Back to Journals » Adolescent Health, Medicine and Therapeutics » Volume 14
Factors Associated with the Human Papillomavirus Vaccine Coverage in Gulu District, Uganda
Authors Odongo Ojok I , Ogwal JB, Wwesige B, Bongomin F , Akello F
Received 24 November 2022
Accepted for publication 30 March 2023
Published 5 April 2023 Volume 2023:14 Pages 87—96
DOI https://doi.org/10.2147/AHMT.S397734
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Alastair Sutcliffe
Isaac Odongo Ojok,1 John Baptist Ogwal,1 Brian Wwesige,1 Felix Bongomin,2 Franceska Akello3
1Faculty of Medicine, Gulu University, Gulu, Uganda; 2Department of Medical Microbiology and Immunology, Faculty of Medicine, Gulu University, Gulu, Uganda; 3Department of Pediatrics and Child Health, Faculty of Medicine, Gulu University, Gulu, Uganda
Correspondence: Franceska Akello, Department of Pediatrics and Child Health, Faculty of Medicine, Gulu University, P. O. Box 166, Gulu, Uganda, Tel +256-774697094, Email [email protected]
Background: Human papillomavirus (HPV) vaccination is an effective public health measure for the prevention of cancer of the cervix. We aimed to assess HPV vaccine coverage and associated factors in Gulu, Uganda.
Methods: In October 2021, we conducted a cross-sectional study among girls aged 9 to 13 years residing in Pece-Laroo Division, Gulu City, Uganda. HPV vaccine coverage was defined as receipt of at least a dose of the HPV vaccine.
Results: A total of 197 girls, with a mean age of 11± 1.4 years, were enrolled. Most participants were of the Acholi tribe (89.3%, n=176), catholic (58.4%, n=115), and in primary 5 level of education (36%, n=71). Overall, 68(35%) participants had received the HPV vaccine. Factors associated with HPV vaccine utilization were, good knowledge of the HPV vaccine (adjusted odds ratio (aOR) =0.233, 95% confidence interval (95CI): 0.037– 0.640, p=0.101), methods of HPV prevention (OR=0.320, 95CI: 0.112– 0.914, p = 0.033), good knowledge of the importance of HPV vaccination (OR=0.458, 95% CI: 0.334– 0.960, p=0.021), knowledge on frequency of HPV vaccination (OR=0.423, 95CI:0.173– 0.733, p=0.059), and good mobilization (OR=0.443, 95% CI: 0.023– 0.923, p=0.012).
Conclusion: In this community-based study, only one third of eligible girls received the HPV vaccine. Public health interventions are recommended exponentially to enhance the utilization of the HPV vaccine in this community.
Keywords: HPV vaccine, uptake, schoolgirls, Uganda
Introduction
Globally, cervical cancer is the fourth most common cancer in women.1 In 2018, there were an estimated 570,000 women diagnosed worldwide and about 311,000 died from cancer of the cervix.2 About 8% of cancers in developing countries, including cancer of the cervix, are caused by high-risk serotypes of the human papillomavirus (HPV).3 Cancer of the cervix is an increasing problem in sub-Saharan Africa exacerbated by the high HIV burden in the region. There are more than 75,000 new cases and 50,000 deaths yearly due to cancer of the cervix.3
Global utilization of HPV vaccination in resource-limited countries is relatively new. Following introduction of HPV vaccine in 2010, only 1.4% of the girls who received the first dose of the vaccine went ahead to complete the full course of the vaccine globally.4 Recent studies show that utilization of HPV vaccine in developed countries is higher at 33.5% as compared to 2.7% in low developed countries.5 In a recent study that was carried out in Sub-Saharan African region involving 64 countries, only 1% of the 118 million women had received and completed the full course of the HPV vaccine.5
In Uganda the Ministry of Health, through its strategic plan for cervical cancer prevention among girls aged 10 years, committed itself to achieving 80% HPV vaccine coverage among eligible girls. However, in one of the cross-sectional studies that was done in Lira district in Uganda, less than 20% of eligible girls received the HPV vaccine.6
The WHO recommends different implementation strategies which include school-based, health facility-based, and outreach-based strategies. Uganda adopted the school-based strategy targeting girls aged 9–13 yrs, with the majority being in primary four or have dropped out of school. The majority of the girls in this age category are not sexually active, and this makes them a target group for prevention of human papillomavirus infection that is a causative agent in 70% of cervical cancer.7 It is therefore worth noting that poor HPV vaccine uptake results in increased HPV infection burden that eventually leads to increased cervical cancer burden and morbidity as well as mortality associated with it.
