Back to Journals » OncoTargets and Therapy » Volume 13
Expression and Clinical Significance of ERCC1 and XPF in Human Hepatocellular Carcinoma
Authors Liao X , Li Y, Li H, Huang W, Wang H, Xie W
Received 9 November 2019
Accepted for publication 20 January 2020
Published 4 February 2020 Volume 2020:13 Pages 1059—1072
DOI https://doi.org/10.2147/OTT.S237916
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Prof. Dr. Nicola Silvestris
Xiaoli Liao,1,* Yongqiang Li,1,* Hualan Li,1,* Wenfeng Huang,2 Hongxue Wang,3 Weimin Xie3
1Department of First Chemotherapy, Affiliated Guangxi Medical University Cancer Hospital, Nanning 530021, People’s Republic of China; 2Department of Oncology, The Second Affiliated Hospital of Guangxi Medical University, Nanning 530007, People’s Republic of China; 3Department of Fifth Chemotherapy, Affiliated Guangxi Medical University Cancer Hospital, Nanning 530021, People’s Republic of China
*These authors contributed equally to this work
Correspondence: Weimin Xie
Department of Fifth Chemotherapy, Affiliated Guangxi Medical University Cancer Hospital, He Di Road 71#, Nanning 530021, People’s Republic of China
Tel +86-771-5332393
Email [email protected]
Objective: To investigate the correlation between the ERCC1 and XPF expression and the clinicopathological parameters of hepatocellular carcinoma (HCC) patients through assessment of the expression of the DNA repair genes ERCC1 and XPF.
Methods: ERCC1 and XPF mRNA expression in HCC (n= 177) and adjacent para-cancer tissues (n=21) were assessed by RT-PCR. The correlation between ERCC1 and XPF expression, clinicopathological features and HCC prognosis were compared.
Results: ERCC1 expression in liver cancer tissues was significantly lower than that of adjacent tissues (9.5% (2/21) vs 38.1% (8/21); P< 0.05). The positive expression rates of XPF in liver cancer tissues was lower than that of adjacent tissues (14.3% (3/21) vs 71.4% (15/21); P< 0.05). ERCC1 and XPF expression were associated with hepatic capsular invasion and microvascular invasion. HCC patients with hepatic capsular invasion and microvascular tumor embolus formation had significantly lower levels of ERCC1 and XPF mRNA than those without hepatic capsular invasion and microvascular tumor embolus formation (P< 0.05). In addition, ERCC1 expression was associated with TNM staging of HCC. The expression of ERCC1 mRNA in patients with stage II and III HCC were lower than that of patients with stage I HCC (P< 0.05). The low levels of ERCC1 and XPF mRNA significantly correlated with relapse-free survival times (RFS) in HCC patients. The median RFS of the low ERCC1 expression group and low XPF expression group were shorter than those of the high expression group (15.0 months vs 32.0 months, P< 0.05) and (19.0 months vs 33.0 months, P< 0.05). The decrease in XPF mRNA expression was significantly associated with the overall survival (OS) of HCC patients. The median OS in the low XPF expression group was shorter than that of the high expression group (46.0 months vs 78.0 months, P< 0.05). However, no significant difference in OS between the low ERCC1 expression group and the high ERCC1 expression groups were observed (63.0 months vs 64.0 months, P> 0.05). Multivariate analysis showed that tumor size and the extent of differentiation were independent factors affecting the RFS in HCC patients (P< 0.05). The extent of differentiation and XPF were independent factors affecting the OS in HCC (P< 0.05).
Conclusion: The expression in ERCC1 and XPF were low in HCC and associated with early relapse after HCC surgery. Low XPF expression may be a potential indicator of a high risk of death.
Keywords: hepatocellular carcinoma, ERCC1, XPF, prognosis
Liver cancer is the 6th most common malignant tumor, ranking 4th for global mortality amongst all human cancers.1 Early postoperative relapse is associated with the prognosis of patients with surgically resectable HCC. Therefore, the identification of molecular markers associated with the risk of HCC relapse is of great clinical significance for effective monitoring of the progression of the tumor and assessment of the prognosis.
