Back to Journals » International Journal of General Medicine » Volume 14
Feasibility of Iodine-131 6β-Methyl-Iodo-19 Norcholesterol (NP-59) Scintigraphy to Complement Adrenal Venous Sampling in Management of Primary Aldosteronism: A Case Series
Authors Lee J , Ha J , Lee SK , Park HL, Kim SH, Lim DJ , Lee JM, Chang SA, Kang MI, Kim MH
Received 29 October 2020
Accepted for publication 8 December 2020
Published 1 March 2021 Volume 2021:14 Pages 673—680
DOI https://doi.org/10.2147/IJGM.S288774
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Scott Fraser
Jeongmin Lee,1 Jeonghoon Ha,2 Sang-Kuon Lee,3 Hye Lim Park,4 Sung-Hoon Kim,5 Dong-Jun Lim,2 Jung Min Lee,1 Sang-Ah Chang,1 Moo Il Kang,2 Min-Hee Kim1
1Division of Endocrinology and Metabolism, Department of Internal Medicine, Eunpyeong St. Mary’s Hospital, College of Medicine, The Catholic University of Korea, Seoul, Republic of Korea; 2Division of Endocrinology and Metabolism, Department of Internal Medicine, Seoul St. Mary’s Hospital, College of Medicine, The Catholic University of Korea, Seoul, Republic of Korea; 3Division of Pancreatobiliary Surgery, Department of Surgery, Seoul St. Mary’s Hospital, College of Medicine, The Catholic University of Korea, Seoul, Republic of Korea; 4Division of Nuclear Medicine, Department of Radiology, Eunpyeong St. Mary’s Hospital, College of Medicine, The Catholic University of Korea, Seoul, Republic of Korea; 5Division of Nuclear Medicine, Department of Radiology, Seoul St. Mary’s Hospital, College of Medicine, The Catholic University of Korea, Seoul, Republic of Korea
Correspondence: Min-Hee Kim
Division of Endocrinology and Metabolism, Department of Internal Medicine, Eunpyeong St. Mary’s Hospital, College of Medicine, The Catholic University of Korea, Seoul, Republic of Korea
Tel +82-2-2030-4348
Fax +82-2-2030-4641
Email [email protected]
Purpose: Primary aldosteronism (PA) is mainly comprised of aldosterone-producing adenoma and bilateral idiopathic adrenal hyperplasia. Current guidelines recommend adrenal venous sampling (AVS) as a gold standard method to classify the subtypes. However, because of technical challenges in AVS including invasiveness of AVS and a wide range of success rate for cannulation, it is not uncommon that appropriate decisions could not be made depending on AVS. The aim of this study is to elucidate the proper role of I‑131‑6β‑iodomethyl‑norcholesterol (NP-59) scintigraphy in management of PA.
Patients and Methods: Between January 2009 and October 2018, patients with PA were retrospectively reviewed for the study. Five patients were included in the study who had NP-59 scintigraphy with non-conclusive AVS results or without AVS. We described the clinical outcome of patients in whom clinical decisions were made according to NP-59 scintigraphy results.
Results: Patients in the presenting cases were diagnosed for PA. AVS, the most reliable test to identify unilateral APA, were not applicable because of hypersensitivity to contrast dye (patient 1), and use of antiplatelet agents after acute cerebral infarction (patient 2). NP-59 scintigraphy was performed in patients 3 and 4 whose result of AVS and CT scan were inconsistent. In patient 5, who had bilateral adrenal adenomas (two in the left and one in the right adrenal gland), both unsuccessful catheterization and coexistence of cortisol overproduction made AVS results unreliable.
Conclusion: Based on clinical outcomes of these case series, it is noticeable that NP-59 scintigraphy could play a substantial role in management of PA in selected cases.
Keywords: hyperaldosteronism, angiography, aldosterone, radionuclide imaging
Introduction
Primary aldosteronism (PA) is a common cause of secondary hypertension (HTN) consisting of about 5–10% of subjects with hypertension.1–4 Prolonged exposure to excessive aldosterone in PA could cause cardiovascular or renal damage. In addition, cardiovascular morbidity and mortality rates were higher in subjects with PA than in subjects with essential HTN, despite similar blood pressure (BP) levels.5 Thus, the early diagnosis and appropriate treatment of PA are crucial to prevent its associated sequelae.
