Back to Journals » OncoTargets and Therapy » Volume 12
Everolimus for the treatment of advanced gastrointestinal or lung nonfunctional neuroendocrine tumors in East Asian patients: a subgroup analysis of the RADIANT-4 study
Authors Yao JC , Oh DY, Qian JM, Park YS, Herbst F, Ridolfi A, Izquierdo M , Ito T, Jia LQ, Komoto I, Sriuranpong V , Shimada Y
Received 1 August 2018
Accepted for publication 9 January 2019
Published 28 February 2019 Volume 2019:12 Pages 1717—1728
DOI https://doi.org/10.2147/OTT.S182259
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr William C. Cho
James C Yao,1 Do-Youn Oh,2 Jiaming Qian,3 Young Suk Park,4 Fabian Herbst,5 Antonia Ridolfi,6 Miguel Izquierdo,5 Tetsuhide Ito,7 Liqun Jia,8 Izumi Komoto,9 Virote Sriuranpong,10 Yasuhiro Shimada11
1Department of Gastrointestinal Medical Oncology, University of Texas MD Anderson Cancer Center, Houston, TX, USA; 2Department of Medical Oncology, Seoul National University Hospital, Seoul National University College of Medicine, Seoul, South Korea; 3Department of Gastroenterology, Peking Union Medical College Hospital, Beijing, China; 4Department of Hematology and Oncology, Samsung Medical Center, Seoul, South Korea; 5Novartis Oncology, Novartis Pharma AG, Basel, Switzerland; 6Novartis Oncology, Novartis Pharma S.A.S, Rueil-Malmaison, Paris, France; 7Department of Gastroenterology, Kyushu University Hospital, Fukuoka, Japan; 8Department of Medical Oncology, China-Japan Friendship Hospital, Beijing, China; 9Department of Surgery, Kansai Electric Power Hospital, Osaka, Japan; 10Department of Medical Oncology, Chulalongkorn University and King Chulalongkorn Memorial Hospital, Bangkok, Thailand; 11Department of Gastrointestinal Medical Oncology, National Cancer Center Hospital, Tsukiji, Chuo-ku, Tokyo, Japan
Background: In RADIANT-4, everolimus showed an improvement of 7.1 months in median progression-free survival (PFS) vs placebo among patients with advanced, well-differentiated, nonfunctional neuroendocrine tumors (NETs) of gastrointestinal (GI) or lung origin. The present analysis focuses on the effect of everolimus on the East Asian-subgroup population of the RADIANT-4 study.
Methods: Patients were randomized to receive everolimus 10 mg/day or matching placebo. The primary end point was PFS (central review). Secondary end points were overall response rate, safety, and tolerability.
Results: Among 302 patients enrolled in RADIANT-4, 46 were included in the East Asian subgroup (everolimus, n=28; placebo, n=18) analysis. Everolimus was associated with an 82% reduction in the relative risk of disease progression or death (HR 0.18, 95% CI 0.09–0.38). The median PFS (central review) in this subgroup was 11.2 months with everolimus vs 3.1 months with placebo. Adverse events (AEs) occurred in all 28 patients treated with everolimus and ten patients receiving placebo. The majority of these AEs were grade 1 or 2. Most commonly reported (≥30% of incidence) drug-related AEs of any grade included stomatitis (75%, n=21) and rash (43%, n=12) in the everolimus arm.
Conclusion: Everolimus demonstrated a clinically meaningful PFS benefit in the East Asian population. The safety findings were consistent with the known safety profile of everolimus. These results support the use of everolimus in the East Asian population with advanced, nonfunctional NETs of GI or lung origin.
