Back to Journals » Journal of Asthma and Allergy » Volume 14
Health-Related Quality of Life and Productivity Among US Patients with Severe Asthma
Authors Soong W, Chipps BE, O'Quinn S , Trevor J, Carr WW, Belton L, Trudo F, Ambrose CS
Received 20 February 2021
Accepted for publication 9 June 2021
Published 25 June 2021 Volume 2021:14 Pages 713—725
DOI https://doi.org/10.2147/JAA.S305513
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Amrita Dosanjh
Weily Soong,1 Bradley E Chipps,2 Sean O’Quinn,3 Jennifer Trevor,4 Warner W Carr,5 Laura Belton,6 Frank Trudo,7 Christopher S Ambrose3
1Alabama Allergy & Asthma Center, Birmingham, AL, USA; 2Capital Allergy & Respiratory Disease Center, Sacramento, CA, USA; 3BioPharmaceuticals Medical, AstraZeneca, Gaithersburg, MD, USA; 4University of Alabama at Birmingham, Birmingham, AL, USA; 5Allergy & Asthma Associates of Southern California, Mission Viejo, CA, USA; 6Biostatistics, AstraZeneca, Cambridge, UK; 7BioPharmaceuticals Medical, AstraZeneca, Wilmington, DE, USA
Correspondence: Weily Soong
Alabama Allergy & Asthma Center, 504 Brookwood Boulevard, Birmingham, AL, 35209, USA
Tel +1 205 871-9661
Fax +1 205 870-1621
Email [email protected]
Background: Health-related quality of life (HRQoL) and productivity of patients with confirmed severe asthma (SA) have not been well characterized in large, real-world populations.
Purpose: To characterize SA impact on HRQoL, work productivity, and activity impairment in a large, real-world cohort in the United States (US).
Methods: CHRONICLE is an observational study of specialist-treated adults (≥ 18 years) in the US with SA receiving biologics or maintenance systemic corticosteroids (mSCS), or those persistently uncontrolled by high-dosage inhaled corticosteroids with additional controllers (HD ICS+). At enrollment, patients completed the St. George’s Respiratory Questionnaire (SGRQ) and Work Productivity and Activity Impairment (WPAI) questionnaire. Results were analyzed for those enrolled between February 2018 and February 2020.
Results: Among patients who completed enrollment questionnaires (n = 1109), mean age was 54 years and most were women (70%). Among SGRQ respondents (n = 960), mean (SD) total score was 43 (23); 51% reported good/very good health. Among WPAI respondents (n = 1057; 566 employed), mean (SD) overall work impairment was 21% (25). Patients receiving biologics (vs mSCS, HD ICS+ only) had better SGRQ total scores (38 vs 59, 48) and lower work impairment (17% vs 34%, 27%). Patients with better SGRQ activity scores relative to symptom scores had better SGRQ impacts scores, total scores, and reported better overall health.
Conclusion: SA significantly affects HRQoL, work productivity, and activity. The SGRQ is a valuable research instrument for evaluating HRQoL in SA. Due to its association with HRQoL and overall health, activity impairment should be a focus when monitoring patients’ disease control.
Study Registration: ClinicalTrials.gov Identifier: NCT03373045.
Keywords: real-world, St. George’s Respiratory Questionnaire, SGRQ, WPAI, symptoms, activity
Introduction
Asthma is a heterogeneous disease for which health care providers (HCPs) use a combination of subjective symptom-based measurements and objective measurements to guide its diagnosis and management.1 For the 5–10% of patients who have severe disease,2 the impact of severe asthma (SA) on health-related quality of life (HRQoL) is significant but has been underappreciated in asthma research, where the focus is often on clinical measures of severity such as exacerbation rate, patient-reported near-term symptom control, and pulmonary function testing.3
Although current asthma treatment guidelines endorse assessment of respiratory symptoms and associated impairments, activity limitation, and asthma reliever medication use to determine asthma control,1 broader measures of patient experience and other assessments of HRQoL are challenging to quantify and often poorly correlate with clinical assessments. As a result, they are often secondary objectives in asthma research.4 However, in real-world clinical practice, patient experience is a critical aspect of asthma treatment and management. Respiratory symptoms, comorbidities, and associated functional impairments have been shown to contribute to overall reduced HRQoL.3,5 With SA, most patients experience functional limitations that result in a reduced ability to perform daily activities and impaired work productivity.5 As a result, an improved understanding of the patient experience through measures of lifestyle impacts, HRQoL, and work productivity could help improve clinical decision-making in the treatment of SA. Furthermore, there is value in understanding whether specific aspects of the patient experience with SA have a disproportionate influence on overall HRQoL and thus warrant greater consideration in SA treatment decisions.
