Back to Journals » Drug Design, Development and Therapy » Volume 11
Facilitated saliva secretion and reduced oral inflammation by a novel artificial saliva system in the treatment of salivary hypofunction
Authors Kang M, Park H, Jun JH, Son M, Kang MJ
Received 1 September 2016
Accepted for publication 15 November 2016
Published 11 January 2017 Volume 2017:11 Pages 185—191
DOI https://doi.org/10.2147/DDDT.S121254
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Frank Boeckler
Minkyung Kang,1 Hyounggeun Park,1 Joon-Ho Jun,1 Miwon Son,1 Myung Joo Kang2
1Pharmaceutical Product Research Laboratories, Dong-A ST Research Institute, Gyeonggi, 2Division of Pharmaceutical Sciences, College of Pharmacy, Dankook University, Cheonan, Chungnam, Korea
Abstract: Saliva substitutes and/or lubricants are commonly employed to lessen dry mouth symptoms by stimulating and/or substituting for the secretion of saliva. In this study, a novel artificial saliva containing inorganic salts, including sodium chloride and potassium chloride, and bactericidal agents, including potassium thiocyanate and lactoperoxidase, was formulated in the form of a solution (DM-sol) or gel (DM-gel). Those in vivo therapeutic efficacies were assessed in terms of saliva secretion and anti-inflammatory activity in rats and mice, respectively. Salivary secretion was promoted by mucosal application of DM-formulations in normal rats. In particular, DM-gel resulted in 2.5- and 1.9-fold greater salivary flow rates compared to normal saline and DM-sol, respectively. In an in vivo efficacy evaluation in diabetic mice with salivary hypofunction, repeated application of DM-formulations alleviated histopathological changes in the buccal mucosa in terms of atrophy and thinning of the epithelium, compared to vehicle, after 4 weeks. Moreover, the DM-sol and DM-gel were comparably effective for relieving periodontal gingivitis, reducing infiltration of inflammatory cells, and normalizing the neutrophil level in the gingival gingiva, after 4 weeks. Therefore, the novel artificial saliva is expected to facilitate salivary secretion and restore physiological conditions in the mouth of patients with salivary hypofunction.
Keywords: saliva substitute, carbopol gel, hypothiocyanite–hydrogen peroxide mixture, antimicrobial activity, diabetic rats
Introduction
Dry mouth, also known as xerostomia, is defined as a subjective feeling of dryness in the oral cavity, as a result of a decline in saliva in the mouth due to salivary gland dysfunction, radiological treatment, Sjögren’s syndrome, or adverse reactions to other medications such as anticholinergic agents, antihistamines, and opioids.1,2 Saliva plays crucial roles not only in lubricating the oral mucosa and teeth, facilitating taste sensation, but also in providing antimicrobial activity in the epithelial membrane. Thus, patients with xerostomia are susceptible to dental caries periodontitis and microbial infections including candidiasis, as well as discomfort and/or dysfunction in speech, chewing, or swallowing, especially in the elderly.3–6 Although the treatment of xerostomia is very individual,7 a first-line medication is to ameliorate the dehydration in the mouth by stimulating and/or substituting for the secretion of saliva. The artificial saliva products available contain several inorganic salts found in human saliva and antimicrobial agents to humidify and/or prevent bacterial infection in the dehydrated mucosa.8–10 However, the therapeutic efficacy of artificial substitutes is often transient, because of rapid elimination from the oral cavity, and thus there is an unmet need to maintain a hydrated mucosa for a prolonged period.11
Mucoadhesive polymers are widely utilized in gel systems to prolong the residence time of the active compounds in the local area, reducing the frequency of application and thus improving patient compliance.12 Furthermore, mucoadhesive polymers can cover oral mucosal surfaces, restoring the damaged epithelium and physiological functions. Hydroxyethyl cellulose (HEC)-based oral products can improve oral dryness and have superior soothing effects compared with placebo in patients with postradiation xerostomia.13 Therefore, a mucoadhesive polymer-based artificial salvia gel could be an alternative for local delivery of inorganic salts and bactericidal agents in dry mouth. However, to our knowledge, there are no previous reports of the therapeutic efficacy of such gel preparations for dry mouth syndrome.