Keywords: mTOR inhibitors, everolimus, RADIANT-4, neuroendocrine tumors, East Asian population
Plain-language summary
Everolimus improved the median progression-free survival by 7.1 months vs placebo among patients with advanced, well-differentiated, nonfunctional neuroendocrine tumors of gastrointestinal or lung origin in the RADIANT-4 study. The present post hoc analysis of the phase III, randomized, placebo-controlled, RADIANT-4 study demonstrates a clinically meaningful improvement in progression-free survival in East Asian patients with advanced, progressive, nonfunctional neuroendocrine tumors of lung or gastrointestinal origin. In addition, the safety of everolimus in the East Asian subgroup was consistent with the known safety profile of everolimus.
Introduction
Neuroendocrine tumors (NETs) are a group of rare heterogeneous malignancies that arise from neuroendocrine cells found throughout the body.1 Tumors associated with hormonal symptoms due to excessive secretion of hormones and peptides are termed functional NETs, whereas those not associated with hormonal symptoms are considered nonfunctional. The majority of NETs are nonfunctional, and most commonly arise in the gastrointestinal (GI) and bronchopulmonary regions.2 An epidemiological analysis revealed that carcinoid syndrome is significantly associated with the primary tumor site, grade, and stage.3 Understanding the complex cell biology and tumor heterogeneity associated with NETs could facilitate a tailored approach to improve patient survival.3 According to the US population-based Surveillance, Epidemiology, and End Results (SEER) database, the annual age-adjusted incidence of NETs showed a 6.4-fold increase – from 1.1 in 100,000 in 1973 to 7.0 in 100,000 in 2012 – and continues to grow irrespective of tumor site, stage, and grade.4 Incidence rates were higher for gastroenteropancreatic (GEP) NETs (3.6 in 100,000) than lung NETs (1.5 in 100,000), followed by unknown primary (0.8 in 100,000) as per the SEER 18 registry (2000–2012).4 Overall survival rates increased from 2000–2004 to 2009–2012, wherein patients had a 21.3% of reduced risk of death (HR 0.79, 95% CI 0.73–0.85). Similar trends have been observed in distant-stage GI NETs.4
The reason for this rise in incidence is unknown, although a variety of underlying factors, including improved diagnostic methods, are suspected.3 In 2010, the prevalence and annual incidence rates of GI NETs in Japanese patients were 6.4 in 100,000 and 3.5 in 100,000, respectively.5 A nationwide survey reported a much higher frequency of GI NETs among Japanese patients than was reported in Western countries.4 The incidence rate of NETs in Taiwan was not well known until Tsai et al published their surveys, which showed a fivefold increase from 0.3 in 100,000 in 1996 to 1.5 in 100,000 in 2008.6 A single-center study7 conducted in South Korea estimated the incidence of NETs to be 24.1 in 100,000, which is comparatively much higher than the population-based rate (2.9–5.9 in 100,000).1,8 These epidemiological data suggest that ethnic differences, including possible differences in hormone production or other characteristics, may have a role in the clinical presentation of disease; however, potential ethnic differences in the evaluation of drug therapies for NETs have not been studied extensively.7
In 2015, the Japan Neuroendocrine Tumor Society established consensus guidelines for the diagnosis and management of Japanese patients with GEP NETs. These guidelines focus primarily on identifying the disease stage and highlight the importance of an evidence-based treatment approach for the treatment of Japanese patients with GEP NETs.8
Surgery is the mainstay of treatment of localized and locoregional NETs of GI or lung origin, and no adjuvant therapy is required. Somatostatin analogues (SSAs), including octreotide and lanreotide, are the current gold standard and a widely used treatment option to control the symptoms of functional NETs.9 Furthermore, long-acting octreotide has also demonstrated antiproliferative activity in patients with well-differentiated, unresectable, metastatic midgut NETs in the randomized, double-blind, placebo-controlled, multicenter PROMID study.10 In addition, findings from the CLARINET study11 led to the approval of long-acting lanreotide 120 mg for the treatment of Japanese patients with GEP NETs.12 Additional treatment options include hepatic arterial embolization, radioembolization, peptide-receptor radionuclide therapy, and chemoembolization.