Liver cancer results from the interaction of the environment and genes. Effective DNA repair systems exist in normal cells, enabling quick recovery in response to DNA damage to maintain normal cell function and genetic stability. DNA damage in human cells leads to a series of cellular responses, including signal transmission, damage and repair, and the induction of cell death. Liver cancer results from a failure of the DNA damage repair system promoting malignant transformation and liver cancer. Nucleotide excision repair (NER) is the most important pathway of DNA damage repair associated with HCC.2,3 The process of NER consists of the identification of the damage site, unwinding of DNA strands, excision of a DNA fragment, and DNA strand filling and connection.4,5 The excision of the damaged DNA fragment mediated by the ERCC1/XPF complex is a key regulatory element of NER.6 Therefore, defect or mutation of the genes coding for ERCC1 and XPF may lead to a severe disorder in NER function, resulting in genomic instability, which is closely associated with carcinogenesis.
Excision repair cross-complementing 1 (ERCC1) gene is a critical gene in the entire process of DNA damage repair;7–9 its function is to cleave the 5ʹ end of the damaged DNA strand. The xeroderma pigmentosum group F (XPF) protein represents another crucial component of the NER mechanism, playing the same role with endonuclease at the DNA 5ʹ end. ERCC1 and XPF are located on chromosome 19q13.2 to 13.3 and p13.1, respectively. The XPF-ERCC1 complex of proteins encoded by these two genes acts as a structurally selective heterodimeric endonuclease. The XPF protein has a catalytic function, while ERCC1 participates in the binding of DNA strands. Together, the complex is involved in the 5ʹ incision of the damaged DNA strand in the NER pathway.9 It has been well-documented that the expression of ERCC1 and XPF is associated with the initiation, development, prognosis, and chemoresistance of diverse malignancies.9
Limited information is available regarding the relationship between the ERCC1 and XPF expression and the tumor relapse and prognosis after the HCC surgery. Therefore, in the present study, the levels of ERCC1 and XPF mRNA were detected by RT-PCR in 177 HCC patients who had undergone radical surgery, with the objective of elucidation the relationship between the expression of these two genes and the clinicopathological features of the tumor and postoperative survival of HCC patients. The expected results could identify novel molecular markers for the prediction of the relapse, metastasis, and patient survival after the HCC surgery.
Materials and Methods
Patients and Specimens
HCC tissue samples were harvested from 177 patients who had the tumor surgically removed at the Department of Hepatobiliary Surgery of the Affiliated Cancer Hospital of Guangxi Medical University. The patients were treated from January 2008 to December 2013, and the diagnosis of HCC was confirmed by histopathology. The para-carcinoma tissue samples were collected from 21 patients. For all patients, complete clinical and pathological data, and follow-up documentation was collected. The patients did not undergo any anticancer treatment prior to the surgery. All tissue specimens were frozen in liquid nitrogen within 30 mins after being harvested and transferred stored at −80 °C until use. The collection and use of the specimens were approved by the Ethics Committee of the Affiliated Cancer Hospital of Guangxi Medical University, and all patients provided written informed consent, in accordance with the Declaration of Helsinki.
Clinical Pathological Data
The age of the patients ranged from 19 to 77 years (median: 51 years; mean: 50.6 years). Eighty-three of them were younger than 50 years, and 94 patients were older than 50. There were 160 males and 17 females. HbsAg was positive in 156 patients and negative in 21, 119 patients had an AFP<400 ng/mL, and 58 had an AFP≥400 ng/mL, 145 had a solitary tumor, and 32 had multiple tumors. The tumor size was ≤5 cm in 80 patients and > 5 cm in 97. Moderate/high differentiation was found in 118 cases, and poor differentiation in 59 cases. Cirrhosis was present in 147 patients and absent in 30. Thirty patients had microvascular tumor embolus that was absent in the remaining 147 patients. Evaluation of the tumor stage according to the TNM staging system for hepatocarcinoma (2010 AJCC Cancer Staging, 7th Edition) revealed that there were 117 cases of stage I, 37 cases of stage II, and 23 cases of stage III. Staging using the Barcelona-Clínic hepatocarcinoma (BCLC) system identified 67 cases of stage A and 110 cases of stage B (Table 1).
 |
Table 1 Primary Clinicopathological Features of 177 Patients with HCC |
Patient Follow-Up
Regular postoperative rechecks to monitor relapse included serum AFP, ultrasound, CT scan, magnetic resonance imaging, or hepatic angiography. Relapse-free survival (RFS) was defined as the time interval from the HCC surgery to the diagnosis of intrahepatic relapse or distant metastasis. Overall survival (OS) was defined as the time from the HCC surgery to all-causes death.