Here, a novel artificial saliva containing inorganic salts and lactoperoxidase system as bactericidal agent was formulated in the form of a solution (DM-sol) or gel (DM-gel), and those in vivo therapeutic efficacies were assessed in terms of the degree of saliva secretion, antimicrobial activity, and anti-inflammatory effect. Salivary secretion was measured after a single dosing of each formula in normal rats. In vitro antimicrobial activity was assessed against several strains that are abundant in dry mouth, such as Staphylococcus aureus, Candida albicans, and Actinomyces viscosus, using the disc agar plate method. Anti-inflammatory effects were assessed in streptozotocin-induced diabetes rats under hyposalivation conditions, by evaluating histological changes in the mucosal membrane and neutrophil levels in the gum tissue.
Materials and methods
Materials
Sodium chloride was purchased from Tomita Pharmaceutical Co., Ltd. (Tokushima, Japan). Potassium chloride, calcium chloride, and magnesium chloride were obtained from Duksan Pure Chemicals Co. (Gyeonggi-do, Korea). Lactoperoxidase and glucose oxidase were provided from Biopole (Les Isnes, Belgium) and Amano enzyme Inc. (Nagoya, Japan), respectively. Lactoferrin and lysozyme were purchased from Tatua (Morrinsville, New Zealand) and Dalian Greensnow Egg Products Development Co., Ltd. (Dalian City, People’s Republic of China), respectively. Xylitol and potassium thiocyanate were supplied from Namyung Commercial Co., Ltd. (Seoul, Korea) and Friends Union Enterprises Ltd. (Tianjin, People’s Republic of China), respectively. Carbopol 934P and HEC hhx250 were provided from Lubrizol Corporation (Wickliffe, OH, USA) and Hercules Inc. (Wilmington, DE, USA), respectively. Streptozotocin was purchased from Sigma-Aldrich Chemical Co. (St Louis, MO, USA). All other reagents were of analytical grade and were used without further purification. Double-distilled water was used for all experiments.
Preparation of DM-sol and DM-gel formulations
DM-sol was prepared by dissolving 0.085 g of sodium chloride, 0.12 g of potassium chloride, 0.02 g of calcium chloride, and 0.005 g of magnesium chloride in 100 g of distilled water using a magnetic stirrer at room temperature. Then, appropriate amounts of antimicrobial agents such as lactoperoxidase, glucose oxidase, lysozyme, potassium thiocyanate, and glucose were added to the solution and gently stirred until transparency was reached.
The amounts of the aforementioned inorganic salts and antimicrobial agents were dissolved in 50 g of distilled water. Separately, 1.5 g of Carbopol 934P and 0.5 g of HEC were added to 50 g of distilled water and stirred in a magnetic stirrer for 2 h at 40°C. The two solutions were then admixed, using an overhead stirrer equipped with a paddle-shaped propeller, for 1 h at room temperature. The pH of the mixture (total 100 g) was adjusted to 6.0 to produce DM-gel, by adding arginine solution dropwise.
Animals
All animal studies were carried out in accordance with Good Laboratory Practice for Nonclinical Laboratory Studies (21 CFR, Part 58, US Food and Drug Administration, Revised as of April 1, 2011), and all protocols were approved by the Committee for Animal Experiments of Dong-A Pharmaceutical (Seoul, Korea). Eight-week-old male Sprague Dawley rats and an 8-week-old female BALB/c mouse were obtained from Samtako (Kyungki-do, Korea). Animals were housed under controlled conditions (temperature of 18°C–26°C, relative humidity of 30%–70%, 12 h light/dark cycle) for at least 1 week before experimental treatment. Tap water and standard laboratory feed were available ad libitum.
Saliva secretion after treatment of DM-sol or DM-gel in rats
Sprague Dawley rats were assigned to three groups (n=10 per each group) to achieve comparable body weights (296–342 g). Rats were fasted for 12 h and then anesthetized by an intraperitoneal injection of urethane (1.5 mg/kg). Approximately 10 μL of normal saline, DM-sol, or DM-gel was applied to the inner surface of the right cheek of sedated rats, using a micropipette. The saliva secreted into the oral cavity during 1 h postdosing of each sample was collected by placing sanitary cotton in the left cheek. The accumulated amount of saliva collected for 1 h was divided by the weight of the rat.14
In vitro antimicrobial activity of DM-sol and DM-gel
Several strains abundant in the dehydrated oral cavity, S.aureus (ATCC 6538P), C. albicans (ATCC 10231), and A.viscosus (ATCC 15987), were obtained from the American Type Culture Collection (Manassas, VA, USA). These strains were cultured on Sabouraud dextrose infusion agar and incubated for 24–48 h under anaerobic conditions. After incubation, bacterial colonies were suspended in sterile brain-heart infusion broth to 1×106 cfu/mL. The disk diffusion method was used to assess the antibacterial activity of DM-sol and DM-gel.15 Samples (200 μL) were added to commercially available discs (Oxoid, Basingstoke, UK) and placed on a plate using sterile tweezers. After anaerobic incubation at 37°C for 48 h, the diameter of the inhibition zone around the test sample was measured in three locations, and the average was recorded.