Low utilization of HPV vaccine in Uganda could be attributed to the knowledge gap of the community regarding HPV and its relation to causation of cancer of the cervix, preventive measures, attitudes toward the vaccine, lack of community engagement and the health system factor like the cost of vaccine.8
Concerns from the community about vaccine utilization need to be unearthed and addressed in order to improve the health promotion according to the sustainable development goal 3, which emphasizes “good health and well-being”. Therefore, we aimed to investigate the factors associated with HPV vaccine coverage in Gulu City, Uganda.
Methods
Study Design
A descriptive, community-based, cross-sectional study was conducted in Pece-Laroo Division, Gulu City, Uganda in October 2021.
Study Setting
This study was conducted in Pece-Laroo Division, Gulu City. Pece-Laroo Division is located on the Western side of Gulu City. It comprises eight parishes; Agwee, Queens Avenue, Iriaga, Pece Prisons, Labour line, Pawel, Tegwana and Vanguard. It has 57 sub-wards with an estimated population of about 79,000. The estimated number of households in the whole division was 6350. Gulu Regional Referral Hospital is the biggest and nearest government health facility that serves the population. A small proportion of the population is served by private health facilities with reproductive health services. The division also has government-headed health facilities namely: Laroo health centre III, Gulu University Health Centre II, Pece Prison Health Centre II.
Study Population
We recruited girls aged 9 to 13 years within Pece-Laroo Division, Gulu, Uganda. Written informed assent was obtained from the parents or legal guardians of the children. Children with speech and language impairment and those with mental retardation such as cerebral palsy or Down’s syndrome were excluded.
Sample Size Estimation
Sample size for a single population was estimated using the assumption of a Z value of 1.96 at 95% confidence interval (95CI), uptake of HPV vaccine of 17.4% (Isabirye et al, 2020), desired precision 5%, and power of 80%. A sample size of 200 was realized.
Study Variables
The main study outcome variable was utilization of the HPV vaccine by girls aged 9 to 13 years in Pece-Laroo Division, Gulu City. The independent variables were sociodemographic characteristics and health system factors.
Data Collection Procedure
Data were collected using a pre-tested, standardized, semi-structured questionnaire in primary-level English. The data collection tool was translated to the local language (Acholi) and administered by a research assistant proficient in both English and Acholi languages. All completed questionnaires were kept out of reach of non-members of this research group. Several data backups were obtained and stored in other computers with password protection.
Data Analysis
Data were analyzed using STATA version 17.0. Numerical data were summarized as mean and standard deviation or median and interquartile ranges. Fischer’s exact test or Chi-square for categorical data and Student’s t-test or Mann–Whitney U tests for numerical data were used to assess for association between categorical variables and utilization of confidence. Binary logistic regression model was constructed to determine factors independently associated with utilization of HPV vaccine. Results were presented as adjusted odds ratio (aOR), with corresponding 95CI. A p < 0.05 was considered statistically significant.
Ethical Considerations
The study was conducted in accordance with the Declaration of Helsinki. The study protocol was approved by the Gulu University Research and Ethics Committee (GUREC-021-21). Parents/legal guardians provided written informed consent for all the minors prior to study commencement. The study participants who had defaulted their vaccination were referred to a nearby health facility or the parent or guardian was advised to take them to a health facility to complete their vaccination.
Results
Socio-Demographic Characteristics of Participants
A total of 197 girls participated in the study, making the percentage of girls who participated 98.5%. The age range of the study population was from 9 to 13 years. The mean +SD age of the respondents was 11 = 1.36 years. Majority (36%) of the participants were in primary five with only 23(11.7%) in primary seven. Overwhelming number (89.3%) of the participants were from the indigenous Acholi tribe. Regarding the religion of the girls, 115 (58.4%) were Catholic (Table 1).
 |
Table 1 Socio-Demographic Characteristics |
Majority (71.6%) of the girls lived with their parents and both parents were responsible for their upkeep (Figure 1).
 |
Figure 1 Care-takers of the girls. |
HPV Vaccination Coverage
Out of the 197 participants registered for the study, the level of HPV vaccination usage was only 35%. The majority never received the HPV vaccine (Figure 2).
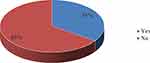 |
Figure 2 HPV utilization level. |
Out of 71 girls in primary five, 70.4% had never received the HPV vaccine. Out of the 115 girls who are Catholic, 65.2% had not been vaccinated at the time of this study. 63.6% of those residing in Queens Avenue ever received HPV vaccine. Majority of the girls reside with both their parents and out of the 141 girls, 64.5% had never received HPV vaccine (Table 2).
