Back to Journals » Neuropsychiatric Disease and Treatment » Volume 16
Functional Connectivity Density with Frequency-Dependent Changes in Patients with Diffuse Axonal Injury: A Resting-State Functional Magnetic Resonance Imaging Study
Authors Xia G , Hu Z, Zhou F , Duan W, Wang M, Gong H, He Y, Guan Y
Received 16 June 2020
Accepted for publication 19 October 2020
Published 12 November 2020 Volume 2020:16 Pages 2733—2742
DOI https://doi.org/10.2147/NDT.S267023
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Yuping Ning
Guojin Xia,1,2 Zhenzhen Hu,3 Fuqing Zhou,1,2 Wenfeng Duan,1,2 Min Wang,1,2 Honghan Gong,1,2 Yulin He,1,2 Yanxing Guan4
1Department of Radiology, The First Affiliated Hospital of Nanchang University, Jiangxi, People’s Republic of China; 2Jiangxi Province Medical Imaging Research Institute, Jiangxi, People’s Republic of China; 3Department of Thoracic Oncology, Jiangxi Cancer Hospital, Jiangxi, People’s Republic of China; 4Department of Nuclear Medicine, The First Affiliated Hospital of Nanchang University, Jiangxi, People’s Republic of China
Correspondence: Yulin He; Yanxing Guan 17 Yongwaizheng Street, Nanchang City, Jiangxi Province, People’s Republic of China
Tel +86-791-88692585
Email [email protected]; [email protected]
Purpose: We explored changes in spontaneous brain connectivity in patients with diffuse axonal injury (DAI), assessed via functional connectivity density (FCD) tests using different frequency bands.
Patients and Methods: In all, 23 patients with DAI (17 males and 6 females) and 23 healthy controls (HCs; 17 males and 6 females) were included. Functional magnetic resonance imaging scans were performed when the participants were in a resting state and the FCD levels in three frequency bands (slow-4: 0.027– 0.073 Hz, slow-5: 0.01– 0.027 Hz, and typical: 0.01– 0.08 Hz) were measured. In addition, Pearson’s correlation coefficient was used to explore the relationship between clinical indices and brain regions with abnormal FCD values.
Results: Compared to HCs, DAI patients had significantly greater FCD values in the right extranuclear/limbic lobe/cingulate gyrus and left limbic lobe/hippocampus/parahippocampal gyrus, and significantly lower FCD values in the left precuneus/posterior cingulate gyrus, in the slow-4 band. In the slow-5 band, the DAI patients had higher FCD values in the left inferior temporal gyrus/superior temporal gyrus, left parahippocampal gyrus/limbic lobe, left extranuclear/cingulate gyrus, and right medial frontal gyrus, and lower values in the right inferior frontal gyrus, right inferior parietal lobule, and left cingulate gyrus/limbic lobe. Moreover, compared to HCs, the values in the typical band were higher in the right extranuclear/limbic lobe/hippocampus/parahippocampal gyrus, but were significantly lower in the right precuneus/posterior cingulate gyrus and right inferior parietal lobule/supramarginal gyrus. The abnormal FCD values of these brain regions were linearly correlated with different clinical scale scores.
Conclusion: DAI patients had abnormal FCD values in various brain regions, indicating disruption to the brain functional network. Moreover, the values were frequency dependent. Our results provide new evidence for the pathogenesis of functional impairment and may explain the neuropathological or compensatory mechanism of the disease.
Keywords: diffuse axonal injury, functional connectivity density, functional magnetic resonance imaging, frequency bands
Introduction
Diffuse axonal injury (DAI) is a common and serious traumatic brain injury (TBI). It accounts for about 20% of severe craniocerebral injuries and 29–43% of deaths from TBI1,2 It is the result of the brain moving when subjected to a severe external force, such as a car accident. Due to differences in the mass and elasticity of gray and white matter, these regions move in different ways, resulting in shear stress. This leads to rupture of nerve axons and capillary damage, ultimately resulting in DAI. The common regions of injury in DAI are where nerve axons are gathered, such as the gray–white matter junction, corpus callosum, brainstem, cerebellum, and internal capsule.3
TBI typically causes persistent cognitive and behavioral dysfunction, such as memory, attention, emotion, and executive dysfunction. However, the clinical manifestations of DAI are a series of nonspecific symptoms, including disturbance of consciousness, pupil changes, coma, restlessness, memory loss, limb movement disorders, and language disorders. The most prominent clinical manifestation of DAI is rapidly progressive consciousness disorder without obvious focal injury. Furthermore, it can occur alone or be accompanied by severe brain contusion and laceration, brainstem blood vessel injury, and deep brain hemorrhage. Because of its nonspecific symptoms and wide range of axonal injuries, DAI is difficult to diagnose and recover from.
