Back to Journals » OncoTargets and Therapy » Volume 7
High expression of 23 kDa protein of augmenter of liver regeneration (ALR) in human hepatocellular carcinoma
Authors Yu H, Zhu M, Xiang D, Li J, Sheng J
Received 29 January 2014
Accepted for publication 7 April 2014
Published 2 June 2014 Volume 2014:7 Pages 887—893
DOI https://doi.org/10.2147/OTT.S61531
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Hai-Ying Yu, Man-Hua Zhu, Dai-Rong Xiang, Jun Li, Ji-Fang Sheng
State Key Laboratory of Infectious Disease and Department of Infectious Disease, First Affiliated Hospital, Zhejiang University School of Medicine, Hangzhou, People's Republic of China
Background: Augmenter of liver regeneration (ALR) is an important polypeptide that participates in the process of liver regeneration. Two forms of ALR proteins are expressed in hepatocytes. Previous data have shown that ALR is essential for cell survival and has potential antimetastatic properties in hepatocellular carcinoma (HCC).
Aims: The study aimed to evaluate the expression levels of two forms of ALR proteins in HCC and their possible significance in HCC development.
Methods: Balb/c mouse monoclonal antibody against ALR protein was prepared in order to detect the ALR protein in HCC by Western blotting and immunohistochemistry. ALR mRNA expression levels were measured by real-time polymerase chain reaction in HCC tissues and compared to paracancerous liver tissues in 22 HCC patients.
Results: ALR mRNA expression in HCC liver tissues (1.51×106 copies/µL) was higher than in paracancerous tissues (1.04×104 copies/µL). ALR protein expression was also enhanced in HCC liver tissues. The enhanced ALR protein was shown to be 23 kDa by Western blotting. Immunohistochemical analysis showed that the 23 kDa ALR protein mainly existed in the hepatocyte cytosol.
Conclusion: The 23 kDa ALR protein was highly expressed in HCC and may play an important role in hepatocarcinogenesis.
Keywords: HCC, ALR mRNA, ALR protein
Introduction
Augmenter of liver regeneration (ALR) is an important polypeptide involved in the process of liver regeneration1 and hepatocyte proliferation.2 It is a member of the newly discovered ALR/Erv1 protein family, with flavin adenine dinucleotide (FAD)-linked sulfhydryl oxidase activity.3,4 ALR is dimerized at the protein level and is alternatively spliced at the transcriptional level. Therefore, two forms of human ALR protein are found in hepatocytes. The shorter protein of 15 kDa, which lacks the amino terminus, consists of 125 amino acids and is mainly located in the nucleus. The longer protein of 23 kDa consists of 205 amino acids, and is located in the cytosol and mitochondrial intermembrane space.5 ALR has been found to be essential for cell survival and biogenesis of cytosolic Fe/S proteins.6 Enhanced ALR mRNA and protein expression were found in the liver tissue samples of patients with liver cirrhosis and hepatic failure.7,8
Hepatocellular carcinoma (HCC) is the third leading cause of death from cancer worldwide.9,10 The incidence of HCC in the People’s Republic of China in the past 25 years has increased, primarily due to the high prevalence of both hepatitis B virus infection11 and liver cirrhosis. Most HCCs develop into liver cirrhosis, and involve hepatocyte apoptosis, atypical hyperplasia, abnormal regulation of oncogenes, and antioncogenes. The process of carcinogenesis of HCC is complex and the molecular pathogenesis is not yet fully understood.12 ALR is a hepatotrophic factor that stimulates hepatocyte proliferation. Human liver diseases, such as chronic hepatitis, liver cirrhosis, and HCC, are all associated with hepatocyte injury and regeneration. It has been shown that the re-expression of 15 kDa ALR in HepG2 cells reduces migration and invasiveness of HCC cells.13 Therefore, the ALR short isoform may be considered as an antimetastatic protein in HCC with the potential to become a marker in HCC diagnosis. In contrast, other studies reported that silencing of the full-length isoform of 23 kDa ALR significantly inhibited hepatoma cell growth and survival, but had almost no effect on hepatocyte cell viability.14 The aim of this study was to investigate the expression and potential clinical importance of the two isoforms of ALR protein in human HCC.
