm) were measured on a microplate reader (Thermo Varioskan Flash). For the different numbers of E. coli in different experiments, the total amount of ROS was normalized to the total bacterial number. The relative ROS level was calculated by normalizing the ROS level between the experimental group and the control group.
Results and Discussion
Morphological Characterization
The morphologies of TiO2 NPs, FeS2 NPs, and TiO2-FeS2 NCs were first characterized by SEM as shown in Figure 1A-C, respectively. In the SEM image of Figure 1A, TiO2 NPs (P25) exhibited a spherical shape and revealed the formation of aggregates. For FeS2 NPs and TiO2-FeS2 NCs, globular aggregates were observed as shown in Figure 1B and C. To further characterize their structures, TiO2 NPs, FeS2 NPs, and TiO2-FeS2 NCs were examined by TEM as, respectively, shown in Figure 1D-F. As shown in the TEM image of Figure 1D, TiO2 NPs clearly revealed a spherical shape with an average size of 25 nm. In Figure 1E, FeS2 NPs exhibited slight aggregates. The blue arrow indicates FeS2 NPs in Figure 1E. Most importantly, as shown in the TEM image of Figure 1F, TiO2-FeS2 NCs exhibited heterostructures composed by TiO2 NPs (red arrow) and FeS2 NPs (blue arrow). High-resolution transmission electron microscopy image of heterostructured TiO2-FeS2 NCs was shown in the supporting information of Figure S1. Moreover, the energy-dispersive X-ray spectroscopy (EDS) was applied to measure weight percentages of FeS2 and TiO2 in TiO2-FeS2 NCs. In the supporting information of Figure S2, EDS spectra showed that weight percentages of FeS2 and TiO2 in TiO2-FeS2 NCs were, respectively, 49.47% and 50.53%. Overall, SEM images, TEM images, and EDS spectra demonstrated the successful preparation of TiO2 NPs, FeS2 NPs and TiO2-FeS2 NCs for subsequent antibacterial applications.
Structural Analysis of TiO2-FeS2 NCs
XRD was utilized to investigate the crystal structure of TiO2-FeS2 NCs. In Figure 2, clear XRD peaks at 25.4°, 37.8°, 48.0°, and 54.5° were accordingly cataloged to the (101), (004), (200) and, (211) planes of the anatase phase of TiO2 (JCPDS 21–1272), and XRD peaks at 27.5° and, 54.4° were accordingly cataloged to the (110) and (211) planes of the rutile phase of TiO2 (JCPDS 21–1276). XRD peaks of FeS2 NPs were matched with approved pyrite FeS2 (JCPDS 42–1340). The principal peaks of FeS2 NPs at 28.5°, 33.1°, 37.1°, 40.8°, 47.4°, and 56.3° were accordingly cataloged to the (111), (200), (210), (211), (220), and (311) planes of pyrite FeS2 as shown in Figure 2. Results of XRD studies also revealed the crystalline nature of TiO2-FeS2 NCs. To sum up, TiO2-FeS2 NCs were composed of TiO2 NPs and FeS2 NPs according to SEM and TEM measurements.
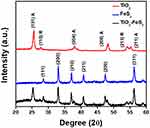 |
Figure 2 XRD spectra of TiO2 NPs, FeS2 NPs, and TiO2-FeS2 NCs. (A: anatase; R: rutile). |
Furthermore, Raman spectra of TiO2 NPs, FeS2 NPs, and TiO2-FeS2 NCs were studied to evaluate and validate their structural properties and confirm the formation of TiO2-FeS2 NCs. As shown in Figure 3, Raman frequencies of TiO2 NPs were 143 (Eg), 395 (B1g), 513 (B1g), and 633 cm−1 (Eg) for the TiO2 anatase phase. For FeS2 NPs, there were two strong peaks located at 338 (Eg) and 377 cm−1 (Ag) corresponding to pyrite FeS2.37 The two weak Raman signals of 220 and 283 cm−1 were attributed to the presence of FeS due to sulfur insufficiency and sulfur vacancies.38,39 For Raman spectra of TiO2-FeS2 NCs, the peak at 143 cm−1 was attributed to the Raman signal of TiO2 NPs (Eg), and the two peaks at 338 and 373 cm−1 were, respectively, attributed to Raman signals of Eg and Ag of FeS2 NPs. Based on the results of Raman spectra, TiO2-FeS2 NCs were composed of TiO2 NPs and FeS2 NPs. Based on examination of SEM images, TEM images, XRD spectra, and Raman spectra, TiO2-FeS2 NCs were successfully prepared by a simple solution process.