The mTOR pathway is an essential cellular signaling pathway that serves as a central modulator of cell metabolism, proliferation, growth, and survival. Aberrant activation of mTOR by PI3K/Akt has been identified in a wide range of cancers, including NETs.13 Everolimus (an oral mTOR inhibitor) has demonstrated significant activity in the randomized phase III studies RADIANT-3 and RADIANT-4.14,15 In the RADIANT-4 study, everolimus showed a 52% reduction in risk of disease progression or death (HR 0.48, 95% CI 0.35–0.67), with median progression-free survival (PFS) improved by 7.1 months in patients with advanced, well-differentiated, nonfunctional NETs of GI or lung origin compared with placebo. Based on this study’s findings, everolimus received approval for the treatment of progressive, unresectable, or metastatic, well-differentiated, nonfunctional NETs of the GI tract or lung.16 Further, everolimus is also approved for the treatment of patients with advanced or metastatic, unresectable, progressive pancreatic NETs.17 This present analysis aimed to explore the efficacy and safety of everolimus in the East Asian subgroup of the RADIANT-4 study.
Methods
Study design and treatment
RADIANT-4 was an international, multicenter, randomized, double blind, placebo-controlled, phase III study (NCT01524783) conducted in 97 centers in 25 countries worldwide. For the East Asian-subgroup analysis, patients were included from 20 centers in five countries (China, Japan, South Korea, Taiwan, and Thailand; Figure 1). Patients aged ≥18 years with pathologically confirmed, advanced, nonfunctional, well-differentiated NETs of lung or GI origin with documented disease progression within 6 months before randomization were eligible for inclusion.
Additional inclusion criteria included measurable disease as per Response Evaluation Criteria in Solid Tumors (RECIST) version 1.0, World Health Organization (WHO) performance status (0 or 1), and adequate bone-marrow, liver, and kidney function. Patients were ineligible if they had a history of or presented with carcinoid syndrome, poorly differentiated histology, or pancreatic NETs, and had received more than one line of chemotherapy, previous treatment with mTOR inhibitors (sirolimus, temsirolimus, or everolimus), hepatic intra-arterial embolization within 6 months of randomization, cryoablation or radiofrequency ablation of hepatic metastases within 2 months of randomization, or chronic treatment with corticosteroids or other immunosuppressive agents. The details of RADIANT-4 inclusion and exclusion criteria have been reported previously.15
The study followed the guiding principles of the Declaration of Helsinki, Good Clinical Practice guidelines, and local regulations. The study was approved by each center’s institutional review boards or independent ethics committees (Supplementary material). All patients provided written informed consent. An independent data-monitoring committee reviewed safety-related data and provided oversight in study conduct.
Patients were randomly allocated (2:1) to receive everolimus (oral, 10 mg/day) or matching placebo, both with best supportive care, until disease progression or occurrence of an intolerable adverse event (AE) or consent withdrawal. Treatment was continued until disease progression or occurrence of an intolerable AE or consent withdrawal. An interactive voice-response system was used to randomize the patients. Randomization was stratified based on prior SSA treatment (defined as continuous SSA treatment for ≥12 weeks), tumor origin (based on prognostic level, grouped into two strata: stratum A [better prognosis], appendix, cecum, jejunum, ileum, duodenum, or NETs of unknown primary; stratum B [worse prognosis], lung, stomach, colon [other than cecum], or rectum), and WHO performance status 0 or 1. Dose reductions and treatment interruptions were permitted for the occurrence of any treatment-related AEs and for a maximum of 28 days. Crossover from the placebo to open-label everolimus upon disease progression was not permitted, and the investigators remained blinded to treatment assignment until the primary analysis.15
Assessments
The primary efficacy end point was PFS as per central review, defined as the time from randomization to death or progression according to RECIST version 1.0. Secondary end points comprised overall response rate (defined as proportion of patients with best overall response of complete response [CR] or partial response [PR]), disease-control rate (defined as the proportion of patients with best overall response of CR, PR, or stable disease, safety, and tolerability. An exploratory analysis of this study included an evaluation of population pharmacokinetic (PK) data.
