Back to Journals » Journal of Pain Research » Volume 13
Fluoroscopy-Guided Percutaneous Sacroplasty for Painful Metastases at the Sacral Ala
Authors Tian QH , Liu HF, Wang T, Wu CG, Cheng YS
Received 8 November 2018
Accepted for publication 9 July 2019
Published 16 January 2020 Volume 2020:13 Pages 151—156
DOI https://doi.org/10.2147/JPR.S193866
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Katherine Hanlon
Qing-Hua Tian, He-Fei Liu, Tao Wang, Chun-Gen Wu, Ying-Sheng Cheng
Department of Diagnostic and Interventional Radiology, Shanghai Sixth People’s Hospital East Affiliated to Shanghai University of Medicine and Health Sciences, Shanghai 201306, People’s Republic of China
Correspondence: Ying-Sheng Cheng; Chun-Gen Wu
Department of Diagnostic and Interventional Radiology, Shanghai Sixth People’s Hospital East Affiliated to Shanghai University of Medicine & Health Sciences, No. 222 West Third Road, Shanghai 201306, People’s Republic of China
Tel +8618917683588
Fax +86021-38297739
Email [email protected]; [email protected]
Objective: Percutaneous sacroplasty (PSP) is widely used in the clinic for osteoporotic sacral insufficiency fractures; however, few reports have described the safety and effectiveness of PSP for painful sacral metastases at the sacral ala under fluoroscopy alone. We aimed to evaluate the safety and efficacy of fluoroscopy-guided PSP for painful metastases at the sacral ala.
Patients and Methods: Thirty-five consecutive patients (median age, 60.74 ± 12.74 years), with a total of 41 metastatic lesions at the sacral ala, were treated with PSP. The patients were followed up for periods ranging from 1 month to 30 months (average, 8.23 ± 6.75 months). The visual analog scale (VAS), Oswestry Disability Index (ODI), and Karnofsky Performance Scale (KPS) were used to evaluate pain, mobility, and quality of life before the procedure and at 3 days and 1, 3, 6, 12, and 18 months after the procedure.
Results: Technical success was achieved in all patients. The minimum follow-up duration was 1 month. The mean VAS scores declined significantly from 7.20 ± 0.93 before the procedure to 3.43 ± 1.38 by day 3 after the procedure, and was 3.13 ± 1.07 at 1 month, 3.17 ± 1.15 at 3 months, 2.91± 1.38 at 6 months, and 2.57 ± 1.51 at 12 months after the procedure (P < 0.001). After PSP, analgesic drug administration had been discontinued in 31 of 35 patients (88.57%). The ODI and KPS also changed after PSP, with significant differences between the baseline scores and those at each follow-up examination (P < 0.001). Extraosseous cement leakage occurred in 12 cases without any major clinical complications.
Conclusion: PSP is a safe and effective technique for the palliative treatment of painful metastases involving the sacral ala under fluoroscopic guidance alone. It can relieve pain, reduce disability, and improve function, and is associated with minimal complications.
Keywords: percutaneous sacroplasty, metastases, sacral ala
Introduction
Percutaneous sacroplasty (PSP), which involves the placement of bone needles and the injection of bone cement into the sacrum, is a minimally invasive, image-guided procedure for pain control and stability restoration in cases with sacral insufficiency fractures (SIF) resulting from both osteoporosis and metastases.1–3 Although PSP has been widely used in cases with osteoporotic insufficiency,3–6 only a few reports have described the safety and effectiveness of PSP in painful sacral metastases.7–10 Moreover, studies have not compared the effect of multiple approaches for metastases involving the specific sacral ala and, because the follow-up periods of the available studies have generally been short, little is known regarding the medium- and long-term improvement in pain and function after PSP in cases with metastases. In this retrospective study, we aimed to evaluate the efficacy and safety of fluoroscopy-guided PSP in the treatment of painful metastases involving the sacral ala with a relatively large sample and medium-term follow-up.