Methods
Ethical considerations
All human and animal protocols and study methods were approved by the Animal Care Ethics Committee and Clinical Investigation Committee of the First Affiliated Hospital, Zhejiang University School of Medicine and Ethics Committee of the First Affiliated Hospital, Zhejiang University School of Medicine (Hangzhou, People’s Republic of China). Written informed consent was obtained from all the patients before surgery to utilize their liver tissues and blood for research purposes.
Liver and serum specimens
A total of 22 patients with histologically proven HCC after liver partial resection surgery performed at the First Affiliated Hospital, Zhejiang University, were prospectively enrolled into our study (16 males and six females; aged 42–63 years, mean age 57.5±9.9 years; all Chinese, and HBsAg positive). Forty-four specimens of liver tissues were collected from these 22 HCC patients, including, for each patient, the carcinoma liver tissues and the paracancerous liver tissues (2 cm away from the carcinoma liver tissue). Tissue samples were fixed in formalin, paraffin-embedded for immunohistochemical analysis, and put into liquid nitrogen for real-time polymerase chain reaction (PCR) and Western blotting. Peripheral blood was obtained from five patients before surgery and centrifuged at 3,000 g for 10 minutes to isolate the serum portion, and stored at −80°C before use.
Preparation of recombinant human ALR (hALR) and anti-hALR monoclonal antibody
The recombinant expression vector of hALR-pPIC9 was constructed by inserting the full-length cDNA encoding hALR into the pPIC9 vector; the recombinant hALR protein of ~24 kDa was expressed in the GS115 line. hALR protein was purified through a chromatographic column (Qiagen GmbH, Hilden, Germany) and displayed using sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE). The molecular weight, isoelectric point, and N- and C-terminal amino acid sequences of the hALR protein were determined by matrix-assisted laser desorption/ionization time of flight mass spectrometry (MALDI-TOF-MS [Bruker BioSpin MRI GmbH, Ettlingen, Germany]) at the Research Centre for Proteome Analysis, Key Lab of Proteomics, Institute of Biochemistry and Cell Biology, Shanghai Institutes for Biological Sciences, Chinese Academy of Sciences (Shanghai, People’s Republic of China).
To generate anti-hALR monoclonal antibody (MAb)-producing cell lines, 0.05 mg of the purified hALR protein with an equal volume of Freund’s complete adjuvant (Sigma-Aldrich Co, St Louis, MO, USA) was injected into five female Balb/c mice separately. The same dosage of proteins emulsified in Freund’s incomplete adjuvant (Sigma-Aldrich Co,) was injected 2, 4, and 6 weeks after the first injection. The spleen was obtained 2 weeks after the last injection. Generation of hybridoma cell lines was performed by fusion of the spleen cells from the immunized mice with myeloma cells, as previously reported.15 The hybridoma cells were distributed in 96-well culture plates with hypoxanthine-aminopterin-thymidine (HAT) medium and cultured to screen the hALR-specific MAb-producing cell lines using the limited dilution method. The MAb was then purified using a ProteinG chromatographic column (Qiagen GmBH); the subtype of the MAb was IgG1 by ELISA assay.
ALR mRNA expression with real-time PCR
The ALR and β-actin standard substances were synthesized in our laboratory and their concentrations were determined by absorbance at 260 nm (SmartSpec™ Plus Spectrophotometer; Bio-Rad Laboratories Inc., Hercules, CA, USA). Ten-fold serial dilutions of the standard substance from 1×100 to 1×1010 copies per mL were prepared.
The 44 liver specimens from the 22 HCC patients corresponded to two subgroups, with 22 specimens each: the HCC group and the para-HCC group. One hundred milligrams of each liver tissue specimen was homogenized in a small earthenware basin with liquid nitrogen, and total RNA was isolated using TRIzol® reagent as described by the manufacturer (Thermo Fisher Scientific, Waltham, MA, USA). RNA concentrations were determined by absorbance at 260 nm, and were transformed into cDNA and stored at −80°C before use.
