Back to Journals » Drug Design, Development and Therapy » Volume 12
Evaluation of the effects of food on levodropropizine controlled-release tablet and its pharmacokinetic profile in comparison to that of immediate-release tablet
Authors Lee S , Nam KY, Oh J , Lee S , Cho SM, Choi YW, Cho JY , Lee BJ , Hong JH
Received 21 July 2017
Accepted for publication 27 January 2018
Published 23 May 2018 Volume 2018:12 Pages 1413—1420
DOI https://doi.org/10.2147/DDDT.S146958
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Frank Boeckler
Soyoung Lee,1,* Kyu-Yeol Nam,2,3,* Jaeseong Oh,1 SeungHwan Lee,1 Sang-Min Cho,3 Youn-Woong Choi,3 Joo-Youn Cho,1 Beom-Jin Lee,2,4,* Jang Hee Hong5,*
1Department of Clinical Pharmacology and Therapeutics, Seoul National University College of Medicine and Hospital, Seoul, Republic of Korea; 2College of Pharmacy, Ajou University, Suwon, Republic of Korea; 3Korea United Pharm Inc., Seoul, Republic of Korea; 4Institute of Pharmaceutical Science and Technology, Ajou University, Suwon, Republic of Korea; 5Department of Pharmacology, Chungnam National University Hospital and College of Medicine, Daejeon, Republic of Korea
*These authors contributed equally to this work
Background: Levodropropizine is a non-opioid antitussive agent that inhibits cough reflex by reducing the release of sensory peptide in the peripheral region. To improve patients’ compliance, a controlled-release (CR) tablet is under development. The aim of this study was to compare the pharmacokinetic (PK) profiles of the CR and immediate-release (IR) tablets of levodropropizine. In addition, the effect of food on the PK properties of levodropropizine CR tablet in healthy subjects was evaluated.
Subjects and methods: A randomized, open-label, multiple-dose, three-treatment, three-period, six-sequence, crossover study was conducted on 47 healthy subjects. All subjects were randomly assigned to one of the six sequences, which involve combinations of the following three treatments: levodropropizine IR 60 mg three times in the fasted state (R), levodropropizine CR 90 mg two times in the fasted state (T), and levodropropizine CR 90 mg two times in the fed state (TF). Serial blood samples were collected up to 24 h after the first dose. Tolerability was assessed based on the vital signs, adverse events (AEs), and clinical laboratory tests.
Results: Levodropropizine CR showed lower maximum drug concentration (Cmax) and similar total exposure compared to levodropropizine IR. The geometric mean ratios (GMRs) (90% confidence intervals [CIs]) of T to R for the Cmax and area under the concentration–time curve from the 0 to 24 h time points (AUC0–24h) were 0.80 (0.75–0.85) and 0.89 (0.86–0.93), respectively. In the fed group, levodropropizine CR showed exposure similar to that in the fasted group. The GMRs (90% CIs) of TF to T for the Cmax and AUC0–24h were 0.90 (0.85–0.97) and 1.10 (1.05–1.14), respectively. No serious AEs occurred with both levodropropizine CR and IR tablets.
Conclusion: Total systemic exposure for levodropropizine was similar in subjects receiving the CR and IR formulations in terms of the AUC. Although food delayed the absorption of levodropropizine CR, systemic exposure was not affected.
Keywords: pharmacokinetics, controlled-release, immediate-release, food effect
Introduction
Cough is a respiratory defense mechanism to protect our body from external materials, such as dust and pathogens. However, persistent and uncontrollable cough can affect the patients’ quality of life by inducing nausea and sleep disturbance.1 To relieve cough, levodropropizine is one of the therapeutic options used to treat acute and chronic bronchitis.2 Levodropropizine is a peripherally acting, non-opioid antitussive agent, which inhibits the cough reflex by reducing the release of sensory peptide in the peripheral region and suppresses the afferent pathway of pulmonary.3–5 It has comparable efficacy and less central sedative adverse effects compared to the centrally acting antitussives.1 Levodropropizine has been approved in some Asian and European countries and has been widely used for adults and children older than 2 years in Republic of Korea.