Materials and Methods
Study Design
This retrospective study was approved by the institutional review board of Shanghai Sixth People’s Hospital East Campus, and written informed consent was obtained from all participants included in the study. From March 2016 to August 2018, patients with painful metastatic lesions of the sacral ala were recruited from our department for treatment with PSP. All patients were referred to our institution due to the persistence of pain that had not responded to conventional treatments such as analgesic drugs, chemotherapy, and radiotherapy. All patients had pathologically confirmed primary cancer and had undergone Computed Tomography (CT) and/or magnetic resonance imaging (MRI) examinations before the procedure to determine the size of the lesion and the part of the sacral segment involved, and to rule out other causes of back pain (such as degenerative facet disease). All patients had severe pain, without any neurological deficit related to the metastatic lesions of the sacral ala. Patients were eligible for inclusion in the study if they had life expectancy ≥3 months, were 18 years of age or older, and were willing to sign the consent form. Patients with pathological fractures and lesions involving the area of the sacral foramina were excluded. The primary tumors in these patients were located in the lung (n=14), thyroid (n=8), liver (n=6), prostate (n=3), biliary (n=2), breast (n=1), and colon (n=1). The number of treated lesions per patient ranged from 1 to 2; in particular, 82.86% (29/35) patients had 1 lesion each and 17.14% (6/35) had 2 lesions each, thus resulting in a total of 41 metastases in the cohort. In addition, 28 patients were treated for other metastatic localizations, including spinal metastases in 19 (54.29%) and pelvic lesions in 9 (25.71%). The baseline characteristics of the 35 patients and the results are summarized in Table 1.
 |
Table 1 Baseline Characteristics and Clinical Outcomes in Patients with Painful Metastases at the Sacral Ala Treated with PSP |
PSP Procedures
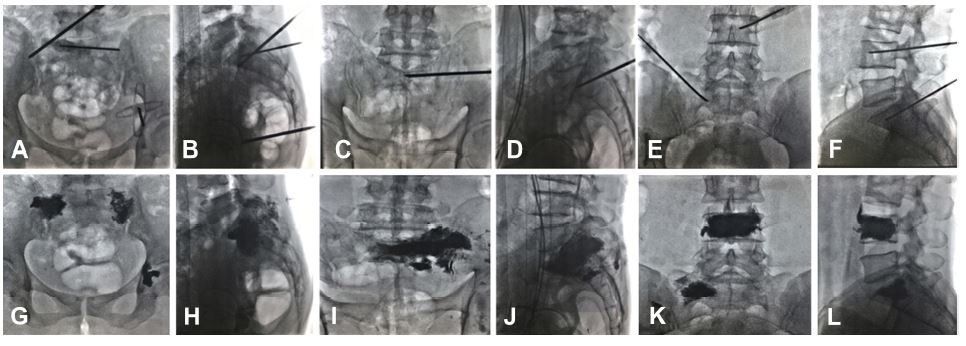
All procedures were performed under fluoroscopic guidance alone with a biplane machine (GE, Innova IGS630, America). Under conscious sedation, the patient was placed in the prone position. Under anteroposterior and lateral fluoroscopic guidance, a 13-gauge bone puncture needle (Cook Inc., Bloomington, IN, USA) was slowly hammered into the metastatic lesions at the sacral ala through the posterior approach, transiliac approach, or anterior-oblique approach under fluoroscopic guidance. As the bone needle was advanced into the S1 sacral ala, continuous anteroposterior and lateral fluoroscopic guidance was used to confirm needle access between the sacroiliac joints, sacral foramina, and anterior margin of the sacral ala, whereas the orientation of the bevel edge was adjusted to avoid penetration of structures such as the anterior surface of the sacral ala and S1 foramen. One or more needles were used if necessary. Thereafter, mixed bone cement (polymethyl methacrylate [PMMA]; Palacos V; Heraeus Medical GmBH, Germany) was injected into the metastatic lesion at the sacral ala. The injection process was monitored continuously under fluoroscopy in the anterior and lateral plane. Injection was stopped when substantial resistance was met or when the PMMA cement reached the margin of the sacroiliac joint or the posterior portion of the sacrum. The technical success of the procedure and any complications that occurred were also recorded. Immediately after PSP, non-contrast CT examination was performed in all patients to assess if there was cement leakage (Figure 1).
Clinical Outcome Evaluation and Data Collection
All patients underwent clinical examination by two of the authors before the procedure; 1 week after the procedure; 1, 3, 6, and 12 months after the procedure; and every 6 months thereafter until patient death. Data on technical success, PMMA volume injected, pain relief, functional outcomes, and complications were evaluated during follow-up consultations or at patient death. Technical success was defined as successful puncture of the sacral lesion with any approach, followed by PMMA injection without any major complications.
Major complications included accidental nerve root injury, cauda equina syndrome, pulmonary embolism, intestinal rupture, or perioperative mortality, whereas minor complications included postoperative urinary retention, wound hematoma, and infection. Pain was measured using the visual analog scale (VAS), where a score of 0 indicated “no pain” and a score of 10 indicated “worst pain ever.” Pain relief was defined as a decrease in the VAS score by ≥3 points from the baseline score. The functional status of patients for walking, standing, and sleeping was measured using the Oswestry Disability Index (ODI). The functional outcomes were measured on a 100-point Karnofsky Performance Scale (KPS) to assess any changes in the quality of life. Data on the use of pain medication (narcotics or nonsteroidal anti-inflammatory drugs) before and after the intervention were evaluated. A decrease in dose or a shift to a lower level of the World Health Organization classification of analgesia was considered to represent a reduction in analgesic use.
