Back to Journals » Neuropsychiatric Disease and Treatment » Volume 11
Evaluation of acute cardiovascular effects of immediate-release methylphenidate in children and adolescents with attention-deficit hyperactivity disorder
Authors Lamberti M, Italiano D, Guerriero L, D'Amico G, Siracusano R, Ingrassia M, Germanò E, Calabrò MP, Spina E, Gagliano A
Received 25 December 2014
Accepted for publication 28 January 2015
Published 8 May 2015 Volume 2015:11 Pages 1169—1174
DOI https://doi.org/10.2147/NDT.S79866
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 4
Editor who approved publication: Dr Roger Pinder
Marco Lamberti,1,2 Domenico Italiano,2 Laura Guerriero,1 Gessica D’Amico,3 Rosamaria Siracusano,1,4 Massimo Ingrassia,5 Eva Germanò,1 Maria Pia Calabrò,3 Edoardo Spina,2 Antonella Gagliano1
1Division of Child Neurology and Psychiatry, Department of Pediatrics, 2Department of Clinical and Experimental Medicine, 3Division of Pediatric Cardiology, Department of Pediatrics, University of Messina, Messina, Italy; 4Institution of Clinical Physiology, CNR, Pisa, 5Division of Psychology, Department of Humanities and Social Sciences, University of Messina, Messina, Italy
Abstract: Attention-deficit hyperactivity disorder is a frequent condition in children and often extends into adulthood. Use of immediate-release methylphenidate (MPH) has raised concerns about potential cardiovascular adverse effects within a few hours after administration. This study was carried out to investigate acute effects of MPH on electrocardiogram (ECG) in a pediatric population. A total of 54 consecutive patients with attention-deficit hyperactivity disorder (51 males and 3 females; mean age =12.14±2.6 years, range 6–19 years), receiving a new prescription of MPH, underwent a standard ECG 2 hours before and after the administration of MPH 10 mg per os. Basal and posttreatment ECG parameters, including mean QT (QT interval when corrected for heart rate [QTc]), QTc dispersion (QTd) interval duration, T-peak to T-end (TpTe) intervals, and TpTe/QT ratio were compared. Significant modifications of both QTc and QTd values were not found after drug administration. QTd fluctuated slightly from 25.7±9.3 milliseconds to 25.1±8.4 milliseconds; QTc varied from 407.6±12.4 milliseconds to 409.8±12.7 milliseconds. A significant variation in blood pressure (systolic blood pressure 105.4±10.3 vs 109.6±11.5; P<0.05; diastolic blood pressure 59.2±7.1 vs 63.1±7.9; P<0.05) was observed, but all the data were within normal range. Heart rate moved from 80.5±15.5 bpm to 87.7±18.8 bpm. No change in TpTe values was found, but a statistically significant increase in TpTe/QTc intervals was found with respect to basal values (0.207±0.02 milliseconds vs 0.214±0.02 milliseconds; P<0.01). The findings of this study show no significant changes in ECG parameters. TpTe values can be an additional parameter to evaluate borderline cases.
Keywords: ADHD, methylphenidate, cardiovascular effects, children, adolescents
Introduction
Attention-deficit hyperactivity disorder (ADHD) is a common behavioral disorder characterized by symptoms of inattention, impulsivity, and hyperactivity that impact several domains of the life of patients, their family, and society.1,2 Patients with untreated ADHD are associated with poorer long-term self-esteem and social functioning outcomes than no-ADHD controls.3,4
Treatments and interventions for ADHD are different and include psychological therapies and pharmacological treatment, especially for severe clinical cases. Stimulants, such as amphetamines and methylphenidate (MPH), represent the mainstay of pharmacological treatment for ADHD.5 Amphetamines act by increasing presynaptic release of dopamine and other biogenic amines in the brain while MPH, a substituted phenethylamine, inhibits the reuptake of dopamine and norepinephrine and therefore its pharmacology is identical to that of amphetamines.6
There is strong evidence that MPH is effective in treating ADHD, but recently, concerns have increasingly been raised about the adverse effects of this drug. In the last few years, several studies about the cardiovascular safety of stimulant medications were carried out.7–12 Stimulants and MPH increase blood pressure (BP) and heart rate (HR) in patients with ADHD,13–17 although these cardiovascular effects do not seem to be severe.18–20 Nevertheless, most evidence is based on clinical reports from small populations, so it remains controversial whether the use of stimulants is potentially associated with serious adverse cardiovascular events or otherwise. A recent nationwide prospective cohort study suggests that cardiovascular events are rare in children and adolescents but twice as likely in stimulant users as in nonusers with a complex, time- and dose-dependent relationship between cardiovascular adverse events and stimulant treatment.21
Stimulant drugs can be proarrhythmogenic because they are closely related to sympathomimetic amines. The autonomic nervous system plays an important role in sudden cardiac death (SCD). However, it is hard to establish a strict connection between drugs and sudden cardiac arrest in children and adolescents because SCD is a very rare event in pediatric populations. It is likely that significant changes of HR variability reflect diminished vagal tone and represent one of the key factors leading to SCD in adults.7 A pilot study by Buchhorn et al reports an improvement of HR variability in children under stimulant medication.16 The authors assume that this effect may be an important predictor of cardiovascular risk.