The primary efficacy analysis was performed on an intent-to-treat basis, whereas the safety set included all patients who had received at least one dose of the study drug with at least one postbaseline safety assessment. AEs were reported as per National Cancer Institute Common Terminology Criteria for AEs version 4.03.15 A predose PK blood sample was collected in all patients by direct venipuncture prior to dosing on day 29. The number of blood samples available for PK analysis was limited, and only valid blood samples (confirmed blood samples collected at steady state, immediately prior to dosing on the same study day, approximately 24±4 hours after the previous dose, and with no vomiting within the first 4 hours following the last dose) were considered.
Statistical analyses
By central radiology review, PFS was analyzed using Kaplan–Meier methods, as reported previously.15 The full-analysis set (all randomized patients) was used for all efficacy analyses. Sample-size calculations were not undertaken for the cohort of patients described in this report, as this analysis was not powered to compare treatment groups. HRs and corresponding CIs were estimated using an unstratified Cox proportional-hazard model.15 Steady-state condition for the minimum plasma concentration (Cmin) sample was defined as continuous administration of the same dose in the last 4 days prior to collection of the Cmin sample. Drug concentrations for the everolimus and best-supportive-care groups were summarized by actual leading dose, ie, the dose taken on the day prior to the PK sampling day. Descriptive statistics included arithmetic and geometric mean, median, SD, CV%, geometric CV%, minimum, and maximum. Concentrations of 0 were not included in the geometric mean calculation. PK analysis was based on the safety set in patients with evaluable samples.
Results
Patient demographics and disposition
Of the 302 patients (everolimus, n=205; placebo, n=97) enrolled in the RADIANT-4 study who received treatment with everolimus 10 mg/day or matching placebo, 46 were included in the East Asian subgroup (everolimus, n=28; placebo, n=18; Figure S1). The median age of patients in the East Asian subgroup was 57 (22–75) years in the everolimus arm and 53 (33–72) years in the placebo arm, which was consistent with the overall RADIANT-4 population.15 The details of baseline demographics and disease characteristics are included in Table 1.
  | Table 1 Baseline demographics, disease characteristics, and prior therapies |
Patient disposition is shown in Table 2. The median duration of exposure was 46.9 (3.1–101.4) and 40.4 (0.7–120.4) weeks for everolimus vs 13.4 (4.0–85.3) and 19.6 (4.0–130.3) weeks for placebo in the East Asian subgroup and the overall RADIANT-4 population, respectively.
  | Table 2 Patient disposition and duration of exposure |
A greater number of patients in the placebo arm discontinued treatment (94% in the East Asian subgroup and 87% in the overall population) compared with everolimus (68% in the East Asian subgroup and 76% in the overall population). In both the East Asian subgroup and the overall population, disease progression was the primary reason for treatment discontinuation. The proportion of patients experiencing disease progression by RECIST version 1.0 was substantially higher in the placebo arm (77.8% in the East Asian subgroup and 72.2% in the overall population) compared with everolimus (42.9% in the East Asian subgroup and 37.1% in the overall population). Other reasons for treatment discontinuation included AEs, consent withdrawal, protocol deviation, or death (Table 2).