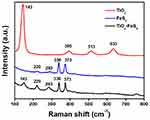 |
Figure 3 Raman spectra of TiO2 NPs, FeS2 NPs, and TiO2-FeS2 NCs. |
Optical Properties of TiO2-FeS2 NCs
UV-Vis-NIR spectra were used to investigate the optical properties of TiO2 NPs, FeS2 NPs, and TiO2-FeS2 NCs. In UV-Vis-NIR spectra of Figure 4, absorption curves of TiO2 NPs, FeS2 NPs and TiO2-FeS2 NCs are depicted. Photoactivitiesof TiO2 NPs were only found in the UV region of the electromagnetic spectrum. Bandgaps of TiO2 (anatase) and TiO2 (rutile) were, respectively, found to be 3.2 and 3.0 eV according to their absorption wavelengths at 388 and 413 nm. Therefore, the absorption spectra of TiO2 NPs suddenly decreased after 413 nm.40 FeS2 NPs were seen to have absorption in a wide range from Vis to NIR due to their band gap of 0.95 eV. The absorption of TiO2-FeS2 NCs was clearly observed from UV to NIR because of the combination of the absorption curves of TiO2 NPs and FeS2 NPs.41 Results of UV-Vis-NIR spectra indicated that the absorption of TiO2-FeS2 NCs was extended from UV to NIR compared to that of TiO2 NPs.
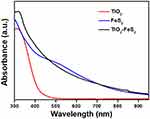 |
Figure 4 UV-Vis-NIR spectra of TiO2 NPs, FeS2 NPs, and TiO2-FeS2 NCs. |
Light-Induced Antibacterial Activity of TiO2-FeS2 NCs
To investigate light-induced antibacterial activities, TiO2 NPs, FeS2 NPs, and TiO2-FeS2 NCs incubated with E. coli solutions were irradiated with simulated AM1.5 sunlight and not irradiated. After light irradiation for 3 min, the OD600 value of the E. coli solution was found to be 0.85 after culturing for 150 min as shown in the growth curve (green line) of Figure 5A. Without light irradiation, the growth curve of E. coli showed no significant change compared to that of E. coli with light irradiation for 3 min. To investigate the light-induced antibacterial activity, TiO2 NPs, FeS2 NPs, and TiO2-FeS2 NCs were incubated with E. coli solutions and then irradiated with light for 3 min. As shown in the growth curves of Figure 5A, after light irradiation for 3 min, OD600 values of E. coli solutions incubated with TiO2 NPs (red line), FeS2 NPs (blue line), and TiO2-FeS2 NCs (black line) were, respectively, 0.52, 0.58, and 0.49 after culturing for 150 min. Bacterial growth results indicated that light-induced antibacterial activities increased in the order of FeS2 NPs, TiO2 NPs, and TiO2-FeS2 NCs. To further examine the light-harvesting capability of FeS2 NPs in TiO2-FeS2 NCs, a 515-nm longpass filter was applied to exclude light wavelengths shorter than 515 nm. As shown in Figure 5A and B, under light irradiation, growth curves of E. coli revealed no changes with and without the 515-nm longpass filter. However, with the 515-nm longpass filter, light-induced antibacterial activities of TiO2 NPs, FeS2 NPs, and TiO2-FeS2 NCs all decreased, as shown in Figure 5B. After light irradiation for 3 min with the 515-nm longpass filter, OD600 values of E. coli solutions incubated with TiO2 NPs (red line), FeS2 NPs (blue line), and TiO2-FeS2 NCs (black line) were, respectively, 0.85, 0.79, and 0.67 after culturing for 150 min. Obviously, with the 515-nm longpass filter, light absorption by TiO2 NPs was cut off, resulting in no light-induced antibacterial activity. For FeS2 NPs, the light-induced antibacterial activity was still exhibited because FeS2 NPs can absorb light wavelengths longer than 515 nm. Moreover, with the light harvester of FeS2 NPs, the light-induced antibacterial activity of TiO2-FeS2 NCs was retained with the use of the 515-nm longpass filter. When using the filter to restrict light wavelengths shorter than 515 nm, the light-induced antibacterial activity of TiO2-FeS2 NCs could be attributed to that the light harvester of FeS2 NPs in the TiO2-FeS2 NCs, which could absorb light wavelengths longer than 515 nm to generate photoinduced electrons, and then those photoelectrons were transferred to TiO2 NPs to generate ROS. Furthermore, after light irradiation for 30 min with the 515-nm longpass filter, the final OD600 value of E. coli incubated with TiO2-FeS2 NCs was found to be 0.39 as shown in Figure 5C. Without light irradiation, the final OD600 value of E. coli incubated with TiO2-FeS2 NCs was 0.83. Most importantly, after light irradiation for 30 min, there was no growth of E. coli in the presence of TiO2-FeS2 NCs as a light-activated antibacterial agent. Overall, results of light-induced antibacterial activity suggested that with TiO2-FeS2 NCs, FeS2 NPs acted as a superior light harvester to absorb light in the Vis and NIR regions to generate photoelectrons, and then the photoelectrons were delivered from FeS2 NPs to TiO2 NPs for improved ROS generation.