Drug-induced SCD is most commonly caused by torsades de pointes, a potentially life-threatening, polymorphic, ventricular tachycardia often associated with a QT interval prolongation. In clinical practice, several drugs are able to induce QT/QT interval when corrected for HR (QTc), prolongation and torsades de pointes.22 QT interval monitoring (QTc) is widely used as a predictor of fatal arrhythmias.23 The QTc dispersion (QTd), a measure of the interlead variation in the QT, is believed to be a more reliable predictor for cardiovascular mortality in the general population. A QTc interval greater than 500 milliseconds, or a QTd greater than 100 milliseconds, is considered a risk factor for fatal arrhythmias.24 More recent studies indicate that prolongation of the interval between the peak and the end of the T wave (T-peak to T-end [TpTe]) on the 12-lead electrocardiogram (ECG) can represent a new marker of ventricular arrhythmogenesis. In fact, prolongation of the TpTe interval, measured in lead V5, is considered independently associated with SCD, and it can be a suitable risk indicator even when the QTc is within range or not measurable due to prolonged QRS duration.25,26
To date, the studies investigating the cardiovascular risk of immediate-release methylphenidate (IR-MPH) have exclusively considered the QTc and QTd variations.27 TpTe and TpTe/QT intervals, along with QTc and QTd modifications have not been investigated in the context of ADHD and drug-related cardiovascular adverse effects. We therefore performed an observational prospective study aimed at examining the modifying effect of IR-MPH on the cardiovascular system in pediatric patients diagnosed with ADHD, in particular by measuring the acute effects of IR-MPH on the ventricular repolarization through TpTe and TpTe/QT intervals, along with QTc and QTd.
Experimental procedures
Study population
Children and adolescent patients (mean age =12.14±2.6 years, range 6–19 years) were enrolled into this observational, prospective study to assess the arrhythmic risk of IR-MPH. Patients were considered eligible for the study if they were drug-naïve ADHD outpatients, had ADHD diagnosed according to Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition (DSM-5) criteria,28 were attending the Unit of Child Neurology and Psychiatry of the University Policlinic of Messina between September 2013 and March 2014, and were new users of IR-MPH. None of the included patients had a history and signs or symptoms of cardiovascular, pulmonary, or endocrine disorders. The study was approved by the local ethics committee. Subjects and parents received detailed information about characteristics, potential adverse effects, and different treatment options for ADHD. All parents gave their written informed consent, and all the patients, when able, gave their consent as well.
Study design
In all enrolled patients, IR-MPH was started at an initial dosage of 5 mg/day and progressively titrated over 4 weeks until the therapeutic dose was achieved. The low and medium doses were 10 mg and 30 mg, respectively, for each child according to his/her weight. The high dose was 60 mg reached by only three adolescents. All patients in the study had body weight higher than 17 kg, and therefore, the range of dose administered was between 0.68 mg/kg body weight and 0.87 mg/kg body weight. Each treatment condition was administered 7 days, twice or three times daily, at breakfast (approximately 7.30 am), at lunch (approximately 12.30 pm), and in some cases, early afternoon (approximately 3.30 pm).