Two large, United States (US)-based, observational studies previously assessed HRQoL in patients with SA using the Asthma Quality of Life Questionnaire (AQLQ). With the AQLQ, a score of 1 indicates severe impairment and 7 indicates no impairment.6 In the most recent Severe Asthma Research Program study conducted in 2012, adults with SA had mean (standard deviation [SD]) total AQLQ score at baseline of 4.6 (1.3), lower than that observed for adults with non-severe asthma (5.5 [1.1]).7 In the TENOR II follow-up assessment in 2013–2014, the mean (SD) overall Mini-AQLQ score was 4.2 (1.3), and domain scores were lowest for the environmental stimuli domain (mean [SD]: 3.7 [1.7]).8 Despite these prior observations, there are no data characterizing the impact of SA on HRQoL in a large, contemporary, US real-world patient population.
Although the AQLQ has been the traditional instrument used to examine HRQoL in asthma, several studies in SA have utilized the Saint George’s Respiratory Questionnaire (SGRQ). Recent randomized controlled trials (RCTs) of biologic therapies have shown that the SGRQ performs well in patients with SA.9–14 The SGRQ was developed for both asthma and chronic obstructive pulmonary disease (COPD), and has been validated in patients with SA.10,15 It has 50 items that cover the aspects of symptoms (and their frequency and severity), activities (eg, those that cause or are caused by breathlessness), and impacts (eg, social, emotional, work/school interference, self-care, medication concerns, overall self-perception on HRQoL), with lower scores reflecting better HRQoL.16 In addition, a total score indicates overall health status.16 The recent use of the SGRQ in clinical trials in patients with SA underscores the need to better understand the HRQoL impairments experienced by patients with SA in a real-world cohort of patients using this assessment. Although global RCTs are the gold standard for establishing treatment safety and efficacy, RCTs in SA do not well represent the overall patient population due to study inclusion and exclusion criteria and limited country-specific samples; Brown et al recently estimated that on average only 10% of patients with SA were eligible for inclusion in RCTs.17 Thus, there is great value in studying a real-world patient population with SA that is less selected than cohorts enrolled in RCTs and more representative of patients treated by SA specialists in day-to-day clinical practice.
In addition to HRQoL impairments, patients with SA may experience losses in work productivity. The Work Productivity and Activity Impairment Asthma (WPAI-Asthma) questionnaire is often used to evaluate work productivity and activity losses due to asthma. The WPAI-Asthma has been found to perform well in patients with SA and correlates strongly with other asthma-specific outcomes.18 As with HRQoL, there are no data characterizing the impact of SA on work productivity in a large, contemporary, real-world patient population.
CHRONICLE is a real-world, prospective, noninterventional study of US patients with confirmed SA who are treated by allergists/immunologists or pulmonologists.19 CHRONICLE is designed to observe routine clinical practice with minimal inclusion and exclusion criteria other than those required to confirm the diagnosis of SA and ongoing subspecialist treatment. To collect data in the most naturalistic manner possible, there are no required tests, imaging, or study visits beyond the initial enrollment visit. The design of CHRONICLE has been described in detail.19 The study uses the SGRQ to evaluate HRQoL and the WPAI-Asthma to assess work productivity. The goal of the present analysis was to utilize data collected in the first 2 years of CHRONICLE to characterize HRQoL, work productivity, and activity impairment in a large, real-world population of US patients with specialist-confirmed and specialist-treated SA.
Methods
Study Participants
Briefly, patients eligible for enrollment in CHRONICLE must have a diagnosis of SA, as determined by subspecialists, for ≥12 months prior to enrollment, consistent with European Respiratory Society/American Thoracic Society guidelines.2,19 Patients must be adults (aged ≥18 years) currently receiving care from subspecialist physicians at a participating site. In addition, patients must meet one or more of the following criteria: (1) current use of US Food and Drug Administration-approved monoclonal antibody therapy for SA; (2) use of systemic corticosteroids or other systemic immunosuppressants for ≥50% of the prior 12 months for SA; or (3) uncontrolled asthma while receiving high-dosage inhaled corticosteroids with additional controllers (HD ICS+ only). To minimize selection bias and reflect a real-world population as much as possible, exclusion criteria were intentionally kept minimal, and specialists at participating sites approached all protocol-eligible patients. Patients were excluded if they were not willing or able to provide written informed consent, were not fluent in English or Spanish, were unable to complete the web-based patient-reported outcome (PRO) questionnaires, or received an investigational therapy (for asthma, allergy, atopic disease, or eosinophilic disease) as part of a clinical trial in the 6 months before enrollment. The CHRONICLE study protocol received central institutional review board (Advarra, Columbia, MD, USA) approval on November 3, 2017, and was registered on ClinicalTrials.gov on December 14, 2017 (NCT03373045).