Quality Control of Total RNA
Due to the prolonged storage of the specimens at −80 °C, the quality control of total RNA was performed. For this purpose, total RNA was extracted from 50 mg of fresh HCC tissue and tissue adjacent to the tumor, and a part of DNA from both aliquots was subjected to agarose gel electrophoresis, respectively. The absence of three RNA bands corresponding to 28S, 18S, and 5S rRNA indicated that the sample was too degraded to be used for RT-PCR and should be discarded. Electrophoresis results demonstrated that although all samples were degraded to a varying extent, they could still be used in RT-PCR assays.
Real-Time Quantitative Polymerase Chain Reaction
The retroviral reverse transcriptase kit was used to synthesize cDNA under the reaction conditions of 37 °C for 15 min, 85 °C for 5 sec, and 4 °C. The primers for the PCR were designed using Premier 5.0 software, and the sequences as follows:
ERCC1 forward 5´-TGCCCATTCACTGCCTCCT-3´,
ERCC1 reverse 5´-GCCTCGGTTCTGTGCCTTT-3´,
XPF forward 5´-GAGAAATAAGCAGTAGCCCAGAAA-3´,
XPF reverse 5´- GAAGCGGATGGATGATAGTGAG-3´,
GAPDH forward 5´-GTCAGCCGCATCTTCTTT-3´, and
GAPDH reverse 5´-CGCCCCGCCCAATACGACCAAAT-3´.
Multiplex PCR reactions were performed using the Roche FastStart Universal SYBR Green Master (ROX) kit and according to the ABl7500 fluorescent PCR system operating guideline. The thermocycling conditions were included denaturation at 95 °C for 15 sec, and annealing and extension at 60 °C for 1 min, for a total of 40 cycles. GAPDH was used as an internal standard to normalize the expression of each gene, and the relative expression level of XPC, ERCC1, and XPF gene was obtained by the comparative 2−△△Ct method.
Western Blotting Study
Tissues from 10 HCC cases were collected and the total proteins were extracted using RIPA lysates. The protein concentration of each extract was determined using a Modified Bradford Protein Assay kit. Aliquots of total protein (25 µg per lane) were boiled at 100°C in loading buffer, and then separated via 10% SDS-PAGE. The separated proteins were transferred onto a PVDF membrane that was then blocked with a 5% lipid-free milk solution for 2h at room temperature with gentle rotation. The membrane was incubated with the appropriate primary antibodies diluted in TBST for 1–2h at room temperature, after which it was washed three times with TBST, and then incubated with goat anti-rabbit horseradish peroxidase (HRP)-conjugated secondary antibody and goat anti-mouse HRP-conjugated secondary antibody at room temperature for 1 h. The independent experiments were performed with GAPDH as the internal standard. Protein expression was quantified using Image J2X software with GAPDH as the loading control. Each experiment was performed in triplicate.
Statistical Analysis
SPSS 21.0 software was used for all data analysis. ROC curves were plotted to determine the threshold values of the relative expression of ERCC1 and XPF mRNA. According to the critical value, values above were considered as positive expression. ERCC1 and XPF expression between cancer and adjacent tissues were compared by McNemar tests. The ERCC1 and XPF expression levels were compared across the groups using the χ2 tests. The correlation between genes was established using the Spearman correlation analysis. Relapse-free survival and overall survival analyses were performed using the Kaplan-Meier method, and the comparison was performed using the logrank test. Cox proportional hazards regression models were adopted for the univariate and multivariate analyses. P<0.05 was considered to indicate the presence of a statistically significant difference.
Results
Determination of the Optimal Threshold of the Level of Expression of ERCC1 and XPF mRNA
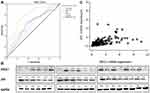
When the cutoffs of the relative expression of ERCC1 and XPF mRNA were 0.89 and 0.27, the [sensitivity + specificity - 1] was the largest, and the two cutoff values were considered to represent the optimal threshold. The sensitivity of ERCC1 and XPF in the evaluation of an early (≤12 months) postoperative relapse in HCC patients was 61.3% and 34.5%, respectively; the corresponding specificity was 64.4% and 63.5%, respectively. The area under curve (AUC) for ERCC1 and XPF was 0.615 and 0.653, respectively (P<0.05). The sensitivity and specificity of combination of ERCC1 and XPF was 77.4% and 70.4%, respectively. The area under curve (AUC) for the combination of ERCC1 and XPF was 0.784 (P<0.001). Statistics showed that prediction performance of combination of two genes was better than individual prediction performance (P<0.05). However this study showed that AFP≥400ng/mL (Chinese liver cancer guideline) or AFP≥20ng/mL (European liver cancer guideline) as the standard had no predictive effect on early recurrence of HCC (AUC = 0.583, P>0.05; AUC = 0.503, P>0.05) (Figure 1A). There were 79 HCC patients with low expression of ERCC1 (≤0.89), 98 HCC patients with high expression of XPC (>0.89), 85 HCC patients with low expression of XPF (≤0.27), and 92 HCC patients with high expression of XPC (>0.27).
