Back to Journals » Journal of Pain Research » Volume 13
Herpes Zoster and Postherpetic Neuralgia in an Elderly Patient with Critical COVID-19: A Case Report
Authors Cao X, Zhang X, Meng W, Zheng H
Received 27 July 2020
Accepted for publication 1 September 2020
Published 22 September 2020 Volume 2020:13 Pages 2361—2365
DOI https://doi.org/10.2147/JPR.S274199
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Michael Schatman
Xueqin Cao,1 Xianwei Zhang,1 Weihua Meng,2 Hua Zheng1
1Department of Anesthesiology and Pain Medicine, Tongji Hospital, Tongji Medical College, Huazhong University of Science and Technology, Wuhan 430030, People’s Republic of China; 2Division of Population Health and Genomics, School of Medicine, University of Dundee, Ninewells Hospital and Medical School, Dundee DD2 4BF, Scotland, UK
Correspondence: Hua Zheng
Department of Anesthesiology and Pain Medicine, Tongji Hospital, Tongji Medical College, Huazhong University of Science and Technology, Wuhan 430030, People’s Republic of China
Tel +86 27 8366 3173
Fax +86 27 8366 2853
Email [email protected]
Abstract: Critical patients with COVID-19 are thought to be at high risk of developing chronic pain. However, the exact nature and mechanisms of COVID-19-related chronic pain remain largely unknown. Here, we describe clinical features, treatments and outcome of herpes zoster as well as postherpetic neuralgia in a 70-year-old woman with critical COVID-19. The patient had a history of type 2 diabetes and myasthenia gravis. She developed herpes zoster in the right 10 to 12 lumbar dermatomes in the recovery period of COVID-19. Intravenous (250 mg 3 times a day) and then oral (400 mg 5 times a day) acyclovir was used for antiviral therapy. Pregabalin (75 mg orally twice a day) and ibuprofen was used for analgesia. Her skin lesions resolved 21 days after the onset of rash. However, she continued to have persistent pain in the same dermatomal distribution. After the dosage of pregabalin was increased to 150 mg orally twice a day, her pain was partially relieved. During the telephone follow-up 4 months after herpes zoster eruption, the patient still complained intermittent pain in the right 10 to 12 lumbar dermatomes. Our case draws attention to postherpetic neuralgia in COVID-19 patients and provides a targeted suggestion for this kind of patients.
Keywords: SARS-CoV-2, COVID-19, herpes zoster, postherpetic neuralgia, elderly patient
Introduction
Caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), the outbreak of coronavirus disease 2019 (COVID-19) has spread quickly across the world. As of July 27, 2020, COVID-19 has been confirmed in 15,785,641 cases and caused 640,016 deaths globally.1 Although SARS-CoV-2 primarily affects the respiratory system, other organ systems such as cardiovascular, urinary and neurological systems are also involved.2 It is likely that COVID-19 survivors will have additional comorbidities requiring further support, including chronic pain.3 Previous studies have suggested that chronic pain is not an uncommon complication following ICU discharge.4 Critical patients with COVID-19 are thought to be at high risk of developing chronic pain.5 However, the exact nature and mechanisms of COVID-19-related chronic pain remain largely unknown.
Postherpetic neuralgia is a chronic painful condition that can occur following herpes zoster, an extremely painful skin rash caused by the varicella zoster virus. Postherpetic neuralgia occurs most often in elderly people and in people with immunocompromise, who are also susceptible to COVID-19.6 Recently, there are case reports of herpes zoster in patients with both symptomatic and asymptomatic SARS-CoV-2 infection.7,8 However, in these cases, no long-term follow up was performed and no postherpetic neuralgia was observed. Here, we report clinical features and treatments of herpes zoster and postherpetic neuralgia in an elderly patient with critical COVID-19. In addition, we review literatures about pathogenesis of COVID-19 and herpes zoster, and the prevention and treatment of postherpetic neuralgia, in order to provide a targeted suggestion for this kind of patient. This case report was approved by the Research and Ethics Committee of Tongji hospital at Tongji Medical College, Huazhong University of Science and Technology to publish the case details. Written informed consent has been provided by the patient to have the case details and any accompanying images published.