PA is mainly categorized into aldosterone-producing adenoma (APA) and bilateral idiopathic adrenal hyperplasia (BAH), which can be managed by surgical removal and medication, respectively.6 Surgical cure rates of APA were reported to be 30–60%.7 In BAH, a mineralocorticoid receptor antagonist is considered as the treatment of choice. As the treatment is completely different depending on the subtypes, discrimination of subtypes of PA is essential for adequate management. The clinical practice guidelines of the Endocrine Society suggest adrenal computed tomography (CT) scan as the initial image approach after screening for PA with plasma aldosterone/renin ratio (ARR).8 Although a CT scan can exclude the large adrenal mass that is suggestive of adrenocortical carcinoma, it has relatively low sensitivity and specificity for detection of an APA.9 Furthermore, CT scans cannot distinguish an APA from a nonfunctioning incidentaloma, which can coexist with bilateral BAH.10 Currently, adrenal vein sampling (AVS) has been advocated as the gold standard for differentiation of APA from BAH. Despite the high sensitivity and specificity of AVS, the procedure is invasive and technically challenging. The rate of successful bilateral renal vein cannulation was reported as in a wide range from 42–98%.11 Thus, a supplementary diagnostic tool for the differentiation of PA subtypes is still required, especially for cases in which AVS could not be performed or where AVS results are inconclusive, including unsuccessful cannulation.
I‑131‑6β‑iodomethyl‑norcholesterol (NP‑59) scintigraphy with dexamethasone suppression was introduced for adrenal cortex imaging.12 NP-59, a radiolabeled cholesterol analog, is a good marker of adrenal cholesterol uptake. With dexamethasone suppression, NP-59 scintigraphy has been suggested as a supplementary tool for the functional localization of PA.12 Currently, it is not frequently performed or recommended for the management of PA due to its limited resolution. In fact, NP-59 scintigraphy has shown low sensitivity for the detection of APA.5 Nonetheless, NP-59 scintigraphy still remains clinically useful because AVS is sometimes insufficient to make a clinical decision.
In this study, we aimed to present the outcomes of PA patients for whom clinical decisions were made based on NP-59 scintigraphy and to elucidate a complementary role of NP-59 scintigraphy to AVS in patients who were either contraindicated to AVS or whose AVS results were inconclusive.
Patients and Methods
Subjects
Patients with PA who had undergone NP-59 scintigraphy between January 2009 and October 2018 were retrospectively reviewed. PA was diagnosed on the basis of the results of the ARR and saline loading test. After the exclusion of patients with conclusive AVS results, five patients were included in the study.
Hormone Assay for Screening and Confirmative Diagnosis
Plasma aldosterone concentration (PAC) (SPAC‐S Aldosterone Kit; Fujirebio, Tokyo, Japan) and cortisol concentrations (Cortisol RIA kit; Shizuoka, Japan) were measured using a radioimmunoassay kit. The ARR was calculated by dividing the plasma aldosterone concentration by plasma renin activity. A combination of the ARR greater than 20 and the plasma aldosterone greater than 15 ng/dL suggests PA.8,13,14 The diagnosis was confirmed by non-suppressed aldosterone after 2 L of 0.9% saline infusion over 4 hours starting at 8:00 AM. Patients stayed in the supine position during the intravenous saline loading. APC and renin were measured at baseline and after 4 hours, together with BP. PA was confirmed if post-saline loading PAC was greater than 5 ng/dL.8
AVS (Adrenal Venous Sampling)
AVS was performed by a single interventional radiologist who had experience of over 10 years in interventional vascular procedures. To avoid diurnal fluctuations, AVS was performed in the morning. Synthetic tetracosapeptide (cosyntropin, 250 mg) was injected intravenously for even stimulation of aldosterone and cortisol secretion. Catheters were located at the inferior vena cava (IVC) and the right/left adrenal veins to obtain blood samples simultaneously. Samples were acquired at 10 and 20 minutes after administration of a bolus dose cosyntropin.15
Preparation Before NP-59 Scintigraphy and NP-59 Scintigraphy Procedures
Seven days before and 5 days after NP-59 intravenous injection, 4 mg/day of oral dexamethasone (1 mg, 4 times daily) was given to patients to suppress the cholesterol uptake in normal adrenal glands. Lugol’s solution was given though oral route for 10 days (from 2 days before injection of radiotracer) to block uptake of free I-131 in the thyroid. Then 37 MBq (1 mCi) of NP-59 was intravenously injected. A whole body scintigraphy was obtained 72, 96, and 120 hours after radiotracer injection. Single-photon emission computed tomography (SPECT) was performed on the fourth or the sixth day after injection.16
Definition of Clinical Outcome After Adrenalectomy
The outcome was assessed 3 months after adrenalectomy. Due to limited data availability, the final clinical outcome was evaluated based on the clinical components, including BP and medications, which were suggested by the Primary Aldosteronism Surgical Outcome study.17 Complete clinical success was defined when patients showed a normal BP without any antihypertensive medications. Partial clinical success was defined as a reduction in BP levels or maintaining the same BP levels before the therapy under the same or a reduced dose of antihypertensive agents. Absent clinical success was when patients showed no clinical improvement.