The level of β-actin mRNA was measured to determine the efficiency of cDNA synthesis and reverse transcription of different mRNA batches. The PCR primers specific for the ALR cDNA were 5′-CCT GTG AGG AGT GTG CTG AAG A-3′ (sense) and 5′-GCT TGC GGT TCA CTT CAT TGT-3′ (antisense) and for β-actin cDNA were 5′-GGC ATC CTC ACC CTG AAG TA-3′ (sense) and 5′-TCG GGT GTT GAA GGT CTC AA-3′ (antisense).
Real-time PCR was carried out on 20 mL of sample using the QuantiTect SYBR Green PCR kit (Qiagen NV, Venlo, The Netherlands) in accordance with the manufacturer’s protocol on an Applied Biosystems StepOne™ Real-Time PCR System (7300/7500; Thermo Fisher Scientific). Real-time PCR data were analyzed using the Applied Biosystems StepOne™ Real-Time PCR System software. The Ct value analysis, PCR efficiency, linearity, slopes of the standard curve, relative quantity of fluorescence, and dissociation curve analysis were calculated by the software program built into the 7300/7500 apparatus. All reactions were carried out in triplicate, and a nontemplate control was performed in every analysis.
ALR protein expression by Western blotting
Total proteins were extracted from the liver specimens (100 mg) of the five patients whose serum was collected before surgery. The total protein in the HCC liver tissues and paracancerous liver tissues was extracted using the Mammalian Protein Extraction Reagent (Thermo Fisher Scientific). The extracted proteins, serum, and the purified recombinant hALR protein (20 μL) were separated on 12% SDS-PAGE and transferred onto a 0.2 μm pore polyvinylidene fluoride (PVDF) membrane (EMD Millipore, Billerica, MA, USA) at 220 mA for 2 hours in a Bio-Rad Trans-Blot Electrophoretic Transfer Cell (Bio-Rad Laboratories Inc.) for Western blotting analysis. The nonspecific sites were blocked with 5% bovine serum albumin (BSA) in Tris-buffered saline and Tween 20 (TBST). The membrane was washed in TBST and incubated with anti-hALR monoclonal antibody (final concentration of 0.5 μg/mL) in blocking buffer or with mouse origin anti-β-actin monoclonal antibody (1:5,000; EarthOx Life Sciences, Millbrae, CA, USA) as a loading control antibody, followed by washing and incubation with horseradish peroxidase (HRP)-conjugated goat anti-mouse immunoglobulins (1:5,000; EarthOx Life Science). The ALR-anti-ALR complex was treated with a mixture of 2× stable peroxide solution and 2× luminol/enhancer solution (Bio-Rad Laboratories Inc.) and chemiluminescence on Fuji X-ray film (Fujifilm Holdings Corporation, Tokyo, Japan).
ALR expression with immunohistochemistry
The liver specimens from HCC patients were embedded in paraffin wax and 4 μm sections were cut. Standard pretreatment was done; anti-hALR monoclonal antibody (1.1 mg/mL) diluted at 1:5,000 in block solution was pipetted onto the sections and incubated overnight at 4°C. The sections were then washed three times with phosphate-buffered saline, and 1:5,000 diluted HRP-conjugated goat anti-mouse immunoglobulins (EarthOx Life Science) were added and incubated at 37°C for 1 hour. Staining was completed by 3,3′-diaminobenzidine (DAB) and counterstaining was performed with hematoxylin. Fluorescent immunohistochemical analysis was also displayed by fluorescein isothiocyanate (FITC) under dark operation.
Statistical analysis
Statistical analysis was performed using SPSS software (v13.0 for Windows; IBM Corporation, Armonk, NY, USA). Dependent t-tests between the HCC and the para-HCC groups were used. P<0.05 was considered as statistically significant.