Case Presentation
A 70-year-old woman presented to the emergency department with fever and dry cough of 3 days’ duration on January 17, 2020. Her medical history was noteworthy for type 2 diabetes and myasthenia gravis, for which she was taking acarbose (25 mg orally 3 times a day), pyridostigmine (120mg orally 3 times a day), prednisone (20mg orally once a day), and tacrolimus (2 mg orally once a day). The patient had a history of muscle weakness, with no respiratory failure, which was well controlled. In addition, the patient had a history of chickenpox but no previous documented episodes of herpes zoster. Chest CT scans showed multiple ground glass opacities and infiltration in both lungs. Blood examination showed a decreased leukocyte count of 3.4 x109/L and lymphocyte count of 0.6 x 109/L (Table 1). The patient was diagnosed as a suspected case of COVID-19 and admitted to an infection isolation ward.
 |
Table 1 Clinical Laboratory Results of the Patient |
After admission, the diagnose of COVID-19 was confirmed based on positive results of real-time reverse transcriptase–polymerase chain reaction (RT-PCR) tests for nucleic acid of SARS-CoV-2 (Novel Coronavirus PCR Fluorescence Diagnostic Kit, BioGerm Medical Biotechnology). She was treated with arbidol (0.2 g orally every 8 h) and moxifloxacin (0.4 g orally every day). On January 27, 2020, she developed respiratory distress with breath rate > 30 per min and oxygen saturation < 93% at rest. Low-flow and then high-flow oxygen therapy via nasal cannula was started, but the symptoms did not improve. The patient was transferred to the intensive care unit and noninvasive mechanical ventilation was implemented. Nine days later, noninvasive mechanical ventilation was stopped and high-flow oxygen therapy via nasal cannula was continued. Her blood examination showed a repeatedly increased leukocyte and neutrophil count, indicating a potential bacterial infection. Thus, tacrolimus was stopped on February 13, 2020. Her clinical condition improved gradually. The patient was transferred to a general ward and oxygen therapy was stopped on February 23, 2020, and tacrolimus was restarted on March 3, 2020.
On March 5, 2020, there were clusters of small red papules occurring on her right side of the waist accompanied by spontaneous sharp pain (rated as 8–9 on an 11-point numeric rating scale (NRS, 0–10), and a clinical diagnosis of herpes zoster was made. The patient received antiviral treatment with intravenous acyclovir (250 mg 3 times a day), and prednisone (20mg orally once a day) was restarted. Meantime, infrared therapy were applied for promoting rash absorption, and pregabalin (75 mg orally twice a day) and ibuprofen was used for analgesia. A follow-up RT-PCR test for nucleic acid of SARS-CoV-2 was performed and the results were positive. On March 7, 2020 (day 3 of herpes zoster), most papules were replaced with clear vesicles and the pain was slightly relieved. On March 20, 2020 (day 16 of herpes zoster), the clear vesicles had become cloudy and some eventually crusted. Intravenous acyclovir was replaced with oral acyclovir (400 mg 5 times a day) and discontinued 7 days later. On March 25, 2020 (day 21 of herpes zoster), her skin lesions resolved (Figure 1). However, she continued to have persistent pain in the right 10 to 12 lumbar dermatomes. She experienced constant burning pain (NRS 5), intermittent electric shocks (NRS 7, >20 times a day, a 5-minute duration each). The dosage of pregabalin was increased to 150 mg orally twice a day. The constant pain decreased to 3/10 within 2 weeks. The intensity of intermittent pain also decreased to 5/10 (3 times a day, a 3-minute duration each). On April 23, 2020, the patient was discharged to an isolation centre for 14 days of quarantine after recovery from COVID-19. On July 6, 2020 (4 months after the onset of rash), the patient received a follow-up telephone call and reported that she still had intermittent pain in the right 10 to 12 lumbar dermatomes (NRS 3, 1–2 times per day, seconds for each episode).