Anti-inflammatory activity of DM-formulations in the oral cavity of diabetic rats
Induction of diabetes in mice and application of DM-formulations
Diabetes causes changes in salivary secretion and composition, usually leading to dry mouth and/or hyposalivation.16–18 Therefore, the anti-inflammatory effects of DM-sol and DM-gel were assessed in streptozotocin-induced diabetic mice after removal of submandibular glands, the major salivary glands in the mouth. Thirty-two female BALB/c mice were assigned to four groups (n=7–8 per group) to balance body weights (22–24 g). Mice were anesthetized by intraperitoneal injection of urethane (1.5 mg/kg), and then the submandibular gland, located beneath the lower jaws, was surgically removed using biopsy forceps. One week after excision of the submandibular gland, mice were intraperitoneally administered 60 mg/kg streptozotocin (0.1 M citrate buffer, pH 4.5). Induction of diabetes was confirmed by monitoring the blood glucose level 72 h after injection of streptozotocin, using a blood glucose analyzer (Roche-Diagnostics 1, Mannheim, Germany). Mice with glucose levels >300 mg/100 mL were regarded as diabetic. About 20 μL of DM-sol, DM-gel, and normal saline as negative control were applied to the inner surface of the cheek using a micropipette four times per day for 4 weeks. The sham group received only normal saline and underwent neither removal of the submandibular gland nor injection of streptozotocin during the same period.
Histological observation
After 4 weeks of treatment, the animals were euthanized by exsanguination under deep anesthesia. Then, for histological examination, a segment of the inner cheek and gum was excised and fixed in 10% neutral formaldehyde solution. Biopsies were embedded in paraffin and were cut into 5-μm-thick sections. Slides were deparaffinized with xylene, followed by dehydration in ethanol. The tissue sections were stained with hematoxylin-eosin dye (Histology Tech Services, Gainesville, FL, USA). The severity of inflammation in the mucosal lesions was graded from 0 to 3, in terms of mucosal atrophy, superficial epithelial degeneration, and submucosal inflammatory infiltration. Neutrophil infiltration in gingival gingiva tissue was further determined after staining with hematoxylin-eosin dye. Total neutrophils were counted by a pathologist under optical microscopic observation, and then neutrophils counted per viewing field (400× magnification) in 10 random foci were averaged for each gingival gingiva tissue.
Statistical analysis
All data are expressed as mean ± standard deviation (SD). Statistical analysis was carried out using Student’s t-test, and differences were considered significant at P<0.05, unless otherwise indicated.
Results and discussion
Formulation development
A mucoadhesive gel system was formulated to prolong the retention of inorganic salts and enzymes in the oral cavity and to keep the mucosa hydrated for an extended period. Both DM-sol and DM-gel were transparent, with no precipitation or phase separation. Both formulas were isotonic, with no significant difference (300–320 mOsm/L) (data not shown). As active ingredients, 85 mg NaCl, 120 mg KCl, 20 mg CaCl2, and 5 mg MgCl2 were included in 100 mL of solution to stimulate saliva secretion and restore the remineralization of the teeth. Antibacterial agents such as lactoperoxidase, glucose oxidase, lactoferrin, lysozyme, and potassium thiocyanate were included to protect the mucosa and/or teeth from bacterial infection. Lysozyme catalyzes the hydrolysis of the cell walls of Gram-positive bacteria and induces a nonspecific innate immune response to bacteria.19 Lactoperoxidase itself has no antimicrobial activity, but in the presence of hydrogen peroxide and thiocyanate ion produces hypothiocyanite, which has potent bacteriostatic activity.20
Two mucoadhesive polymers, HEC and carbopol, which are generally recognized as safe materials, were employed as gelling agents. HEC, a cellulose derivative, relieved intraoral dryness, and exhibited superior palliative effects in patients with postradiation xerostomia.13,21 Carbopol, an anionic polymer of acrylic acid, was selected in view of its excellent gel-forming ability at low concentrations and mucoadhesive properties through interacting with mucus glycoproteins. The carboxyl group of the polymer is dissociated at pH 6.8, resulting in the formation of a swollen gel.22 These mucoadhesive polymers form viscous gels on the epithelium, lubricating and physically protecting the dehydrated mucosa and providing relief from the symptoms of xerostomia.