Results
Features of recombinant hALR protein and anti-hALR monoclonal antibody
SDS-PAGE analysis showed that the size of the expected recombinant hALR protein was ~24 kDa after purification (Figure 1). The molecular weight of the hALR protein was 24.712 kDa as shown by MALDI-TOF-MS. The isoelectric point of hALR was 4.63 (Figure 2). The C-terminal amino acid sequence was Arg-Asp-Gly-Trp-Lys-Asp-Gly-Ger-Cys-Asp (RDGWKDGSCD) and the N-terminal amino acid sequence was Asp-Asn-Glu-Gla-Pro-Val-Phe-Ile (DNEAPVFI), which was consistent with the sequence of ALR cDNA (accession: BC002429.1).
Two cell lines with anti-hALR monoclonal antibody with steady secretion were selected; the anti-hALR monoclonal antibody was purified using a ProteinG chromatographic column and the final concentration was 1.1 mg/μL.
ALR mRNA expression was higher in cancerous versus para-cancerous liver tissues
The absolute values of ALR and β-actin mRNA levels in the 44 liver specimens from the 22 HCC patients are shown in Table 1. Much higher ALR mRNA expression levels were observed in the HCC liver tissues (1.51×106 copies/μL) as compared to the paracancerous liver tissues (1.04×104 copies/μL; P<0.05).
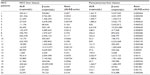  | Table 1 The absolute value of ALR and β-actin mRNA levels of the 22 patients |
The enhanced ALR protein was the 23 kDa isoform
The anti-hALR monoclonal antibody can immunoreact with the recombinant hALR protein during Western blotting. Two bands of anti-hALR monoclonal antibody-positive proteins were present in the extract prepared from the liver tissues: a band of approximately 23 kDa and a band of 15 kDa. One band of anti-hALR monoclonal antibody-positive protein of 23 kDa was present in the serum line.
The 23 kDa protein was 3.6 times stronger (band optical intensity) in the HCC than in the paracancerous liver tissue extracts. The 15 kDa protein band was similar for the two types of extracts. The protein loading control could be detected in the liver tissue extract as a positive 42 kDa band of β-actin.
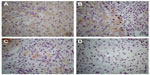
The 23 kDa ALR isoform was mainly present in the hepatocyte cytosol
Positive immunostaining could be essentially seen in the cytoplasm of the HCC liver cells but seldom within the nucleus. Positive immunostaining was more intense in the HCC liver tissue than in the paracancerous liver tissue. No positive immunostaining was found in the negative controls (Figure 3).
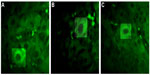
Fluorescent immunohistochemical analysis showed positive fluorescence signals in the cytoplasm and nucleus in the HCC liver tissues (Figure 4). The staining was in a dotted pattern.
Discussion
The role of ALR as a stimulator of hepatocyte proliferation in liver regeneration has been systematically investigated since the 1970s.16,17,18 ALR and its homolog Erv1 (essential for respiratory and vegetative growth) are essential for the survival of hepatocytes and Saccharomyces cerevisiae, respectively.18,19 ALR acts as an FAD-linked sulfhydryl oxidase belonging to the new Erv1p/Alrp family, which is constitutively expressed in hepatocytes in an inactive form and released from cells in an active form during liver regeneration.20 Two isoforms of ALR with molecular weights of about 15 kDa and 23 kDa were found predominantly in the liver. The 23 kDa full-length isoform of ALR has an N-terminal mitochondrial-targeting sequence located in the hepatocyte cytosol and mitochondrial intermembrane space.21 It has been shown to be involved in the export of Fe/S clusters from the mitochondrial matrix, contributing to the biogenesis of Fe/S proteins and cellular iron homeostasis.6,22 The 15 kDa short isoform of ALR lacks the N-terminal 80 amino acids located in the hepatocyte nucleus.21 It appears to regulate mitochondrial gene expression by inducing the transcription and translation of the nuclear-encoded mitochondrial transcription factor A,23 and displays redox active properties by formation of dimers via mediation of cytochrome c in oxidase reactions.24
In the present study, we observed that ALR mRNA and protein expression levels were upregulated in HCC patients in accordance with previously reported data.7 Our data indicate for the first time that both 15 kDa and 23 kDa ALR isoforms were detected in HCC and para-HCC liver tissues, with higher expression in HCC tissues. Moreover, the enhanced 23 kDa ALR existed predominantly in the hepatocyte cytosol. As to the 15 kDa ALR isoform, it was detected within the hepatocyte nucleus and little difference was found between the HCC and para-HCC liver tissues. All these results converge to demonstrate that the increased ALR expression is most likely to be related to the 23 kDa isoform in HCC liver tissues. Furthermore, the fact that the 23 kDa ALR was detected in the peripheral serum of HCC patients strongly suggests the sequential involvement of ALR synthesis, post-translational modification transport into and out of mitochondria, as well as extracellular secretion.