Computed tomography (CT) is often applied to DAI early after presentation. It has a short examination time and can quickly differentiate whether acute patients have brain parenchyma injury and hemorrhage. But its sensitivity is poor for the detection of microhemorrhage, and its resolution for soft tissue is poor. Compared to CT, conventional magnetic resonance imaging (MRI) sequences offer higher resolution and do not emit radiation. However, neither option can directly reveal the extent of damaged axons, and thus many clinical symptoms cannot be explained.
In recent years, the rapid development of neuroimaging technology has provided more possibilities for studying the pathogenesis of neuropsychiatric diseases. Notably, resting-state functional MRI (rs-fMRI) can provide evidence of changes in spontaneous activity closely related to various nervous system diseases. The technology takes advantage of the different magnetisms of oxyhemoglobin and deoxyhemoglobin to provide signals that depend on the blood oxygen levels in different brain regions. In particular, signals from spontaneous neural activity in the resting state are good biomarkers for the study of brain-related clinical conditions. The use of rs-fMRI to assess functional connectivity density (FCD) is one method to assess brain trauma injury.4,5 FCD is a voxel data-driven method of whole-brain analysis, which has the advantage of relatively fast processing speed. In this method, the number of connections among voxels in the whole brain below a certain threshold are detected. Different from the traditional functional connectivity method which focus on connection strength between two voxels or two regions of interest, FCD reflects the relation between one region to the whole brain. Therefore, FCD can indirectly reflect the role and status of a region in brain functional network. The FCD method has been widely used in Alzheimer’s disease,6 schizophrenia,7 Parkinson’s disease,8 and anisometropic amblyopia.9 However, it has not been used in DAI patients. In this study, we applied the FCD method at different frequency bands to DAI patients, and then attempted to explain the pathological mechanism of their clinical symptoms.
Patients and Methods
Participants
We recruited 23 patients with DAI (17 males and 6 females) and 23 healthy controls (HCs; 17 males and 6 females). The education level and age of the two groups were matched. All of the subjects were right-handed. All DAI patients were diagnosed according to the imaging results and clinical manifestations in the First Affiliated Hospital of Nanchang University, People’s Republic of China. The inclusion criteria for DAI patients were a history of closed head trauma (acceleration–deceleration effect or high-speed rotation); brain MRI showing possible diffuse pathology without large focal lesions (over 10 mm3); hemodynamically stable and 18–60 years old.
The exclusion criteria were CT showing obvious injuries (such as cerebral contusion and laceration, subdural/epidural hematoma, or intracerebral hemorrhage, with a volume greater than 10 mL); systemic diseases such as a history of brain trauma or cerebrovascular disease, Parkinson’s disease, intracranial space occupation, epilepsy, Alzheimer’s disease, multiple sclerosis, psychological disorders, alcoholism, drug abuse, encephalitis, or diabetes; a midline offset >3 mm; bilateral pupillary reaction deficiency; and/or contraindications for MRI. In the same period, 23 healthy volunteers, matched in age, sex, and education, were recruited according to the following inclusion criteria: no abnormalities in a routine head MRI examination and no diabetes, alcoholism, neuropsychiatric diseases, or other systemic disease that might cause an abnormal brain condition.
All participants were informed of the method, purpose, content, precautions, and possible problems of the study and signed a consent form. The study was performed in accordance with the principles of the Helsinki Declaration and received approval from the Medical Research Ethics Committee of The First Affiliated Hospital of Nanchang University.