 |
Table 2 HPV Utilization by Socio-Demographic Factors |
Factors Associated with Utilization of HPV Vaccine
At bivariate analysis, the knowledge factors statistically associated with HPV vaccine use included good knowledge about the meaning of HPV (OR=0.233, 95% CI: 0.037–0.640), various good methods of HPV prevention (OR=0.320, 95% CI: 0.112–0.914), adequate information on the importance of HPV vaccination (OR=0.458, 95% CI: 0.334–0.960) and adequate knowledge about dosage of HPV vaccines (OR=0.423, 95% CI: 0.173–0.733) (Table 3). Mobilization factors statistically associated with HPV vaccine utilization included mobilization (OR=0.443, 95% CI: 0.023–0.923), time of mobilization (OR=2.528, 95% CI: 1.145–5.580), and how long the vaccination takes (OR=0.379, 95% CI: 0.288–0.684) (Table 4). Distance variable associated with HPV vaccine use revealed no significant variable associated with HPV vaccine use at 0.2 p-values (Table 5).
 |
Table 3 Bivariate Analysis of Knowledge Factors |
 |
Table 4 Bivariate Analysis of Mobilization Factors |
 |
Table 5 Bivariate Analysis of Distance Factors |
Multivariate Analysis of Factors Associated with Utilization of HPV Vaccine Among Girls Aged 9–13 Years in Pece-Laroo Division
Variables that were significant for HPV utilization at bivariate analysis were considered for multivariable analysis. These included knowledge about HPV, prevention of HPV, importance of HPV vaccine, the dosage, mobilization, time of mobilization and duration of vaccination. In multivariable analysis, factors found to be significantly associated with HPV vaccine utilization at 5% level of significance included knowledge about what HPV is (OR=0.580, 95% CI: 0.127-0.0.189), HPV prevention (OR=1.227, 95% CI: 1.077–4.669), the importance of HPV vaccine (OR=0.571, 95% CI: 0.290–0.899), time of mobilization (OR=4.534, 95% CI: 1.802–11.40) and the time taken for vaccination (OR=0.521, 95% CI: 0.144–0.906) (Table 6).
 |
Table 6 Multivariable Analysis of Factors Associated with Utilization of HPV Vaccine |
Attitudes of the Girls Toward Factors Associated with HPV Vaccination
In order to determine the girls’ perceptions of factors relating to their attitude that influenced HPV vaccine utilization, five statements were used (Table 7). The results show that 96.4% (n = 190) of the respondents indicated they have received information on HPV vaccine which is a good predictor of HPV utilization. The belief that HPV vaccine is safe was indicated by 198 (98%) girls. Additionally, 172 (87.3%) respondents agreed that HPV vaccine was not against their religious beliefs. 186 (94.4%) girls were willing to vaccinate if the vaccine could be provided indicating that the presence of the vaccine is a good predictor of HPV vaccine utilization. Incurring costs by the girls for HPV vaccination could affect HPV utilization negatively since only 39 (19.8%) expected the service to be at a cost.
 |
Table 7 Participants’ Perceptions of Factors Relating to Attitude That are Associated with HPV Vaccine Utilization |
Discussion
This study aimed to determine the level and factors associated with HPV vaccine utilization among girls aged 9 to 13 years in Pece-Laroo Division.
Out of the 197 participants registered for the study, the level of HPV vaccination usage was only 35% as the majority never received the HPV vaccine. This is higher than existing cross-sectional evidence for Lira district that established low coverage (17.4%) of HPV vaccination pointing to the urgent need to establish the predictors of HPV vaccine uptake.6 This slightly higher coverage was probably due to good knowledge and attitude, short distance to health facility, and good strategy especially outreach for vaccination and good mobilization.
In multivariable analysis, knowledge about HPV vaccine was found to be significantly associated with HPV vaccine utilization. Girls who did not know the purpose of HPV vaccine were 442% less likely to use HPV vaccine compared to girls who knew that HPV causes cervical cancer.
This is in line with a pilot study in Uganda carried out in two districts of Ibanda and Nakasongola which showed sub-optimal adolescent girls’ knowledge of HPV vaccine, where only 17.6% of the adolescents overall were knowledgeable. And a comparative study found that vaccinated girls were more knowledgeable about the vaccine than the unvaccinated.9
The results show that 96.4% (n = 190) of the respondents indicated they have received information on HPV vaccine, which is a good predictor of HPV utilization. It was indicated that 198 (98%) girls believed that HPV vaccine is safe. Additionally, 172 (87.3%) respondents agreed that HPV vaccine was not against their religious belief. 186 (94.4%) girls were willing to vaccinate if the vaccine could be provided, indicating that the presence of the vaccine is a good predictor of HPV vaccine utilization. Incurring costs by the girls for HPV vaccination could affect HPV utilization negatively since only 39 (19.8%) expected the service to be at a cost.