Previous studies have shown that silencing of the 23 kDa ALR had almost no effect on hepatocyte cell viability but significantly inhibited hepatoma cell growth and survival,14 which revealed a crucial correlation of 23 kDa ALR with HCC in vitro. Our results, by showing that in HCC patients, the 23 kDa ALR expression was higher in HCC cytoplasm and was present in peripheral serum, indicated an extracellular secretion of 23 kDa ALR from hepatoma to serum. Reexpression of the ALR in the cytosol of HepG2 cells reduced migration and invasiveness of HCC cells, suggesting that the metastatic potential of HCC cells with enhanced growth activity and increased levels of ALR depends on the ratio of mitochondrial and cytosolic-localized ALR.13
ALR may stimulate hepatocyte proliferation through two signaling pathways. On one hand, intracellular ALR interacted with Jun activation domain-binding protein 1 (JAB1), triggering activating protein-1 (AP-1) transcriptional activity. On the other hand, extracellular ALR autocrined from hepatoma cells activated the mitogen-activated protein kinase (MAPK) signaling pathway.25 AP-1 plays an important role in carcinogenesis by stimulating cell proliferation, inhibiting cell differentiation, and promoting carcinoma invasion and metastasis. More efforts should be carried out concerning the transcription and post-transcriptional regulation of ALR, and its interaction with receptor protein and signal protein in cells. Whether the 23 kDa ALR isoform would be more specific of liver carcinoma than purely regenerative lesions remains to be explored. HCC now represents the fifth most frequent malignant tumor in the world and the third cause of death due to cancer.9 There is a growing understanding of the molecular mechanisms inducing hepatocarcinogenesis, which almost never occurs in healthy liver, but most often develops in response to chronic liver injury at the cirrhosis stage. A more in-depth understanding of epidemiologic factors and molecular mechanisms associated with HCC, including a better knowledge of the involvement of the 23 kDa ALR isoform, should ultimately improve our current concepts for screening and treating this life-threatening disease.
Acknowledgments
We are grateful to Lin-Fu Zhou of the Institute of Medical Biotechnology, Faculty of Basic Medicine, Zhejiang University School of Medicine, Hangzhou, People’s Republic of China for experimental assistance.
Disclosure
The authors report no conflicts of interest in this work.
References
Gandhi CR. Augmenter of liver regeneration. Fibrogenesis Tissue Repair. 2012;5(1):10. | |
Dayoub R, Thasler WE, Bosserhoff AK, et al. Regulation of polyamine synthesis in human hepatocytes by hepatotrophic factor augmenter of liver regeneration. Biochem Biophys Res Commun. 2006;345(1):181–187. | |
Senkevich TG, White CL, Koonin EV, Moss B. A viral member of the ERV1/ALR protein family participates in a cytoplasmic pathway of disulfide bond formation. Proc Natl Acad Sci U S A. 2000;97(22):12068–12073. | |
Senkevich TG, White CL, Koonin EV, Moss B. Complete pathway for protein disulfide bond formation encoded by poxviruses. Proc Natl Acad Sci U S A. 2002;99(10):6667–6672. | |
Gatzidou E, Kouraklis G, Theocharis S. Insights on augmenter of liver regeneration cloning and function. World J Gastroenterol. 2006;12(31):4951–4958. | |
Lange H, Lisowsky T, Gerber J, Mühlenhoff U, Kispal G, Lill R. An essential function of the mitochondrial sulfhydryl oxidase Erv1p/ALR in the maturation of cytosolic Fe/S proteins. EMBO Rep. 2001;2(8):715–720. | |