Questionnaires
Before performing MRI scanning, all subjects were assessed with different clinical scales, including the Glasgow Coma Scale (GCS), Disability Rating Scale (DRS), Mini-mental State Examination (MMSE), Activities of Daily Living (ADL), Motor Assessment Scale (MAS), Agitation Behavior Scale (ABS), Beck Depression Inventory (BDI), and Hamilton Anxiety Scale (HAMA). GCS was used to evaluate patients’ consciousness levels; ABS, DRS, and MAS were used to evaluate body-related functions; ADL was used for patients’ quality of life after injury; and MMSE and HAMA were used for cognitive functions. These scales are widely used in patients with nervous system diseases and can comprehensively evaluate various brain functions of patients.
MRI Acquisition
All subjects received MRI scans with a 3.0 T scanner (Trio TIM, Siemens, Erlangen, Bavaria, Germany) in the resting state with eyes closed while remaining awake. Firstly, each participant underwent a conventional T1-weighted and T2-weighted MRI scan, with the same parameters described in previous study.6 Images of 240 functional volumes were obtained with the echo planar imaging sequence (TR = 2000 ms, TE = 30 ms, matrix = 64 × 64, flip angle = 90°, FOV = 220 mm × 220 mm, slices = 30, and slice thickness = 4 mm). Oriented, three-dimensional, T1 images were also acquired (TR = 1900 ms, TE = 2.26 ms, matrix = 240 × 256, flip angle = 9°, FOV = 215 × 230 mm, slices = 176, and slice thickness = 1.0 mm).
Resting-State Date Preprocessing
The Data Processing Assistant for Resting-State fMRI v2.1 (http://www.restfmri.net/forum/dparsf) on MATLAB 7.14.0 (Mathworks, Natick, MA, USA) and SPM12 (http://www.fil.ion.ucl.ac.uk/spm) were used to preprocess the data. Removing the initial 10 volumes and applying slice-timing correction and realignment to the remaining images. A subject was excluded if his or her head motion had a maximum spin (x, y, or z) >2.0°. Then, the data were normalized to the Montreal Neurological Institute space. Finally, bandpass filtering (ranging from 0.01 to 0.08 Hz) was used to reduce the effects of noise.
FCD Analyses
The FCD of each voxel was calculated using an in-house script, as described by Tomasi and Volkow.10,11 Pearson’s linear correlation coefficient, r, was calculated to identify the strength of the functional connectivity among voxels. Next, the FCD values were spatially smoothed using a Gaussian kernel with a full-width at half-maximum of 8 mm.
FCD is useful for exploring the strength of functional connections in the brain. To detect DAI-related brain connections, we calculated the FCD for three frequency bands: slow-4 (0.027–0.073 Hz), slow-5 (0.01–0.027 Hz), and typical (0.01–0.08 Hz).
Statistical Analyses
We used the SPSS 20.0 software (SPSS Inc, Chicago, IL, USA) to analyze demographic and clinical differences. The chi-square test was used to compare the classification data (handedness and sex). Two-sample t-tests were applied for the differences in the age and clinical scales; p < 0.05 was considered significantly different. The SPM12.0 software was used to compare FCD values of different brain areas between DAI and HC group. The data were collected with a voxel level of p < 0.01 and a cluster level of p < 0.05, based on the Gaussian random field theory. Then, the relationships between the FCD values and clinical scales were analyzed using Pearson correlation analysis.
Results
Demographic and Behavior Measurements
No significant differences were found in sex (p > 0.99) or age (p = 0.928) between the DAI patients and HCs. The mean duration of DAI was 78.63 days. As for the clinical scales, the scores for GCS, MMSE, and MAS were significantly lower in DAI patients than in the HC group, while the scores for DRS, ADL, ABS, BDI, and HAMA were significantly higher (p < 0.05); Further details are shown in Table 1. The results indicated DAI patients had a variety of brain dysfunction, such as disturbance of consciousness, cognitive impairment and emotional disorders.