Levodropropizine is rapidly absorbed into the gastrointestinal tract, with time required to reach the maximum drug concentration (Tmax) ranging from 0.25 to 0.75 h.6 This absorption profile is due to high bioavailability (>75%) and rapid distribution of levodropropizine.7 The mean terminal half-life (t1/2) of levodropropizine is 2.73 h, which suggests rapid elimination after systemic absorption. Levodropropizine has linear pharmacokinetic (PK) characteristics at doses ranging from 30 to 90 mg.8 The effects of food on the PK characteristics of levodropropizine have not been evaluated yet.
The food intake can change the systemic exposure of drug by altering the absorption and/or elimination of drug. For instance, food can alter either the gastric emptying time or pH conditions of the gastrointestinal tract, which can affect the absorption kinetics of the drug. The dietary intake can also change the elimination profile of the drug by increasing the blood flow and by stimulating the bile production.9,10 Especially in the case of a sustained release drug, it is necessary to evaluate the interaction between food and drug, since the patients are more likely to consume food during the dosing interval.
The currently available dosage forms of levodropropizine worldwide are syrup, capsule, and tablet. The levodropropizine immediate-release (IR) tablet 60 mg is prescribed three times daily (a total daily dose of 180 mg) for adults. To improve the dosing convenience and patients’ compliance, levodropropizine controlled-release (CR) tablet is under development by Korea United Pharm Inc. (Seoul, Republic of Korea).11–13
The aim of this study is to compare the PK profiles of CR and IR formulations of levodropropizine in the fasted state. In addition, the effects of food on the PK of levodropropizine CR formulation in healthy male subjects are evaluated.
Subjects and methods
Subjects
Korean male subjects aged 20–55 years, who were evaluated as healthy based on medical history, 12-lead electrocardiography, vital signs, physical examination, and clinical laboratory tests, were enrolled in this study. Subjects were determined healthy if they had none of the following medical histories: known hypersensitivity to levodropropizine or other antitussive agent and a history of cardiovascular, respiratory, renal, metabolic, hematologic, gastrointestinal, neurologic, psychiatric, oncologic, or hepatic disease. All subjects provided a written informed consent before participating in the study. This clinical study (Clinical Research Information Service registration number: KCT0002383) was approved by the institutional review board (IRB) of Chungnam National University Hospital (IRB number: CNUH 2011-12-019) and was conducted in accordance with the Declaration of Helsinki as implemented in the Good Clinical Practice guidelines.
Study design
A randomized, open-label, multiple-dose, three-treatment, three-period, six-sequence Williams design crossover study was conducted. The randomization was performed using the PROC PLAN procedure of SAS® software. Subjects were randomly assigned to one of the six treatment sequences, which involve combinations of the following three treatments: levodropropizine IR 60 mg three times daily with 6-h interval in the fasted state, levodropropizine CR 90 mg two times daily with 12-h interval in the fasted state, and levodropropizine CR 90 mg two times daily with 12-h interval in the fed state (Figure 1). The total daily dose of levodropropizine was 180 mg in all treatment groups. For the fasted groups, at least 10 h of fasted state were required prior to drug administration. For the fed group, subjects received high-fat meal (946 kcal; fat content, 35.5%) prior to drug administration. One-week washout period was set between the different treatment periods based on the t1/2 of levodropropizine. For PK analysis of levodropropizine IR, blood samples were obtained at 0, 0.25, 0.5, 0.75, 1, 2, 4, 6, 6.25, 6.5, 6.75, 7, 8, 10, 12, 12.25, 12.5, 12.75, 13, 14, 16, 18, and 24 h post-dose. For levodropropizine CR, serial blood samples were collected at 0, 0.25, 0.5, 0.75, 1, 2, 4, 6, 8, 12, 12.25, 12.5, 12.75, 13, 14, 16, 18, and 24 h post-dose.
Determination of levodropropizine plasma concentrations
The blood samples were centrifuged at 3,000 rpm for 10 min at 4°C, and the plasma samples were stored at −70°C until assay. Plasma concentrations of levodropropizine were determined by using high-performance liquid chromatography (1260 series; Agilent Technologies, Santa Clara, CA, USA) coupled with a mass spectrometer (6410A Triple quad mass spectrometer; Agilent Technologies). The blood samples were treated with acetonitrile for precipitation of proteins, and terazosin was used as an internal standard (IS) for quantitation of levodropropizine. Levodropropizine was separated on an Eclipse XDB-C18 column (3.0 × 100 mm, 3.5 μm; Agilent Technologies) and detected using the positive electrospray ionization and multiple reaction monitoring modes. The mobile phase consisted of 0.2% formic acid in 5 mM ammonium formate and acetonitrile (40:60, v/v), and the flow rate was 0.35 mL/min. The lower limit of quantification (LLOQ) was 5 ng/mL, and the calibration curve for levodropropizine was linear over the range of 5–1,000 ng/mL with a coefficient of determination of ≥0.99. The intraday and interday precision were ≤3.6 and ≤6.7%, respectively. The intraday and interday accuracy ranged from 95.3 to 103.5% and 98.2 to 99.2%, respectively.