Efficacy
In the East Asian subgroup, everolimus was associated with an 82% reduction in estimated risk of disease progression or death (HR 0.18, 95% CI 0–0.38; Figure 2A), with improved median PFS of 11.2 months (95% CI 7.33–not available; 15 events) with everolimus vs 3.1 months (95% CI 1.81–3.71; 17 events) with placebo arms. These results were consistent with the overall RADIANT-4 population, wherein everolimus was associated with a 52% reduction in risk of disease progression or death (HR 0.48, 95% CI 0.35–0.67; Figure 2B), with improved median PFS of 11.0 months (95% CI 9.23–13.31; 113 events) with everolimus vs 3.9 months (95% CI 3.58–7.43; 65 events) with placebo arms.15 The disease-control rate (greater than or equal to stable disease) was 93% and 44% in the everolimus and placebo arms, respectively, for the East Asian subgroup compared with 82% and 65%, respectively, for the everolimus and placebo arms in the overall population.15 In the East Asian subgroup, stable disease as best response was noted in 26 (93%) and eight patients (44%) in the everolimus and placebo arms, respectively. Progressive disease as best response occurred in one patient (4%) treated with everolimus and eight patients (44%) in the placebo arm. None of the patients in the East Asian subgroup achieved CR or PR, which was consistent with efficacy for the overall RADIANT-4 population (Table 3). From the results of the best percentage change from baseline in the East Asian subgroup, it was observed that a greater proportion of patients experienced reduction in size of the target lesion in the everolimus arm vs placebo (72% vs 21%; Figure 3A). This difference was slightly greater than observed for the overall population (64% vs 26% in the everolimus and placebo arms, respectively; Figure 3B).
  | Figure 2 Kaplan–Meier plot of progression-free survival. |
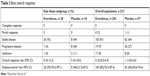  | Table 3 Best overall response |
  | Figure 3 Percentage changes from baseline in size of target lesion: central review (full-analysis set). |
Safety
In the East Asian subgroup, AEs suspected to be related to the study drug occurred in all 28 patients (100%) who were treated with everolimus and ten (56%) who received placebo. The most frequently reported (≥30% of incidence) drug-related any-grade AEs in patients receiving everolimus in the East Asian subgroup vs the overall population were stomatitis (21 of 28 [75%] vs 127 of 202 [63%]), and rash (12 of 28 [43%] vs 55 of 202 [27%]). The majority of the AEs reported were of grade 1 or of grade 2 in severity (Table 4). Drug-related grade 3 or 4 AEs were reported in ten patients in the everolimus arm and two patients receiving placebo. The most common drug-related grade 3 or 4 AEs in the everolimus arm were stomatitis, anemia, and increased levels of ALT (each two of 28 [7%]).
  | Table 4 Drug-related adverse events in the East Asian subgroup and the overall population (≥10% of incidence in either arm) |
In the overall population, the most common drug-related any-grade AEs occurring in ≥30% of patients given everolimus were stomatitis (127 of 202 [63%]), diarrhea (63 of 202 [31%]), and fatigue (62 of 202 [31%]). The majority of these AEs were grade 1 or 2. The most common drug-related grade 3 or 4 AEs with everolimus included stomatitis (18 of 202 [9%]), diarrhea (15 of 202 [7%]), infections (14 of 202 [7%]), fatigue (seven of 202 [3%]), anemia (eight of 202 [4%]), and hyperglycemia (seven of 202 [4%]).15
Pharmacokinetics
A summary of everolimus PK parameters after 5 mg and 10 mg daily doses is presented in Table S1. Considering the high interpatient variability in both East Asian and non–East Asian patients, there was no apparent difference in Cmin observed in the two subgroups after daily administration of 10 mg everolimus. Further, the everolimus Cmin exposure among the East Asian subgroup population was found to be consistent with the overall population.
Discussion
RADIANT-4 is the largest clinical study to demonstrate significant improvement in PFS in patients with advanced, progressive NETs of GI or lung origin.15 However, considering the heterogeneous nature of patient population, there is a need to conduct a subgroup analysis to better understand the effect of everolimus on geographical population-based distribution.
In the present post hoc analysis of the RADIANT-4 East Asian subgroup, everolimus was associated with an 82% reduction in estimated risk of progression or death (HR 0.18, 95% CI 0.09–0.38), with improved median PFS of 11.2 months with everolimus vs 3.1 months with placebo, in patients with advanced, progressive, nonfunctional NETs of GI or lung origin. In the overall RADIANT-4 population, everolimus showed a 52% reduction in the estimated risk of progression or death (HR 0.48, 95% CI 0.35–0.67), and median PFS was prolonged by a clinically meaningful 7.1 months for patients treated with everolimus. Further, the safety of everolimus in the overall RADIANT-4 population was found to be consistent with the established safety profile.15 Based on the findings from this study, everolimus has been approved worldwide for the treatment of patients with advanced, nonfunctional NETs of GI, lung, and pancreatic origin.