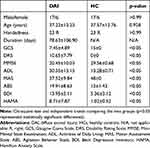 |
Table 1 Demographics and Behavioral Results of DAI and HCs |
FCD Results
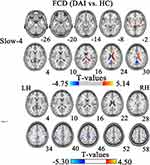
Figure 1 shows the group mean FCD values in DAI patients and healthy controls. In the slow-4 band, DAI patients had significantly higher FCD values in the right extranuclear/limbic lobe/cingulate gyrus and left limbic lobe/hippocampus/parahippocampal gyrus than HCs. However, the values in the left precuneus/posterior cingulate gyrus in that band were significantly lower (Figure 2 and Table 2). In the slow-5 band, the values were significantly higher in the left inferior temporal gyrus/superior temporal gyrus, left parahippocampal gyrus/limbic lobe, left extranuclear/cingulate gyrus, and right medial frontal gyrus, and lower in the right inferior frontal gyrus, right inferior parietal lobule, and left cingulate gyrus/limbic lobe (Figure 2 and Table 3). Furthermore, in the typical range, DAI patients had significantly higher values in the right extranuclear/limbic lobe/hippocampus/parahippocampal gyrus, but significantly lower values in the right precuneus/posterior cingulate gyrus and right inferior parietal lobule/supramarginal gyrus (Figure 3 and Table 4).
 |
Table 2 Brain Regions with Significant Differences in FCD Between DAI Patients and HCs in Slow-4 Band |
 |
Table 3 Brain Regions with Significant Differences in FCD Between DAI Patients and HCs in Slow-5 Band |
 |
Table 4 Brain Regions with Significant Differences in FCD Between DAI Patients and HCs in Typical Band |
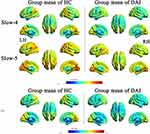 |
Figure 1 Whole-brain voxel-wise functional connectivity density (FCD) patterns in diffuse axonal injury patients and healthy controls. |
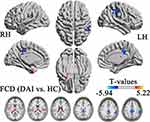 |
Figure 3 Areas with significantly different functional connectivity density in the typical frequency band (0.01–0.08 Hz) between patients with diffuse axonal injury and healthy controls. |
Correlation Analyses
In correlation analyses, the elevated FCD values in the right extra-nuclear/limbic lobe/cingulate gyrus and left limbic lobe/hippocampus/parahippocampal gyrus of DAI patients using the slow-4 band were positively correlated with DRS score and the lower values in the left precuneus/posterior cingulate were positively correlated with ABS score. For the slow-5 band, the elevated values in the left parahippocampal gyrus/limbic lobe were positively correlated with DRS score, and the lower values in the right inferior frontal gyrus were negatively correlated with DRS score; the lower values in the right inferior parietal lobule were positively correlated with ADL score. Finally, the abnormal FCD values for the typical band in the right extranuclear/limbic lobe/hippocampus/parahippocampal gyrus and right precuneus/posterior cingulate gyrus were positively correlated with DRS score and negatively correlated with MMSE score. See the details in Table 5.
 |
Table 5 Correlations Between Abnormal FCD of Brain Regions in DAI Patients and Clinical Scales |
Discussion
In the past several years, great progress has been made in the field of functional MRI. For example, diffusion-weighted imaging (DWI) and susceptibility-weighted imaging (SWI) are useful for showing white matter pathology and microhemorrhage, respectively. However, rs-fMRI provides a better understanding of brain dysfunction by examining changes in spontaneous brain activity when subjects are at rest. Previous studies have used the amplitude of low-frequency fluctuation (ALFF) and regional homogeneity (ReHo) to assess DAI patients.12,13 FCD is a useful tool for exploring the connections in the brain, being noninvasive and highly repeatable in the resting state. To the best of our knowledge, this is the first time that the FCD method has been applied to patients with DAI.