PKs and statistical analysis
The PK parameters were calculated through a non-compartmental analysis using Phoenix® WinNonlin® software version 1.4 (Certara, St Louis, MO, USA). To compare the total exposure to levodropropizine CR and IR, the maximum plasma concentration (Cmax) and area under the concentration–time curve from the 0 to 24 h time points (AUC0–24h) were calculated. In addition, to determine the PK parameters of levodropropizine IR, the 0–6 h data were used because they represent the data set after the first dose. Similarly, to determine the PK parameters of levodropropizine CR, the 0–12 h data were used. The following PK parameters were calculated for both levodropropizine IR and CR: Cmax, Tmax, AUC from 0 to dose interval time (AUC0–tau), AUC from 0 to infinity (AUCinf), t1/2, and apparent clearance (CL/F). The AUC was calculated using the linear trapezoidal–linear interpolation method. The Tmax and Cmax were determined directly based on the observed values. The values below the LLOQ prior to the Tmax were regarded as 0, and the values after the Tmax were regarded as missing data. No imputations were made for the missing data when calculating PK parameters.
Statistical analyses were performed using SAS® software version 6.4 (SAS Institute Inc., Cary, NC, USA). A mixed-effects analysis of variance (ANOVA) model was fit by using the natural logarithm-transformed Cmax and AUC0–24h. Based on the ANOVA model, PK comparisons between different formulations were assessed by calculating the geometric mean ratio (GMR) and 90% confidence interval (90% CI) of levodropropizine CR to IR for the Cmax and AUC0–24h. In addition, to evaluate the food effect on the PK of levodropropizine CR, the GMR and 90% CI of the fed group to the fasted group for the Cmax and AUC0–24h were calculated. Furthermore, the GMR and 90% CI of AUC0–tau/D and AUCinf/D were calculated. The ANOVA test was used to determine the statistical difference in demographic baseline between six sequences.
Tolerability assessment
Tolerability was evaluated in all subjects who received at least one dose of the study drug throughout the entire study period. Tolerability was evaluated based on the assessment of the vital signs (blood pressure, heart rate, and body temperature), physical examination, 12-lead electrocardiogram, clinical laboratory tests (hematology, blood chemistry, and urinalysis), and adverse events (AEs).
Results
Demographics
Forty-eight subjects were enrolled in this study, and one subject dropped out because of consent withdrawal. The remaining 47 subjects completed the study as planned and were included in the PK analysis set. The mean ± standard deviation values of the demographic characteristics of the enrolled subjects were as follows: age, 24.44 ± 1.63 years; height, 175.98 ± 6.60 cm; and body weight, 64.94 ± 8.54 kg. There were no statistically significant differences in demographic characteristics between six-sequence groups (Table S1).
PK results
PK characteristics of levodropropizine CR formulation
The AUC0–24h, which represents the total daily exposure, was similar for both formulations (Table 1). The GMR and 90% CI for the AUC0–24h of levodropropizine CR to IR were within the conventional bioequivalence range. Levodropropizine CR showed delayed Tmax, prolonged t1/2, and lower Cmax compared to that of levodropropizine IR (Figure 2, Tables 1 and 2). The median Tmax values were 1.00 and 0.75 h, the mean t1/2 values were 3.10 and 2.30 h, and the mean Cmax values during 24 h were 261.22 and 325.46 μg/L for levodropropizine CR and IR, respectively. The GMRs and 90% CIs for the Cmax and dose-normalized AUC0–tau of levodropropizine CR to IR were within the conventional bioequivalence range after the first single dose administration (Table 2). Individual comparisons of the Cmax and AUC0–24h between levodropropizine CR and IR are shown in Figure 3.
