Back to Archived Journals » Orphan Drugs: Research and Reviews » Volume 5
Fahr's disease: current perspectives
Received 2 March 2015
Accepted for publication 4 May 2015
Published 16 July 2015 Volume 2015:5 Pages 43—49
DOI https://doi.org/10.2147/ODRR.S63388
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 4
Editor who approved publication: Dr Lise Aagaard
Xin You Tai, Amit Batla
UCL Institute of Neurology, Queen Square, London, UK
Abstract: Based on original descriptions of brain calcification by Theodor Fahr, brain calcification, and more specifically basal ganglia calcification, is referred to as Fahr's syndrome. Recent identification of genetic mutations has concerted the description of this erstwhile heterogeneous condition which we refer to here as Fahr's disease. Fahr's disease refers to idiopathic calcification of the basal ganglia without a secondary (non-genetic) cause. Idiopathic basal ganglia calcification (IBGC) is another term, which offers a more accurate description of this condition. Within the last 2 years, genetic mutations for IBGC have been described in SLC20A2, PDGFB, and PDGFRB. These findings broaden our understanding of the pathophysiology and encourage a search for specific treatment options in this rare but disabling condition. Clinically, parkinsonism, dystonia, and other movement disorders are the most common clinical features, but psychiatric features can predominate. Cases with confirmed genetic mutations reveal new clinical features linked with Fahr's disease, such as headaches. Computed tomography or magnetic resonance imaging is usually the trigger for suspecting Fahr's disease and important for identifying the calcification pattern. Symmetric calcification of the globus pallidus, thalamus, and dentate nucleus is the most common pattern in IBGC, but other parts of the brain such as the cerebellum are involved. Treatment of Fahr's disease is currently limited and is largely symptomatic. A better understanding of this condition in light of genetic findings is important to improve the clinical diagnosis and develop specific treatment options.
Keywords: Fahr's disease, basal ganglia calcification, parkinsonism, calcium
Historical perspective
Idiopathic calcification of cerebral vessels or “Idiopathische Verkalkung der Hirngefässe” was described by the German neurologist Theodor Fahr in 1930.1 His report was based on an 81-year-old patient with dementia and “immobility without paralysis”. This later led to the eponym “Fahr’s disease” to describe idiopathic brain calcification. However, for historical accuracy, it was actually Delacour from France in 18502 who first recognized “ossification of brain capillaries” in a 56-year-old man whom he described as having “stiffness and weakness of the lower extremities with tremor”.
After Delacour’s report but before Fahr’s description, Bamberger wrote an article in 1855 entitled “Observations and comments about brain diseases”, in which he described a woman with mental retardation and seizures who was found to have calcifications of the cerebral vessels.3 Some authors have argued that “Fahr’s disease” is a misnomer because Fahr was neither the first to describe calcification in the brain nor did he contribute significantly to the understanding of this disorder of arteriosclerotic calcification, which is well recognized and common.4 However, Fahr’s description was the first of what has also been labeled as “idiopathic non-arteriosclerotic cerebral calcification”. It is also interesting to note that medical parlance at that time was not conditioned to so-called “parkinsonism”, which became much more commonly used in the early 20th century.
Radiologically, using roentgenography, calcification “mainly in the basal ganglia” was described by Fritzsche in 1924.5 Interest was drawn again to this disease when Boller et al described nine members of the same family with familial idiopathic brain calcification in 1977.6
There are at least 35 additional names used in the literature to describe the same condition of idiopathic basal ganglia calcification (IBGC),7 a term used to support genetic terminology. Another commonly used term is bilateral striopallidodentate calcinosis.8 However, “Fahr’s disease” has become closely associated with the condition and is frequently used in the literature to describe it. Despite popular reference, IBGC is a more appropriate term for this condition and will be used in this article interchangeably with Fahr’s disease. The choice of the term IBGC is additionally based on the fact that earlier studies localized the genetic basis of Fahr’s disease as IBGC1, IBGC2, IBGC3, and IBGC4.9–12
In this review, we aim to disentangle what should or should not be considered IBGC, describe the underlying genetics, and focus on confirmed genetic descriptions to offer a unique insight into the clinical features specific to IBGC. An approach to diagnosis, radiological identification, and management of IBGC is discussed.