Results
Characteristics of Cases
Table 1 showed the characteristics of cases included in the study. Median follow-up duration was 5.0 months (range=2.5–39 months). Postoperative histologic results presented adrenal cortical adenomas in all five patients.
 |
Table 1 Patients’ Characteristics |
Case 1
A 61-year-old female who presented refractory HTN (systolic BP (SBP) >160 mmHg), despite multiple antihypertensive medications for many years, was referred in order to evaluate the secondary cause of HTN. From the results of PAC (29.6 ng/dL), plasma renin activity (PRA) (0.01 ng/mL/h), and an ARR exceeding 30, PA was suggested. Confirmative diagnosis for PA was induced from the saline loading test which was shown as non-suppressed production of aldosterone (10.96 ng/dL). An adrenal CT scan showed a 1.3 cm-sized mass in the right adrenal gland. However, she had experienced a hypotensive event causing near syncope during adrenal CT scanning using contrast dye. Because of hypersensitivity to contrast dye and patient’s refusal, AVS could not be performed. Instead, NP-59 scintigraphy was performed to evaluate the functional status of the adenoma found in an adrenal CT scan. A hot spot was observed in the right supra-renal area on 96-hour images. It became more intense and discrete on the 120-hour images than prior images. According to NP-59 scintigraphy results, she had right adrenalectomy. The patient showed partial clinical success. BP was well controlled without hypokalemia, though spironolactone was reduced from 75 mg to 12.5 mg after surgery. She was planned to stop spironolactone during follow-up, but was not followed up thereafter.
Case 2
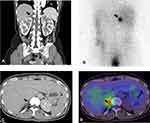
A 54-year-old female who presented with dysarthria was diagnosed with acute cerebral infarction at the left basal ganglia. She was treated with antiplatelet agents after thrombolytic therapy. High BP (SBP and diastolic BP [DBP]; 224/149) and hypokalemia (2.1 mEq/L) were evident at presentation. PAC (60.0 ng/dL), PRA (0.05 ng/mL/h), and ARR (1,200) were suggestive of PA. As her DBP was above 100 mmHg, despite taking antihypertensive agents, and she was in the acute phase of cerebral infarction, the saline loading test, which could cause a steep increase in BP, was avoided. On the adrenal CT scan, a 2.7 cm-sized mass was detected in the right adrenal gland (Figure 1A). For the identification of PA subtypes, NP-59 scintigraphy was applied instead of AVS to avoid the risk of bleeding due to antiplatelet therapy. NP-59 scintigraphy displayed an intense uptake on 72-hours images, which became more intense and discrete on 96-hour images, corresponding to the right adrenal gland lesion detected in the CT scan (Figure 1B). This patient underwent right adrenalectomy based on NP-59 scintigraphy and adrenal CT scan results.
The patient achieved complete clinical success after adrenalectomy. The potassium levels remained within the normal range without potassium supplements.
