Back to Journals » Infection and Drug Resistance » Volume 14
Evaluation of the Efficacy of Alcohol-Based Hand Sanitizers Sold in Southwest Ethiopia
Authors Manaye G , Muleta D, Henok A, Asres A , Mamo Y, Feyissa D , Ejeta F , Niguse W
Received 11 December 2020
Accepted for publication 28 January 2021
Published 12 February 2021 Volume 2021:14 Pages 547—554
DOI https://doi.org/10.2147/IDR.S288852
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 4
Editor who approved publication: Dr Sahil Khanna
Gizachew Manaye,1 Desalegn Muleta,1 Andualem Henok,2 Abyot Asres,2 Yitagesu Mamo,3 Desalegn Feyissa,3 Fikadu Ejeta,3 Wondwossen Niguse4
1Department of Medical Laboratory Science, College of Medicine and Health Sciences, Mizan-Tepi University, Mizan Teferi, Ethiopia; 2Department of Public Health, College of Medicine and Health Sciences, Mizan-Tepi University, Mizan Teferi, Ethiopia; 3Department of Pharmacy, College of Medicine and Health Sciences, Mizan-Tepi University, Mizan Teferi, Ethiopia; 4Department of Nursing, College of Medicine and Health Sciences, Mizan-Tepi University, Mizan Teferi, Ethiopia
Correspondence: Gizachew Manaye
Department of Medical Laboratory Science, College of Medicine and Health Sciences, Mizan-Tepi University, P.O. Box: 206, MizanTefri, Ethiopia
Tel +251928444333
Email [email protected]
Background: Regarding the protection of community and health professionals suffering from a COVID-19 outbreak, currently different alcohol-based hand sanitizers have been distributed. Even though for effective protection effective alcohol-based hand sanitizers are mandatory. Their efficacy was not evaluated. This is the reason why this research was designed to assess the antimicrobial efficacy of hand sanitizers that have been sold in southern parts of Ethiopia.
Methods: Six test organisms (Staphylococcus aureus, Escherichia coli, Pseudomonas aeruginosa, Klebsiella pneumoniae, Salmonella typhimurium and Shigella boydii) were selected from different clinical specimens. Then seven locally made products of alcohol-based hand sanitizers (MTU, Folium Fine, Epharm, Harego, Taflen and Sheba) were purchased and the disc diffusion, minimum inhibition concentration and minimum bactericidal concentration test were done against selected test organisms. Quality control measures throughout the whole process of the laboratory work were implemented and descriptive parameters were analyzed.
Results: The zones of inhibition of the hand sanitizers at their claim concentration were ranged from noninhibition zone (Folium) to 27mm (Sheba). The minimum inhibitory concentration against all selected test organisms was observed at 45%, 55%, 65% and Taflen on undiluted form. The growth of test organisms was decreased across increasing the concentration gradient of different hand sanitizers. MTU except against E. coli, Fine, Hargo, Ephra and Sheba hand sanitizers were showed growth below 60% concentrations for all test micro-organisms. But a Folium product against all selected test micro-organisms and MTU product against E. coli were not bactericidal.
Conclusion: Fine, Hargo, Ephra, Sheba and Taflen sanitizers were the products that were effective in inhibiting the growth of all the selected test organisms, and they were having effective bactericidal activity in vitro at their claim concentration. But MTU product against E. coli and Folium product against all test microorganisms were not having an effective bactericidal activity. So, regulatory authorities and manufacturers should implement strict quality control measures and regular observations throughout the production to ensure the efficacy of hand sanitizers.