Saliva secretion after treatment of DM-sol or DM-gel in rats
Although xerostomia is the subjective feeling of oral dryness, a decrease in saliva flow rate is detected. While the average resting and stimulated whole saliva flow rates in normal individuals are 0.35 and 1.5 mL/min, they are less than 0.1 and 0.5 mL/min, respectively, in patients with xerostomia.23–26 Thus, saliva substitutes contain inorganic salts to stimulate saliva secretion and humidify the dehydrated mucosa. Increasing the salt content increased the early salivary flow rate, which peaked at 300 mEq of inorganic salts.27
The saliva flow rates after application of normal saline, DM-sol, and DM-gel are shown in Figure 1. Salivary secretion was promoted by mucosal application of inorganic salts, as evidenced by flow rates of greater than 0.32 μL/min/100 g in all groups. The DM-gel-treated group exhibited the greatest elevation in secretion levels, and exhibited 2.5- and 1.9-fold greater salivary secretion than the normal saline- and DM-sol-treated groups, respectively. The mucoadhesive gel prolonged the residence time of the inorganic salts on the mucosal membrane, thus increasing salivary secretion. The duration of the humidification effect after application of the saliva substitute has been reported to be only 15.3 min in patients with xerostomia.28 Therefore, the prolonged mucosal retention by the mucoadhesive system might extend the duration of the humidification effect of inorganic salts, thus improving patient satisfaction.
In vitro antimicrobial activity of DM-formulations
The influence of saliva composition and/or secretion on oral bacterial or fungal communities has been reported. Almståhl and Wikstrom29 revealed that the levels of mutans streptococci and lactobacilli were increased under hyposalivation conditions. Moreover, the numbers of C.albicans and Enterococci were significantly increased in subjects with postradiation xerostomia.30 Therefore, the antimicrobial activity of DM-formulations against S.aureus, C.albicans, and A.viscosus was assessed.
The inhibition zone diameters of DM-sol and DM-gel are shown in Table 1. The blank disc did not inhibit bacterial growth. However, application of DM-formulations markedly suppressed the growth of all strains. Both DM preparations exhibited inhibition zone diameters over 10, 11, and 49 mm against S.aureus, C.albicans, and A.viscosus, respectively. DM-gel produced smaller zones of inhibition against S.aureus and C.albicans, compared to DM-sol, likely owing to the retarded release of antimicrobial agents such as lactoperoxidase, glucose oxidase, lactoferrin, or lysozyme in the more viscous gel system. Nevertheless, both DM-sol and DM-gel were still effective, suppressing every strain tested, and further in vivo anti-inflammatory effect was performed in diabetic mice with xerostomia.
Anti-inflammatory activity of DM-formulations in diabetic rats
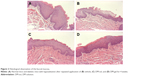
Streptozotocin-induced diabetic mice were used as a model of xerostomia. In a previous study, the salivary flow rate was reduced by 75% in diabetic mice compared to normal mice (P<0.05), while the amylase and peroxidase activities were increased in the diabetic mice.31 In addition, the submandibular glands were removed to produce secretory dysfunction in the mouth. Micrographs of the buccal mucosa of sham, negative control, DM-sol, and DM-gel-treated groups are shown in Figure 2. And the severity scores for mucosal atrophy, epithelial degeneration, and inflammatory infiltration are depicted in Figure 3. In the control group, the buccal mucosa manifested noticeable mucosal atrophy and epithelial thinning. Compared to the sham group, the severity score was significantly elevated in the vehicle-treated group after 4 weeks (average, 2.1). However, repeated application of DM-sol and DM-gel led to marked amelioration of these symptoms, in terms of a >50% lower score. Xerostomia-induced mucosal thinning was ameliorated by the saliva substitute, as evidenced by 50% and 47% increases in epithelium thickness, respectively (Figure 4).