Case 3
A 54-year-old female was referred due to an adrenal incidentaloma. She had HTN, hypokalemia (3.3 mEq/L), and a 2.0 cm-sized left adrenal mass on adrenal CT scan without evidence of adrenal thickening on both sides. PA was suggested from PAC (32.4 ng/dL), PRA (0.54 ng/mL/h), and ARR (60.1). PAC was not suppressed (14.7 ng/dL) in the saline loading test. Catheterization of bilateral adrenal veins was successful based on the ratio of cortisol concentration for each adrenal vein and IVC. In the lateralization index (2.6 after ACTH stimulation) using aldosterone per cortisol (AC) ratio of the left (dominant side) divided by AC ratio of the right (non-dominant side), BAH was suggested (less than 3).13 In contrast, when the AC ratio of the non-dominant side was divided by AC ratio of the IVC (0.38 after ACTH stimulation), the results favored an APA in the left adrenal gland (less than 1). As contradictory results were observed between adrenal CT scan and AVS, NP-59 scintigraphy was planned. An active lesion in the left adrenal gland on the 72- and 96-hour images was shown, which was concordant with adrenal CT findings. Adrenalectomy of the left adrenal gland was performed and patient 3 achieved complete clinical success after adrenalectomy.
Case 4
A 56-year-old female was referred for severe hypokalemia (1.6 mEq/L) and paralysis. She had resistant HTN despite three types of antihypertensive medications including thiazide. PAC was 119.2 ng/dL, PRA was 0.06 ng/mL/h, and ARR was 1,987. PAC measured in saline loading test was 60.8 ng/dL. A 1.5 cm-sized adrenal mass was detected in the right adrenal gland on adrenal CT scan. However, BAH was suggested on the first AVS result. The lateralization index after ACTH stimulation was 29 and contralateral suppression index was 1.8 BAH was suggested according to the results of AVS. Potassium replacement and aldosterone antagonist were prescribed on the basis of the AVS result. However, there was no correction of hypokalemia (2.2 mEq/L), and HTN was persistent with an increased dosage of the antihypertensive agent. In clinical suspicion of PA, the second AVS was performed but it also showed a result consistent with BAH (lateralization index after ACTH stimulation; 2.3). Though the patient was treated with an aldosterone antagonist for 10 months according to repeated AVS results, moderate-to-severe hypokalemia was not resolved (1.9–2.5 mEq/L). NP-59 scintigraphy was performed to identify whether a dominant lesion existed. A focal hot uptake was noticed in the right adrenal gland on 72-hours images and a right adrenalectomy was performed.
The patient also showed less than 140 mmHg of SBP and 90 mmHg of DBP with a reduced dose of antihypertensive agents, which did not include an aldosterone antagonist after adrenalectomy.
Case 5
A 31-year-old female presented with uncontrolled BP and severe hypokalemia (1.9 mEq/L). On evaluation of secondary HTN, PAC was 49.7 ng/dL and PRA was undetectable, which strongly suggested PA. In addition, Cushing syndrome was also suspected based on 24-hour urine free cortisol (75.8 µg/day, using a radioimmunoassay kit, reference range 13.7–75.3 µg/day) and 16.09 µg/dL of serum cortisol levels after 1 mg overnight dexamethasone suppression test. Subsequently, the standard low dose dexamethasone suppression test (2 mg per day for 48 hours) was performed and serum cortisol levels (24.24 µg/dL) after low dose dexamethasone suppression test confirmed adrenal hypersecretion of cortisol. Adrenal CT scan revealed three adenomas (3.0 cm- and 1.7 cm-sized adenomas in the left and a 1.3 cm-sized adenoma in the right adrenal gland). Though AVS was performed to lateralize the source of aldosterone overproduction, interpretation was not available due to coexisting hypersecretion of cortisol, which could cause an increase in peripheral to adrenal cortisol ratio suggestive of unsuccessful catheterization. To provide the functional status of each adenoma in an adrenal CT scan, NP-59 scintigraphy was performed. Two dominant hot uptakes were observed in the left adrenal gland on the first NP-59 scintigraphy images, the left unilateral adrenalectomy was planned firstly considering a sequential therapy. The second NP-59 scintigraphy with SPECT/CT was performed to decide the extent of surgery on the remaining right adrenal gland (Figure 1C); whether to remove the whole right adrenal gland or only the adenoma with partial adrenalectomy. NP-59 scintigraphy with SPECT/CT, which was performed after the left adrenalectomy, revealed a localized hot uptake in the upper area of the right adrenal gland (Figure 1D). After left adrenalectomy based on the first NP-59 scintigraphy, partial right adrenalectomy was performed to preserve the rest of the right adrenal gland.