Keywords: alcohol-based hand sanitizers, hand sanitizers, sanitizers, COVID-19
Background
Hospital-associated and community-acquired infections being a serious public health problem all over the world and have become a major concern.1 Hospital-acquired infections are infections developing in hospitalized patients,2 whereas community-acquired infection on the other hand is an infection acquired anywhere other than a healthcare facility.1 These infections have considerable impacts on individuals such as prolonged hospitalization, disability, increased risk of antimicrobial resistance, huge financial burden and deaths.3
The Center for Disease Control and Prevention (CDC) showed that about 2 million people acquire hospital-associated infections every year and about 90,000 of these patients die as a result of their infections.4 Currently, severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) is a viral infectious agent of both Hospital-associated and community-acquired infections, and it becomes a serious issue in the world. Greater than 2 million people were infected and 139,378 peoples died with it exclusively until this proposal was developed.5
Different organizations such as WHO, CDC, and other professional document that practicing hand hygiene is the simplest and preferable technique in the prevention of the spread of the current serious issue of SARS-CoV-2 and other community-acquired and hospital-associated infections.3,6 Hand hygiene can be done by several methods; the most important method is frequent hand washing with soap and plain water or sanitizing hands with alcohol-based hand sanitizer (ABHS) in the absence of water and soap.7–9
The use of ABHS has been reported as the common recommended means of hand hygiene.10 The most regularly utilized ABHS should have an alcohol concentration of 60 to 85%.11 The effectiveness of these ABHS depends on the concentration of alcohol and the time of rubbing. Rubbing hands with ABHS (60 to 85% concentrations) for 25–30 seconds was reported to kill 99.99% of microorganisms on hand. For instance, rubbing sanitizers with 70% alcohol is suggested to kill 99.9% of the microorganisms on hands.12
Currently, in Ethiopia, different ABHS have been distributed throughout the community and the market, regarding the protection of community and health professionals suffering from a coronavirus disease 2019 (COVID-2019) outbreak. But still now, the efficacy of them was not evaluated even though, for effective protection, effective ABHS is mandatory. This is the reason why this research was designed to evaluate the antimicrobial efficacy of alcohol-based hand sanitizer that has been sold in southern parts of Ethiopia.
The finding of this study will be used for better understanding of status of ABHS that have been sold and distributed in the community. The result will be helpful for ministry of health, policy- makers, and health professionals, other stakeholders and community partners working on COVID-19 outbreak prevention strategy. It may also help as a baseline data for further related studies and early evaluating of alcohol-based hand sanitizer.
Methods and Materials
Test Organisms
Sanitizers containing an alcohol concentration between 60–85% are suggested to kill 99.99% of microorganisms on hands. The sanitizers which are effective on bacteria are effective in virus and vice versa. Therefore, we were forced to assess the efficacy of alcohol-based hand sanitizers only on selected bacteria, but not on virus (SARS-CoV-2) due to our laboratory set up constraint for viral isolation and growth. Following this assumption and reviewing related literature, Staphylococcus aureus, Escherichia coli, Pseudomonas aeruginosa, Klebsiella pneumoniae, Salmonella typhimurium and Shigella boydii were selected at the department of microbiology laboratory at Mizan Tepi University from different left over clinical specimen (Urine, stool and wound) which were collected with laboratory technologists for laboratory test at Mizan Tepi teaching hospital. The microorganisms were isolated from clinical specimens with culture, gram stain, and biochemical test. During the study, clinical specimens were inoculated on chocolate agar, blood agar, MacConkey agar, and Mannitol salt agar, and incubated overnight (35°C - 37°C) for colony isolation. Then on the second day gram stain test was examined to differentiate gram positive from negative bacteria for all colonies. At the 3rd day test organisms were isolated with biochemical tests using their biochemical characteristics after overnight incubation (35°C - 37°C). The isolated test organisms were stored on storage media and kept at 2–8° C. It were refreshed on nutrient agar and used when needed.
Hand Sanitizers
Seven locally made products of alcohol-based hand sanitizers were purchased from local vendors in Southwest, Ethiopia (Table). Sanitizers that have been sold on markets southern parts of Ethiopia were included and tear or not sealed expired and unlabeled (unknown manufacturer) hand sanitizers were excluded from the study (Table 1).