When is brain calcification not Fahr’s disease?
Intracranial calcification on brain computed tomography (CT) is a common finding, with studies reporting variable incidence rates. Two studies performed in the 1980s examining 7,040 and 6,348 patient scans revealed that 1.02% and 1.1% of patients, respectively, had symmetric intracranial calcification,13,14 while a study by Yamada et al reported symmetric brain calcification in 17.2% and 20.6% of CT brain images over 1 year across two sites.15 This significant variation may be explained by improved access to CT imaging and an aging scanned population, but also reflects the need to better understand what is and what is not Fahr’s disease.4
Basal ganglia calcification can be secondary to a long list of causes, as outlined described in Table 1. The calcification can be unilateral or bilateral, and should not be called Fahr’s disease because they are not idiopathic. It is a common misconception that any brain calcification can be called Fahr’s disease, and stems from the fact that Fahr described a case of brain calcification without an underlying cause. This is one argument for using the term IBGC instead of Fahr’s disease. Considering its idiopathic label, the etiology of IBGC is not completely unknown and several responsible genes have been identified.16 However, like some other “idiopathic” disorders, such as Parkinson’s disease, the term idiopathic is often applied to genetic cases.
Pathophysiology and genetics
Our understanding of the pathophysiology of IBGC has evolved significantly since the initial suspicion of “ossification” of blood vessels1 and the description by Fahr. Genetic advances have revealed that IBGC is genetically heterogeneous, with four genetic loci described based on linkage and haplotype analyses. IBGC-related gene loci have been mapped to 14q11.2–21.3 (IBGC1),9 2q37 (IBGC2),10 8p21.1–q11.13 (IBGC3),11 and 5q32 (IBGC4).12 SLC20A2 was the first gene reported (in the IBGC3 region), and so far 218 patients from 29 IBGC families are documented to have mutation in SLC20A2.17,18 The fourth locus was identified in 2013 and linked to PDGFRB using exome sequencing in two affected family members with IBGC.12 In a more recent report of genetic analysis in six families, nonsense and missense mutations in the gene encoding platelet-derived growth factor-beta (PDGFB), the main ligand for PDGFRB, was reported in familial IBGC.19 Experimental mice expressing 50% less endothelial PDGFB were shown to developed substantial brain calcification at 1 year of age. Thus, genetic mutations in SLC20A2, PDGFRB, and PDGFB have been found so far and additional genes will likely be identified. This opens up an era of more advanced understanding and possible therapeutic targets to prevent progressive calcification changes in IBGC.
The genetic mutations identified so far have different pathogenic mechanisms. Loss of endothelial PDGFB is the proposed mechanism, and this correlates with the degree of pericyte and blood–brain barrier deficiency. SLC20A2, however, is a member of the type III sodium-dependent phosphate transporter family, and loss-of-function mutations may lead to regional accumulation of inorganic phosphate in the brain, causing deposition of calcium phosphate.
Combined, these facts suggest that IBGC is not a monogenic disorder, and several mechanisms of calcium or phosphate transport are involved in causing the end result of brain calcification. Most of these mechanisms are related to the vascular endothelium or blood–brain barrier physiology. This helps to explain why calcium and other mineral deposits in pathological cases have been predominantly in the walls of capillaries, arterioles, small veins, and perivascular spaces,20 and also helps, in retrospect, to accredit Fahr’s original observation of “idiopathic calcification of cerebral vessels”. This calcification may contain several other trace elements and may be complicated by neuronal degeneration and gliosis.21
Clinical presentation
A wide clinical spectrum of IBGC is described in the literature, and there are several reasons for this. First, most of the data are derived from individual case reports of this rare condition. Second, there is marked heterogeneity in the clinical presentation of genetically proven cases of IBGC. Even within the same family, individuals present with different clinical features or may be asymptomatic despite brain calcification seen on brain imaging. In one purpose-built registry of patients labeled as having bilateral striopallidodentate calcinosis, 38 patients were studied and 30 had autosomal dominant inheritance.8 The authors reported their mean patient age as 43±21 years, and found the mean age of published cases to be 38±17 years. The male to female ratio was 2:1. In the combined data (n=99), 32 were asymptomatic. Movement disorders were the most common features, and were seen in 55% of symptomatic patients with parkinsonism (57%), chorea (19%), tremor (8%), dystonia (8%), athetosis (5%), and orofacial dyskinesia (3%). Other features included cognitive impairment, cerebellar signs, gait disorders, speech disorder, pyramidal signs, psychiatric features, sensory changes, and pain.