The reduction in risk of disease progression or death in the East Asian subgroup (82%) was greater than observed in the overall RADIANT-4 population (52%). In addition, a slightly higher proportion of patients in the East Asian subgroup experienced tumor shrinkage than the overall population. Tumor shrinkage occurred more frequently in the everolimus than placebo arms (East Asian, 72% vs 21%; overall, 64% vs 26%).15
The efficacy of everolimus was also assessed in the Japanese subgroup of the RADIANT-3 study, wherein median PFS was 19.4 months (95% CI 8.31–not available) with everolimus vs 2.8 months (95% CI 2.46–8.34) with placebo, corresponding to an 81% reduction in risk of disease progression or death (HR 0.19, 95% CI 0.08–0.48).18 On the other hand, in the overall RADIANT-3 population, median PFS was 11.0 months with everolimus vs 4.6 months with placebo (HR 0.35, 95% CI 0.27–0.45), resulting in a 65% reduction in estimated risk of disease progression or death.14 The favorable efficacy outcomes and tolerable safety profile in the RADIANT-3 Japanese subgroup and the RADIANT-4 East Asian subgroup populations were consistent with their respective overall study populations.
In the present East Asian subgroup of RADIANT-4, which included patients with NETs of GI and lung origin, study-drug-related AEs occurred in all patients who received everolimus and in approximately 55% of patients receiving placebo. The majority of these events were of grade 1 or of grade 2 (Table 4). Drug-related pneumonitis is a class-effect toxicity of rapamycin derivatives, including everolimus.19 This effect has been studied in several studies conducted on patients with advanced renal-cell carcinoma, non-small-cell lung carcinoma, and advanced NETs.14,20,21 The reported incidence rates of pneumonitis in patients with advanced renal-cell carcinoma, non-small-cell lung carcinoma, and advanced NETs were 23%–30%, 25%, and 12%, respectively.14,20–23
In a phase III study of everolimus in combination with long-acting octreotide in patients with advanced NETs, the drug-related pneumonitis rate was 8%.24 The incidence of pneumonitis in the everolimus arm of the East Asian subgroup of RADIANT-4 was 18%; however, none of these was severe in nature. The incidence of pneumonitis in the everolimus arm of the overall population was 14%. The majority of these were either of grade 1 or of 2 in severity. Grade 3 pneumonitis was observed in three patients. The incidence of grade 3 or 4 AEs in the East Asian patients was comparatively lower than the overall population. In general, everolimus toxicity was manageable, with no new safety signals observed during the study (incidence ≥30%; East Asian subgroup, stomatitis [75%], rash [43%], and pneumonitis [18%]; overall population, stomatitis [63%], rash [27%], and pneumonitis [14%]).
Among the East Asian, non–East Asian, and overall RADIANT-4 populations, no differences in exposure in terms of Cmin were noted after daily administration of 10 mg everolimus. However, a reduced risk of progression or death was observed in the East Asian subgroup (HR 0.18) vs the overall population (HR 0.48). A possible explanation for this reduced risk in the East Asian subset could be the longer duration of everolimus exposure in these patients (47 weeks) compared with the overall population (40 weeks) or differences in distribution of disease characteristics, such as primary site (rectum as primary site, East Asian 41% [19 of 46] vs overall 13% [40 of 302]).
Although this study was a post hoc analysis from a randomized phase III study, it is the first study of its kind specifically to include patients from the East Asian region (China, Japan, South Korea, Taiwan, and Thailand) from a phase III study on NETs. As with most subgroup analyses, this study carries some limitations: it was not powered to compare the treatment groups, was not a planned subgroup analysis, and the sample was small. Therefore, careful interpretation of data and extrapolation should be considered.