Expression of ERCC1 and XPF in Hepatocellular Carcinoma
The positive expression rates of ERCC1 in liver cancer tissues were significantly lower than those of adjacent tissues [9.5% (2/21) vs 38.1% (8/21); P<0.05]. Similarly, the expression of XPF in liver cancer tissue was significantly lower than that of adjacent tissues. The positive expression rates of XPF in cancer and adjacent tissues were 14.3% (3/21) and 71.4% (15/21), respectively, suggesting significant differences between the groups (P<0.05). To determine whether the changes in mRNA levels altered protein expression, 10 pairs of cancerous and paracancerous tissues were randomly harvested from 21 HCC patients and ERCC1 and XPF expression was assessed by Western blot. The protein expression levels of ERCC1 and XPF in tumor tissues was 0.24 ± 0.23, 0.32 ± 0.22, respectively. The protein expression levels of ERCC1 and XPF in adjacent tissues was 0.31 ± 0.28, 0.58 ± 0.35, respectively. The protein expression levels of XPF in tumor tissues was lower than that in adjacent tissues (P <0.05). But there was no expression difference of ERCC1 protein between tumor tissue and adjacent tissues (P>0.05) (Figure 1B).
Correlation Between ERCC1 and XPF Levels and the Clinicopathological Features of Patients with Hepatocellular Carcinoma
The correlation between the expression in HCC and the common clinicopathological features of 177 HCC patients were analyzed. ERCC1 and XPF expression in cancer tissues was associated with the invasion of hepatic capsules and microvascular tumor embolus. The positive expression rates of ERCC1 in HCC patients with or without hepatic capsular invasion were 2.8% (5/177) and 46.3% (82/177), respectively, showing significant differences across the groups (P<0.05). The positive expression rates of ERCC1 in HCC patients with and without microvascular tumor embolus were 5.1% (9/177) and 44.1% (78/177), respectively, showing significant differences across the groups (P<0.05). The positive expression rates of XPF in HCC patients with and without hepatic capsular invasion were 2.3% (4/177) and 47.5% (84/177), respectively, showing significant differences across the groups (P<0.05). The positive expression rates of XPF in HCC patients with microvascular tumor embolus with and without microvascular tumor embolus were 4.5% (8/177) and 45.2% (80/177), respectively, showing significant differences across the groups (P<0.05). The positive expression rates of ERCC1 in patients with stage II-III liver cancer were significantly lower than those of stage I patients [12.4% (22/177) vs 36.7% (65/177); P<0.05]. XPF was not associated with TNM staging [13.5% (24/177) vs 36.2% (64/177); P>0.05]. The expression of ERCC1 and XPF mRNA in hepatocarcinoma tissues did not correlate with age, gender, smoking history, tobacco and alcohol abuse history, HBsAg, ALT, AST, AFP, the extent of differentiation, liver cirrhosis, tumor size, number of tumors, or BCLC staging (P>0.05). (Tables 2–3).
 |
Table 2 Correlation Between ERCC1 Expression and Clinicopathological Features in 177 Patients with HCC |
 |
Table 3 Correlation Between XPF Expression and Clinicopathological Features in HCC Patients |
Correlation Among the Expression of ERCC1 and XPF mRNA in HCC
Spearman correlation analysis determined that the expression of ERCC1 and XPF mRNA in 177 samples of HCC tissues was positively correlated (γ>0, P<0.001) (Figure 1C), indicating that the reduction in the expression levels of ERCCl and XPF mRNA similarly regulated in HCC tissues.
Correlation of ERCC1, and XPF Expression with HCC Prognosis
The follow-up ended on October 30, 2018.
The median follow-up time of HCC patients was 63.0 months (range: 2.0 to 135.0 months). By the end of the follow-up period, 151 of the 177 HCC patients had a relapse, accounting for 85.6%. Fifty-one patients survived, 109 (62.4%) died, and 17 (9.4%) were lost to follow-up. Lower levels of ERCC1 and XPF mRNA were significantly associated with RFS in HCC patients. The median RFS in the low ERCC1 expression group and the low XPF expression group was shorter than in the corresponding high expression groups (ERCC1: 15.0 vs 32.0 months, P<0.05; XFP: 19.0 vs 31.0 months, P<0.05) (Figure 2A and B). Additionally, the decrease in XPF mRNA expression was significantly associated with OS. The median OS in the low XPF expression group was shorter than in the high expression group (46.0 vs 78.0 months, P<0.05). However, OS was similar in the low and high ERCC1 expression groups (63.0 vs 64.0 months, P>0.05) (Figure 2C and D).