Data Collection
Data, including patient demographics, medical history, and current asthma management, are collected from the specialist at enrollment via an electronic case report form. After enrollment, the specialist updates the patient’s medical history and medical management every 6 months. Enrolled patients receive emails to complete web-based questionnaires at enrollment and at subsequent regular intervals. The WPAI-Asthma is completed every 3 months, and the SGRQ is completed every 6 months. In addition, the specialist rates the patient’s asthma control status (controlled vs uncontrolled) at enrollment and every 6 months during the study.
For the present study, we used data from patients enrolled during the first 2 years of CHRONICLE (February 2018–February 2020) to assess HRQoL based on SGRQ responses, as well as work productivity and activity based on WPAI-Asthma responses. As noted above, the SGRQ measures HRQoL in individuals with airway disease.10,11 There are 50 items divided into 3 components: (1) respiratory symptoms (Symptoms); (2) activity limitations (Activity); and (3) psychosocial impacts (Impacts) in the prior 3 months.11 Scores range from 0 to 100, with 0 representing perfect health and 100 representing maximum impairment. Overall health is captured via a separately scored item that uses a 5-point verbal rating (ie, very good, good, fair, poor, very poor). In addition, a free-text question captures unstructured patient responses about other important activities that their respiratory condition prevents them from doing beyond those mentioned in the questionnaire items.
The WPAI-Asthma is an asthma-specific, patient-reported measure of work absenteeism, work presenteeism (reduced productivity while working due to illness), total work productivity loss (absenteeism plus presenteeism), and overall activity impairment over the prior 7 days.20 The WPAI questionnaire score represents the percentage of impairment, from 0 to 100%. Activity limitation is evaluated in all respondents, whereas questions about work productivity are applied only to patients who report being employed.
Statistical Analysis
Descriptive statistics (eg, percentage, mean, SD) were calculated for patient demographic and clinical characteristics at enrollment for all enrolled and those who completed the enrollment questionnaires. There was no imputation done for missing data. Mean (SD) scores were calculated for the SGRQ and WPAI at enrollment. SGRQ and WPAI scores were also assessed by enrollment treatment class (ie, biologics, maintenance systemic corticosteroids [mSCS], or HD ICS+ only).
Additional analyses of SGRQ scores at baseline assessed the correlation between total summary score and patients’ overall self-assessment of their health and the frequency of specific disease effects captured by true/false questions. A word cloud was used to visualize the frequency of concepts from the free-text question. Patient clusters were identified in an exploratory fashion based on concordance or discordance of SGRQ component scores (ie, Symptoms, Activity, and Impacts).
SGRQ and WPAI scores were also analyzed for subgroups of interest utilizing questionnaire responses collected at or subsequent to enrollment. Among patients not receiving biologics or systemic corticosteroids (SCS) during the study, scores were assessed by highest blood eosinophil count (<150, 150- <300, ≥300 cells/μL) and highest total immunoglobulin E level (<30, 30–700, >700 IU/mL). For the subset of patients with continuous biologic use for ≥12 months prior to enrollment, SGRQ and WPAI scores were assessed among patient groups stratified by exacerbation rate during the year prior to enrollment (≥1 versus 0) and HCP control assessment (controlled versus uncontrolled). SGRQ and WPAI scores were also assessed by clinical remission status among patients who received biologics for ≥12 months during the study. Clinical remission was defined consistent with the recent framework by Menzies-Gow et al using available study endpoints: During the latest 12 months of biologic use, the absence of SCS use (for maintenance or acute exacerbation treatment), all (and at least 2) patient Asthma Control Test scores ≥20, and confirmation from the patient (via the Asthma Control Test) and HCP that the patient’s asthma was controlled in the most recent time interval.21
All analyses were conducted using SAS version 9.4 (SAS Institute, Cary, NC, USA).
Results
A total of 1884 patients enrolled in CHRONICLE from February 2018 to February 2020 with all enrollment data entry forms completed. Of these patients, 1109 completed at least one online questionnaire at enrollment. Supplementary Table 1 shows the characteristics of all patients who were approached for inclusion (n = 2722) and enrolled (n = 2026). Patients who were approached but did not enroll were less likely to have commercial health insurance (57% vs 60%) and more likely to be uncontrolled on HD ICS+ only (38% vs 30%) than those who enrolled; both groups had a similar mean number of exacerbations in the prior year. Supplementary Table 2 shows the characteristics of enrolled patients who did (n = 1109) or did not (n = 775) complete the enrollment PRO questionnaires. Among enrolled patients, those who did not complete enrollment questionnaires were more likely to report Black race (23% vs 15%) or Hispanic or Latino ethnicity (11% vs 7%), be receiving care from a pulmonologist (58% vs 47%), have Medicaid insurance (14% vs 7%), and were less likely to be employed full-time (36% vs 42%) or be receiving biologic therapy (61% vs 67%) than enrollees who completed enrollment questionnaires; the mean number of exacerbations in the prior year was slightly higher among those not completing questionnaires (1.3 vs 1.1).