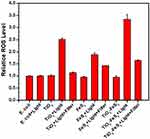
Investigation of ROS Generation
TiO2-based materials were found to possess remarkable antibacterial activities due to ROS generation under UV light irradiation.42–44 Therefore, to investigate antibacterial activities, ROS generation activities of TiO2 NPs, FeS2 NPs, and TiO2-FeS2 NCs incubated with E. coli were measured by an H2DCFDA assay with and without light irradiation. In Figure 6, for the control experiment, the ROS level of E. coli without light irradiation was set to 1.0. Compared to E. coli with light irradiation for 3 min, the relative ROS level revealed no significant change due to no light-induced antibacterial agent being produced. Moreover, without light irradiation, neither TiO2 NPs, FeS2 NPs, nor TiO2-FeS2 NCs showed any obvious increases in ROS. With light irradiation for 3 min, relative ROS levels of TiO2 NPs, FeS2 NPs, and TiO2-FeS2 NCs were, respectively, 2.51-, 1.88-, and 3.34-fold higher compared to that of the control experiment. However, under light irradiation with a 515-nm longpass filter, relative ROS levels of TiO2 NPs, FeS2 NPs, and TiO2-FeS2 NCs were, respectively, 1.13-, 1.43-, and 1.63-fold higher compared to that of the control experiment. For TiO2 NPs with light irradiation, relative ROS levels dramatically decreased from 2.51-fold (without the filter) to 1.13-fold (with the filter) because the light absorption of TiO2 NPs was reduced by the filter. For FeS2 NPs under light irradiation, relative ROS levels only dropped from 1.88-fold (without) to 1.13-fold (with the filter). The reason can be attributed to FeS2 NPs still being able to absorb light at wavelengths longer than 515 nm to produce photoelectrons for ROS generation. For TiO2-FeS2 NCs, relative ROS levels decreased from 3.34- (with light irradiation) to 1.64-fold (with light irradiation and the filter). Under light irradiation with the 515-nm filter, the light-induced antibacterial activity of TiO2-FeS2 NCs suggested that photoelectrons generated by FeS2 NPs in TiO2-FeS2 NCs were transferred to the conduction band of TiO2 NPs to generate more ROS (1.63-fold) compared to that of only FeS2 NPs (1.43-fold). For TiO2 NPs, FeS2 NPs, and TiO2-FeS2 NCs, results of the ROS tests were consistent with measurements of antibacterial activities.