We documented abnormal FCD values in various brain regions (eg, limbic lobe, cingulate gyrus, extra-nuclear, hippocampus, and parahippocampal gyrus) of DAI patients. The limbic lobe consists of the cingulate gyrus, parahippocampal gyrus, hippocampus, and other structures; it is located inside the cerebral hemisphere and surrounds the upper end of the brainstem and the corpus callosum. The limbic lobe, cingulate gyrus, hippocampus, and parahippocampal gyrus are important components of the limbic system. The aforementioned structures are connected to each other through the Papez circuit and have extensive connections with other brain structures. Through connections with the hypothalamus and the autonomic nervous system, the limbic system is involved in mediating instinct and emotional behavior, and regulation of visceral activity. Moreines et al14 reported abnormal activity in the limbic lobe in patients with depression and found that increased connectivity of limbic lobe was associate with negative emotions. Similarly, we found higher FCD values in limbic lobe and cingulate gyrus in DAI patients in three bands, which were speculated to be one of the pathogenic factors of emotional disorder in DAI. In addition, the extra-nuclear region plays an important role in emotional processing. Depressive patients who experienced childhood neglect exhibit decreased voxel-based morphometry in the bilateral extra-nuclear. What is more, the decreased voxel-based morphometry was found to be negatively correlated with HAMA.15 Lei et al16 found that white matter in the bilateral extra-nuclear was damaged in rats model with deficit schizophrenia, suggesting extra-nuclear impairment was important for the occurrence of greater negative symptoms and anhedonia. Contrary to the findings presented above, the FCD values of extra-nuclear in DAI patients were increased in both slow-4 band and typical band, and it might be a compensatory mechanism for mental disorder in DAI. A study conducted by Slobounov et al17 concluded that athletes with concussion have more activity in the hippocampus when performing spatial memory tasks, in contrast to HCs. Increased ReHo and ALFF values in the bilateral hippocampus and parahippocampal gyrus have also been observed in patients with mild cognitive impairment without lacunar infarction.18 That can be explained compensation in patients with cognitive impairment. In the current study, we found that FCD values in the extra-nuclear, limbic lobe, cingulate gyrus, hippocampus, and parahippocampal gyrus were significantly higher in DAI patients. Furthermore, the increased values in the right extranuclear/limbic lobe/cingulate gyrus and left limbic lobe/hippocampus/parahippocampal gyrus in slow-4 band were positively correlated with DRS score and the increased values in the right extranuclear/limbic lobe/hippocampus/parahippocampal gyrus in typical band were positively correlated with DRS score and negatively correlated with MMSE score. The increased FCD values in these brain regions might be related to the mechanism of emotional disorders and the activation of brain regions working as a compensatory mechanism for brain function after injury. It is worth noting that in the slow-5 band, the FCD value of the left limbic lobe/left cingulate gyrus was significantly lower, indicating severe damage in this region. The reason for this phenomenon may be the dominant hemisphere of individual were more fragile to be damaged.
The default mode network (DMN) is an important brain network in humans, with strong, spontaneous activity in the resting state. It is involved in introspection, recalling the past, thinking about the future, and introversion thinking activities under the basic or default state. As important parts of the DMN system, the precuneus and posterior cingulate gyrus are responsible for consciousness, learning, memory, and other cognitive functions by automatically collecting and integrating information from the human body and surrounding environment. Johnson et al19 reported a reduced number of connections and detectable connection strengths in the DMN of patients with subconcussive head trauma. Another study that used rs-fMRI found a significantly lower ALFF value in the precuneus of DAI patients, which also correlated with GCS score.12 Therefore, it can be speculated that the DMN may be associated with disorder of consciousness. Another study observed different degrees of injury in the cingulum, including the posterior cingulate gyrus, in TBI patients.20 That might be the reason for the impairment of sustained attention in TBI patients. Hence, the deterioration of attention is related to posterior cingulate gyrus injury, which can also be predicted by the reduction of functional connections in the DMN. A previous study discovered lower functional connectivity in the posterior cingulate gyrus in patients with Parkinson’s disease with dementia and increased functional connectivity in the posterior cingulate gyrus of those with Parkinson’s disease with mild cognitive impairment (compared to Parkinson’s disease patients with no cognitive impairment).21 The authors considered this region to be severely damaged in the former, and part of a compensatory mechanism in the latter. The precuneus and posterior cingulate gyrus are also closely related to emotional processing. Considering the posterior cingulate gyrus/anterior cuneiform lobe as the region of interest, Bluhm et al22 detected changes in brain activity in 17 patients with post-traumatic stress disorder. The data showed a noticeably reduced functional connection between the posterior cingulate gyrus and other areas of the brain. The authors speculated that it might be closely related to symptoms such as lack of pleasure and emotional retardation. In patients with borderline personality disorder, reduced ALFF and ReHo values in the precuneus and posterior cingulate gyrus are thought to be the cause of their unstable self-identification characteristics.23 The precuneus and posterior cingulate gyrus can also influence behavior by changing decision-making and motivation. We observed significantly reduced FCD values in the precuneus/posterior cingulate gyrus in both the slow-4 and typical bands in this study, which indicates network dysfunction and impairment of the precuneus/posterior cingulate gyrus in DAI patients. Moreover, the decreased FCD values in the precuneus/posterior cingulate gyrus were positively correlated with ABS and DRS scores, and negatively correlated with MMSE score. According to evidence from earlier, alterations in the precuneus/posterior cingulate gyrus can be considered as one of neuropathological mechanisms of disturbance of consciousness, cognitive impairment and mental disorder of DAI patients.