For each patient, two standard 12-lead ECGs were obtained at a paper speed of 25 mm/second with the same instrument (Cardioline delta 3 plus), on the same day and under similar conditions. The first (predose) ECG examination was performed before the administration of the first daily dose of IR-MPH; the second (postdose) ECG was executed 2 hours after drug intake, simultaneously with the serum peak of MPH.29 Predose ECG was executed at least 16 hours after the administration of the last MPH dose on the previous day; therefore, MPH serum concentrations were minimal at the time of the examination. The duration of QT intervals was measured manually, by an experienced cardiologist, in all leads in which the onset of the QRS complex and the return of T wave to baseline were clearly identified. The QT duration was corrected for HR according to the Bazett’s formula to produce QTc and averaged for all assessed leads (mean QTc). If respiratory sinus arrhythmia was present, the QT interval was measured in all leads where RR intervals were almost equal. TpTe interval was measured from the peak of the T wave to the end of T wave in lead V5.26 The TpTe/QT ratio was calculated as the ratio of TpTe in that lead to the corresponding QT interval.25,30,31 HR, RR, PR, and QRS intervals were also measured. QTd was calculated as the difference between the longest and shortest individual lead QTc. QTc intervals >450 milliseconds or 60 milliseconds longer than at baseline and QTd >100 milliseconds were considered abnormal.32,33 Normal values for TpTe interval and for TpTe/QT ratio, calculated on V5 leads, were ranged from 4 milliseconds to 100 milliseconds (mean 63.3±11.38 milliseconds, median 60 milliseconds) and from 0.12 to 0.29 (mean 0.195±0.0344, median 0.188), respectively.25 Basal and 2-hour postintake ECG parameters, including QTc, QTd, and TpTe ratio interval duration, were compared.
Statistical analysis
The baseline and 2-hour postintake ECG parameters were presented as mean ± standard deviation. Ranges were also presented for the respective measurements. The baseline/posttreatment measures were compared using the paired-samples, two-sided t-test. Statistical significance was accepted at a P<0.05 level. Data analyses were performed using SPSS® for Windows® package.
Results
A total of 54 subjects completed the study (51 males and 3 females, mean age 12.14±2.6 years, range 6–19 years). Therapeutic dosages were obtained within a month after the beginning of MPH treatment, in all enrolled patients. The average final dose of IR-MPH was 18.5 mg/day, administered in two to three divided doses (range 10–60 mg/day) according to clinical individual needs.
Electrocardiographic changes
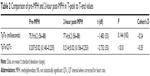
The mean, standard deviation, ranges, t, P, and d values of the ECG measurements before and 2 hours postintake of IR-MPH are shown in Table 1. No abnormal findings were observed in the ECG results at both times, including any changes in voltages, axes, or morphology. Moreover, there was no significant difference noted in QTc and QTd intervals between baseline and 2-hour postintake determination. No patient exhibited a QTc interval greater than 500 milliseconds or a QTd greater than 100 milliseconds, at both determinations. However, in all patients, a significant variation in BP (systolic BP 105.4±10.3 vs 109.6±11.5; P<0.05; diastolic BP 59.2±7.1 vs 63.1±7.8; P<0.05), as well as in HR values (80.5±15.5 bpm vs 87.7±18.4 bpm), was observed between baseline and 2-hour postintake values. No change in TpTe values was found (Table 2), but a statistically significant increase in TpTe/QTc ratio was found with respect to basal values (0.207±0.02 milliseconds vs 0.214±0.02 milliseconds; P<0.01).
Discussion
Our data suggest a relative cardiovascular safety of IR-MPH used to treat ADHD. In our patients, the administration of a standard dosage of IR-MPH is not associated with a statistically significant increase of QTc and QTd values compared to baseline. Nevertheless, treatment with IR-MPH was associated with a slight increase of systolic and diastolic BP. However, no patient exhibited values of BP exceeding the normal range, suggesting that MPH resulted in no consistent or clinically meaningful, short-term BP changes. Furthermore, we did not find significant differences between the acquired TpTe interval measurements. Even if we reported a TpTe/QT ratio increase between basal and posttreatment values, this value was within the normal range.