Conclusion
Everolimus demonstrated a clinically meaningful improvement of PFS in East Asian patients with advanced, progressive, nonfunctional NETs of lung or GI origin enrolled in RADIANT-4. The safety profile in the East Asian subgroup was consistent with the known safety of everolimus, and no new safety signals were observed. These findings support the use of everolimus as a valuable treatment option in East Asian patients with advanced, progressive, nonfunctional NETs of lung or GI origin.
Data-sharing statement
Novartis is committed to sharing with qualified external researchers access to patient-level data and supporting clinical documents from eligible studies. These requests are reviewed and approved by an independent review panel on the basis of scientific merit. All data provided are anonymized to respect the privacy of patients who have participated in the trial in line with applicable laws and regulations. This trial-data availability is according to the criteria and process described on www.clinicalstudydatarequest.com.
Acknowledgments
We thank the patients, their families, and their caregivers, as well as the investigators and research nurses. The study was sponsored by Novartis Pharmaceuticals Corporation. In addition, we thank Charu Pundir (Novartis Healthcare Pvt Ltd) for providing medical editorial assistance with this article.
Author contributions
JCY, DYO, JQ, YSP, TI, LJ, IK, VS, and YS were responsible for patient recruitment, JCY and DYO for study design, and AR for statistical analysis. All authors contributed toward data interpretation, drafting and critically revising the paper, provided final approval to the version to be published, and agree to remain accountable for all aspects of the work.
Disclosure
The authors report the following types of declarations of interest: consultant/advisory relationship (C/A), employment (E), honoraria received (H), intellectual property rights/inventor/patent holder (IP), leadership position (L), ownership interest (OI), research funding (RF), speaker’s bureau (SB), and travel and accommodation expenses (TAE). JCY: Novartis (H, C/A), Ipsen (C/A). VS: Novartis (RF). YS: Novartis (H). FH: Novartis (E). AR: Novartis (E). MI: Novartis (E). The other authors report no conflicts of interest in this work.
References
Hallet J, Law CH, Cukier M, Saskin R, Liu N, Singh S. Exploring the rising incidence of neuroendocrine tumors: a population-based analysis of epidemiology, metastatic presentation, and outcomes. Cancer. 2015;121(4):589–597. | ||
Vinik AI, Woltering EA, Warner RR, et al. NANETS consensus guidelines for the diagnosis of neuroendocrine tumor. Pancreas. 2010;39(6):713–734. | ||
Halperin DM, Shen C, Dasari A, et al. Frequency of carcinoid syndrome at neuroendocrine tumour diagnosis: a population-based study. Lancet Oncol. 2017;18(4):525–534. | ||
Dasari A, Shen C, Halperin D, et al. Trends in the incidence, prevalence, and survival outcomes in patients with neuroendocrine tumors in the United States. JAMA Oncol. 2017;3(10):1335–1342. | ||
Ito T, Igarashi H, Nakamura K, et al. Epidemiological trends of pancreatic and gastrointestinal neuroendocrine tumors in Japan: a nationwide survey analysis. J Gastroenterol. 2015;50(1):58–64. | ||
Tsai HJ, Wu CC, Tsai CR, Lin SF, Chen LT, Chang JS. The epidemiology of neuroendocrine tumors in Taiwan: a nation-wide cancer registry-based study. PLoS One. 2013;8(4):e62487. | ||
Lim CH, Lee IS, Jun BY, et al. Incidence and clinical characteristics of gastroenteropancreatic neuroendocrine tumor in Korea: a single-center experience. Korean J Intern Med. 2017;32(3):452–458. | ||