Univariate analysis showed that tumor size, extent of differentiation, and expression of ERCC1 and XPF were associated with RFS (Figure 3).
 |
Figure 3 Forest plot for subgroup univariate analysis of recurrence-free survival. |
Univariate analysis demonstrated that tumor size, the extent of differentiation, and expression of ERCC1 and XPF were associated with RFS (Figure 3). With larger tumor and low degree of differentiation, the RFS was shorter (ERCC1: HR=1.48, 95% CI=1.07 to 2.05, P<0.05; XPF: HR=2.79, 95% CI=1.96 to 3.95, P<0.05). Moreover, the RFS in the groups with low ERCC1 and XPF expression was shorter than the high expression groups (ERCC1: HR=1.49, 95% CI=1.08 to 2.06, P<0.05; XPF: HR=1.51, 95% CI: 1.09–2.08, P<0.05). The number of tumors, degree of differentiation, and XPF expression were associated with OS (Figure 4). Larger number of tumors and worse degree of differentiation correlated with a shorter OS (number of tumors: HR=1.63, 95% CI=1.03 to 2.57, P<0.05; degree off differentiation: HR=2.08, 95% CI=1.41 to 3.08, P<0.05). The OS of the low XPF expression group was shorter than in the high XPF expression group (HR=1.48, 95% CI=1.01 to 2.17, P<0.05).
 |
Figure 4 Forest plot for subgroup univariate analysis of overall survival. |
Multivariate analysis showed that tumor size and the degree of differentiation were independent factors affecting RFS in HCC patients. The risk of relapse in HCC patients with tumors >5 cm and poorly differentiated tumors was 2.20 and 2.84 times higher than in patients with tumors with ≤5 cm and moderately or highly differentiated tumors, respectively (tumor size: HR=2.20, 95% CI=1.33 to 3.63, P<0.05; degree of differentiation: HR=2.84, 95% CI=1.98 to 4.06; P<0.05) (Figure 5). The degree of differentiation and the level of XPF expression were independent factors affecting OS in HCC patients with HCC. The risk of death in patients with poorly differentiated tumors and low expression of XPF was 2.16 times and 2.00 times higher than in patients with highly differentiated tumor and high expression of XPF, respectively (degree of differentiation: HR=2.16, 95% CI=1.42 to 3.28, P<0.05; XPF expression: HR=2.00, 95% CI=1.25 to 3.20; P<0.05) (Figure 6).
 |
Figure 5 Forest plot for subgroup multivariate analysis of recurrence-free survival. |
 |
Figure 6 Forest plot for subgroup multivariate analysis of overall survival. |
Discussion
Primary HCC continues to be one of the most common malignant tumors posing a serious problem to humans. After a radical resection of HCC, new tumors may relapse due to the growth factors present in the microenvironment of liver cancer, and the persistence of HBV and HCV infection and other cancer-promoting factors. Additionally, cancer cells from the originally resected lesion may become disseminated pre-operatively and intraoperatively through the portal vein, giving rise to intrahepatic recurrence and extrahepatic metastasis. At present, the commonly used indicators for the prediction of the relapse of HCC have limitations. Further research is required to identify new effective, sensitive, and specific biomarkers that could serve as predictors of HCC relapse and metastasis. Such markers would be invaluable in the prognosis of HCC, guiding the clinical treatment, and monitoring its results.