This is consistent with a study which was done in northern Uganda by Katagwa which found that HPV vaccine was highly acceptable with positive attitudes among primary school girls though mixed reactions and wrong perceptions still exist.10
This study indicates that the majority of respondents, 35 (83.3%), reported that HPV vaccines were available at the facility, 27(64.3%) health workers got their vaccine from the DHO’s office. This is not in line with a study conducted by Janice and Amerita which found that despite effective cervical cancer prevention, vaccines are often unavailable to people in developing countries.8 In their conclusion, delivery of vaccine in developing countries faces a wide range of socio-technical challenges that include lack of institutional capacity to monitor vaccine delivery to the population in the rural areas.
About 15(35.7%) and 14(33.3%) health workers had health education and media as their main source of information about HPV vaccine, respectively. This is inconsistent with a study conducted among University students in Malaysia,11 where 10.3% were aware of HPV vaccine, half of them had received the information from the newspaper and only 1.5% had information from health professionals despite the fact that the vaccine had been available for at least 5 years by that time.
According to our findings, stock outs, and misconception of HPV vaccine were the main hindrances to HPV vaccine utilization. This is in line with a study conducted by Nabirye which found that inconsistency of vaccine supply to health facilities was one of the health system factors associated with low levels of utilization of HPV vaccine among girls aged 9–13 years in Uganda.12
Conclusion
About one third of eligible girls received the HPV vaccine in Gulu City due to lack of knowledge, vaccine stock outs and misconception, meanwhile attitude, perception of the HPV vaccine, mobilization and distance had no significant effect. To increase HPV vaccination coverage among high-risk population, the government should increase the HPV vaccine supply especially in peripheral facilities so as to avoid vaccine stock outs and increase continuous health education to reduce misconception of the vaccine.
Author Contributions
All authors made a significant contribution to the work reported, whether that is in the conception, study design, execution, acquisition of data, analysis and interpretation, or in all these areas; took part in drafting, revising or critically reviewing the article; gave final approval of the version to be published; have agreed on the journal to which the article has been submitted; and agree to be accountable for all aspects of the work.
Funding
There is no funding to report.
Disclosure
Felix Bongomin and Franceska Akello are co-senior authors for this study. The authors report no conflicts of interest in this work.
References
1. Zhang X, Zeng Q, Cai W, Ruan W. Trends of cervical cancer at global, regional, and national level: data from the Global Burden of Disease study 2019. BMC Public Health. 2021;21:1–10.
2. WHO. Draft Global Strategy Towards Eliminating Cervical Cancer as a Public Health Problem. WHO; 2018.
3. Theirry P, Therese L, Mohammad-Ali J, Jean-Francois M, Laurent B, Bélec L. Cervical cancer in sub-Saharan Africa: a preventable noncommunicable disease. Expert Rev Anti Infect Ther. 2017;15(6):613–627. doi:10.1080/14787210.2017.1322902
4. Dempsey AF, Patel DA. HPV vaccine acceptance, utilization and expected impacts in the U.S.: where are we now? Hum Vaccin. 2010;6(9):715–720. doi:10.4161/hv.6.9.12730
5. Bruni L, Diaz M, Barrionuevo-Rosas L, et al. Global estimates of human papillomavirus vaccination coverage by region and income level: a pooled analysis. Lancet Glob Health. 2016;4(7):e453–e463. doi:10.1016/S2214-109X(16
6. Isabirye A, Mbonye M, Asiimwe JB, Kwagala B. Factors associated with HPV vaccination uptake in Uganda: a multi-level analysis. BMC Womens Health. 2020;20(1):1–11. doi:10.1186/s12905-020-01014-5
7. Olateju A, Cazier C. Improving data for decision-making: a toolkit for cervical cancer prevention and control programmes. 2016.
8. Graham JE, Mishra A. Global challenges of implementing human papillomavirus vaccines. Int J Equity Health. 2011;10(1):27. doi:10.1186/1475-9276-10-27
9. Turiho A, Muhwezi WW, Okello ES. Human papillomavirus (HPV) vaccination and adolescent girls’ knowledge and sexuality in Western Uganda: a comparative cross-sectional study. PLoS One. 2015;10(9):e0137094. doi:10.1371/journal.pone.0137094
10. Katagwa V. Acceptability of human papilloma campaign in Northern Uganda. PLoS Negl Trop Dis. 2014;6(10):e1844.
11. Wong LP, Wong P-F, Megat Hashim MA. Multidimensional social and cultural norms influencing HPV vaccine hesitancy in Malaysia. Hum Vaccin Immunother. 2011;16(7):1611–1622. doi:10.1080/21645515.2020.1756670
12. Nabirye J, Okwi LA, Nuwematsiko R, et al. Health system factors influencing uptake of Human Papilloma Virus (HPV) vaccine among adolescent girls 9–15 years in Mbale District, Uganda. BCM Public Health. 2020;20(1):1–11.
 © 2023 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2023 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