Currently, with the advent of genetic testing, reports of IBGC confirm mutations in the SLC20A2, PDGFB, or PDGFRB genes. While it is likely that more causative genes will be found, it may be useful to examine clinical features in cases with genetic confirmation to attempt a focused clinical description of IBGC.
In one series of genetically confirmed cases with SLC20A2 mutation (n=33), almost half (n=15) were asymptomatic. Pallilalia, tremor, paroxysmal dyskinesia, dizziness, headaches, seizures, and psychiatric features were commonly described in the remainder.18
In another series with SLC20A2 mutation, including 108 affected patients from 28 families, movement disorders, parkinsonism, cognitive deficits, and psychiatric disturbances were seen most often.17
Similarly, in genetically proven cases with PDGFRB (n=15), almost half (n=7) were asymptomatic. Migrainous headaches, seizures, and psychiatric features, including attention deficit hyperactivity disorder and anxiety, were reported to be common features.12
It is interesting to note that some gene carriers are asymptomatic, while others have had migraine/headache with or without movement disorders.2 Considering that headaches are quite common and Fahr’s can be asymptomatic, it is difficult to ascribe certainty to this association.
A review of the clinical features in all published case series and case reports is found in Table 2, which collates clinical features described in IBGC patients with confirmed mutation in SLC20A2, PDGFRB, or PDGFB. While clinical heterogeneity is evident, a movement disorder such as parkinsonism is reported with the highest frequency followed by psychiatric symptoms of psychosis and depression. There is also a moderate frequency of headache reported, which is not a usual feature associated with IBGC.
  | Table 2 Summary of main clinical features described in patients with idiopathic basal ganglia calcification and confirmed PDGFRB, PDGFB, or SCL20A2 mutation |
Radiological findings
The main radiological investigation in IBGC is CT imaging, which is considered more sensitive than magnetic resonance imaging.8 All reports of IBGC with confirmed genetic mutation in the literature have radiological findings from CT imaging. Prior to the availability of CT scanning, skull roentgenogram or post-mortem analysis were used to identify intracranial calcification.
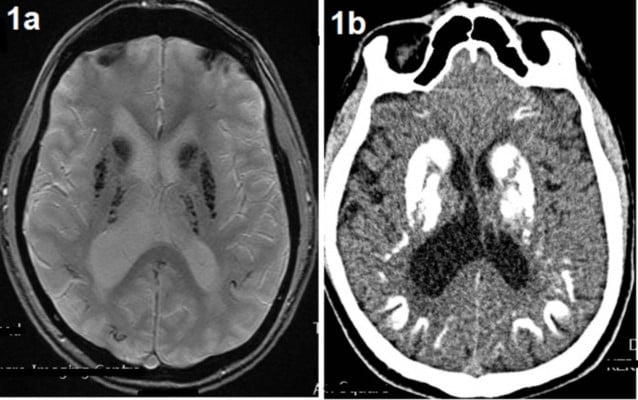
As with clinical features, the radiological findings are heterogeneous, and the most commonly observed is calcification of the basal ganglia, including globus pallidus, thalamus, and dentate nucleus in a symmetric pattern. Other areas of calcification less consistently reported include the brainstem, cerebellum, and the cortical and subcortical white matter (Figure 1).