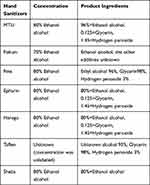 |
Table 1 Hand Sanitizers Used in the Study and Their Ingredients |
McFarland (0.5 Turbidity Standard) Preparation
A 0.5 McFarland turbidity standard was prepared for the standardization of selected test organisms that are needed for the determination of the minimum inhibitory and bactericidal concentration, for performing the agar diffusion test, and other tests necessary for the study such as biochemical tests. This 0.5 McFarland turbidity standard was prepared into a test tube from the mixture of barium chloride dehydrate (BaCl2. 2H2 0) solution and sulfuric acid (H2 SO4) with verification of the mixture absorbance (0.08–0.10) density precision via a spectrophotometer at a wavelength of 625nm. The prepared tube was stored in a sealed container at room temperature in a dark place.13
Test Organism Standardization
Each selected test organism was standardized before the research was performed by taking a loopful of inoculum using a sterile loop from a pure culture, transferring and suspending it into tubes of sterile normal saline. Then, the suspension density was adjusting by comparing it with the 0.5 McFarland turbidity standards by either adding additional bacteria or sterile saline.14
Agar Diffusion Test
To determine the susceptibility test of selected test organisms for each hand sanitizer well variant agar diffusion method was used.15 This agar diffusion method was done in duplicates for each hand sanitizer by inoculated sterile Mueller Hinton agar plates using sterile cotton swab which was immersed into a tube contain standardized test organisms. After Mueller Hinton agar was inoculated, it was allowed to remain at room temperature to dry for only some minutes and 8 equally spaced holes were bored in the agar plate with the 8th hole at the center of the plate with the aid of a sterile 6mm cork borer. The 7 holes were filled with 50µL of the hand sanitizer at the same time as the central hole (8th) was filled with an equal volume of sterile water for control purposes. The Mueller Hinton agar was incubated for 24 hours at 37° C. The zones of inhibition (susceptibility or resistance) of the hand sanitizers to each test organism were examined with the help of a ruler in millimeter by evaluating the average of 2 readings that were found from duplicates of agar diffusion test for each hand sanitizers.
Minimum Inhibitory Concentration (MIC) Determination
In this study MIC (the lowest concentration of a definite antimicrobial or hand sanitizers required to inhibit the growth of a known test organism in vitro16) was done on nutrient broth for each hand sanitizer against the selected test organisms. It was done by preparing various concentrations of each hand sanitizers (10%, 20%, 30%, 40%, 50%, 60%, 70%, and 80%). Then, one milliliter from the different concentrations (10%, 20%, 30%, 40%, 50%, 60%, 70%, and 80%) of each sanitizer was introduced into the tube containing equal volumes (1 mL) of nutrient broth inoculated with standardized test organism. A tube, containing only nutrient broth and bacteria without sanitizer and a tube containing just the sanitizer and broth without bacteria was used as negative control and positive control, respectively. Finally, the tubes were incubated for 18–24 hours and visible growth (turbidity) was assessed. When compared with the controls, the concentration of the sanitizers at which no visible growth was regarded as MIC.
Minimum Bactericidal Concentration (MBC) Determination
During this study MBC (the lowest concentration of a specific antimicrobial substance that can kill 99.9% of a given bacterial strain16) was determined from the MIC tests that showed no visible growth by taking a loopful of inoculum living test organisms from the MIC tubes by streaked on fresh Mueller Hinton agar. The streaked Mueller Hinton agar plates were incubated at 37°C for 24 hours and were observed for growth. Streaked Mueller Hinton agar plates that cannot show any growth indicates a 99.9% bactericidal effect of the sanitizer at that concentration or MBC.
Data Analysis
Data analysis was done by descriptive methods.
Quality Control
The reliability of the study findings was guaranteed by implementing strict quality control (QC) methods throughout the whole process of the laboratory work, such as pre-analytical (specimen collection from hospital laboratory room, transportation from hospital laboratory to testing laboratory, etc.), analytical (reagent preparation, sample inoculation, gram stain, bacterial isolation, biochemical test, MIC, MBC agar diffusion test, etc.) and post-analytical (reporting MIC, MBC result, etc.) quality control. During MBC and MIC only broth as a negative control to test the sterility of the media and equipment, and culture inoculated broth without antibiotics as a positive control to test the growth ability of the medium was also used.