To further examine the effect of ROS, the morphologies of E. coli incubated with TiO2-FeS2 NCs before and after light irradiation were characterized by SEM. As shown in Figure 7A, after incubation with TiO2-FeS2 NCs, E. coli revealed compact membrane structure without light irradiation. However, with light irradiation for 3 min, E. coli incubation with TiO2-FeS2 NCs appeared slight membrane rupture as indicated by the yellow arrows in Figure 7B. Furthermore, the complete rupture of E. coli membrane was observed as indicated by the white arrows. In high-resolution SEM image of Figure 7C, the slight membrane rupture and complete rupture of E. coli membrane were respectively indicated by the yellow arrow and the white arrows. The results indicated that in the system of TiO2-FeS2 NCs, under light irradiation, the increases of ROS generation enhanced for the destruction of bacterial membrane.45–47
Mechanism of Light-Induced Antibacterial Activity of TiO2-FeS2 NCs
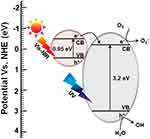
Bandgaps of TiO2 NPs and FeS2 NPs were, respectively, found to be 3.2 and 0.95 eV in the UV-Vis-NIR region of the electromagnetic spectrum. The combination of TiO2 NPs and FeS2 NPs was demonstrated to have increased photoactivity. Herein, TiO2-FeS2 NCs were also demonstrated to have superior light-induced antibacterial activity compared to that of only TiO2 NPs or only FeS2 NPs. A schematic of the mechanism of light-induced antibacterial activity of TiO2-FeS2 NCs is shown in Figure 8. With reference to the normal hydrogen electrode (NHE), values of the conduction band (CB) and valence band (VB) of TiO2 NPs were −0.2 and 3 eV, respectively. Therefore, TiO2 NPs can absorb UV light to generate photoinduced electrons and holes for ROS production, including ⋅O2− (−0.16 eV) and ⋅OH (2.32 eV). For FeS2 NPs, CB and VB were, respectively, −0.5 and 0.45 eV. In TiO2-FeS2 NCs, FeS2 NPs acted as light harvesters to absorb light from Vis to NIR to produce photoinduced electrons and holes. Furthermore, the photoinduced electrons in the CB of FeS2 NPs were transferred to the CB of TiO2 NPs to facilitate ROS generation. Overall, the light-induced antibacterial activity of TiO2-FeS2 NCs was enhanced by the light harvesters of FeS2 NPs due to the broad range of light absorption from UV to NIR.
Conclusions
TiO2-FeS2 NCs were successfully synthesized by a simple solution process, and their structural and optical properties were validated by SEM, TEM, XRD, Raman spectroscopy, and UV-Vis-NIR spectroscopy. TiO2-FeS2 NCs exhibited a broad range of light absorption from UV to NIR, because of the combination of the absorptions of TiO2 NPs and FeS2 NPs. In TiO2-FeS2 NCs, FeS2 NPs acted as a superior light harvester to increase light absorption from the Vis and NIR ranges. With light irradiation for 3 min, OD600 values of E. coli solutions incubated with TiO2 NPs, FeS2 NPs, and TiO2-FeS2 NCs were, respectively, 0.52, 0.58, and 0.49 after culturing for 150 min, indicating that the best antibacterial activity was with TiO2-FeS2 NCs. Light-induced antibacterial activities of FeS2 NPs, TiO2 NPs, and TiO2-FeS2 NCs can be attributed to ROS generation. Under light irradiation, relative ROS levels increased in the decreasing order of FeS2 NPs (1.88-fold), TiO2 NPs (2.51-fold), and TiO2-FeS2 NCs (3.34-fold). In TiO2-FeS2 NCs, TiO2 NPs absorbed UV light to generate photoinduced electrons and holes for ROS generation, including ⋅O2− and ⋅OH. Furthermore, FeS2 NPs in TiO2-FeS2 NCs harvested the light from Vis to NIR to produce photoinduced electrons, and then the photoinduced electrons from FeS2 NPs were transferred to TiO2 NPs to facilitate ROS generation. Our work demonstrated that TiO2-FeS2 NCs with superior light-induced antibacterial activity could be a potential antibacterial agent for future antibacterial applications in fields such as medicine, food, and water quality.
Acknowledgments
This work was financially supported by MOST109-2636-E-038-001, MOST 109-2113-M-038-005-MY2, Taipei Medical University and the Higher Education Sprout Project by the Ministry of Education (MOE) in Taiwan. We would like to acknowledge Yuan-Chin Hsiung, Chun-Chih Liu and Dr. Chi-Ming Lee for her/his excellent technical support at the TMU Core Facility Center.
Disclosure
The authors declare no conflicts of interest.