Clinical correlation with radiological findings is similarly not straightforward. Members of IBGC families may demonstrate characteristic clinical features, including movement disorder and psychiatric symptoms, or be asymptomatic despite showing a similar pattern of calcifications. This is a consistent finding in families with PDGFB, PDGFRB, or SCL20A2 gene mutations.
Single proton emission CT using technetium-99m hexamethylpropyleneamine performed in patients with IBGC shows decreased perfusion to the basal ganglia bilaterally and to the cerebral cortices, which correlates with the clinical presentation.22 Interestingly, there was no significant difference between IBGC and controls on positron emission tomography scanning using fluorodopa.8
Approach to a patient with basal ganglia calcification or suspected Fahr’s disease
The usual clinical scenario in which a clinician starts to suspect IBGC is when a CT/magnetic resonance scan shows calcification in the brain, in particular the basal ganglia. It is important to interpret the findings with a clear consideration of age. People younger than 40 years almost always have a secondary cause, so it is likely that the calcification is secondary.23 The history is crucial to exclude drug-induced or poisoning-related brain calcifications, and may also help exclude central nervous system (CNS) infection, radiation exposure, or a metabolic cause. A general examination may help identify neurofibromas or other phakomatosis.24 Skeletal defects, such as a small fourth or fifth digit, point to a parathyroid disorder.25 The presence of psychiatric features can point to a parathyroid disorder and Fahr’s disease.26
The radiological pattern of calcification may guide clinical diagnosis. Usually, physiological calcification in the basal ganglia is limited to the globus pallidus and is bilateral and faint; other areas that may be involved include the pineal gland, falx, arachnoid granulation, and choroid plexus.27 If the calcification is denser, involves the thalamus and dentate nucleus in a symmetric pattern, and includes the brainstem, cerebellum, and areas of cortical and subcortical white matter, then IBGC should be suspected. Dystrophic calcification can be seen with previous brain injuries due to a variety of causes, such as brain trauma, surgery, previous stroke, or hemorrhage.28 The location of dystrophic calcification is often a clue that the diagnosis is not IBGC but secondary calcification in a previously injured part of the brain, and previous scans may be useful in confirming this. Calcification due to phakomatosis also follows a specific pattern, and if present, the changes may be diagnostic. Subependymal nodular calcification points to tuberous sclerosis,29 gyriform cortical calcifications suggest Sturge Weber syndrome,30 while nodular calcification in the cerebellum, periventricular region and disproportionate calcification in the choroid plexus suggests neurofibromatosis.31 With regard to congenital CNS infection, the onset and clinical presentation are diagnostic in most cases, but periventricular calcification is common. More specifically, leptomeningeal calcification suggests cryptococcosis or tuberculosis.32 Calcific granulomas can be seen anywhere in the brain, and may be due to neurocysticercosis, tuberculosis, or sarcoidosis, and are characteristically in the optic chiasm or pituitary stalk.33
When the clinical and radiological picture do not fit with phakomatosis, tumor, or CNS infection, blood tests may help in identifying the cause of basal ganglia calcification, the most common of which is hypoparathyroidism.25 Disorders of calcium metabolism must be excluded before a diagnosis of IBGC is made. Table 3 provides guidance on interpretation of blood results for calcium, phosphate, and parathyroid hormone levels to help diagnose underlying defects of calcium metabolism.
  | Table 3 Interpretation of blood results to diagnose parathyroid disorders |
If routine blood tests of metabolism and endocrine function are negative, it is important to consider idiopathic or familial brain calcification. Genetic testing for IBGC is possible, but is not routinely available and must be limited to selected cases where genetic confirmation is needed after excluding secondary etiologies of basal ganglia calcification. If performed, testing for all three gene mutations should be done to prevent ambiguity and further confusion with a negative result from only one of the known gene mutations.
Management
Symptomatic or secondary basal ganglia calcification is best treated by addressing the underlying cause. IBGC is best managed with a multidisciplinary team approach. After diagnosis, patients should undergo regular follow-up with the neurology team to monitor development or progression of symptoms. Other services offer invaluable input, eg, physiotherapy and occupational therapy for patients with a movement disorder and psychiatric services for mood disorders and psychosis.