All materials and equipment are controlled and Standard Operating Procedures (SOPs) were strictly followed.
Results
Hand Sanitizers’ Agar Diffusion Test
In our study, efforts were made to assess the efficiency of seven hand sanitizers (MTU, Folium Fine, Epharm, Harego, Taflen and Sheba) by using agar disc inhibitory activity against the selected test organisms (Staphylococcus aureus, Escherichia coli, klebsiella pneumoniae, Pseudomonas aeruginosa, Salmonella typhi and Shigella boydii). The zones of inhibition of the hand sanitizers at their claim concentration including the unknown (unlabeled) concentration Taflen products were ranged from no inhibition zone (Folium) to 27mm (Sheba). For instance, 9mm (Folium) to 17mm (MTU) on Staphylococcus aureus; 8mm (Folium) to 20mm (Fine) on Escherichia coli; 6mm (Folium) to 20mm (Fine) on K. pneumoniae; 6mm (Folium) to 27mm (Sheba) on Ps. aeruginosa; 8mm (Folium) to 15mm (Hargo) on Salmonella typhi; whereas on Shigella boydii it ranged from 8mm (Folium) to 17mm (Ephram) (Table 2).
 |
Table 2 The Susceptibility Pattern of the Selected Test Organisms to the Hand Sanitizers in the Agar Diffusion Test |
Minimum Inhibitory Concentration (MIC)
All hand sanitizers were further tested to determine their MIC and MBC values at 10%, 20%, 30%, 40%, 50%, 60%, 70% and 80% (at claim concentration). The minimum inhibitory concentration against all selected test organisms was observed at 45%, 55%, and 65% except Taflen which have had unknown concentration but it was bacteriostatic at undiluted form (Table 3).
 |
Table 3 Minimum Inhibitory Concentration (MIC) of Selected Test Organisms to MTU, Folium, Fine, Epharm, Harego, Taflen and Sheba Hand Sanitizers |
Minimum Bactericidal Concentration (MBC)
The content of the concentrations tubes of MIC was further plated out on sterile Mueller Hinton agar plates in order to determine the bactericidal effect of concentration. MTU (except against E. coli), Fine, Hargo, Ephra and Sheba hand sanitizers were showed growth on 10%, 20%, 30%, 40%, 50%, and 60% concentrations against the selected test organisms. The growth of selected test organisms was decreased in colony-forming unit across increasing the concentration gradient of different hand sanitizers (Table 4). But a Folium product was indicating that the product was only bacteriostatic against the selected test organisms at 65% but not bactericidal. Similarly, MTU product had a MIC value of 65% concentration against E. coli but was not bactericidal (Table 5).
 |
Table 4 Counting Colony Forming Unit of Selected Test Organisms at Increasing Concentration of Different Hand Sanitizers |
 |
Table 5 Minimum Bactericidal Concentration (MBC) of Selected Test Organisms to MTU, Folium, Fine, Epharm, Harego, Taflen and Sheba Hand Sanitizers |
Discussion
Now the use of hand sanitizers in the community has mostly gained recognition in the world, including Ethiopia since the emergence of COVID-19. This has led to the development, production and importation of several hand sanitizers by various companies with the intend of profit-making as well as supporting the health-care system and community in preventing transmission of disease especially COVID-19. But there is no information about laboratory-based efficacy evaluation.