Statistical Analysis
All statistical analyses were performed using commercially available software (SPSS Version 16, SPSS Inc., Chicago, IL, USA). Data are expressed as means ± standard deviation. The paired t-test was used to compare the mean VAS, ODI, and KPS scores between the different study time points. A P value of ≤0.05 was considered statistically significant.
Results
Thirty-five consecutive patients (median age: 60.74 ± 12.74 years, including 24 men and 11 women) with painful metastatic lesions at the sacral ala underwent PSP in our medical center. PSP was technically successful and well tolerated in all the patients through three different approaches: the posterior approach (n=18), transiliac approach (n=7), or anterior-oblique approach (n=10). The mean amount of cement injected per lesion was 5.20 ± 1.55 mL (range, 2–8 mL). The mean number of puncture needles used during PSP was 1.49 ± 0.84 (range, 1–4) per lesion. No major complications were observed during the procedure. The only minor complication encountered was PMMA leakage, which was noted in 34.29% (12/35) patients. Leakages occurred into the sacral venous plexus (n=5), puncture path (n=2), anterior epidural space (n=2), or anterior sacral space (n=3), but were all asymptomatic and did not require any special treatment. In addition, there was no significant difference in PMMA leakage among the three approaches (P > 0.05).
The average follow-up duration was 8.23 ± 6.75 months (range, 1–30 months). The changes in the VAS, ODI, and KPS values following PSP are shown in Figure 2. Of the 35 patients, 31 (88.57%) reported pain relief and the other 4 (11.43%) experienced no obvious regression of pain immediately after the intervention. Four patients did not receive regular treatment, such as systematic chemotherapy or radiotherapy. The mean VAS scores declined significantly from 7.20 ± 0.93 before the procedure to 3.43 ± 1.38 by day 3 after the procedure, and was 3.13 ± 1.07 at 1 month, 3.17 ± 1.15 at 3 months, 2.91± 1.38 at 6 months, and 2.57 ± 1.51 at 12 months after the procedure; the scores remained low throughout the follow-up period. There was a significant difference between the pre-procedure VAS score and that at each study time point after the procedure (P < 0.001). As shown in Figure 2, the average ODI and KPS scores also changed after the procedure, with significant differences between the baseline scores and those at each follow-up examination (P < 0.001). In addition, no significant differences were observed in the VAS, ODI, and KPS values among the three approaches and among the three types of bone destruction (P > 0.05). Prior to the PSP procedure, all patients were prescribed analgesic drugs, such as strong opioid analgesics (n=12), weak opioid analgesics (n=15), and nonsteroidal anti-inflammatory drugs (NSAIDs; n=8). After the PSP procedure, the administration of analgesic drugs was discontinued or reduced in 31 of 35 patients (88.57%). In 31 of the 35 patients, post-procedural pain was controlled by using strong opioid analgesics (n=1), weak opioid analgesics (n=4), and NSAIDs (n=12); in the remaining 14 patients, no analgesic therapy was necessary after the procedure.
Discussion
Metastatic tumors are the most common malignant lesions occurring in the sacrum, especially in the sacral ala, and account for 1–7% of all spinal tumors.11,12 The sacrum is a weight-bearing structure, and the sacral ala dissipates vertical axial forces from the lumbar to iliac region, thus aiding in spinal stability. Symptomatic sacral metastases usually manifest as debilitating local pain that can radiate into the buttocks, perineum, and posterior thigh. In addition to severe pain, sacral metastasis can also lead to pathological fractures and neurological deficits, which often limit the mobility, quality of life, and tolerance for further necessary cancer treatment in patients.
The currently available treatments for sacral metastases combine systemic and local therapies, including pain medication, radiotherapy, chemotherapy, and endovascular embolization in cases with highly vascular tumors.13,14 When a localized painful lesion is identified, open surgery has limited application because it is often too invasive for a fragile patient. Radiotherapy is a good option but has certain limitations, such as a delayed effect and tissue tolerance.15 It is estimated that at least 45% of the patients with bone metastases develop intractable pain due to the lack of sufficient treatment.16 Therefore, researchers have sought to develop novel therapies to relieve pain and improve mobility.