Pharmacological therapy can be divided into two broad categories of symptom control tailored to the individual and disease modification. Treatment of movement disorders is a main consideration in IBGC. Levodopa is useful for patients with parkinsonism, while those with dystonia may benefit from trihexiphenidyl or treatment of focal symptoms with Botulinum toxin. Functionally disabling chorea may be improved with antichoreic medication such as sulpiride or tetrabenazine. Patients who develop psychiatric features can be offered a mood stabilizer or an antipsychotic. Antiepileptic medication should be considered in the presence of seizures.
Currently, there is no treatment regimen to modify the disease process. There has been no success in targeting the deposition or removal of cerebral calcium (and other associated minerals). Disodium etidronate was used in one patient, who demonstrated symptomatic relief but with no change in calcification on imaging.34 Use of nimodipine, a CNS-specific calcium channel blocking agent, has been unsuccessful.4 Increasing understanding of the underlying genetic pathophysiology of the disease may bring new avenues of treatment in the future.
Prognosis
It is understandably difficult to predict the prognosis for patients with IBGC due to the variable clinical presentation. Further, there appears to be little correlation between radiological findings, patient age, and clinical features.
Conclusion
Fahr’s disease (IBGC) is a rare condition with basal ganglia calcification and a varied clinical picture, predominantly of movement disorders and behavioral or cognitive changes. Causative genetic mutations have been identified in recent years that help characterize this as a distinct disorder and not just a syndrome. The new developments also provide insight into the underlying pathophysiology, but this needs further research to become useful in terms of therapeutic goals.
Disclosure
The authors report no conflicts of interest in this work.
References
Fahr T. [Idiopathische Verkalkung der Hirngefäße]. Zentralbl Allg Pathol. 1930;50:129–133. German. | |
Delacour A. [Ossification des capillaires du cerveau]. Ann Med Psychol. 1850;2:458–461. French. | |
Bamberger H. [Beobachtungen und bemerkungen uber hirnkrankheiten]. Verhandl Phy Med Ges. 1855;6:325–328. German. | |
Manyam BV. What is and what is not “Fahr”s disease”. Parkinsonism Relat Disord. 2005;11:73–80. | |
Fritzsche R. [Eine familiar auftrentende From vonolidophrenie mit vontgenologish nachveisbaren symmetrischein Kalkoablagerungenim Geheirn besounders in den Stammganglien Schweiz]. Arch Neurol Neurochir Psychiatr. 1924;1:29–33. | |
Boller F, Boller M, Gilbert J. Familial idiopathic cerebral calcifications. J Neurol Neurosurg Psychiatry. 1977;40:280–285. | |
Kuroiwa Y, Boller F, Boller M. Computed tomographic demonstration of idiopathic familial basal ganglia calcification. Comput Radiol. 1983;7:141–143. | |
Manyam BV, Bhatt MH, Moore WD, Devleschoward AB, Anderson DR, Calne DB. Bilateral striopallidodentate calcinosis: cerebrospinal fluid, imaging, and electrophysiological studies. Ann Neurol. 1992;31:379–384. | |
Geschwind DH, Loginov M, Stern JM. Identification of a locus on chromosome 14q for idiopathic basal ganglia calcification (Fahr disease). Am J Hum Genet. 1999;65:764–772. | |
Volpato CB, De Grandi A, Buffone E, et al. 2q37 as a susceptibility locus for idiopathic basal ganglia calcification (IBGC) in a large South Tyrolean family. J Mol Neurosci. 2009;39:346–353. | |
Dai X, Gao Y, Xu Z, et al. Identification of a novel genetic locus on chromosome 8p21.1-q11.23 for idiopathic basal ganglia calcification. Am J Med Genet B Neuropsychiatr Genet. 2010;153B:1305–1310. | |