References
1. Mutalik C, Wang D-Y, Krisnawati DI, Jazidie A, Yougbare S, Kuo T-R. Light-activated heterostructured nanomaterials for antibacterial applications. Nanomaterials. 2020;10(4):643. doi:10.3390/nano10040643
2. Luo J, Deng W, Yang F, Wu Z, Huang M, Gu M. Gold nanoparticles decorated graphene oxide/nanocellulose paper for NIR laser-induced photothermal ablation of pathogenic bacteria. Carbohydr Polym. 2018;198:206–214. doi:10.1016/j.carbpol.2018.06.074
3. Teng CP, Zhou T, Ye E, et al. Effective targeted photothermal ablation of multidrug resistant bacteria and their biofilms with NIR‐absorbing gold nanocrosses. Adv Healthcare Mater. 2016;5:2122–2130. doi:10.1002/adhm.201600346
4. Khan SA, Singh AK, Senapati D, Fan Z, Ray PC. Bio-conjugated popcorn shaped gold nanoparticles for targeted photothermal killing of multiple drug resistant salmonella Dt104. J Mater Chem. 2011;21:17705–17709. doi:10.1039/c1jm13320a
5. de Kraker ME, Stewardson AJ, Harbarth S. Will 10 million people die a year due to antimicrobial resistance by 2050? PLoS Med. 2016;13(11):e1002184. doi:10.1371/journal.pmed.1002184
6. Matsunaga T, Tomoda R, Nakajima T, Wake H. Photoelectrochemical sterilization of microbial cells by semiconductor powders. FEMS Microbiol Lett. 1985;29:211–214. doi:10.1111/j.1574-6968.1985.tb00864.x
7. Mills A, Le Hunte S. An overview of semiconductor photocatalysis. J Photochem Photobiol A Chem. 1997;108:1–35. doi:10.1016/S1010-6030(97)00118-4
8. Nadtochenko V, Denisov N, Sarkisov O, Gumy D, Pulgarin C, Kiwi J. Laser kinetic spectroscopy of the interfacial charge transfer between membrane cell walls of E. Coli and TiO2. J Photochem Photobiol A Chem. 2006;181:401–407. doi:10.1016/j.jphotochem.2005.12.028
9. Caratto V, Ball L, Sanguineti E, et al. Antibacterial activity of standard and n-doped titanium dioxide-coated endotracheal tubes: an in vitro study. Revista Brasileira De Terapia Intensiva. 2017;29:55–62. doi:10.5935/0103-507X.20170009
10. Kangwansupamonkon W, Lauruengtana V, Surassmo S, Ruktanonchai U. Antibacterial effect of apatite-coated titanium dioxide for textiles applications. Nanomedicine. 2009;5:240–249. doi:10.1016/j.nano.2008.09.004
11. Deepagan V, You DG, Um W, et al. Long-Circulating Au-TiO2 nanocomposite as a sonosensitizer for ROS-mediated eradication of cancer. Nano Lett. 2016;16:6257–6264. doi:10.1021/acs.nanolett.6b02547
12. Liu R, Wang X, Ye J, et al. Enhanced antibacterial activity of silver-decorated sandwich-like mesoporous silica/reduced graphene oxide nanosheets through photothermal effect. Nanotechnology. 2018;29:105704. doi:10.1088/1361-6528/aaa624
13. Prakash J, Sun S, Swart HC, Gupta RK. Noble metals-TiO2 nanocomposites: from fundamental mechanisms to photocatalysis, surface enhanced Raman scattering and antibacterial applications. Appl Mater Today. 2018;11:82–135. doi:10.1016/j.apmt.2018.02.002
14. Hamzah MQ, Mezan SO, Tuama AN, Jabbar AH, Agam MA. Study and characterization of polystyrene/titanium dioxide nanocomposites (PS/TiO2 NCS) for photocatalytic degradation application: a review. Int J Eng Technol. 2018;7:538–543. doi:10.14419/ijet.v7i4.30.28172
15. Nabiyouni G, Ghanbari D. Simple preparation of magnetic, antibacterial and photo-catalyst Nife2o4@ TiO2/Pt nanocomposites. J Nanostruct. 2018;8:408–416.