PSP, analogous to percutaneous vertebroplasty (PVP), has been widely reported as a safe and effective option for managing osteoporotic sacral insufficiency fractures.4–6 Moreover, case and case-series reports have indicated that PSP provides pain relief and mobility improvement in patients with sacral metastases, although the number of cases described is small.7–10,13,17 In a multicenter study with 243 patients, including 204 with painful sacral insufficiency fractures and 39 with symptomatic sacral lesions, 24 patients with sacral metastases experienced remarkable and prompt pain relief.1 Pereira et al described promising outcomes and safety data in the largest published sample of 42 patients with sacral metastases undergoing PSP.2 In the present retrospective study with a relatively large sample, most patients experienced significant changes in the VAS, ODI, and KPS immediately and longitudinally after PSP, consistent with the previously published reports. Collectively, these studies with a relatively large sample appear to strongly indicate that PSP is an effective procedure for providing pain relief and function recovery in patients who are unresponsive to conservative management and those who are not candidates for surgery. In our study, four patients did not experience any apparent pain reduction. This finding may be related to the fact that the four patients were not under regular treatment, such as systematic chemotherapy or radiotherapy, which might have helped control the cancer progression. Moreover, this finding suggests that successful immediate and long-term pain relief is achievable in patients with sacral metastasis who require regular anticancer therapy in addition to PVP.
Although PSP is a variation of PVP, the puncture and injection techniques of PSP completely differ from those of PVP due to the complex anatomy of the sacrum. The sacral ala – classified as zone I by Denis et al18– is adjacent to the sacral foramina at its internal side and to the sacroiliac joint at its external side. Therefore, the adjacent nature of the sacral ala and the convex course complicate needle positioning and cement injection. Moreover, the iliac bones of the pelvis prevent adequate visualization of the sacral ala and require lateral fluoroscopic evaluation. In addition, it can be difficult to identify anatomic landmarks in destructive bone under fluoroscopy.
However, there are still three primary approaches for PSP at the sacral ala: the posterior approach, transiliac approach, and anterior-oblique approach.1,13,19 In particular, the posterior approach, including a long-axis approach and a short-axis approach, is the most commonly used approach under fluoroscopic or CT guidance. In the present study, the above three approaches were all used with 100% technical success. We also compared the safety and efficacy of the different approaches for metastases of the sacral ala, and did not observe any differences, consistent with the findings of another study.20 Although a combination of CT and fluoroscopic guidance may be the best option at present, our procedures were all performed under continuous fluoroscopy alone without any major complications. We primarily preferred fluoroscopic guidance due to the potential for real-time imaging during needle placement and cement injection. We believe that an experienced doctor with a thorough understanding of the radiologic anatomy of the sacrum can achieve both precise needle placement and real-time visualization of cement delivery under continuous anteroposterior and lateral fluoroscopy alone. Furthermore, sacro-kyphoplasty has been described as an effective treatment in patients with sacral metastasis to reduce pain and the rate of cement leakage.8 However, in the present study, we used sacro-vertebroplasty due to the lower associated cost with the relatively similar clinical efficacy.
This study has certain limitations. First, it was conducted at a single center and was retrospective in nature. Second, the sample size was not sufficiently large. Third, we did not compare PSP with other therapeutic options such as surgical treatment or radiotherapy. Moreover, 28 patients were treated for other metastatic localizations, which may bias the effect of PSP.
In conclusion, PSP is an effective, safe, and minimally invasive procedure for the treatment of painful metastases of the sacral ala that are refractory to conservative treatment. This method can achieve a marked reduction in pain, as well as improvement in function and the quality of life. Nevertheless, large-scale prospective research is required to confirm our findings.
Acknowledgements
This work was sponsored by the National Natural Scientific Fund of China [grant number 81701798], as well as Natural Fund from the Shanghai Science and Technology Commission [grant numbers 18ZR1429400, 19411971800], Shanghai Sailing Program [grant number 17YF1414600], and Shanghai Jiao Tong University Affiliated Sixth People’s Hospital East Campus [grant number 2016014] in China.
Statement of Informed Consent
Informed consent was obtained from all participants included in the study.
Statement of Human and Animal Rights
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Disclosure
The authors declare that they have no conflict of interest.