Nicolas G, Pottier C, Maltête D, et al. Mutation of the PDGFRB gene as a cause of idiopathic basal ganglia calcification. Neurology. 2013;80:181–187. | |
Kazis AD. Contribution of CT scan to the diagnosis of Fahr’s syndrome. Acta Neurol Scand. 2009;71:206–211. | |
König P. Psychopathological alterations in cases of symmetrical basal ganglia sclerosis. Biol Psychiatry. 1989;25:459–468. | |
Yamada M, Tanaka M, Takagi M, et al. Evaluation of SLC20A2 mutations that cause idiopathic basal ganglia calcification in Japan. Neurology. 2014;82:705–712. | |
Batla A, Bhatia KP. A new gene for Fahr’s syndrome – PDGF-B. Mov Disord. 2014;29:307–317. | |
Hsu SC, Sears RL, Lemos RR, et al. Mutations in SLC20A2 are a major cause of familial idiopathic basal ganglia calcification. Neurogenetics. 2013;14:11–22. | |
Chen W-J, Yao X-P, Zhang Q-J, et al. Novel SLC20A2 mutations identified in southern Chinese patients with idiopathic basal ganglia calcification. Gene. 2013;529:159–162. | |
Keller A, Westenberger A, Sobrido MJ, et al. Mutations in the gene encoding PDGF-B cause brain calcifications in humans and mice. Nat Genet. 2013;45:1077–1082. | |
Duckett S, Galle P, Escourolle R, Poirier J, Hauw JJ. Presence of zinc, aluminum, magnesium in striopalledodentate (SPD) calcifications (Fahr’s disease): electron probe study. Acta Neuropathol. 1977;38:7–10. | |
Kozik M, Kulczycki J. Laser-spectrographic analysis of the cation content in Fahr’s syndrome. Arch Psychiatr Nervenkr (1970). 1978;225:135–142. | |
Kostic VS, Lukić-Ječmenica M, Novaković I, et al. Exclusion of linkage to chromosomes 14q, 2q37 and 8p21.1-q11.23 in a Serbian family with idiopathic basal ganglia calcification. J Neurol. 2011;258:1637–1642. | |
Koller WC, Cochran JW, Klawans HL. Calcification of the basal ganglia: computerized tomography and clinical correlation. Neurology. 1979;29:328–333. | |
Boyd KP, Korf BR, Theos A. Neurofibromatosis type 1. J Am Acad Dermatol. 2009;61:1–14. | |
Marx SJ. Hyperparathyroid and hypoparathyroid disorders. N Engl J Med. 2000;343:1863–1875. | |
Velasco PJ, Manshadi M, Breen K, Lippmann S. Psychiatric aspects of parathyroid disease. Psychosomatics. 1999;40:486–490. | |
Daghighi MH, Rezaei V, Zarrintan S, Pourfathi H. Intracranial physiological calcifications in adults on computed tomography in Tabriz, Iran. Folia Morphol (Warsz). 2007;66:115–119. | |
Kachewar SG, Kulkarni DS. Distant perijoint calcifications: sequel of non traumatic brain injury – a review and case report. J Clin Diagn Res. 2013;7:2606–2609. | |
Inoue Y, Nemoto Y, Murata R, et al. CT and MR imaging of cerebral tuberous sclerosis. Brain Dev. 1998;20:209–221. | |
Wu J, Tarabishy B, Hu J, et al. Cortical calcification in Sturge-Weber syndrome on MRI-SWI: relation to brain perfusion status and seizure severity. J Magn Reson Imaging. 2011;34:791–798. | |
Bydder GM, Steiner RE, Young IR, et al. Clinical NMR imaging of the brain: 140 cases. AJR Am J Roentgenol. 1982;139:215–236. | |
Bernaerts A, Vanhoenacker FM, Parizel PM, et al. Tuberculosis of the central nervous system: overview of neuroradiological findings. Eur Radiol. 2003;13:1876–1890. | |
Ooi WW, Wijemanne S, Thomas CB, Quezado M, Brown CR, Nash TE. Short report: a calcified Taenia solium granuloma associated with recurrent perilesional edema causing refractory seizures: histopathological features. Am J Trop Med Hyg. 2011;85:460–463. | |
Loeb JA. Functional improvement in a patient with cerebral calcinosis using a bisphosphonate. Mov Disord. 1998;13:345–349. |
 © 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.