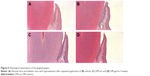
Histopathological changes in gum tissue (periodontal gingiva) were also examined (Figure 5). Swelling and infiltration of inflammatory cells were evident in the vehicle-treated group after 4 weeks. Moreover, a large number of neutrophils were present in the gingiva in the control group (Figure 6). In the presence of inflammation, neutrophils migrate to periodontal tissues such as the gingival crevice and epithelium, to protect the infected tissue by forming a mechanical barrier between the junctional epithelium and dental plaque.32 In contrast, the DM-formulations reduced the neutrophil count to less than half that of the negative control group. Peroxidase combined with potassium thiocyanate might promote generation of hypothiocyanate ions, which inhibit plaque and/or caries-forming bacteria.33 The peroxidase system is effective against oral bacteria including mutans streptococci, lactobacilli, fungi, and viruses.33 The changes in epithelial thickness, mucosal atrophy, and the level of eosinophils after repeated application of DM-sol and DM-gel were comparable. After 4 weeks of treatment, these parameters were normalized and did not differ between the DM-sol and DM-gel. Patients with salivary hypofunction commonly suffer from secondary ailments including dental caries and mucosal injection including candidiasis.3–6 Karbach et al34 demonstrated a strong association between the saliva flow rate and fungal colonization in the oral cavity in subjects with postradiation, as saliva predominantly regulates the composition and population of the oral microflora with its antibacterial and antifungal activities. Thus, a novel saliva substitute containing hypothiocyanate ion-based antimicrobial system could be beneficial to alleviate mucosal infection and/or inflammation in patients with hyposalivation.
In summary, the primary finding of the study was that mucoadhesive polymers enhanced humidification of the dehydrated oral cavity and maintained the antimicrobial activity of the saliva substitute. The DM-gel system resulted in 2.5- and 1.9-fold greater salivary flow than that effected by normal saline and DM-sol, respectively. Moreover, the DM-sol and DM-gel alleviated mucosal inflammation in diabetic mice in terms of mucosal epithelial thickness and neutrophil infiltration in gingival gingiva. From these findings, we expect that the mucoadhesive gel is favorable to formulate artificial saliva, as it exerts a lubricating effect and prolongs the residence time of the active ingredients in the oral cavity of patients with xerostomia.
Disclosure
The authors report no conflicts of interest in this work.
References
Alimi D. Xerostomia induced by radiotherapy. Ther Clin Risk Manag. 2015;11:1149–1152. | ||
Kanehira T, Yamaguchi T, Asano K, et al. A screening test for capsaicin-stimulated salivary flow using filter paper: a study for diagnosis of hyposalivation with a complaint of dry mouth. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2011;112:73–80. | ||
Samarawickrama DY. Saliva substitutes: how effective and safe are they? Oral Dis. 2002;8:177–179. | ||
Van der Reijden WA, van der Kwaak JS, Veerman ECI, Nieuw Amerongen AV. Analysis of the concentration and output of whole salivary constituents in patients with Sjogren’s syndrome. Eur J Oral Sci. 1996;104:335–340. | ||
Greenspan D. Xerostomia: diagnosis and management. Oncology (Williston Park). 1996;10:7–11. | ||
Pedersen AM, Bardow A, Jensen SB, Nauntofte B. Saliva and gastrointestinal functions of taste, mastication, swallowing and digestion.Oral Dis. 2002;8:117–129. | ||
Momm F, Volegova-Neher NJ, Schulte-Mönting J, Guttenberger R. Different saliva substitutes for treatment of xerostomia following radiotherapy. A prospective crossover study. Strahlenther Onkol. 2005; 181:231–236. | ||
Sreebny LM. Recognition and treatment of salivary induced conditions. Int Dent J. 1989;39:197–204. | ||
Levine MJ. Development of artificial salivas. Crit Rev Oral Biol Med. 1993;4:279–286. | ||
Tenovuo J, Söderling E. Chemical aids in the prevention of dental diseases in the elderly. Int Dent J. 1992;42:355–364. | ||