Indeed, although cardiovascular safety of MPH has been extensively explored in adults, in the last decade, few studies investigated the safety of these drugs in pediatric patients. Even though recent studies focusing on long-term safety have been performed, to date, findings about the association between stimulant use and adverse cardiovascular outcomes are conflicting.20,21,34 A systematic review by Westover and Halm35 found evidence that the statistical power of studies performed on children and adolescents is limited by the sample size, although the absolute risk of an event is low. Studies on adults found an increased risk for transient ischemic attack and sudden death/ventricular arrhythmia, which was more suggestive of a genuine safety issue. However, their interpretation was limited due to differences in population, cardiovascular outcome selection/ascertainment, and methodology.35 In a meta-analysis by Mick et al about the cardiovascular effects of various Central Nervous System (CNS) stimulants in adults with ADHD, based on data from 10 placebo-controlled trials conducted in a pooled sample of 2,665 adults, the use of stimulants, but not of placebo, was associated with a statistically significant increase in mean HR.17 These modifications are thought to be mediated by the sympathomimetic effects, as well as the central and peripheral catecholaminergic effects, of stimulant medications.36,37 Minor but statistically significant changes in systolic BP (3.3 mmHg), diastolic BP (1.5 mmHg), and HR (3.9 bpm) were observed over a 1-year period in a sample of 432 children with ADHD aged 6–13 years, participating in an open-label extension trial of MPH–osmotic-controlled release oral delivery system, 18–54 mg.38 However, findings from the majority of randomized, controlled, and open-label studies coincide in considering that changes in BP and HR most commonly occur during the initial titration phase.39
At typical therapeutic doses, in all age groups, several studies confirmed that cardiovascular changes were minor or of negligible clinical significance.11,35,40–43 Apart from one study44 with possible methodological issues,45 available epidemiological studies have not shown a significant association between ADHD drugs use and serious cardiovascular events.46 A recent large study47 on 1,200,438 children and young adults between the ages of 2 years and 24 years found no evidence that use of current medications for ADHD was associated with an increased risk of severe cardiovascular events (SCD, acute myocardial infarction, and stroke). Nevertheless, possible underreporting and rare deaths with the initiation of the medication remain reasons for concern. Two other large studies41,48 performed on 443,198 adults and on 241,417 children (3–17 years of age), respectively, support the findings of Cooper et al47 thus confirming that ADHD drug use is not associated with increased risk of severe cardiovascular events.
Our data further confirm the safety of MPH in pediatric population and provide one more cardiovascular parameter (TpTe), possibly useful to identify borderline cases. In fact, preexisting conditions and other interindividual differences may raise the risk of harmful adverse effects, which need to be investigated as carefully as possible, particularly with regard to cardiac sequelae.19 In a recent study, Shahani et al screened 691 ECGs of patients with ADHD during a 2-year period.49 Differences in ECG parameters were compared in patients already on ADHD medications compared to those starting a new ADHD treatment, as well as in the stimulant vs nonstimulant drugs group. According to our data, these authors conclude that screening ECGs rarely resulted in management changes for patients taking or starting ADHD medications. In view of these negative studies, it is probably not useful to monitor ECG parameters during routine psychostimulant treatment of children and adolescents with ADHD, unless there are risk factors or comorbid cardiovascular abnormalities. When ECG parameters need to be monitored for information regarding the repolarization, TpTe could be useful. TpTe is the result of the global distribution of the repolarization process. In a recent study, it was significantly and independently associated with increased odds of SCD in subjects with coronary artery disease. Thus, TpTe measurement may extend the value of repolarization beyond the QTc, particularly in situations where QTc is either normal or not valid due to prolongation of QRS duration. Prolonged TpTe has potential for enhancement of SCD risk stratification and warrants evaluation in additional, larger populations.26 This new ECG marker can be very useful when patients have instead cardiological pathologies (such as hypertension, Brugada syndrome, or others)50 or metabolic diseases (like diabetes) before starting psychiatric pharmacological treatment. In fact, normal values of TpTe/QT ratio are relatively narrow, so it can be easier to find patients with high risks of torsades de pointes.
The most important limitation of this study includes the lack of a long-term follow-up. A causal link between the drug and the cardiovascular outcomes could not be definitively confirmed, as observational studies allow the investigation of association rather than causality. Nevertheless, given that ECGs were recorded 2 hours before and after the administration of MPH, it is improbable that the outcomes observed were not due to drug exposure. More studies are needed to confirm the cardiovascular safety during long-term therapy.43 Furthermore, the study design does not permit the investigation of a time- and dose-dependent relationship between cardiovascular adverse events and stimulant treatment in children and adolescents, even though this was not within the scope of the study.