Genes responsible for the repair of DNA damage have become a central focus of cancer research. The loss of function of the DNA repair machinery is closely associated with the initiation and development of tumors, including HCC.10 NER is a complicated system involving multiple enzymes. The NER pathway involves at least 20 repair genes, including ERCC1 and XPF. The XPF-ERCC1 complex is a structurally selective heterodimeric endonuclease. The XPF has primarily catalytic function, while ERCC1 binds the DNA strands. Together, these proteins are involved in the incision of the damaged DNA at the 5ʹ end strand and are rate-limiting factors in the NER pathway.9
ERCC1 was the first discovered human DNA repair gene. It has a critical function in the NER pathway, and its activity is closely related to the ability of cells to repair damaged DNA. Numerous studies have demonstrated that the level of ERCC1 expression correlates tightly with the curative effect on treating a variety of tumors and the prognosises, in particular in non-small cell lung cancer (NSCLC),11–14 but the role of ERCC1 in HCC is less frequently studied. There is no consensus regarding the relationship between the ERCC1 expression and HCC prognosis. The present study indicated that ERCC1 was expressed at a low level in HCC, and was associated with early relapse of the tumor. The work by Li suggested that the downregulation of ERCC1 might be associated with early relapse,15 but reports published by Fautrel16 and Bassullu et al17 demonstrated high expression of ERCC1 in HSS. Bassullu’s study determined that the biological characteristics of malignant tumors, including ERCC1 overexpression and vascular lymphatic invasion by HCC, are associated with the poor prognosis, a finding that is inconsistent with the results of this present study. This discrepancy can be explained by different levels of expression of ERCC1 at different developmental stages of various solid tumors such as lung cancer and colorectal cancer. The study from the group of Olaussen found that in patients with early NSCLC, the prognosis was better when the expression of ERCC1 was high, while the patients with low ERCC1 expression were more likely to benefit from platinum-based adjuvant chemotherapy.11 However, a Phase III clinical study conducted by Vilmar et al14 suggested that patients with advanced NSCLC and high expression of ERCC1 were resistant to platinum-based chemotherapy, and their prognosis was poor. The same is true in colorectal cancer. Shirot et al18 measured by RT-PCR the expression of ERCC1 mRNA in advanced colorectal cancer tissues of patients treated with an oxaliplatin-based regimen. They found that the OS in patients with low expression of ERCC1 was significantly longer than in patients with high expression. Liang Jing et al19 demonstrated that patients with stage II colorectal cancer characterized by low ERCC1 expression had a poor prognosis, but could benefit from adjuvant chemotherapy based on an oxaliplatin-based regimen. Thus, in a manner similar to NSCLC and colorectal cancer, ERCC1 appears to be expressed at distinct levels during different stages of HCC development. In the early stage, the amount of DNA damage is relatively limited, and ERCC1 expressed at a low level. At this time, the ability to repair DNA damage is reduced; failure to repair the damage completely results in an ongoing accumulation of DNA lesions resulting in the continuous growth of tumor cells. However, as the tumor progresses, the NER repair capacity becomes enhanced as a compensatory reaction, and the expression of DNA repair genes is also increased. This possibility explains the low expression of ERCC1 at the early stage of HCC and high expression at the advanced stages. Additionally, the aforementioned works of Fautrel and Bassullu16,17 were performed on patients of an ethnicity different from those included in the present study. Since genetic variability among human populations and different liver backgrounds may affect the expression of genetic information, the levels of ERCC1 may be impacted as well, further affecting the biological properties of HCC and prognosis of the disease. Numerous investigations have demonstrated that abnormal expression of the XPF gene was associated with the tumorigenesis of a variety of tumors, including lung, bladder, and gastric cancers.20–22 However, there is no agreement between the studies regarding the relationship between the expression of XPF in various tumors and tumorprognosis. In NSCLC and gastric cancer, XPF is expressed at high levels,20,21 and XPF overexpression was shown to be an independent risk factor for poor prognosis of gastric cancer. Conversely, a study by Zhao et al22 demonstrated low expression of XPF in bladder cancer. The current investigation also showed that XPF mRNA was expressed at a low level in HCC, and this characteristic was associated with poor prognosis. Similarly, Li’s findings also documented the low level of XPF mRNA in HCC. By immunohistochemistry, Zhao23 established that XPF protein was highly expressed in HCC tissues, and this upregulation was associated with the formation of portal vein tumor embolus. Additionally, high expression of XPF protein was indicative of a poor prognosis, a finding inconsistent with the results of the present study. Two major causes underlying this discrepancy can be hypothesized. First, Zhao used immunohistochemistry to determine the protein product encoded by the XPF gene, and the current work utilized RT-PCR to determine the transcription level of XPF. After transcription, a variety of distinct mRNAs can be formed, producing different proteins in the process of translation. However, not all of the encoded products necessarily have the function of nucleotide excision repair. Therefore, changes in the transcription level and translation level of the genes may have a different impact on DNA repair machinery. Moreover, immunohistochemistry has its own limitations. This methodology is greatly influenced by human factors, including the quality of the prepared sections, the quality of staining, and the subjectivity in the evaluation of the results. Therefore, when a study is performed on the differences in XPF expression in hepatocarcinoma tissues and adjacent tissues, the detection of XPF protein content by immunohistochemistry may not serve as a direct indicator of the mRNA expression level of ERCCl gene; The quantitative detection of mature mRNA by RT-PCR is highly specific and sensitive, and the results obtained may be more convincing. Second, since XPF and ERCC1 cooperate in DNA fragment excision in NER, and both are expressed at a low level in HCC, XPF may have a low expression at the early stage of tumor development and be upregulated at a more. In this regard, the present study enrolled patients with stage I–III HCC, with stage I being the predominant type, while the patients included in Zhao’s investigation had stage I-IV HCC. The discrepancy in the proportion of patients with different stages of tumor staging between the two studies might be one of the reasons for the divergence in the obtained results.