In the patient, after the left adrenalectomy, hypersecretion of cortisol was completely resolved. However, the PAC continued to rise along with suppressed PRA and hypokalemia. The remaining right adrenal mass on adrenal CT scan was suspected as a source of aldosterone hypersecretion. After right partial adrenalectomy, she took 5 mg of prednisone per day without any antihypertensive agent or potassium supplement.
Discussion
AVS is considered as a gold standard method in discriminating the subtypes of PA.2 However, the invasiveness of the procedure, use of contrast dye and operator dependency (requiring a highly experienced operator to obtain reliable data by proper catheterization) are potential disadvantages of AVS. In addition, AVS could be unavailable in certain conditions such as bleeding diathesis, impending renal failure or presence of contrast dye hypersensitivity. In this study, we presented the feasibility of NP-59 scintigraphy to complement AVS in management of PA.
Though NP-59 scintigraphy is not used in the western countries such as the US, it is considered as a supplementary or alternative diagnostic tool in some Asian countries.18,19 NP-59 scintigraphy has several disadvantages. The accuracy of identifying the subtype of PA has been reported from 47–94%. 20,21 Additionally, resolution of the image is relatively low for detection of small sized adrenal mass (usually less than 1.5 cm) on planar scintigraphy.22 NP-59 scintigraphy is also inconvenient because it requires several preparations. Other than the discontinuation of antihypertensive agents such as spironolactone to prevent interferences, dexamethasone suppression for a certain period to reduce uptake of normal adrenal gland is needed.23 Thus, use of NP-59 scintigraphy has been restricted in the management of PA; however, this method could still prove useful. Recent studies have been refocused on the feasibility of NP-59 scintigraphy with SPECT/CT.23,24 SPECT-CT with NP-59 scintigraphy has been contended to improve resolution and detection of smaller nodules. Simultaneous use of SPECT-CT with NP-59 scintigraphy improved the accuracy of NP-59 scintigraphy.25 Moreover, Chen et al26 demonstrated that NP-59 scintigraphy with SPECT/CT had a role in early diagnosis for subclinical or atypical feature of PA or PA combined chronic kidney disease. In fact, NP-59 scintigraphy was suggested in inconclusive cases of AVS and in prediction of the existence of subclinical Cushing syndrome in adrenal incidentaloma.27,28 Unilateral uptake of NP-59 scintigraphy has been associated with surgical cure in PA and Cushing syndrome compared to nodules without NP-59 uptake.29 In BAH, bilateral symmetrical NP-59 uptake is presented. However, bilateral asymmetrical uptake of NP-59 suggested contralateral suppression of normal adrenal gland or hyperfunctioning adenoma in BAH.30 In case 5, initial NP-59 scintigraphy showed symmetric uptake in the bilateral adrenal gland. Sequential adrenalectomy was performed based on NP-scintigraphy with SPECT/CT. Bilateral adenoma was confirmed by histopathology.
In the current cases, successful visualization of functioning lesions by NP-59 scintigraphy (in conjunction with SPECT/CT scan in case 5) showed a pivotal role in the management of PA patients. AVS was essential to provide the correct treatment strategies considering the low diagnostic accuracy of CT in patients above 35 years of age. However, hypersensitivity to contrast dye and high risk of bleeding made performing AVS difficult. Of course, use of Gadolinium contrast dye instead of the iodinated radiocontrast dye in AVS could be considered as described in a previous report. 29
This study has some limitations. First, data were collected and analyzed retrospectively. Second, due to the absence of AVS (case 1.2) and incomplete interpretation (case 3.5) of AVS results, the accuracy of AVS and NP-59 scintigraphy could not be compared precisely. Third, clinical outcomes were decided by clinical responses, not by biochemical measurements such as serum aldosterone levels and ARR. Finally, because all the adrenal masses (except one in case 5) were larger than 1.0 cm in size, it is not possible to suggest the role of NP-59 scintigraphy in management of small adenomas. The simultaneous SPECT-CT was performed in one case. A further prospective study might be needed to validate our suggestions.