Food effect on levodropropizine CR
The total exposure and peak concentration of levodropropizine CR were similar in both the fasted and fed groups. The GMRs and 90% CIs for the Cmax and AUC0–24h of levodropropizine CR fed to the fasted groups were within the conventional bioequivalence range (Table 1). In addition, the GMRs and 90% CIs for the Cmax and dose-normalized AUC0–tau of levodropropizine CR fed to the fasted groups were within the conventional bioequivalence acceptance range (Table 2). The fed group showed delayed absorption compared to the fasted group, and the median Tmax values were 3.00 and 1.00 h, respectively (Figure 4 and Table 2). Individual comparisons of the Cmax and AUC0–24h are shown in Figure 5.
Tolerability assessment
Sixteen AEs occurred in 14 subjects. Of these, five AEs were observed in the levodropropizine IR 60 mg group, and 11 in the levodropropizine CR 90 mg group. All AEs were mild in severity, except for one subject who experienced urticaria after test drug. The AEs that occurred in the levodropropizine CR 90 mg group were the foreign body, dizziness, headache, presyncope, and abrasion. In the levodropropizine IR 60 mg group, diarrhea, rash pustular, pain in extremity, dizziness, headache, pyuria, contusion, dermatitis atopic, pruritus, and urticaria occurred as AEs. Of these, dizziness, headache, and diarrhea were reported as frequent AEs of levodropropizine on the drug label. No serious AEs occurred in either levodropropizine CR or IR administrated group. In addition, there were no clinically significant changes from the baseline of vital signs, physical examinations, electrocardiogram, and clinical laboratory tests in all subjects.
Discussion
In this study, we aimed to compare the total daily exposure to levodropropizine CR and IR formulations at equal total daily dose of levodropropizine. Therefore, we measured the Cmax and AUC0–24h as primary PK parameters to compare the PK characteristics of the two formulations. In addition, we calculated the single-dose PK parameters to compare the absorption and elimination profiles of the different formulations.
This study showed that at equal total daily dose, the total daily systemic exposure to levodropropizine was comparable between the CR and IR formulations. The GMR (90% CI) of AUC0–24h was 0.89 (0.86–0.93) although the GMR (90% CI) of Cmax was 0.80 (0.75–0.85). The PK differences between the 90 mg CR and 60 mg IR tablets might not be clinically significant if the efficacy of levodropropizine is dependent on the total daily systemic exposure. Based on the results of a previous study, the onset of cough inhibitory action of levodropropizine was 1 h post-dose.14 Therefore, a minimal delay in Tmax with the CR formulation would be clinically insignificant regarding the onset time of levodropropizine action. Further clinical study will be needed to evaluate the efficacy and safety of the CR formulation compared to the IR formulation.
The US Food and Drug Administration guidance recommends conducting a food-effect study to evaluate the bioavailability of modified-release formulations.9 This is because food can affect the PK of drug in various ways. In this study, food delayed the Tmax of levodropropizine CR formulation. This could be attributed to alteration of the gastric pH because of food intake. For instance, weakly basic drugs, such as levodropropizine (pKa1=7.12 and pKa2=10.58), generally exhibit reduced dissolution at high pH.7,15,16 Because the gastric pH can easily increase by food intake, this can result in decreased absorption rate of weakly basic drugs. In addition, food delays the gastric emptying time, which leads to delayed oral absorption of drugs.17 Moreover, food can affect the action of hydroxypropyl methylcellulose (HPMC), which constitutes the matrix that modulates the drug release. For example, HPMC release was more rapid when the pH condition was similar to the fasted state of the stomach. However, when the pH increased from 1.20 to 6.80, it showed slow dissolution. Since the pH of the stomach after food intake is ~4.90, HPMC would dissolve faster in the fasted state compared to that in the fed state.18 These mechanisms might contribute to the delay in Tmax of levodropropizine CR when it was administered in the fed state. However, the GMRs and 90% CIs of the Cmax and AUC0–24h were within the conventional bioequivalence range; thus, the effect of food on the bioavailability of levodropropizine CR would be minimal.
Conclusion
The total systemic exposure to levodropropizine was comparable between the CR and IR formulations in terms of the AUC. Although food delayed the absorption of levodropropizine CR, systemic exposure was not affected. Both levodropropizine CR and IR formulations were well tolerated in healthy subjects.