The inferior parietal lobule and supramarginal gyrus also belong to the DMN. The connectivity between the bilateral inferior parietal lobules and whole-brain function are lower in patients with TBI, and this has been thought to be the reason for cognitive impairment.24 Consistently, we found significantly lower FCD values in slow-5 and typical bands in those regions in DAI patients. Furthermore, the significantly lower FCD value in the slow-5 band in the inferior parietal lobule was positively correlated with ADL score. These results indicate the dysfunction and reduction in importance of the inferior parietal lobule and supramarginal gyrus. To some extent, it is helpful to understand this pathophysiological mechanism of cognitive impairment after extensive axonal injury.
We also found that DAI patients exhibited abnormal FCD values in the superior temporal gyrus/inferior temporal gyrus. The temporal lobe principally handles visual and auditory information but also relates to emotional function. One study showed that the ReHo value of the left inferior temporal gyrus of Parkinson’s disease patients in the resting state is significantly increased.25 Even if there are no clinically relevant visual symptoms in patients with Parkinson’s disease, the metabolic activity of the inferior temporal gyrus will change to compensate for motor disorders by improving their ability to process visual information. Abnormal signals in the temporal gyrus may relate to the process of negative automatic thinking and negative emotional immersion in patients with depression. In one study,26 TBI patients with depression showed increased activities in the bilateral inferior temporal gyrus and left superior temporal compared to those without depression. Previous studies have also demonstrated increased functional connectivity in the left and right temporal gyrus, related to depression, aggressive behavior, and stress-related symptoms in patients with mild TBI.27 Consistent with these results, we found increased FCD values in the superior temporal gyrus/inferior temporal gyrus in slow-5 band. The data supported the view that the increased FCD values in the superior temporal gyrus/inferior temporal gyrus were the cause of abnormal emotional responses and compensation for the motor disorder after extensive axonal injury.
The frontal lobe plays a major role in executive function, including selective attention, behavior planning, information manipulation, and emotional processing. For instance, the interruption of functional connections of the bilateral inferior frontal gyrus had been observed in TBI patients with poor working memory.28 However, Smits et al29 reported the activation of the inferior frontal gyrus in concussion patients during attention tasks. In addition, the posterior part of the inferior frontal gyrus is crucial for the integration of language information and the understanding of the meaning of sentences. The results of diffusion tensor imaging have shown that the loss of fiber bundles in the inferior frontal and temporal gyrus of DAI patients results in the interruption of frontal lobe information, thus disturbing the production of language.30 Similarly, the study displayed decreased FCD values in inferior frontal gyrus in slow-5 band, which was negatively correlated with DRS score. The results indicated disturbed intrinsic brain function compared with HCs and can be considered as neurological basis for cognitive dysfunction in DAI patients. However, an analysis of the ReHo values of 32 manic-depressive patients found that the ReHo of the right middle frontal gyrus was higher than that of HCs,31 pointing out the role of middle frontal gyrus in emotion processing. In this study, we observed significantly higher FCD values in the inferior frontal gyrus in DAI patients, suggesting the involvement of the neuropathological compensatory mechanism for mental disorder.
The study showed abnormal FCD values in all three bands. We also found abnormal FCD values in the extranuclear, limbic lobe, cingulate gyrus, hippocampus, and parahippocampal gyrus in both the slow-4 and slow-5 bands, while the abnormal FCD values in the frontal and temporal lobes were only found in the slow-5 band and the abnormal FCD values in the inferior parietal lobule were only found in slow-5 and typical bands. These results indicate that the internal brain activity pattern is sensitive to a specific frequency. Slow-5 band is more suitable for studying large neural networks, such as the frontal and temporal lobes, which may be due to the higher energy in this lower frequency band. While slow-4 band which has lower power is more suitable for the study of changed brain activity of small neurons. In view of the fact that different frequency bands may have specific physiological and pathological significance, the influence of the frequency band should be considered when studying the FCD of DAI patients. In brief, our results highlighted the importance of FCD values within different frequency bands in understanding the mechanism of DAI.