Conclusion
This study underlines the relative cardiac safety of IR-MPH in childhood, even if stimulants may exert a cardiovascular effect on BP and HR. MPH-induced changes seem to be modest and comparable with changes in cardiovascular functioning associated with normal daily activities. Thus, there appears to be a wide margin of cardiovascular safety when MPH is taken orally in usual doses by hyperactive children. However, particular caution should be exercised by physicians in prescribing these drugs to patients with a genetic predisposition to arrhythmias. It might be useful to carry out an ECG examination in all patients starting MPH therapy.10 Additionally, both QTc and QTd should be measured in order to warrant a reliable assessment of drug-induced QT prolongation. TpTe can be an additional useful parameter. In children with known cardiac disease, arrhythmia, or risk factors for cardiac disease, ADHD treatment should be undertaken in consultation with a medical specialist with competence in pediatric cardiology.23,40
Disclosure
The authors report no conflicts of interest in this work.
References
Klassen AF, Miller A, Fine S. Health-related quality of life in children and adolescents who have a diagnosis of attention-deficit/hyperactivity disorder. Pediatrics. 2004;114(5):e541–e547. | ||
Spencer TJ, Biederman J, Mick E. Attention-deficit/hyperactivity disorder: diagnosis, lifespan, comorbidities, and neurobiology. J Pediatr Psychol. 2007;32(6):631–642. | ||
Harpin V, Mazzone L, Raynaud JP, Kahle J, Hodgkins P. Long-term outcomes of ADHD: a systematic review of self-esteem and social function. J Atten Disord. Epub 2013 May 22. | ||
Shaw M, Hodgkins P, Caci H, et al. A systematic review and analysis of long-term outcomes in attention deficit hyperactivity disorder: effects of treatment and non-treatment. BMC Med. 2012;10:99. | ||
Wolraich M, Brown L, Brown RT, et al; Subcommittee on Attention-Deficit/Hyperactivity Disorder; Steering Committee on Quality Improvement and Management. ADHD: clinical practice guideline for the diagnosis, evaluation, and treatment of attention-deficit/hyperactivity disorder in children and adolescents. Pediatrics. 2011;128(5):1007–1022. | ||
Reddy DS. Current pharmacotherapy of attention deficit hyperactivity disorder. Drugs Today (Barc). 2013;49(10):647–665. | ||
Buchhorn R, Conzelmann A, Willaschek C, Stork D, Taurines R, Renner TJ. Heart rate variability and methylphenidate in children with ADHD. Atten Defic Hyperact Disord. 2012;4(2):85–91. | ||
Faraone SV. Using meta-analysis to compare the efficacy of medications for attention-deficit/hyperactivity disorder in youths. Pharm Ther. 2009;34(12):678–694. | ||
Safer DJ. Relative cardiovascular safety of psychostimulants used to treat attention-deficit hyperactivity disorder. J Child Adolesc Psychopharmacol. 1992;2(4):279–290. | ||
Silva RR, Skimming JW, Muniz R. Cardiovascular safety of stimulant medications for pediatric attention-deficit hyperactivity disorder. Clin Pediatr (Phila). 2010;49(9):840–851. | ||
Stiefel G, Besag FM. Cardiovascular effects of methylphenidate, amphetamines and atomoxetine in the treatment of attention-deficit hyperactivity disorder. Drug Saf. 2010;33(10):821–842. | ||
Winterstein AG, Gerhard T, Kubilis P, et al. Cardiovascular safety of central nervous system stimulants in children and adolescents: population based cohort study. BMJ. 2012;345:e4627. | ||
Findling RL, Short EJ, Manos MJ. Short-term cardiovascular effects of methylphenidate and adderall. J Am Acad Child Adolesc Psychiatry. 2001;40(5):525–529. | ||
Samuels JA, Franco K, Wan F, Sorof JM. Effect of stimulants on 24-h ambulatory blood pressure in children with ADHD: a double-blind, randomized, cross-over trial. Pediatr Nephrol. 2006;21(1):92–95. | ||