Among enrollees who completed at least one online PRO questionnaire at enrollment (n = 1109), the mean age was 54 years, and most patients were women (70%) and reported White race (80%) (Table 1). Similar percentages of patients received care from pulmonologists (47%) and allergists/immunologists (43%). At enrollment, 52% of patients were employed. On average, patients experienced 1.1 exacerbations in the 12 months prior to enrollment. Patient demographic and clinical characteristics were generally similar among those who completed enrollment questionnaires and the overall enrolled population, although patients who completed the questionnaires were slightly more likely to report White race (80% vs 75% of the overall study population), receive care from an allergist/immunologist (43% vs 39%), and have commercial health care insurance (64% vs 60%). A total of 960 patients fully completed the SGRQ at baseline. The mean (SD) total SGRQ score for the overall study population was 43 (23) and 51% of patients rated their health as good or very good. SGRQ scores by treatment category at enrollment are shown in Figure 1. SGRQ scores were best for patients receiving biologics and worst for those receiving mSCS. Aligned with SGRQ scores, the percentage of patients who reported their health to be good or very good was 55% for patients receiving biologics, 46% for patients receiving HD ICS+, and 22% for patients receiving mSCS.
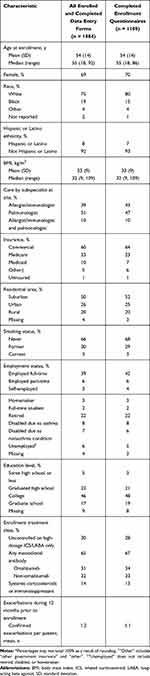 |
Table 1 Demographics and Clinical Characteristics of Enrolled Patients* |
The WPAI was completed at enrollment by 1057 patients, of whom 566 (54%) reported being employed. For mean (SD) impairment due to asthma among employed patients, 4% (14) of work time was missed, and there was a 19% (24) loss in productivity while at work, resulting in 21% (25) overall work impairment (Figure 2). Among all patients completing the WPAI, mean (SD) activity impairment due to asthma was 34% (31). WPAI results by treatment category at enrollment are shown in Figure 2. Across all categories, patients receiving mSCS reported the highest impairment and patients receiving biologics reported the lowest impairment.
SGRQ data were investigated for further insights. SGRQ total and component scores (ie, Symptoms, Activity, and Impacts) were consistent with patient-reported health; however, there was significant variability in individual patient scores (Figure 3). The most-reported activity impairments and psychosocial impacts were shortness of breath from hills, sports or exercise, severe exertion, and stairs (Figure 4). Patients’ free-text responses regarding additional factors impacting their quality of life (beyond those summarized in Figure 4) reinforced the importance of physical activity limitations (eg, going places, walking, playing, exercise, running) (Figure 5). Other major themes included environmental exposures (eg, pollen, perfume, smoke, humidity, weather) and relationships with people (eg, time spent with family and/or friends, and sexual relationships).
 |
Figure 5 Word cloud of patient-reported free-text responses to the SGRQ prompt for “other activities that your respiratory problems may prevent you from doing.”*. Abbreviations: SGRQ, St. George’s Respiratory Questionnaire. Notes: *The more frequently a specific word was reported, the bigger and bolder it appears in the word cloud. Adapted from WordItOut (http://worditout.com). |
Among SGRQ components, there were strong correlations between Symptoms and Impacts scores and between Activity and Impacts scores. The greatest interpatient variability occurred when contrasting Symptoms and Activity scores (median difference: 14.6). Therefore, we identified 3 patient clusters using a threshold of the median difference (15 points) between Symptoms and Activity scores (Figure 6A). Cluster 1, which represented most patients, were those with similar Symptoms and Activity scores (difference <15). Cluster 2 patients had better Activity scores relative to their Symptoms scores (difference ≥15), and Cluster 3 patients had better Symptoms scores relative to their Activity scores (difference ≥15). When assessed by cluster (Figure 6B), Cluster 2 patients had better mean Impacts scores and total scores than those in Cluster 1 and Cluster 3. A higher percentage of Cluster 2 patients reported good or very good health (66% vs 50% and 39% in clusters 1 and 3, respectively). Taken together, those with better Activity scores relative to Symptoms score on average had better HRQoL and overall health compared with those with better Symptoms scores relative to Activity Scores.