Conclusion
NP-59 scintigraphy could play a supplementary role in differential diagnosis of PA patients when AVS could not provide an appropriate management. In several exceptional conditions such as the presence of contrast dye hypersensitivity, use of anticoagulation agents or prior unilateral adrenalectomy, NP-59 scintigraphy might be an alternative option to guide adequate management of PA.
Ethics and Consent
This study complied with the ethical standards of the Helsinki Declaration and was approved by the Catholic University of Korea, Catholic Medical Center, Seoul St. Mary’s Hospital Institutional Review Board (IRB approval No. KC11RISI0163). The written informed consent was waived by IRB due to use of anonymized data without subject information. The author ensures the confidentiality of patient information and data which were stored in the form of encrypted files and were accessible only to the responsible investigator.
Consent for Publication
Written informed consent was obtained from the patients to have the case details and any accompanying images published.
Acknowledgments
There was no financial support for the research, authorship, and/or publication of this article.
Disclosure
The authors declare that there is no conflict of interest.
References
1. Mulatero P, Stowasser M, Loh K-C, et al. Increased diagnosis of primary aldosteronism, including surgically correctable forms, in centers from five continents. J Clin Endocrinol Metab 2004; 89: 1045–1050. 10.1210/jc.2003-031337
2. Douma S, Petidis K, Doumas M, et al. Prevalence of primary hyperaldosteronism in resistant hypertension: a retrospective observational study. Lancet 2008; 371: 1921–1926. 10.1016/S0140-6736(08)60834-X
3. Sica DA Endocrine causes of secondary hypertension. J Clin Hypertens 2008; 10: 534–540 10.1111/j.1751-7176.2008.08097.x
4. Thomas RM, Ruel E, Shantavasinkul PC, et al. Endocrine hypertension: an overview on the current etiopathogenesis and management options. World J Hypertens 2015; 5: 14–27. 10.5494/wjh.v5.i2.14
5. Chou CH, Chen YH, Hung CS, et al. Aldosterone impairs vascular smooth muscle function: from clinical to bench research. J Clin Endocrinol Metab 2015; 100: 4339–4347. 10.1210/jc.2015-2752
6. Sawka AM, Young WF
7. Funder JW, Carey RM, Mantero F, et al. The management of primary aldosteronism: case detection, diagnosis, and treatment: an endocrine society clinical practice guideline. J Clin Endocrinol Metab 2016; 101: 1889–1916. 10.1210/jc.2015-4061
8. Magill SB, Raff H, Shaker JL, et al. Comparison of adrenal vein sampling and computed tomography in the differentiation of primary aldosteronism. J Clin Endocrinol Metab 2001; 86: 1066–1071. 10.1210/jcem.86.3.7282
9. Kempers MJ, Lenders JW, van Outheusden L, et al. Systematic review: diagnostic procedures to differentiate unilateral from bilateral adrenal abnormality in primary aldosteronism. Ann Intern Med 2009; 151: 329–337. 10.7326/0003-4819-151-5-200909010-00007
10. Stewart PM, Allolio B Adrenal vein sampling for Primary Aldosteronism: time for a reality check. Clin Endocrinol 2010; 72: 146–148. 10.1111/j.1365-2265.2009.03714.x
11. Sarkar SD, Beierwaltes H, Ice RD, et al. A new and superior adrenal scanning agent, NP-59. J Nucl Med 1975; 16: 1038–1042.