Arcieri R, Germinario EA, Bonati M, et al; Italian Attention-Deficit/Hyperactivity Disorder Regional Reference Centers. Cardiovascular Measures in Children and Adolescents with Attention-Deficit/Hyperactivity Disorder Who Are New Users of Methylphenidate and Atomoxetine. J Child Adolesc Psychopharmacol. 2012;22(6):423–431. | ||
Buchhorn R, Muller C, Willaschek C, Norozi K. How to predict the impact of methylphenidate on cardiovascular risk in children with attention deficit disorder: methylphenidate improves autonomic dysfunction in children with ADHD. ISRN Pharmacol. 2012;2012:170935. | ||
Mick E, McManus DD, Goldberg RJ. Meta-analysis of increased heart rate and blood pressure associated with CNS stimulant treatment of ADHD in adults. Eur Neuropsychopharmacol. 2013;23(6):534–541. | ||
Graham J, Banaschewski T, Buitelaar J, et al; European Guidelines Group. European guidelines on managing adverse effects of medication for ADHD. Eur Child Adolesc Psychiatry. 2011;20(1):17–37. | ||
Graham J, Coghill D. Adverse effects of pharmacotherapies for attention-deficit hyperactivity disorder: epidemiology, prevention and management. CNS Drugs. 2008;22(3):213–237. | ||
Vitiello B, Elliott GR, Swanson JM, et al. Blood pressure and heart rate over 10 years in the multimodal treatment study of children with ADHD. Am J Psychiatry. 2012;169(2):167–177. | ||
Dalsgaard S, Kvist AP, Leckman JF, Nielsen HS, Simonsen M. Cardiovascular safety of stimulants in children with attention-deficit/hyperactivity disorder: a nationwide prospective cohort study. J Child Adolesc Psychopharmacol. 2014;24(6):302–310. | ||
Roden DM. Drug-induced prolongation of the QT interval. N Engl J Med. 2004;350(10):1013–1022. | ||
Berg A, Bratane E, Odland HH, Brudvik C, Rosland B, Hirth A. Kardiovaskulær risikovurdering ved bruk av AD/HD-medikamenter hos barner hos barn [Cardiovascular risk assessment for the use of ADHD drugs in children]. Tidsskr Nor Laegeforen. 2014;134(7):710–714. Norwegian | ||
Schwartz PJ, Moss AJ, Vincent GM, Crampton RS. Diagnostic criteria for the long QT syndrome. An update. Circulation. 1993;88(2): 782–784. | ||
Bieganowska K, Sawicka-Parobczyk M, Bieganowski M, Piskorski J. Tpeak-Tend interval in 12-lead electrocardiogram of healthy children and adolescents Tpeak-Tend interval in childhood. Ann Noninvasive Electrocardiol. 2013;18(4):344–351. | ||
Panikkath R, Reinier K, Uy-Evanado A, et al. Prolonged Tpeak-to-Tend interval on the resting ECG is associated with increased risk of sudden cardiac death. Circ Arrhythm Electrophysiol. 2011;4(4):441–447. | ||
Ilgenli TF, Congologlu A, Ozturk C, Turkbay T, Akpinar O, Kilicaslan F. Acute effect of methylphenidate on QT interval duration and dispersion in children with attention deficit hyperactivity disorder. Adv Ther. 2007;24(1):182–188. | ||
APA. In: APA, editor. Diagnostic and Statistical Manual of Mental Disorders. 5th ed. Arlington: American Psychiatric Publishing; 2013. | ||
Wargin W, Patrick K, Kilts C, et al. Pharmacokinetics of methylphenidate in man, rat and monkey. J Pharmacol Exp Ther. 1983;226(2): 382–386. | ||
Antzelevitch C, Sicouri S, Di Diego JM, et al. Does Tpeak-Tend provide an index of transmural dispersion of repolarization? Heart Rhythm. 2007;4:1114–1116. [author reply 1116–1119]. | ||
Shimizu M, Ino H, Okeie K, et al. T-peak to T-end interval may be a better predictor of high-risk patients with hypertrophic cardiomyopathy associated with a cardiac troponin I mutation than QT dispersion. Clin Cardiol. 2002;25(7):335–339. | ||