The results of subgroup analyses that were conducted to examine associations of SGRQ and WPAI scores with patients’ clinical status are shown in Supplementary Tables 3 and 4, respectively. Among patients not receiving biologics or SCS, SGRQ and WPAI scores were generally similar regardless of highest blood eosinophil count or total immunoglobulin E levels. Small sample sizes prevented robust comparisons on several endpoints. Among patients receiving biologics, HRQoL and work productivity were better for those who had no exacerbations (vs those with exacerbations), had controlled disease per HCP assessment (vs those with uncontrolled disease), or met the definition of clinical remission while receiving biologic treatment (vs those who did not).
Discussion
The current analysis represents the largest contemporary characterization of HRQoL and work productivity among US patients with specialist-confirmed SA. The findings demonstrate the substantial impact of SA on these aspects of the patient experience and provide further evidence suggesting that the SGRQ and WPAI are valuable tools for characterizing HRQoL and work productivity of patients with SA. These analyses also highlight differential impairment by treatment and provide insights into the relationships between impairment types in the SA population.
HRQoL and work productivity were higher among patients receiving biologic therapies and lower among those receiving mSCS and those uncontrolled while receiving HD ICS+ without biologic or mSCS use. The lower patient-reported HRQoL, employment, and work productivity among those receiving mSCS therapy may be a result of poorly controlled disease, greater symptom burden, and/or short-term and longer-term adverse consequences and comorbidities associated with mSCS therapy. Conversely, the higher patient-reported HRQoL and work productivity among those receiving biologic therapy may be attributable to better disease control and reduced side effects compared with SCS treatments. These conclusions are supported by patient responses to several individual SGRQ questions. Compared with patients receiving biologics and HC ICS+ only, patients receiving mSCS at enrollment were more likely to agree with the statements, “I have unpleasant side effects from my medication” (49% vs 19%, 17%) and “My treatment interferes with my life a lot” (27% vs 12%, 11%). In addition, only 9% of biologic recipients at enrollment agreed with the statement, “My treatment doesn’t help me very much,” compared with 23% and 19% of patients receiving mSCS and HD ICS+ only, respectively.
In the SGRQ components results, we noted that Impacts scores tended to be better than Symptoms or Activity scores, regardless of enrollment treatment group. We also found that patient-reported overall health did not correspond with HRQoL impairment, as patient-reported overall health was similar among patients treated with biologic and HD ICS+ only therapies, despite substantial HRQoL and productivity differences. Patients living with chronic diseases or disabilities can experience a process of behavioral, functional, and psychosocial adaptation to their limitations over time.22 Use of coping mechanisms, social comparison, and reframing personal expectations may influence perceptions of HRQoL.23 As a result, true disease impact on aspects of a patient’s well-being may differ from their global perception of disease impact.
Our findings are consistent with those of the Identification and Description of sEvere Asthma patients in a cross-sectionaL (IDEAL) study, a recent, multinational real-world study of patients with SA. In the IDEAL study, patients who had uncontrolled asthma based on exacerbations and the Asthma Control Questionnaire had worse work productivity and HRQoL by SGRQ than those with controlled asthma,24 with absolute scores similar to those observed in the CHRONICLE cohort. Both studies also demonstrated that work impairment from SA is primarily driven by productivity impairment while working (ie, presenteeism) rather than absenteeism.
The WPAI has been used to assess numerous chronic diseases, including SA. In a systematic review, overall work productivity impairment ranged from 6–40% and activity impairment ranged from 6–50% in 11 studies of allergies and asthma.25 The overall work impairment (21%) and activity impairment (34%) observed in our study population were within these broad ranges. Using an approach similar to that of Sadatsafavi et al,26 based on the median weekly income for full-time employees reported by the US Bureau of Labor Statistics for Q1 2020,27 the annual economic value of this work productivity impairment can be estimated at approximately $10,400 per full-time employed patient with SA. In addition, the differences observed by treatment group suggest increased economic productivity of $4900 and $8400 per full-time employed patient receiving biologics relative to those receiving HD ICS+ only or mSCS, respectively.
Furthermore, our cluster analysis of SGRQ component scores suggests that limitation of physical activity, especially exertional activity, significantly affects HRQoL among patients with SA. When engaging in treatment decision-making, clinicians should consider which aspects of HRQoL affect individual patients the most, with a focus on activity impairment.
There is no single, ideal instrument to measure HRQoL in asthma that covers all possible categories. Given the increasing use of the SGRQ in clinical trials in patients with SA, our findings help contextualize SGRQ results by characterizing the impairments experienced by patients with SA in a real-world setting and examining the relationships between domains. The findings of this and other studies support the use of the SGRQ, which has been previously validated in SA,10,15 as an appropriate measure of HRQoL in SA. The SGRQ captures a wide range of asthma experiences that may help support shared decision making between the HCP and patient in a real-world setting.