16. Tang S, Wang Z, Li P, et al. Degradable and photocatalytic antibacterial Au-TiO2/Sodium alginate nanocomposite films for active food packaging. Nanomaterials. 2018;8:930. doi:10.3390/nano8110930
17. Mathew S, Ganguly P, Rhatigan S, et al. Cu-doped TiO2: visible light assisted photocatalytic antimicrobial activity. Appl Sci. 2018;8:2067. doi:10.3390/app8112067
18. Kőrösi L, Bognár B, Horváth M, et al. Hydrothermal evolution of PF-Co-doped TiO2 nanoparticles and their antibacterial activity against carbapenem-resistant klebsiella pneumoniae. Appl Catal B. 2018;231:115–122. doi:10.1016/j.apcatb.2018.03.012
19. Ghilini F, Rodríguez González MC, Miñán AG, et al. Highly stabilized nanoparticles on poly-L-lysine-coated oxidized metals: a versatile platform with enhanced antimicrobial activity. ACS Appl Mater Interfaces. 2018;10:23657–23666. doi:10.1021/acsami.8b07529
20. Huang S-M, Weng C-H, Tzeng J-H, et al. Photocatalytic inactivation of Klebsiella Pneumoniae by visible-light-responsive N/C-Doped and N-Tourmaline/Palladium-C-Codoped TiO2. Chem Eng J. 2020;379:122345. doi:10.1016/j.cej.2019.122345
21. Meekins BH, Kamat PV. Got TiO2 nanotubes? Lithium ion intercalation can boost their photoelectrochemical performance. ACS Nano. 2009;3:3437–3446. doi:10.1021/nn900897r
22. Sivakami R, Thiyagarajan P. Synthesis and luminescence properties of Zns: ce3+, Li+, Mn2+ nanophosphors. Nano Struct Nano Objects. 2016;6:59–66. doi:10.1016/j.nanoso.2016.03.001
23. Asahi R, Morikawa T, Irie H, Ohwaki T. Nitrogen-doped titanium dioxide as visible-light-sensitive photocatalyst: designs, developments, and prospects. Chem Rev. 2014;114:9824–9852. doi:10.1021/cr5000738
24. Ananpattarachai J, Seraphin S, Kajitvichyanukul P. Formation of hydroxyl radicals and kinetic study of 2-chlorophenol photocatalytic oxidation using C-Doped TiO2, N-Doped TiO2, and C, N Co-Doped TiO2 under visible light. Environ Sci Poll Res. 2016;23:3884–3896. doi:10.1007/s11356-015-5570-8
25. Ismail AA, Kandiel TA, Bahnemann DW. Novel (and better?) titania-based photocatalysts: brookite nanorods and mesoporous structures. J Photochem Photobiol A Chem. 2010;216:183–193. doi:10.1016/j.jphotochem.2010.05.016
26. Ksibi M, Rossignol S, Tatibouët J-M, Trapalis C. Synthesis and solid characterization of nitrogen and sulfur-doped TiO2 photocatalysts active under near visible light. Mater Lett. 2008;62:4204–4206. doi:10.1016/j.matlet.2008.06.026
27. Li L, Yang Y, Liu X, et al. Synthesis of B-doped TiO2 and its photocatalytic performance on degradation of RHB. Appl Surf Sci. 2013;265:36–40. doi:10.1016/j.apsusc.2012.10.075
28. Nainani R, Thakur P, Chaskar M. Synthesis of silver doped TiO2 nanoparticles for the improved photocatalytic degradation of methyl orange. J Mater Sci Eng B. 2012;2:52–58.