In this study, the efficiency of the claim concentration of the seven hand sanitizers was assessed. The seven hand sanitizers MTU, Folium, Fine, Ephra, Hargo, and Sheba with claimed concentration of 80% ethanol alcohol, and Taflen with unknown (unlabeled) concentration of ethanol alcohol as the main active ingredient were displayed bacteriostatic activity against all the selected test organisms at a concentration of 45%, 55%, and 65%. Moreover, all Fine, Harego, Epharm and Sheba hand sanitizers showed bactericidal activity against all selected test organisms, with MBC values of 65% and 75%. Generally, the growth of selected test organisms was decreased in colony-forming unit across increasing the concentration gradient of different hand sanitizers. This bacteriostatic/bactericidal activity more probably due to alcohol components of alcohol-based hand sanitizer which is the major active ingredients intended to exert disinfectant activity in bacteria by causing protein denaturation, disruption of tissue membranes and dissolution of several lipids.17
Folium which contained 70% denatured ethanol was not active against all selected test organisms in bactericidal activity with no to narrow rim of inhibition zone in disk diffusion agar. The lack of bactericidal activity and more or less no disk inhibition zone observed in this product could be due to the relatively decrement of the concentration (70%) denatured alcohol compared with others since the efficacy of alcohol-based hand sanitizers is affected by several factors such as the type of alcohol used, concentration of alcohol or amount of alcohol used, the possible contact time18 and absence of active ingredient in product (hydrogen peroxide) which may limit the cidal effect of the alcohol from attainment the bacterial cells.
MTU product was the other hand sanitizer product which was not active against E. coli test organisms in our study. This could be poor or prolonged storage of the products which could lead to increased temperature causing evaporation of the active ingredient and characteristic of the E. coli strain since the strain is bacteria, which can easily develop resistance.
Limitation
There are limitations in our study; we could assess the efficacy of only seven locally made but not imported sanitizers. Only bacterial strain was used because of laboratory setup constraint for virus strain growth.
Conclusion
We evaluated the antibacterial efficacy of the seven most popular brands of hand sanitizers sold in Mizan-Aman town. Of the seven the six sanitizers except MTU product for E. coli and Folium were the products that were effective in inhibited growth of all the selected test organisms in vitro, and they were had effective bactericidal activity on their claim concentration. But MTU products against E. coli and Folium against all selected test organisms were not had effective bactericidal activity.
Recommendation
Even though alcohol-based hand sanitizers are easy to prepare, they must be used after proper testing of their efficacy at their producer claimed concentration to protect consumers from buying poor quality products. The company should have to follow strict standard operational procedures to produce standardized products. The regulatory authorities should enforce strict quality control measures during production, distribution and routine laboratory-based inspections to ensure the efficacy of products. Lastly, consumers should be aware of the availability of poor quality sanitizers on the shelves of some retail outlets.
Abbreviations
CDC, the Center for Disease Control and Prevention; WHO, World Health Organization; MTU, Mizan-Tepi University; MIC, minimum inhibitory concentration; MBC, minimum bactericidal concentration.
Data Sharing Statement
The data used to support the findings of this study are available from the corresponding author upon request.
Ethics Approval and Consent to Participate
We conduct our study in compliance with recognized international standards and the principles of the Declaration of Helsinki. Ethical clearance was acquired from the Research and Ethical Review Committee of College of Medicine and Health Sciences, at Mizan Tepi University. The permission letter was taken from the clinical director of the Mizan-Tepi University referral hospital and laboratory head. Data were collected after full written consent had been obtained from each participant. For the purpose of the study, codes were used instead of any personal identifiers which were labeled by laboratory technologist on the specimen tubes during specimen collection. Specimens with abnormal test results were informed to physicians for further diagnosis and treatment accordingly.
Acknowledgments
We would like to thank the College of Medicine and Health Science and Mizan-Tepi University for giving us this chance to conduct this research. Moreover, we would like to express our appreciation to Mizan-Tepi Teaching Hospital for willingness to give leftover patient specimens and the staff of the outpatient department (OPD) unit for their support during data collection.
Author Contributions
All authors made substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data; took part in drafting the article or revising it critically for important intellectual content; agreed to submit to the current journal; gave final approval of the version to be published; and agree to be accountable for all aspects of the work.
Funding
Mizan-Tepi University.
Disclosure
The authors declare that they do not have any conflict of interests.