In this study we found that ERCC1 and XPF both had predictive effect on recurrence of HCC, further more we also found the combination of two genes had better predictive effect on recurrence of HCC than alone. However neither AFP≥400ng/mL (Chinese liver cancer guideline) nor AFP≥20ng/mL (European liver cancer guideline) can effectively predict recurrence of HCC. The possible causes of negative predictive effect of AFP as followed: (1) Only 177 patients were enrolled in this study, and the lost follow-up rate was 9%. So the negative results may be related to insufficient sample size. (2) The patients included in this study were almost from Guangxi, China, and most of them had HBV infection or cirrhosis. It is possible that AFP has no predictive effect on postoperative recurrence in this cohort, which needs to be verified by further expanding the sample size.
The present study only detected two of the multiple genes in the NER pathway of DNA repair. However, there are more than 20 genes implicated in the NER pathway, and all of them form complex regulatory networks with the genes and their regulatory factors of other DNA repair pathways. Given the complexity of the upstream and downstream regulatory systems, future investigations should not only focus on several individual genes, but also on the numerous networked mechanisms functioning in vivo. A growing number of studies has revealed that XPC, ERCC1, and XPF are closely associated with the initiation and development of various types of tumors and their prognosis. The present study also suggested that the down-regulation of the two genes might affect the progression and prognosis of HCC, but the underlying mechanism of this process is not been fully understood, necessitating more stringent investigations and analyses involving rigorously designed, high-quality, large-sample, multi-center studies.
Some limitations of the present study have to be acknowledged. (1) The current investigation is a single-center retrospective study. The enrolled patients reflect the population of Guangxi Zhuang Autonomous Region, and the limited representativeness may generate bias in population and regional data (2) A small number of HCC patients were treated with prophylactic TACE, and the differences in the number of treatments and the treatment after tumor relapse might have had some impact on the prognosis. This variable was not subject to further stratified analysis in the present study.
In conclusion, the present study suggests that ERCC1 and XPF may serve as biomarkers for predicting early relapse of HCC, and low expression of XPF may be indicative of poor survival prognosis in HCC patients.
Acknowledgments
The present study was supported by the National Natural Science Foundation of China(grant no. 81060169) and Guangxi Scientific Research and Technology Development Plan(grant no.10124001A-31) and the Guangxi Natural Science Foundation (grant nos. 0542064, 2017GXNSFAA198103, and 2017GXNSFBA198041) and Key Planning Development Research Program of Guangxi (grant no.guikeAB16380215) and Pilot Project of Clinical Collaboration of Traditional Chinese Medicine and Western Medicine for Guangxi Major Difficult Diseases (HCC) in 2019.