Hearnden V, Sankar V, Hull K, et al. New developments and opportunities in oral mucosal drug delivery for local and systemic disease. Adv Drug Deliv Rev. 2012;64:16–28. | ||
Perchyonok VT, Shengmiao Z, Theunis O. Chitosan and gelatin based prototype delivery systems for the treatment of oral mucositis: from materials to performance in vitro. Curr Drug Deliv. 2013;10:144–150. | ||
Warde P, Kroll B, O’Sullivan B, et al. A phase II study of Biotene in the treatment of postradiation xerostomia in patients with head and neck cancer. Support Care Cancer. 2000;8:203–208. | ||
Tai Y, Inoue H, Sakurai T, et al. Protective effect of lecithinized SOD on reactive oxygen species-induced xerostomia. Radiat Res. 2009;172:331–338. | ||
Clinical and Laboratory Standards Institute. Performance Standards for Antimicrobial Disk Susceptibility Tests; Approved Standard. 7th ed., CLSI document M02-A11. Wayne, PA: CLSI; 2012. | ||
Bernardi MJ, Reis A, Loguercio AD, Kehrig R, Leite MF, Nicolau J. Study of the buffering capacity, pH and salivary flow rate in type 2 well-controlled and poorly controlled diabetic patients. Oral Health Prev Dent. 2007;5:73–78. | ||
Sandberg GE, Sundberg HE, Fjellstrom CA, Wikblad KF. Type 2 diabetes and oral health: a comparison between diabetic and non-diabetic subjects. Diabetes Res Clin Pract. 2000;50:27–34. | ||
Edblad E, Lundin SA, Sjödin B, Aman J. Caries and salivary status in young adults with type 1 diabetes. Swed Dent J. 2001;25:53–60. | ||
Jollès P, Jollès J. What’s new in lysozyme research? Always a model system, today as yesterday. Mol Cell Biochem. 1984;63:165–189. | ||
Thomas EL, Milligan TW, Joyner RE, Jefferson MM. Antibacterial activity of hydrogen peroxide and the lactoperoxidase-hydrogen peroxide-thiocyanate system against oral streptococci. Infect Immun. 1994;62:529–535. | ||
Epstein JB, Emerton S, Le ND, Stevenson-Moore P. A double-blind crossover trial of Oral Balance gel and Biotene1 toothpaste versus placebo in patients with xerostomia following radiation therapy. Oral Oncol. 1999;35:132–137. | ||
Bonacucina G, Martelli S, Palmieri GF. Rheological, mucoadhesive and release properties of carbopol gels in hydrophilic cosolvents. Int J Pharm. 2004;282:115–130. | ||
Llena-Puy C. The role of saliva in maintaining oral health and as an aid to diagnosis. Med Oral Patol Oral Cir Bucal. 2006;11:449–455. | ||
Fox PC. Xerostomia: recognition and management. Dent Assist. 2008;77:18–20. | ||
Tenovuo J, Lagerlöf F. Saliva. In: Thylstrup A, Fejerskov O, editors. Textbook of Clinical Cardiology. 2nd ed. Copenhagen, Denmark: Munksgaard; 1994. | ||
Axelsson P. Diagnosis and Risk Prediction of Dental Caries. Vol 2. Hanover Park, IL: Quintessence books; 2000. | ||
Neyraud E, Prinz J, Dransfield E. NaCl and sugar release, salivation and taste during mastication of salted chewing gum. Physiol Behav. 2003;79:731–737. | ||
Silvestre FJ, Minguez MP, Suñe-Negre JM. Clinical evaluation of a new artificial saliva in spray form for patients with dry mouth. Med Oral Patol Oral Cir Bucal. 2009;1:8–11. | ||
Almståhl A, Wikstrom M. Oral microflora in subjects with reduced salivary secretion. J Dent Res. 1999;78:1410–1416. | ||
Almståhl A, Wikstrom M, Fagerberg-Mohlin B. Microflora in oral ecosystems in subjects with radiation-induced hyposalivation. Oral Dis. 2008;14:541–549. | ||
Romero AC, Ibuki FK, Nogueira FN. Sialic acid reduction in the saliva of streptozotocin induced diabetic rats.Arch Oral Biol. 2012;57:1189–1193. | ||
Vitkov L, Klappacher M, Hannig M, Krautgartner WD. Extracellular neutrophil traps in periodontitis. J Periodontal Res. 2009;44:664–672. | ||
Tenovuo J, Lumikari M, Soukka T. Salivary lysozyme, lactoferrin and peroxidases: antibacterial effects on cariogenic bacteria and clinical applications in preventive dentistry. Proc Finn Dent Soc. 1991;87:197–208. | ||
Karbach J, Walter C, Al-Nawas B. Evaluation of saliva flow rates, Candida colonization and susceptibility of Candida strains after head and neck radiation. Clin Oral Investig. 2012;16:1305–1312. |
 © 2017 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2017 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.