References
1. Kortman K, Ortiz O, Miller T, et al. Multicenter study to assess the efficacy and safety of sacroplasty in patients with osteoporotic sacral insufficiency fractures or pathologic sacral lesions. J Neurointerv Surg. 2013;5:461–466. doi:10.1136/neurintsurg-2012-010347
2. Pereira LP, Clarençon F, Cormier E, et al. Safety and effectiveness of percutaneous sacroplasty: a single-centre experience in 58 consecutive patients with tumors or osteoporotic insufficient fractures treated under fluoroscopic guidance. Eur Radiol. 2013;23:2764–2772. doi:10.1007/s00330-013-2881-3
3. Gupta AC, Chandra RV, Yoo AJ, et al. Safety and effectiveness of sacroplasty: a large single-center experience. Am J Neuroradiol. 2014;35:2202–2206. doi:10.3174/ajnr.A4027
4. Talmadge J, Smith K, Dykes T, et al. Clinical impact of sacroplasty on patient mobility. J Vasc Interv Radiol. 2014;25:91–915. doi:10.1016/j.jvir.2014.02.007
5. Heo DH, Park CK. Percutaneous sacroplasty for non-neoplastic osteoporotic sacral insufficiency fractures. Pain Physician. 2017;20:89–94.
6. Yang SC, Tsai TT, Chen HS, et al. Comparison of sacroplasty with or without balloon assistance for the treatment of sacral insufficiency fractures. J Orthop Surg (Hong Kong). 2018;26:2309499018782575. doi:10.1177/2309499018782575
7. Sun G, Jin P, Li M, et al. Three-dimensional C-arm computed tomography reformation combined with fluoroscopic-guided sacroplasty for sacral metastases. Support Care Cancer. 2012;20:2083–2088. doi:10.1007/s00520-011-1317-3
8. Andresen R, Radmer S, Lüdtke CW, et al. Balloon sacroplasty as a palliative pain treatment in patients with metastasis-induced bone destruction and pathological fractures. Rofo. 2014;186:881–886. doi:10.1055/s-00000066
9. Moussazadeh N, Laufer I, Werner T, et al. Sacroplasty for cancer-associated insufficiency fractures. Neurosurgery. 2015;76:446–450. doi:10.1227/NEU.0000000000000658
10. Burgard CA, Dinkel J, Strobl F, et al. CT fluoroscopy-guided percutaneous osteoplasty with or without radiofrequency ablation in the treatment of painful extraspinal and spinal bone metastases:technical outcome and complications in 29 patients. Diagn Interv Radiol. 2018;24:158–165. doi:10.5152/dir
11. Nader R, Rhines LD, Mendel E. Metastatic sacral tumors. Neurosurg Clin N Am. 2004;15:453–457. doi:10.1016/j.nec.2004.04.009
12. Perrin RG, Laxton AW. Metastatic spine disease: epidemiology, pathophysiology, and evaluation of patients. Neurosurg Clin N Am. 2004;15:365–373. doi:10.1016/j.nec.2004.04.018
13. Zhang J, Wu CG, Gu YF, et al. Percutaneous sacroplasty for sacral metastatic tumours under fluoroscopic guidance only. Korean J Radiol. 2008;9:572–576. doi:10.3348/kjr.2008.9.6.572
14. Quraishi NA, Giannoulis KE, Edwards KL, et al. Management of metastatic sacral tumours. Eur Spine J. 2012;21:1984–1993. doi:10.1007/s00586-012-2394-9
15. Tran Thang NN, Abdo G, Martin JB, et al. Percutaneous cementoplasty in multiple myeloma: a valuable adjunct for pain control and ambulation maintenance. Support Care Cancer. 2008;16:891–896. doi:10.1007/s00520-007-0344-6
16. Meuser T, Pietruck C, Radbruch L, et al. Symptoms during cancer pain treatment following WHO-guidelines: a longitudin al follow-up study of symptom prevalence, severity and etiology. Pain. 2001;93:247–257. doi:10.1016/S0304-3959(01)00324-4
17. Cho S, Park HS, Kim DY, et al. Percutaneous sacroplasty under fluoroscopic guidance combined with epidurogram for sacral insufficiency fracture resulting from metastatic tumor and osteoporosis. Pain Physician. 2016;19:E473–80.
18. Denis F. The three column spine and its significance in the classification of acute thoracolumbar spinal injuries. Spine (Phila Pa 1976). 1983;8:817–831. doi:10.1097/00007632-198311000-00003
19. Dmytriw AA, Talla K, Smith R. Percutaneous sacroplasty for the management of painful pathologic fracture in a multiple myeloma patient: case report and review of the literature. Neuroradiol J. 2017;30:80–83. doi:10.1177/1971400916678642
20. Waites MD, Mears SC, Richards AM, et al. A biomechanical comparison of lateral and posterior approaches to sacroplasty. Spine (Phila Pa 1976). 2008;33(20):E735–738. doi:10.1097/BRS.0b013e31817ecc22
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.