Acknowledgments
This research was supported by a grant of the Korea Health Technology R&D Project through the Korea Health Industry Development Institute (KHIDI), funded by the Ministry of Health & Welfare, Republic of Korea (grant numbers: HI14C1063 and HI14C1069). This work was supported by research fund of Chungnam National University.
Disclosure
Kyu-Yeol Nam, Sang-Min Cho and Youn-Woong Choi are employees of Korea United Pharm Inc., Seoul, Republic of Korea. The authors report no other conflicts of interest in this work.
References
Dicpinigaitis PV, Morice AH, Birring SS, et al. Antitussive drugs – past, present, and future. Pharmacol Rev. 2014;66(2):468–512. | ||
Bolser DC. Cough suppressant and pharmacologic protussive therapy: ACCP evidence-based clinical practice guidelines. Chest. 2006;129(1 suppl):238S–249S. | ||
Daffonchio L, Clavenna G, Fedele G, et al. Levodropropizine. Drugs Today. 1995;31:299–305. | ||
Shams H, Daffonchio L, Scheid P. Effects of levodropropizine on vagal afferent C-fibres in the cat. Br J Pharmacol. 1996;117:853–858. | ||
Lavezzo A, Melillo G, Clavenna G, Omini C. Peripheral site of action of levodropropizine in experimentally-induced cough: role of sensory neuropeptides. Pulm Pharmacol. 1992;5:143–147. | ||
Jeon S, Lee J, Hong T, et al. Pharmacokinetics and safety of levodropropizine controlled-release tablet after repeated dosing in healthy male volunteers. J Korean Soc Clin Pharmacol Ther. 2013;21:113–119. | ||
Narin I, Sarioglan S, Anilanmert B, et al. pKa determinations for montelukast sodium and levodropropizine. J Solution Chem. 2010;39:1582–1588. | ||
Borsa M, Glavenna G, Ferrari MP, Canali S, Giachetti C, Zanolo G. Linearity of levodropropizine, a new antitussive drug, in the healthy volunteer. Eur J Drug Metab Pharmacokinet. 1991;3:178–184. | ||
U.S. Department of Health and Human Services. Food and Drug Administration. Guidance for Industry: Food-Effect Bioavailability and Fed Bioequivalence Studies. FDA, Center for Drug Evaluation and Research (CDER); 2002. Available from: https://www.fda.gov/downloads/Drugs/GuidanceComplianceRegulatoryInformation/Guidances/UCM070241.pdf. Accessed March 19, 2018. | ||
Won CS, Oberlies NH, Paine MF. Mechanisms underlying food–drug interactions: inhibition of intestinal metabolism and transport. Pharmacol Ther. 2012;136:186–201. | ||
Park J-H, Shin Y, Lee S-Y, Lee SI. Antihypertensive drug medication adherence and its affecting factors in South Korea. Int J Cardiol. 2008;128:392–398. | ||
Hong JS, Kang HC. Relationship between oral antihyperglycemic medication adherence and hospitalization, mortality, and healthcare costs in adult ambulatory care patients with type 2 diabetes in South Korea. Med Care. 2011;49:378–384. | ||
Coleman CI, Limone B, Sobieraj DM, et al. Dosing frequency and medication adherence in chronic disease. J Manag Care Pharm. 2012;18:527–539. | ||
Fumagalli G, Cordaro CI, Vanasia M, et al. A comparative study of the antitussive activity of levodropropizine and dropropizine in the citric acid-induced cough model in normal subjects. Drugs Exp Clin Res. 1992;18:303–309. | ||
Batchelor HK. Influence of food on pediatric gastrointestinal drug absorption following oral administration: a review. Children. 2015;2:244–271. | ||
European Pharmacopoeia Commission, Medicines EDftQo, Healthcare. European Pharmacopoeia 5.0. Vol. 1. Renouf Publishing Company Limited; Council of Europe, Strasbourg, France: 2005. | ||
Welling PG. Influence of food and diet on gastrointestinal drug absorption: a review. J Pharmacokinet Biopharm. 1977;5:291–334. | ||
Guiastrennec B, Söderlind E, Richardson S, Peric A, Bergstrand M. In vitro and in vivo modeling of hydroxypropyl methylcellulose (HPMC) matrix tablet erosion under fasting and postprandial status. Pharm Res. 2017;34:847–859. |
Supplementary material
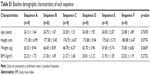  | Table S1 Baseline demographic characteristics of each sequence |
 © 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.