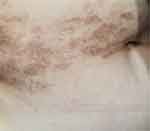 |
Figure 1 Herpes zoster in a right-sided T10-12 dermatomal distribution. The vesicular crusts has fallen off leaving temporary pigmentation. |
Discussion
Postherpetic neuralgia is one of the most common causes of neuropathic pain, which affects up to a third of patients with herpes zoster.9 It is reported that herpes zoster cases substantially increase during COVID-19 pandemic.8 Because of limited knowledge about pathogenesis of SARS-CoV-2 infection, it is challenging to treat acute herpes zoster and prevent postherpetic neuralgia in COVID-19 patients. Here, we report the clinical features, treatment and prognosis of herpes zoster in an elderly patient with critical COVID-19, indicating the importance of early antiviral therapy.
Herpes zoster is caused by reactivation of latent varicella zoster virus. A decline in cell-mediated immune response is thought to play an important role in the pathogenesis of herpes zoster.10 Decreased cell-mediated immune responses can be induced by advancing age, medical treatments or immunosuppressive illnesses.11 In COVID-19 patients, SARS-CoV-2 infection may decrease primarily T lymphocytes, especially CD4+ and CD8+ T cells.12 In addition, the current patient had a history of myasthenia gravis and had been receiving tacrolimus therapy. Myasthenia gravis is the most common humoral and cellular immune-mediated neuromuscular disorder. Dysregulation of regulatory T cells and elevated levels of pre-inflammatory cytokines such as IL-6 have been noted in myasthenia gravis patients.13 Tacrolimus, a calcineurin inhibitor, is one of the most effective agents in controlling the overall regulatory T cells, which can result in the inhibition of expression of IL-6. Earlier reports have described herpes zoster occurs before or after onset of COVID-19 symptoms with lymphopenia.7,8 However, in this patient, herpes zoster occurred in the recovery period of COVID-19 and the results of clinical laboratory measurements were almost normal. This observation indicates the variety of COVID-19 and herpes zoster co-occurrence, and the complexity of the underling mechanisms.
Although herpes zoster is mostly self-limiting, antiviral therapy needs to be considered in patients with severe diseases such as COVID-19. Among antivirals for treatment of herpes zoster licensed by the Food and Drug Administration (FDA), acyclovir is most commonly used. In the previous studies, acyclovir has been shown to reduce virus shedding, stop new lesion formation and hasten resolution of lesions.14,15 In the current report, the administration of acyclovir was started within 24 hours of rash onset and lasted for 21 days. It is recommended that antiviral treatment initiate within 72 hours of onset of the rash, and is usually given for 7 days.14 The principle of antiviral treatment in COVID-19 patients with herpes zoster might be the same as in other patients with the condition. In patients with complications of herpes zoster, which might be more common in COVID-19 patients, the course of treatment needs to be extended. Considering the side effects of acyclovir such as hepatotoxicity and nephrotoxicity, liver and kidney function should be monitored regularly. Under certain circumstances such as contraindications of acyclovir, vitamin C and quercetin might be considered as alternative treatments, which have been proved to display immunomodulatory function and anti-viral effect.16 In addition, the patients with herpes zoster should be placed on contact precautions to avoid varicella zoster virus transmission to susceptible persons such as other COVID-19 patients.
Pain management is one of the cornerstones of treatment of both acute herpes zoster and postherpetic neuralgia. For mild to moderate pain, analgesics such as paracetamol and nonsteroidal anti-inflammatory drugs (NSAIDs) are appropriate. For severe pain, opioid analgesics such as oxycodone or morphine are options.15 In the first line treatment of postherpetic neuralgia, tricyclic antidepressants (like nortriptyline or amitriptyline), anticonvulsant (like gabapentin or pregabalin) and topically administered lidocaine may be used.17 Because the patient in the current report has a history of myasthenia gravis, drugs affecting the neuromuscular junction like anticonvulsants need to be used with caution. Although one anticonvulsant gabapentin has been widely reported to induce an exacerbation of myasthenic symptoms,18–20 another anticonvulsant pregabalin is well tolerant in the current case. Thus, in patients with COVID-19, the comorbidity and the side effects of analgesics should worth further attention to and be treated case by case. Where a single treatment alone is ineffective, combined modalities are recommended.
In summary, this case draws attention to postherpetic neuralgia in COVID-19 patients. For COVID-19 patients with herpes zoster, antiviral treatment should be started as early as possible and lastlonger than a typical course. The use of analgesics should be based on the dermatome involved, severity of pain and the comorbidities. Our report indicates that longer term outcomes of pain management in COVID-19 patients should be monitored in future studies.