12. Sarkar SD, Cohen EL, Beierwaltes WH, et al. A new and superior adrenal imaging agent, 131I-6beta-iodomethyl-19-nor-cholesterol (NP-59): evaluation in humans. J Clin Endocrinol Metab 1977; 45: 353–362. 10.1210/jcem-45-2-353
13. Young WF Primary aldosteronism: renaissance of a syndrome. Clin Endocrinol 2007; 66: 607–618. 10.1111/j.1365-2265.2007.02775.x
14. Tiu S-C, Choi C-H, Shek -C-C, et al. The use of aldosterone-renin ratio as a diagnostic test for primary hyperaldosteronism and its test characteristics under different conditions of blood sampling. J Clin Endocrinol Metab 2005; 90: 72–78. 10.1210/jc.2004-1149
15. Rossi GP, Barisa M, Allolio B, et al. The Adrenal Vein Sampling International Study (AVIS) for identifying the major subtypes of primary aldosteronism. J Clin Endocrinol Metab 2012; 97: 1606–1614. 10.1210/jc.2011-2830
16. Avram AM, Fig LM, Gross MD, Adrenal gland scintigraphy. Semin Nucl Med 2006; 36: 212–227. 10.1053/j.semnuclmed.2006.03.004
17. Williams TA, Lenders JW, Mulatero P, et al. Outcomes after adrenalectomy for unilateral primary aldosteronism: an international consensus on outcome measures and analysis of remission rates in an international cohort. Lancet Diabetes Endocrinol 2017; 5: 689–699. 10.1016/S2213-8587(17)30135-3
18. Nishikawa T, Omura M, Satoh F, et al. Guidelines for the diagnosis and treatment of primary aldosteronism-The Japan Endocrine Society 2009. Endocrine 2011; 58: 711–721. 10.1507/endocrj.EJ11-0133
19. Lee JM, Kim MK, Ko SH, et al. Clinical guidelines for the management of adrenal incidentaloma. Endocrinol Metab 2017; 32: 200–218. 10.3803/EnM.2017.32.2.200
20. Yen RF, Wu VC, Liu KL, et al. 131I-6β-iodomethyl-19-norcholesterol SPECT/CT for primary aldosteronism patients with inconclusive adrenal venous sampling and CT results. J Nucl Med 2009; 50: 1631–1637. 10.2967/jnumed.109.064873
21. Marcello DM, García SI, Muñoz JL, et al. NP-59 test for preoperative localization of primary hyperaldosteronism. Langenbecks Arch Surg 2017;402(2):303–308. 10.1007/s00423-017-1561-1
22. Nomura K, Kusakabe K, Maki M, et al. Iodomethylnorcholesterol uptake in an aldosteronoma shown by dexamethasone-suppression scintigraphy: relationship to adenoma size and functional activity. J Clin Endocrinol Metab 1990; 71: 825–830. 10.1210/jcem-71-4-825
23. Gross M, Valk T, Swanson D, et al. The role of pharmacologic manipulation in adrenal cortical scintigraphy. Semin Nucl MEd 1981,
24. Cava GL, Imperiale A, Olianti C, et al. Spect semiquantitative analysis of adrenocortical 131i-6b-iodomethyl-norcholesterol uptake to discriminate subclinical and preclinical functioning adrenal incidentaloma. J Nucl Med 2003; 44: 1057–1064.
25. Wu MH, Liu FH, Lin KJ, et al. Diagnostic value of adrenal iodine-131 6-beta-iodomethyl-19-norcholesterol scintigraphy for primary aldosteronism: a retrospective study at a medical center in North Taiwan. Nucl Med Commun. 2019;40:568–575. 10.1097/MNM.0000000000000987
26. Chen YC, Su YC, Wei CK, et al. Diagnostic value of I-131 NP-59 SPECT/CT scintigraphy in patients with subclinical or atypical features of primary aldosteronism. J Biomed Biotechnol. 2011;2011:209787. 10.1155/2011/209787
27. Wong KK, Komissarova M, Avram AM, et al. Adrenal cortical imaging with I-131 NP-59 SPECT-CT. Clin Nucl Med 2010; 35: 865–869. 10.1097/RLU.0b013e3181f4a16d
28. Sasamura H, Hashimoto S, Kuribayashi S, et al. Use of gadolinium contrast adrenal venography for the assessment of primary aldosteronism in a patient with iodine allergy. Endocrine 2004; 51: 487–492. 10.1507/endocrj.51.487
29. Miller BS The role of radiologic studies in the evaluation and management of primary hyperaldosteronism. Surgery 2010; 147: 174–175.
30. Chan H, Cheng L Use of NP-59 (131I-iodocholesterol) scan as a potential alternative to adrenal venous sampling in the investigation of primary aldosteronism: a 5-year retrospective study. Hong Kong J Radiol 2014; 17: 176–181 10.12809/hkjr1414230
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.