References
1. Hassan AO, Hassan RO, Muhibi MA, Adebimpe WO. A survey of Enterobacteriaceae in hospital and community acquired infections among adults in a tertiary health institution in Southwestern Nigeria. Afr J Microbiol Res. 2012;6(24):5162–5167.
2. Jain A, Singh K. Recent advances in the management of nosocomial infections. JK Sci. 2007;9(1):3–8.
3. World Health Organisation. WHO guidelines in hand hygiene in health care. WHO/IER/PSP/2009/01. Geneva, Switzerland: World Health Org anisation; 2009
4. Zerr DM, Garrison MM, Allpress AL, Heath J, Christakis DA. Infection control policies and hospital-associated infections among surgical patients: variability and associations in a multicenter pediatric setting. Pediatrics. 2005;115(4):387–392. doi:10.1542/peds.2004-2014
5. World Health Organization. 2020. Coronavirus disease 2019 (COVID-19): situation report 88. World Health Organization. Available from: https://apps.who.int/iris/handle/10665/331851.
6. Boyce JM, Pitted D. Guideline for hand hygiene in health-care settings: recommendations of the Health care Infection Control Practices Advisory Committee and the HICPAC/SHEA/APIC/IDSA Hand Hygiene Task Force. Am J Infect Control. 2002;30(8):S1–46.
7. Verma DK, Tesfu K, Getachew M, Workineh Y, Mekuriaw F, Tilahun M. Evaluation of antibacterial efficacy of different hand gel sanitizers in University of Gondar Students, North–West Ethiopia. J Glob Biosci. 2013;2(6):166–173.
8. Zaragoza M, Sallés M, Gomez J, Bayas JM, Trilla A. Hand washing with soap or alcoholic solutions? A randomized clinical trial of its effectiveness. Am J Infect Control. 1999;27(3):258–261. doi:10.1053/ic.1999.v27.a97622
9. Kramer A, Rudolph P, Kampf G, Pittet D. Limited efficacy of alcohol-based hand gels. Lancet. 2002;359(9316):1489–1490. doi:10.1016/S0140-6736(02)08426-X
10. Wolfe MK, Gallandat K, Daniels K, Desmarais AM, Scheinman P, Lantagne D. Handwashing and Ebola virus disease outbreaks: a randomized comparison of soap, hand sanitizer, and 0.05% chlorine solutions on the inactivation and removal of model organisms Phi6 and E. coli from hands and persistence in rinse water. PLoS One. 2017;12(2):e0172734. doi:10.1371/journal.pone.0172734
11. Kramer A, Rudolph P, Kampf G, Pittet D. Showing classic G to A mutation at nucleotide 1896. Lancet. 2002;359:1489–1490.
12. Rotter M. Hand washing and disinfection. In: Mayhall CG, editor. Hospital Epidemiology and Infection Control.
13. Cheesbrough M. District Laboratory Practice in Tropical Countries, Part 2. Cambridge university press; March 2, 2006.
14. Vandepitte J, Engbaek K, Rohner P, Piot P, Heuck CC; World Health Organization. Basic laboratory procedures in clinical bacteriology/ J. Vandepitte … [et al.]. In: Basic Laboratory Procedures in Clinical Bacteriology/ J. Vandepitte … [et al.]; 2003
15. Valgas C, Souza SM, Smânia EF, Smânia JA. Screening methods to determine antibacterial activity of natural products. Braz J Microbiol. 2007;38(2):369–380. doi:10.1590/S1517-83822007000200034
16. Nester EW, Anderson DG, Roberts CE, Nester MT. Microbiology: A Human Perspective, 6th Edition. 2009.
17. Kar A. Pharmaceutical Microbiology. Delhi: New Age International (P) Ltd Publishers; 2008:216–217.
18. Harmanci H. Guideline for Hand Hygiene in Health-Care Setting Recommendations of the Healthcare Infection Control Practices Advisory Committee and the HICPAC/SHEA/APIC/IDSA Hand Hygiene Task Force. Marmara Med J. 2016;15(4):281–286.
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