Author Contributions
Conception and design: Xiaoli Liao, Weimin Xie. Collection and assembly of data: All authors. Data analysis and interpretation: Xiaoli Liao, Yongqiang Li, Hualan Li, Weimin Xie. All authors contributed to data analysis, drafting and revising the article, gave final approval of the version to be published, and agree to be accountable for all aspects of the work.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Bray F, Ferlay J, Soerjomataram I, et al. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68(6):394–424. doi:10.3322/caac.21492
2. Hanaoka F, Egly J, Riedl T. The comings and goings of nucleotide excision repair factors on damaged DNA. EMBO J. 2003;22(19):5293–5303. doi:10.1093/emboj/cdg489
3. Reardon JT, Sancar A. Molecular anatomy of the human excision nuclease assembled at sites of DNA damage. Mol Cell Biol. 2002;22(16):5938–5945. doi:10.1128/MCB.22.16.5938-5945.2002
4. Friedberg EC. How nucleotide excision repair protects against cancer. Nature Rev Cancer. 2001;1(1):22–33. doi:10.1038/35094000
5. Melis JPM, Luijten M, Mullenders LHF, et al. The role of XPC: implications in cancer and oxidative DNA damage. Mutat Res Rev Mutat Res. 2011;728(3):107–117. doi:10.1016/j.mrrev.2011.07.001
6. Houtsmuller AB. Action of DNA repair endonuclease ERCC1/XPF in living cells. Science. 1999;284(5416):958–961. doi:10.1126/science.284.5416.958
7. Wilson MD, Ruttan CC, Koop BF, et al. ERCC1: a comparative genomic perspective. Environ Mol Mutagen. 2001;38(2–3):209–215. doi:10.1002/em.1073
8. Staresincic L, Orelli B, Ivanov D, et al. Structural basis for the recruitment of ERCC1-XPF to nucleotide excision repair complexes by XPA. EMBO J. 2007;26(22):4768–4776. doi:10.1038/sj.emboj.7601894
9. Al-Minawi AZ, Lee YF, Hakansson D, et al. The ERCC1/XPF endonuclease is required for completion of homologous recombination at DNA replication forks stalled by inter-strand cross-links. Nucleic Acids Res. 2009;37(19):6400–6413. doi:10.1093/nar/gkp705
10. Wei-Feng LI, Shan-Shan YU, Guan-Jun CH, et al. DNA damage checkpoint, damage repair, and genome stability. Yi Chuan Xue Bao. 2006;33(5):381–390. doi:10.1016/S0379-4172(06)60064-4
11. Olaussen KA, Dunant A, Fouret P, et al. DNA repair by ERCC1 in non–small-cell lung cancer and cisplatin-based adjuvant chemotherapy. N Engl J Med. 2006;355:10. doi:10.1056/NEJMoa060570
12. Friboulet L, Olaussen KA, Pignon JP, et al. ERCC1 isoform expression and DNA repair in non – small-cell lung cancer. N Engl J Med. 2013;368(12):1101–1110. doi:10.1056/NEJMoa1214271
13. Besse B, Massard C, Haddad V, et al. ERCC1 influence on the incidence of brain metastases in patients with non-squamous NSCLC treated with adjuvant cisplatin-based chemotherapy. Ann Oncol. 2011;22(3):575–581. doi:10.1093/annonc/mdq407
14. Vilmar AC, Santoni-Rugiu E, Sorensen JB. ERCC1 and histopathology in advanced NSCLC patients randomized in a large multicenter phase III trial. Ann Oncol. 2010;21(9):1817–1824. doi:10.1093/annonc/mdq053
15. Li SS. The Expression and Significance of DNA REPAIR Genes XPC, ERCC1 and XPF in Hepatocellular Carcinoma. Guangxi Medical University; 2016.
16. Fautrel A, Andrieux L, Musso O, et al. Overexpression of the two nucleotide excision repair genes ERCC1 and XPC in human hepatocellular carcinoma. J Hepatol. 2005;43(2):288–293. doi:10.1016/j.jhep.2005.02.020
17. Bassullu N, Turkmen I, Dayangac M, et al. The predictive and prognostic significance of c-erb-B2, EGFR, PTEN, mTOR, PI3K, p27, and ERCC1 expression in hepatocellular carcinoma. Hepat Mon. 2012;12:10. doi:10.5812/hepatmon.7492
18. Shirota Y, Stoehlmacher J, Brabender J, et al. ERCC1 and thymidylate synthase mRNA levels predict survival for colorectal cancer patients receiving combination oxaliplatin and fluorouracil chemotherapy. J Clin Oncol. 2001;19(23):4298. doi:10.1200/JCO.2001.19.23.4298
19. Liang J, Wu WA, Bai L, et al. Expression of ERCC1 in stage Ⅱ colorectal carcinoma and its clinical significance. J Xi’an Jiaotong Univ. 2017;38(06):898–903.
20. Liu C, Zhou S, Begum S, et al. Increased expression and activity of repair genes TDP1 and XPF in non-small cell lung cancer. Lung Cancer. 2007;55(3):303–311. doi:10.1016/j.lungcan.2006.10.019
21. Li P, Ma Y. Correlation of xeroderma pigmentosum complementation group F expression with gastric cancer and prognosis. Oncol Lett. 2018;16(6):6971–6976. doi:10.3892/ol.2018.9529
22. Zhao YG, Yang J, Zeng YJ, et al. Expression level of xeroderma pigmentosum group F protein in bladder cancer tissue and its clinical significance. Chin J Biol. 2010;23(3):303–306.
23. Zhao P. Expression and Significance of XPF in Primary Hepatocellular Carcinoma. Guangxi Medical University; 2010.
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.