SGRQ scores correlated with patient-reported health, and free-text descriptions of specific impairments generally aligned with the concepts captured by the SGRQ. Furthermore, as seen in this study, the SGRQ provides sufficient detail to capture the specific patient experiences that might be overlooked by a single summary HRQoL question. However, free-text responses also highlighted the relevance of environmental exposures and relationship activities, which are largely absent from the SGRQ.16 The exploratory subgroup analyses further confirmed the performance of the SGRQ in patients with SA, as SGRQ scores were associated with exacerbation rate, HCP assessment of asthma control, and remission status. The SGRQ can effectively evaluate the impacts of SA on patient experiences in the research setting. In addition, as noted above, the current findings provide important insights for the contemporary clinical care of patients with SA. The goal should be to personally tailor treatment for each patient with SA based on the patient’s individual disease management and quality-of-life goals.
Limitations
Patients who did not complete online enrollment questionnaires were not included in the analysis, and some patients had missing data for the SGRQ and WPAI. The broader generalizability of the current results is unknown given these missing data as well as the differences observed in patients approached for enrollment, those enrolled, and those completing PRO questionnaires after enrollment. The available data suggest that those not completing questionnaires had more poorly controlled disease, suggesting that the reported SGRQ and WPAI results may underestimate the true burden of disease among the broader population of US patients with specialist-treated SA. For feasibility and data quality, data collection for several outcomes of interest was limited to 12 months prior to enrollment. Although efforts were undertaken to include diverse study sites, selection of sites was nonrandom; therefore, the generalizability of the results is unknown. However, we have previously demonstrated that CHRONICLE site and provider characteristics are generally similar to other characterized samples of US asthma specialists.19 The study does not include children, adolescents, and patients with SA treated by non-subspecialists, and therefore cannot describe these populations. As the purpose of this study was descriptive, analyses comparing treatment groups were not adjusted for potential baseline differences between the groups. Furthermore, it was not possible to compare the performance of the SGRQ with that of other HRQoL instruments because SGRQ is the only HRQoL questionnaire used in CHRONICLE.
A final important point is that the SGRQ does not address the risk of long-term and potentially permanent side effects of SCS treatment. Patients treated with mSCS or repeated bursts of SCS for exacerbations can experience future adverse consequences,28 which can result in functional limitations, reduced ability to perform daily activities, and reduced HRQoL in the future.1 From this perspective, the full HRQoL effects of SA may be underestimated by the SGRQ or any other instrument that does not account for the future effects of current/recent SCS treatment.
Conclusions
SA has a significant impact on HRQoL, work productivity, and activity impairment, despite subspecialist treatment. The SGRQ is a valuable research tool for evaluating HRQoL in SA. Study findings highlight the utility of understanding which specific aspects of HRQoL most affect a patient (symptoms predominant vs activity impairment predominant) when setting patient asthma management goals. Activity limitations should be a focus when monitoring disease control and treatment effectiveness among patients with SA. Furthermore, clinicians should consider the potential psychosocial context and adaptations to health status when evaluating patients with SA. Future research from CHRONICLE will provide data from a larger patient population and longer follow-up period to further elucidate factors affecting HRQoL and work productivity in patients with SA.
Abbreviations
AQLQ, Asthma Quality of Life Questionnaire; COPD, chronic obstructive pulmonary disease; HCPs, health care providers; HD ICS+, high-dosage inhaled corticosteroids with additional controllers; HRQoL, health-related quality of life; mSCS, maintenance systemic corticosteroids; SA, severe asthma; SCS, systemic corticosteroids; SD, standard deviation; SGRQ, St. George’s Respiratory Questionnaire; TENOR, The Epidemiology and Natural History of Asthma: Outcomes and Treatment Regimens; US, United States; WPAI, Work Productivity and Activity Impairment.
Data Sharing Statement
CHRONICLE is an ongoing study; individual de-identified participant data cannot be shared until the study concludes. The full study protocol is available upon request of the corresponding author. Individuals who were or were not involved in the study may submit publication proposals to the study’s Publication Steering Committee by contacting the corresponding author.
Ethics Approval and Informed Consent
The CHRONICLE study protocol received central institutional review board (Advarra, Columbia, MD, USA) approval on November 3, 2017, and was registered on ClinicalTrials.gov on December 14, 2017 (NCT03373045). Patients completed written informed consent. The CHRONICLE study is being performed according to ethical principles consistent with the Declaration of Helsinki, International Conference on Harmonisation of Technical Requirements for Registration of Pharmaceuticals for Human Use (ICH) Good Clinical Practices, Good Pharmacoepidemiology Practices, the Health Insurance Portability and Accountability Act (HIPAA), and applicable legislation for observational studies.