Acknowledgments
The authors thank the patient for providing informed consent and all the healthcare workers involved in the care of the patient.
Disclosure
The authors report no conflicts of interest in this work.
References
1. WHO. Coronavirus disease 2019 (COVID-19) situation report-188; 2020. Available from: https://www.who.int/docs/default-source/coronaviruse/situation-reports/20200726-covid-19-sitrep-188.pdf?sfvrsn=f177c3fa_2.
2. Zheng KI, Feng G, Liu WY, Targher G, Byrne CD, Zheng MH. Extra-pulmonary complications of COVID-19: a multi-system disease? J Med Virol. 2020. doi:10.1002/jmv.26294
3. Carfì A, Bernabei R, Landi F. Persistent symptoms in patients after acute COVID-19. JAMA. 2020;324(6):603–605. doi:10.1001/jama.2020.12603
4. Kemp HI, Laycock H, Costello A, Brett SJ. Chronic pain in critical care survivors: a narrative review. Br J Anaesth. 2019;123(2):e372e84. doi:10.1016/j.bja.2019.03.025
5. Kemp HI, Corner E, Colvin LA. Chronic pain after COVID-19: implications for rehabilitation. Br J Anaesth. 2020.
6. Guan WJ, Ni ZY, Hu Y, et al. Clinical characteristics of coronavirus disease 2019 in China. N Engl J Med. 2020;382(18):1708–1720. doi:10.1056/NEJMoa2002032
7. Elsaie ML, Youssef EA, Nada HA. Herpes zoster might be an indicator for latent COVID 19 infection. Dermatol Ther. 2020;e13666.
8. Ertugrul G, Aktas H. Herpes zoster cases increased during COVID-19 outbreak. Is it possible a relation? J Dermatolog Treat. 2020;1. doi:10.1080/09546634.2020.1789040
9. Weaver BA. The burden of herpes zoster and postherpetic neuralgia in the United States. J Am Osteopath Assoc. 2007;107(3 Suppl 1):S27.
10. Arvin AM. Humoral and cellular immunity to varicella-zoster virus: an overview. J Infect Dis. 2008;197(Suppl 2):S5860. doi:10.1086/522123
11. Gnann JW
12. Chen G, Wu D, Guo W, et al. Clinical and immunological features of severe and moderate coronavirus disease 2019. J Clin Invest. 2020;130(5):2620–2629. doi:10.1172/JCI137244
13. Danikowski KM, Jayaraman S, Prabhakar BS. Regulatory T cells in multiple sclerosis and myasthenia gravis. J Neuroinflammation. 2017;14(1):117.
14. Cohen JI. Clinical practice: herpes zoster. N Engl J Med. 2013;369(3):255–263. doi:10.1056/NEJMcp1302674
15. Le P, Rothberg M. Herpes zoster infection. BMJ. 2019;364:k5095. doi:10.1136/bmj.k5095
16. Colunga Biancatelli RML, Berrill M, Catravas JD, Marik PE. Quercetin and Vitamin C: an experimental, synergistic therapy for the prevention and treatment of SARS-CoV-2 related disease (COVID-19). Front Immunol. 2020;11:1451.
17. Argoff CE. Review of current guidelines on the care of postherpetic neuralgia. Postgrad Med. 2011;123(5):134–142. doi:10.3810/pgm.2011.09.2469
18. Scheschonka A, Beuche W. Treatment of post-herpetic pain in myasthenia gravis: exacerbation of weakness due to gabapentin. Pain. 2003;104(12):423–424. doi:10.1016/S0304-3959(03)00004-6
19. Boneva N, Brenner T, Argov Z. Gabapentin may be hazardous in myasthenia gravis. Muscle Nerve. 2000;23(8):1204–1208. doi:10.1002/1097-4598(200008)23:8<1204::AID-MUS7>3.0.CO;2-H
20. Sheen VL, Ohaegbulam C, Rencus T, Tandon D. Gabapentin-induced exacerbation of myasthenia gravis. Muscle Nerve. 2010;42(1):149. doi:10.1002/mus.21708
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