Correll CU, Harris J, Figen V, Kane JM, Manu P. Antipsychotic drug administration does not correlate with prolonged rate-corrected QT interval in children and adolescents: results from a nested case-control study. J Child Adolesc Psychopharmacol. 2011;21(4):365–368. | ||
Germanò E, Italiano D, Lamberti M, et al. ECG parameters in children and adolescents treated with aripiprazole and risperidone. Prog Neuropsychopharmacol Biol Psychiatry. 2014;51:23–27. | ||
Clavenna A, Bonati M. Safety of medicines used for ADHD in children: a review of published prospective clinical trials. Arch Dis Child. 2014;99(9):866–872. | ||
Westover AN, Halm EA. Do prescription stimulants increase the risk of adverse cardiovascular events? A systematic review. BMC Cardiovasc Disord. 2012;12:41. | ||
Negrao BL, Crafford D, Viljoen M. The effect of sympathomimetic medication on cardiovascular functioning of children with attention-deficit/hyperactivity disorder. Cardiovasc J Afr. 2009;20(5):296–299. | ||
Volkow ND, Wang GJ, Fowler JS, et al. Cardiovascular effects of methylphenidate in humans are associated with increases of dopamine in brain and of epinephrine in plasma. Psychopharmacology (Berl). 2003;166(3):264–270. | ||
Wilens TE, Biederman J, Lerner M. Effects of once-daily osmotic-release methylphenidate on blood pressure and heart rate in children with attention-deficit/hyperactivity disorder: results from a one-year follow-up study. J Clin Psychopharmacol. 2004;24(1):36–41. | ||
Hammerness P, Wilens T, Mick E, et al. Cardiovascular effects of longer-term, high-dose OROS methylphenidate in adolescents with attention deficit hyperactivity disorder. J Pediatr. 2009;155(1):84–89, 89.e81. | ||
Cortese S, Holtmann M, Banaschewski T, et al; European ADHD Guidelines Group. Practitioner review: current best practice in the management of adverse events during treatment with ADHD medications in children and adolescents. J Child Psychol Psychiatry. 2013;54(3): 227–246. | ||
Habel LA, Cooper WO, Sox CM, et al. ADHD medications and risk of serious cardiovascular events in young and middle-aged adults. JAMA. 2011;306(24):2673–2683. | ||
Martinez-Raga J, Knecht C, Szerman N, Martinez MI. Risk of serious cardiovascular problems with medications for attention-deficit hyperactivity disorder. CNS Drugs. 2013;27(1):15–30. | ||
Awudu GA, Besag FM. Cardiovascular effects of methylphenidate, amphetamines and atomoxetine in the treatment of attention-deficit hyperactivity disorder: an update. Drug Saf. 2014;37(9):661–676. | ||
Gould MS, Walsh BT, Munfakh JL, et al. Sudden death and use of stimulant medications in youths. Am J Psychiatry. 2009;166(9):992–1001. | ||
Johnson MR. Increased risk of sudden death among youths and stimulant use: determining alternate potential factors. Am J Psychiatry. 2010;167:214. [author reply 214–215]. | ||
Hammerness PG, Perrin JM, Shelley-Abrahamson R, Wilens TE. Cardiovascular risk of stimulant treatment in pediatric attention-deficit/hyperactivity disorder: update and clinical recommendations. J Am Acad Child Adolesc Psychiatry. 2011;50(10):978–990. | ||
Cooper WO, Habel LA, Sox CM, et al. ADHD drugs and serious cardiovascular events in children and young adults. N Engl J Med. 2011; 365(20):1896–1904. | ||
Schelleman H, Bilker WB, Strom BL, et al. Cardiovascular events and death in children exposed and unexposed to ADHD agents. Pediatrics. 2011;127(6):1102–1110. | ||
Shahani SA, Evans WN, Mayman GA, Thomas VC. Attention deficit hyperactivity disorder screening electrocardiograms: a community-based perspective. Pediatr Cardiol. 2014;35(3):485–489. | ||
Karaagac K, Yontar OC, Tenekecioglu E, et al. Evaluation of Tp-Te interval and Tp-Te/QTc ratio in patients with coronary artery ectasia. Int J Clin Exp Med. 2014;7(9):2865–2870. |
 © 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.