Acknowledgments
The authors would like to thank Sheila Brillhart, a patient with severe asthma, for her review and suggestions regarding the manuscript. Medical writing support was provided by Karen Kurtyka, MPH, of Oxford PharmaGenesis Inc. (Newtown, PA, USA), which was in accordance with Good Publication Practice (GPP3) guidelines and funded by AstraZeneca (Wilmington, DE, USA). The abstract of this paper was presented at the 2020 American Academy of Allergy, Asthma, & Immunology Annual Meeting as a poster presentation with interim findings. The poster’s abstract was published in “Programs and Abstracts of Papers to be Presented During Scientific Sessions: 2020 AAAAI Annual Meeting” in the Journal of Allergy and Clinical Immunology: (DOI: 10.1016/j.jaci.2018.12.971).
Author Contributions
All authors contributed to the conception and design of the CHRONICLE study and the current study. WS, BEC, SO, JT, WC, FT, and CSA contributed to the acquisition of the data. All authors contributed to data analysis, drafting or revising the article, have agreed on the journal to which the article was submitted, gave final approval of the version to be published, and agree to be accountable for all aspects of the work.
Funding
This work was supported by and the CHRONICLE study is funded by AstraZeneca. Four named authors are employees of AstraZeneca; therefore, AstraZeneca was involved in the study design, collection, analysis, and interpretation of data and the development and review of the manuscript. The decision to submit the manuscript for publication was made by the authors.
Disclosure
Christopher S. Ambrose, Laura Belton, Sean O’Quinn, and Frank Trudo are employees of AstraZeneca. Bradley E. Chipps is an advisor for, received consultancy fees from, and is on the speakers’ bureau for AstraZeneca, Boehringer Ingelheim, Genentech, GlaxoSmithKline, Novartis, Regeneron, and Sanofi Genzyme. Weily Soong is a consultant for and reports grants from Amgen, Avillion, Roche, AstraZeneca, Genentech, GlaxoSmithKline, Mandala, Novartis, Regeneron, Sanofi, and Teva; and is a speaker for AstraZeneca, GlaxoSmithKline, Regeneron, Sanofi, and Optinose. Jennifer Trevor is a consultant for AstraZeneca and is on advisory boards for AstraZeneca, GlaxoSmithKline, and Boehringer Ingelheim. Warner W. Carr is a speaker for AstraZeneca, Teva, Boehringer Ingelheim, Regeneron, and Sanofi; and a consultant for AstraZeneca, Teva, Boehringer Ingelheim, Regeneron, Sanofi, Circassia, CSL Behring, Genentech, GlaxoSmithKline, Horizon Pharma, Kaleo, Mylan, Pfizer, Shire, Meda, Baxalta, Novartis, Greer Laboratories, Alcon Laboratories, Valeant Pharmaceuticals, Grifols, Optinose, and Aerocrine. The authors report no other conflicts of interest in this work.
References
1. Global Initiative for Asthma (GINA). Global strategy for asthma management and prevention; 2020. Available from: https://ginasthma.org/wp-content/uploads/2020/04/GINA-2020-full-report_-final-_wms.pdf.
2. Chung KF, Wenzel SE, Brozek JL, et al. International ERS/ATS guidelines on definition, evaluation and treatment of severe asthma. Eur Respir J. 2014;43(2):343–373. doi:10.1183/09031936.00202013
3. McDonald VM, Hiles SA, Jones KA, et al. Health-related quality of life burden in severe asthma. Med J Aust. 2018;209(S2):S28–S33. doi:10.5694/mja18.00207
4. Moy ML, Israel E, Weiss ST, et al. Clinical predictors of health-related quality of life depend on asthma severity. Am J Respir Crit Care Med. 2001;163:924–929. doi:10.1164/ajrccm.163.4.2008014
5. Stubbs MA, Clark VL, McDonald VM. Living well with severe asthma. Breathe (Sheff). 2019;15(2):e40–e49. doi:10.1183/20734735.0165-2019
6. Juniper EF, Guyatt GH, Ferrie PJ, et al. Measuring quality of life in asthma. Am Rev Respir Dis. 1993;147(4):832–838. doi:10.1164/ajrccm/147.4.832
7. Teague WG, Phillips BR, Fahy JV, et al. Baseline features of the Severe Asthma Research Program (SARP III) cohort: differences with age. J Allergy Clin Immunol Pract. 2018;6(2):545–554 e4. doi:10.1016/j.jaip.2017.05.032
8. Chipps BE, Haselkorn T, Paknis B, et al. More than a decade follow-up in patients with severe or difficult-to-treat asthma: the epidemiology and natural history of asthma: outcomes and treatment regimens (TENOR) II. J Allergy Clin Immunol. 2018;141(5):1590–1597 e9. doi:10.1016/j.jaci.2017.07.014