Guideline for Pancreatic and Gastroenteric Neuroendocrine Tumor. edited by Japan neuroendocrine tumor Society. Tokyo: Kanbara publishing. Japanese. | ||
Öberg K, Hellman P, Ferolla P, Papotti M, ESMO Guidelines Working Group. Neuroendocrine bronchial and thymic tumors: ESMO clinical practice guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2012;23(suppl 7):vii120–vii123. | ||
Rinke A, Müller H-H, Schade-Brittinger C, et al. Placebo-controlled, double-blind, prospective, randomized study on the effect of octreotide LAR in the control of tumor growth in patients with metastatic neuroendocrine midgut tumors: a report from the PROMID Study Group. J Clin Oncol. 2009;27(28):4656–4663. | ||
Caplin ME, Pavel M, Ćwikła JB, et al. Lanreotide in metastatic enteropancreatic neuroendocrine tumors. N Engl J Med. 2014;371(3):224–233. | ||
Pharmaceuticals and Medical Devices Agency. Available from: https://www.pmda.go.jp/files/000224507.pdf. Accessed January 24, 2019. Japanese. | ||
Faivre S, Kroemer G, Raymond E. Current development of mTOR inhibitors as anticancer agents. Nat Rev Drug Discov. 2006;5(8):671–688. | ||
Yao JC, Shah MH, Ito T, et al. Everolimus for advanced pancreatic neuroendocrine tumors. N Engl J Med. 2011;364(6):514–523. | ||
Yao JC, Fazio N, Singh S, et al. Everolimus for the treatment of advanced, non-functional neuroendocrine tumours of the lung or gastrointestinal tract (RADIANT-4): a randomised, placebo-controlled, phase 3 study. Lancet. 2016;387(10022):968–977. | ||
National Cancer Institute. NCI dictionary of cancer terms. Available from: https://www.cancer.gov/publications/dictionaries/cancer-terms/search?contains=false&q=afinitor. Accessed April 07, 2018. | ||
Afinitor (everolimus) prescribing information. East Hanover, NJ, USA: Novartis pharmaceuticals Corporation; February 2016. Available from: https://www.pharma.us.novartis.com/sites/www.pharma.us.novartis.com/files/afinitor.pdf. Accessed April 07, 2018. | ||
Ito T, Okusaka T, Ikeda M, et al. Everolimus for advanced pancreatic neuroendocrine tumours: a subgroup analysis evaluating Japanese patients in the RADIANT-3 trial. Jpn J Clin Oncol. 2012;42(10):903–911. | ||
Bouabdallah K, Ribrag V, Terriou L, Soria JC, Delarue R. Temsirolimus in the treatment of mantle cell lymphoma: frequency and management of adverse effects. Curr Opin Oncol. 2013;25(Suppl 2):S1–S12. | ||
Soria JC, Shepherd FA, Douillard JY, et al. Efficacy of everolimus (RAD001) in patients with advanced NSCLC previously treated with chemotherapy alone or with chemotherapy and EGFR inhibitors. Ann Oncol. 2009;20(10):1674–1681. | ||
Atkinson BJ, Cauley DH, Ng C, et al. Mammalian target of rapamycin (mTOR) inhibitor-associated non-infectious pneumonitis in patients with renal cell cancer: predictors, management, and outcomes. BJU Int. 2014;113(3):376–382. | ||
Dabydeen DA, Jagannathan JP, Ramaiya N, et al. Pneumonitis associated with mTOR inhibitors therapy in patients with metastatic renal cell carcinoma: incidence, radiographic findings and correlation with clinical outcome. Eur J Cancer. 2012;48(10):1519–1524. | ||
Maroto JP, Hudes G, Dutcher JP, et al. Drug-related pneumonitis in patients with advanced renal cell carcinoma treated with temsirolimus. J Clin Oncol. 2011;29(13):1750–1756. | ||
Pavel ME, Hainsworth JD, Baudin E, et al. Everolimus plus octreotide long-acting repeatable for the treatment of advanced neuroendocrine tumours associated with carcinoid syndrome (RADIANT-2): a randomised, placebo-controlled, phase 3 study. Lancet. 2011;378(9808):2005–2012. |
Supplementary materials
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.