29. Pal NK, Kryschi C. Improved photocatalytic activity of gold decorated differently doped TiO2 nanoparticles: a comparative study. Chemosphere. 2016;144:1655–1664. doi:10.1016/j.chemosphere.2015.10.060
30. Sadeghi-Niaraki S, Ghasemi B, Habibolahzadeh A, Ghasemi E, Ghahari M. Nanostructured Fe2O3@ TiO2 pigments with improved NIR reflectance and photocatalytic ability. Mater Chem Phys. 2019;121769. doi:10.1016/j.matchemphys.2019.121769
31. Yan D, Wu X, Pei J, Wu C, Wang X, Zhao H. Construction of G-C3n4/Tio2/Ag composites with enhanced visible-light photocatalytic activity and antibacterial properties. Ceramics Int. 2020;46:696–702. doi:10.1016/j.ceramint.2019.09.022
32. Siwińska-Stefańska K, Kubiak A, Piasecki A, et al. Hydrothermal synthesis of multifunctional TiO2-Zno oxide systems with desired antibacterial and photocatalytic properties. Appl Surf Sci. 2019;463:791–801. doi:10.1016/j.apsusc.2018.08.256
33. Fang JH, Wu JW, Su LY, Zhang XY, Lu ZH. Modification of TiO2 microporous electrode with quantum-sized FeS2 particles. Chem Lett. 1997;26:149–150. doi:10.1246/cl.1997.149
34. Xin Y, Li Z, Wu W, Fu B, Zhang Z. Pyrite FeS2 sensitized TiO2 nanotube photoanode for boosting near-infrared light photoelectrochemical water splitting. ACS Sustain Chem Eng. 2016;4:6659–6667. doi:10.1021/acssuschemeng.6b01533
35. Han E, Hu F, Zhang S, et al. Worm-like FeS2/TiO2 nanotubes for photoelectrocatalytic reduction of CO2 to methanol under visible light. Energy Fuels. 2018;32:4357–4363. doi:10.1021/acs.energyfuels.7b03234
36. Kuo T-R, Liao H-J, Chen Y-T, et al. Extended visible to near-infrared harvesting of earth-abundant FeS2–TiO2 heterostructures for highly active photocatalytic hydrogen evolution. Green Chem. 2018;20:1640–1647. doi:10.1039/C7GC03173D
37. Chang CC, Li SR, Chou HL, et al. Photoactive earth‐abundant iron pyrite catalysts for electrocatalytic nitrogen reduction reaction. Small. 2019;15:1904723. doi:10.1002/smll.201904723
38. Balachandran U, Eror N. Raman spectra of titanium dioxide. J Solid State Chem. 1982;42:276–282. doi:10.1016/0022-4596(82)90006-8
39. Wang D-Y, Li C-H, Li -S-S, et al. Iron Pyrite/Titanium dioxide photoanode for extended near infrared light harvesting in a photoelectrochemical cell. Sci Rep. 2016;6:20397. doi:10.1038/srep20397
40. Li X, Wang D, Cheng G, Luo Q, An J, Wang Y. Preparation of polyaniline-modified TiO2 nanoparticles and their photocatalytic activity under visible light illumination. Appl Catal B. 2008;81:267–273. doi:10.1016/j.apcatb.2007.12.022
41. Wang DY, Jiang YT, Lin CC, et al. Solution‐processable Pyrite FeS2 nanocrystals for the fabrication of heterojunction photodiodes with visible to NIR photodetection. Adv Mater. 2012;24:3415–3420. doi:10.1002/adma.201200753
42. Ma S, Zhan S, Jia Y, Zhou Q. Superior antibacterial activity of Fe3O4-TiO2 nanosheets under solar light. ACS Appl Mater Interfaces. 2015;7:21875–21883.
43. Li H, Zhong J, Zhu H, et al. Hybrid Cu2o/TiO2 nanocomposites with enhanced photocatalytic antibacterial activity toward acinetobacter baumannii. ACS Appl Bio Mater. 2019;2:4892–4903. doi:10.1021/acsabm.9b00644
44. Akhavan O. Lasting antibacterial activities of Ag–TiO2/Ag/a-TiO2 nanocomposite thin film photocatalysts under solar light irradiation. J Colloid Interface Sci. 2009;336:117–124. doi:10.1016/j.jcis.2009.03.018
45. Su H-L, Chou -C-C, Hung D-J, et al. The disruption of bacterial membrane integrity through ROS generation induced by nanohybrids of silver and clay. Biomaterials. 2009;30:5979–5987. doi:10.1016/j.biomaterials.2009.07.030
46. Xiang Y, Mao C, Liu X, et al. Rapid and superior bacteria killing of carbon quantum dots/ZNO decorated injectable folic acid‐conjugated PDA hydrogel through dual‐light triggered ROS and membrane permeability. Small. 2019;15:1900322. doi:10.1002/smll.201900322
47. Wang B, Zhang H, An J, et al. Sequential intercellular delivery nanosystem for enhancing ROS-induced antitumor therapy. Nano Lett. 2019;19:3505–3518. doi:10.1021/acs.nanolett.9b00336
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.