9. Farne HA, Wilson A, Powell C, et al. Anti-IL5 therapies for asthma. Cochrane Database Syst Rev. 2017;9:CD010834.
10. Nelsen LM, Cockle SM, Gunsoy NB, et al. Impact of exacerbations on St George’s Respiratory Questionnaire score in patients with severe asthma: post hoc analyses of two clinical trials and an observational study. J Asthma. 2020;57(9):1006–1016. doi:10.1080/02770903.2019.1630640
11. O’Quinn S, Xu X, Hirsch I. Daily patient-reported health status assessment improvements with benralizumab for patients with severe, uncontrolled eosinophilic asthma. J Asthma Allergy. 2019;12:21. doi:10.2147/JAA.S190221
12. Panettieri RA
13. Chupp GL, Bradford ES, Albers FC, et al. Efficacy of mepolizumab add-on therapy on health-related quality of life and markers of asthma control in severe eosinophilic asthma (MUSCA): a randomised, double-blind, placebo-controlled, parallel-group, multicentre, phase 3b trial. Lancet Respir Med. 2017;5(5):390–400. doi:10.1016/S2213-2600(17)30125-X
14. Pavord ID. Mepolizumab, quality of life, and severe eosinophilic asthma. Lancet Respir Med. 2017;5(5):362–363. doi:10.1016/S2213-2600(17)30132-7
15. Nelsen LM, Vernon M, Ortega H, et al. Evaluation of the psychometric properties of the St George’s Respiratory Questionnaire in patients with severe asthma. Respir Med. 2017;128:42–49. doi:10.1016/j.rmed.2017.04.015
16. Jones PW, Quirk FH, Baveystock CM. The St George’s Respiratory questionnaire. Respir Med. 1991;85(Suppl B):
17. Brown T, Jones T, Gove K, et al. Randomised controlled trials in severe asthma: selection by phenotype or stereotype. Eur Respir J. 2018;52(6):1801444. doi:10.1183/13993003.01444-2018
18. Chen H, Blanc PD, Hayden ML, et al. Assessing productivity loss and activity impairment in severe or difficult-to-treat asthma. Value Health. 2008;11(2):231–239. doi:10.1111/j.1524-4733.2007.00229.x
19. Ambrose CS, Chipps BE, Moore WC, et al. The CHRONICLE study of US adults with subspecialist-treated severe asthma: objectives, design, and initial results. Pragmat Obs Res. 2020;11:77–90. doi:10.2147/POR.S251120
20. Reilly MC, Zbrozek AS, Dukes EM. The validity and reproducibility of a work productivity and activity impairment instrument. Pharmacoeconomics. 1993;4(5):353–365. doi:10.2165/00019053-199304050-00006
21. Menzies-Gow A, Bafadhel M, Busse WW, et al. An expert consensus framework for asthma remission as a treatment goal. J Allergy Clin Immunol. 2020;145(3):757–765. doi:10.1016/j.jaci.2019.12.006
22. Livneh H, Wilson LM, Duchesneau A, et al. Psychosocial adaptation to epilepsy: the role of coping strategies. Epilepsy Behav. 2001;2(6):533–544. doi:10.1006/ebeh.2001.0284
23. Sprangers MA, Schwartz CE. Integrating response shift into health-related quality of life research: a theoretical model. Soc Sci Med. 1999;48(11):1507–1515. doi:10.1016/S0277-9536(99)00045-3
24. Mullerova H, Cockle SM, Gunsoy NB, et al. Clinical characteristics and burden of illness among adolescent and adult patients with severe asthma by asthma control: the IDEAL study. J Asthma. 2020;1–12.
25. Miller PS, Hill H, Andersson FL. Nocturia work productivity and activity impairment compared with other common chronic diseases. Pharmacoeconomics. 2016;34(12):1277–1297. doi:10.1007/s40273-016-0441-9
26. Sadatsafavi M, Rousseau R, Chen W, et al. The preventable burden of productivity loss due to suboptimal asthma control: a population-based study. Chest. 2014;145(4):787–793. doi:10.1378/chest.13-1619
27. US Bureau of Labor Statistics. Median usual weekly earnings of full-time wage and salary workers by sex; 2020. Available from: https://www.bls.gov/charts/usual-weekly-earnings/usual-weekly-earnings-over-time-total-men-women.htm#.
28. Bleecker ER, Menzies-Gow AN, Price DB, et al. Systematic literature review of systemic corticosteroid use for asthma management. Am J Respir Crit Care Med. 2020;201(3):276–293. doi:10.1164/rccm.201904-0903SO
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.