Conclusion
The FCD method has shown great potential and advantages in studying brain functional connections and has provided researchers with a powerful tool to measure the abnormal brain functional connectivity of DAI patients. Our results indicate that, in DAI patients, many brain regions exhibit abnormal FCD values. Impairment of consciousness and cognition in such patients may be related to changes in brain network activity and connection patterns. Therefore, we speculate that abnormal functional connectivity between brain regions is a crucial neuropathological or compensatory factor for DAI-related symptoms and even a biomarker for distinguishing DAI patients from healthy people. Furthermore, these findings emphasize the key role of aberrant FCD patterns in DAI patients.
Acknowledgments
This study was supported by the National Natural Science Foundation of China (81260217 and 81460263).
Disclosure
The authors report no conflicts of interest in this work.
References
1. Thompson FJ, Hou J, Bose PK. Closed-head TBI model of multiple morbidity. Methods Mol Biol. 2016;1462:521–536. doi:10.1007/978-1-4939-3816-2_28
2. Pearn ML, Niesman IR, Egawa J, et al. Pathophysiology associated with traumatic brain injury: current treatments and potential novel therapeutics. Cell Mol Neurobiol. 2017;37(4):571–585. doi:10.1007/s10571-016-0400-1
3. Jeong JH, Kim YZ, Cho YW, Kim JS. Negative effect of hypopituitarism following brain trauma in patients with diffuse axonal injury. J Neurosurg. 2010;113(3):532–538. doi:10.3171/2009.10.JNS091152
4. Tomasi D, Volkow ND. Ultrafast method for mapping local functional connectivity hubs in the human brain. Conf Proc IEEE Eng Med Biol Soc. 2010;2010:4274–4277. doi:10.1109/IEMBS.2010.5626180
5. Tomasi D, Shokri-Kojori E, Volkow ND. High-resolution functional connectivity density: hub locations, sensitivity, specificity, reproducibility, and reliability. Cereb Cortex. 2016;26(7):3249–3259. doi:10.1093/cercor/bhv171
6. Balthazar ML, Pereira FR, Lopes TM, et al. Neuropsychiatric symptoms in Alzheimer’s disease are related to functional connectivity alterations in the salience network. Hum Brain Mapp. 2014;35(4):1237–1246. doi:10.1002/hbm.22248
7. Huang H, Jiang Y, Xia M, et al. Increased resting-state global functional connectivity density of default mode network in schizophrenia subjects treated with electroconvulsive therapy. Schizophr Res. 2018;197:192–199. doi:10.1016/j.schres.2017.10.044
8. Hu X, Jiang Y, Jiang X, et al. Altered functional connectivity density in subtypes of Parkinson’s disease. Front Hum Neurosci. 2017;11:458. doi:10.3389/fnhum.2017.00458
9. Wang T, Li Q, Guo M, et al. Abnormal functional connectivity density in children with anisometropic amblyopia at resting-state. Brain Res. 2014;1563:41–51. doi:10.1016/j.brainres.2014.03.015
10. Tomasi D, Volkow ND. Functional connectivity density mapping. Proc Natl Acad Sci U S A. 2010;107(21):9885–9890. doi:10.1073/pnas.1001414107
11. Tomasi D, Volkow ND. Association between functional connectivity hubs and brain networks. Cereb Cortex. 2011;21(9):2003–2013. doi:10.1093/cercor/bhq268
12. Yao S, Song J, Gao L, et al. Thalamocortical sensorimotor circuit damage associated with disorders of consciousness for diffuse axonal injury patients. J Neurol Sci. 2015;356(1–2):168–174. doi:10.1016/j.jns.2015.06.044
13. Wu L, Zhou F, Zhang Y, et al. Thalamic atrophy and dysfunction in patients with mild-to-moderate traumatic diffuse axonal injury: a short-term and mid-term MRI study. Neuroreport. 2018;29(15):1282–1287. doi:10.1097/WNR.0000000000001106
14. Moreines JL, McClintock SM, Kelley ME, Holtzheimer PE, Mayberg HS. Neuropsychological function before and after subcallosal cingulate deep brain stimulation in patients with treatment-resistant depression. Depress Anxiety. 2014;31(8):690–698. doi:10.1002/da.22263