Thasler WE, Schlott T, Thelen P, et al. Expression of augmenter of liver regeneration (ALR) in human liver cirrhosis and carcinoma. Histopathology. 2005;47(1):57–66. | |
Yu HY, Xiang DR, Huang HJ, Li J, Sheng JF. Expression level of augmenter of liver regeneration in patients with hepatic failure and hepatocellular carcinoma. Hepatobiliary Pancreat Dis Int. 2010;9(5):492–498. | |
Forner A, Llovet JM, Bruix J. Hepatocellular carcinoma. Lancet. 2012;379(9822):1245–1255. | |
Metwaly HA, Al-Gayyar MM, Eletreby S, Ebrahim MA, El-Shishtawy MM. Relevance of serum levels of interleukin-6 and syndecan-1 in patients with hepatocellular carcinoma. Sci Pharm. 2012;80(1):179–188. | |
Xu Y, Liu Y, Pan S, et al. IL-23R polymorphisms, HBV infection, and risk of hepatocellular carcinoma in a high-risk Chinese population. J Gastroenterol. 2013;48(1):125–131. | |
El-Serag HB, Rudolph KL. Hepatocellular carcinoma: epidemiology and molecular carcinogenesis. Gastroenterology. 2007;132(7):2557–2576. | |
Dayoub R, Wagner H, Bataille F, et al. Liver regeneration associated protein (ALR) exhibits antimetastatic potential in hepatocellular carcinoma. Mol Med. 2011;17(3–4):221–228. | |
Cao Y, Fu YL, Yu M, et al. Human augmenter of liver regeneration is important for hepatoma cell viability and resistance to radiation-induced oxidative stress. Free Radic Biol Med. 2009;47(7):1057–1066. | |
Rudenko NV, Abbasova SG, Grishin EV. [Preparation and characterization of monoclonal antibodies to Bacillus anthracis protective antigen]. Bioorg Khim. 2011;37(3):354–360. Russian. | |
LaBrecque DR, Pesch LA. Preparation and partial characterization of hepatic regenerative stimulator substance (SS) from rat liver. J Physiol. 1975;248(2):273–284. | |
Francavilla A, Hagiya M, Porter KA, Polimeno L, Ihara I, Starzl TE. Augmenter of liver regeneration: its place in the universe of hepatic growth factors. Hepatology. 1994;20(3):747–757. | |
Thirunavukkarasu C, Wang LF, Harvey SA, et al. Augmenter of liver regeneration: an important intracellular survival factor for hepatocytes. J Hepatol. 2008;48(4):578–588. | |
Lisowsky T. Dual function of a new nuclear gene for oxidative phosphorylation and vegetative growth in yeast. Mol Gen Genet. 1992;232(1):58–64. | |
Gandhi CR, Kuddus R, Subbotin VM, et al. A fresh look at augmenter of liver regeneration in rats. Hepatology. 1999;29(5):1435–1445. | |
Li Y, Wei K, Lu C, et al. Identification of hepatopoietin dimerization, its interacting regions and alternative splicing of its transcription. Eur J Biochem. 2002;269(16):3888–3893. | |
Daithankar VN, Farrell SR, Thorpe C. Augmenter of liver regeneration: substrate specificity of a flavin-dependent oxidoreductase from the mitochondrial intermembrane space. Biochemistry. 2009;48(22):4828–4837. | |
Polimeno L, Capuano F, Marangi LC, et al. The augmenter of liver regeneration induces mitochondrial gene expression in rat liver and enhances oxidative phosphorylation capacity of liver mitochondria. Dig Liver Dis. 2000;32(6):510–517. | |
Farrell SR, Thorpe C. Augmenter of liver regeneration: a flavin-dependent sulfhydryl oxidase with cytochrome c reductase activity. Biochemistry. 2005;44(5):1532–1541. | |
Lu C, Li Y, Zhao Y, et al. Intracrine hepatopoietin potentiates AP-1 activity through JAB1 independent of MAPK pathway. FASEB J. 2002;16(1):90–92. |
 © 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.