15. Peng H, Ning Y, Zhang Y, et al. White-matter density abnormalities in depressive patients with and without childhood neglect: a voxel-based morphometry (VBM) analysis. Neurosci Lett. 2013;550:23–28. doi:10.1016/j.neulet.2013.06.048
16. Lei W, Li N, Deng W, et al. White matter alterations in first episode treatment-naïve patients with deficit schizophrenia: a combined VBM and DTI study. Sci Rep. 2015;5(1):12994. doi:10.1038/srep12994
17. Slobounov SM, Zhang K, Pennell D, Ray W, Johnson B, Sebastianelli W. Functional abnormalities in normally appearing athletes following mild traumatic brain injury: a functional MRI study. Exp Brain Res. 2010;202(2):341–354. doi:10.1007/s00221-009-2141-6
18. Ni L, Liu R, Yin Z, et al. Aberrant spontaneous brain activity in patients with mild cognitive impairment and concomitant lacunar infarction: a resting-state functional MRI study. J Alzheimers Dis. 2016;50(4):1243–1254. doi:10.3233/JAD-150622
19. Johnson B, Neuberger T, Gay M, Hallett M, Slobounov S. Effects of subconcussive head trauma on the default mode network of the brain. J Neurotrauma. 2014;31(23):1907–1913. doi:10.1089/neu.2014.3415
20. Leech R, Sharp DJ. The role of the posterior cingulate cortex in cognition and disease. Brain. 2014;137(Pt 1):12–32. doi:10.1093/brain/awt162
21. Zhan ZW, Lin LZ, Yu EH, et al. Abnormal resting-state functional connectivity in posterior cingulate cortex of Parkinson’s disease with mild cognitive impairment and dementia. CNS Neurosci Ther. 2018;24(10):897–905. doi:10.1111/cns.12838
22. Bluhm RL, Williamson PC, Osuch EA, et al. Alterations in default network connectivity in posttraumatic stress disorder related to early-life trauma. J Psychiatry Neurosci. 2009;34(3):187–194.
23. Lei X, Zhong M, Liu Y, et al. A resting-state fMRI study in borderline personality disorder combining amplitude of low frequency fluctuation, regional homogeneity and seed based functional connectivity. J Affect Disord. 2017;218:299–305. doi:10.1016/j.jad.2017.04.067
24. Bonnelle V, Leech R, Kinnunen KM, et al. Default mode network connectivity predicts sustained attention deficits after traumatic brain injury. J Neurosci. 2011;31(38):13442–13451. doi:10.1523/JNEUROSCI.1163-11.2011
25. Jiang S, Wang M, Zhang L, et al. Regional homogeneity alterations differentiate between tremor dominant and postural instability gait difficulty subtypes of Parkinson’s disease. J Neural Transm (Vienna). 2016;123(3):219–229.
26. Fischer BL, Parsons M, Durgerian S, et al. Neural activation during response inhibition differentiates blast from mechanical causes of mild to moderate traumatic brain injury. J Neurotrauma. 2014;31(2):169–179. doi:10.1089/neu.2013.2877
27. Nathan DE, Oakes TR, Yeh PH, et al. Exploring variations in functional connectivity of the resting state default mode network in mild traumatic brain injury. Brain Connect. 2015;5(2):102–114. doi:10.1089/brain.2014.0273
28. Kasahara M, Menon DK, Salmond CH, et al. Altered functional connectivity in the motor network after traumatic brain injury. Neurology. 2010;75(2):168–176. doi:10.1212/WNL.0b013e3181e7ca58
29. Smits M, Dippel DW, Houston GC, et al. Postconcussion syndrome after minor head injury: brain activation of working memory and attention. Hum Brain Mapp. 2009;30(9):2789–2803. doi:10.1002/hbm.20709
30. Mao H, Polensek SH, Goldstein FC, Holder CA, Ni C. Diffusion tensor and functional magnetic resonance imaging of diffuse axonal injury and resulting language impairment. J Neuroimaging. 2007;17(4):292–294. doi:10.1111/j.1552-6569.2007.00146.x
31. Yang H, Li L, Peng H, et al. Alterations in regional homogeneity of resting-state brain activity in patients with major depressive disorder screening positive on the 32-item hypomania checklist (HCL-32). J Affect Disord. 2016;203:69–76. doi:10.1016/j.jad.2016.05.004
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.