Back to Journals » Neuropsychiatric Disease and Treatment » Volume 15
Evaluating optic nerve diameter as a possible biomarker for disability in patients with multiple sclerosis
Authors Koraysha NA, Kishk N , Hassan A , Samy El Gendy NM, Shehata HS , Al-Azayem SA, Kamal YS
Received 17 May 2019
Accepted for publication 14 August 2019
Published 6 September 2019 Volume 2019:15 Pages 2571—2578
DOI https://doi.org/10.2147/NDT.S216079
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Roger Pinder
Noha Abo Koraysha,1 Nirmeen Kishk,1 Amr Hassan,1 Nehal M Samy El Gendy,2 Hatem S Shehata,1 Salsabil Abo Al-Azayem,1 Yasmine Shawki Kamal1
1Neurology Department, Kasr Al Ainy Hospital, Cairo University, Cairo, Egypt; 2Ophthalomology Department, Kasr Al Ainy Hospital, Cairo University, Cairo, Egypt
Correspondence: Nehal M Samy El Gendy
Ophthalomology Department, Kasr Al Ainy Hospital, Cairo University, 43th Gameat El Dewal Street, Cairo 12411, Egypt
Tel +20 100 108 5164
Email [email protected]
Background and purpose: The development of new biomarkers for multiple sclerosis (MS) is of paramount importance to improve our ability to predict disease progression and disability. The aim of this study was to evaluate the potential role of the optic nerve diameter (OND) measured by ultrasonography as a biomarker of early disability in patients with relapsing-remitting multiple sclerosis (RRMS).
Methods: Forty-nine RRMS patients, 23 with a history of optic neuritis (MS-ON) and 26 without a history of optic neuritis (MS N-ON), and 50 age- and sex-matched healthy control subjects were included in the study. The OND and optic nerve sheath diameter (ONSD) were measured by transorbital sonography (TOS), and the retinal nerve fibre layer (RNFL) and ganglion cell complex (GCC) thicknesses were measured by optical coherence tomography (OCT) using the Optovue RTVue™ system (Optovue, Inc., Fremont, CA, USA).
Results: There was no significant difference between the patient (whether ON or N-ON eyes) and control groups in either the OND (p=0.979) or the ONSD (p=0.911). However, patients with an expanded disability status scale (EDSS) score >2 had a significantly lower OND and RNFL thicknesses (p=0.014, p=0.010 respectively) than patients with an EDSS score ≤2. Statistical logistic regression revealed that OND was an independent predictor of EDSS>2 (p=0.044, OR =0.000, 95% CI=0.000–0.589).
Conclusion: The OND, as measured by ultrasonography, could be potentially used as a biomarker for the detection of early disability in RRMS patients.
Keywords: multiple sclerosis, biomarker, OCT, transorbital sonography, RNFL
Introduction
Multiple sclerosis (MS) is one of the leading disabling neurological diseases in young adults. Characteristically reliable biomarkers for every independent MS pathogenic factor are extremely important.1 Increasing evidence has demonstrated that neuronal and axonal damage within the central nervous system (CNS) contributes substantially to the development of permanent disability in patients with MS.2 Thus, reliable, economic and easily assessable complementary surrogate biomarkers for axonal degeneration and consequently disability remain to be identified.3 The optic nerve can serve as a useful clinical tool for studying these characteristics and can be used to measure and monitor the pathological process of the disease.4 Optical coherence tomography (OCT) is a non-contact machine that allows precise measurement of retinal layers thickness using infrared waves. Spectral domain (SD) technology has been used in more recent generations of ophthalmic OCT machines. SD-OCT systems are capable of providing more than 100 times faster scan speed with lower acquisition time. SD-OCT systems use an 850 nm wavelength, which provides better depth resolution. The higher speed, deeply penetrating wavelength provided high resolution, which, using special software algorithm allowed RNGL and GCC analysis.5 OCT is a potential tool for monitoring axonal loss in MS patients and is being investigated in several clinical researches.6,7
The most important parameters in OCT studies are the retinal nerve fibre layer (RNFL) thickness, which is a good measure of the axonal thickness at the optic disc, and the ganglion cell complex (GCC) thickness, which allows the quantification of both axonal loss and neuronal degeneration at the macula,8 probably because of retrograde trans-synaptic degeneration and the progressive loss of retinal ganglion cells, in addition to the more pronounced thinning caused by optic neuritis (ON), if present;9 Ganglion Cell Complex (GCC) revealed the summation of macular RNFL, GCL (ganglion cell layer: formed by ganglion cell body), and IPL(inner plexiform layer).10
The optic nerve is most commonly assessed by ophthalmoscopy and magnetic resonance imaging (MRI), but measurement of the optic nerve diameter (OND) by a simple ultrasound examination might permit a rough estimation of the extent of brain parenchymal involvement and the consequent global cerebral atrophy and disability in relapsing-remitting MS (RRMS) patients. The analysis of the diameter of the optic nerve showed that it is possible to detect its atrophy in the affected eyes (with ON) and, to a lesser extent, in the unaffected eyes of MS patients.11 The aim of this study was to evaluate the potential role of the OND determined by ultrasonography as a biomarker of early axonal loss and disability in patients with RRMS.
Subjects and methods
Subjects
This cross-sectional case-control study was conducted in 49 patients (17 males and 32 females; mean age: 28.96±9.5 years) diagnosed with RRMS according to the revised McDonald’s criteria with (MS-ON) or without a history of unilateral ON (MS N-ON).12 The patients were recruited from the Kasr Al-Ainy Multiple Sclerosis Unit (KAMSU), Cairo University Hospital, from January 2017 to September 2017. Fifty age- and sex-matched healthy volunteers were recruited from medical students and hospital employees as a control group. We excluded patients with progressive MS (whether primary or secondary), patients with a history of relapse in the last three months prior to enrolment, patients with a history of ocular surgery, glaucoma, diabetes, or hypertension, as these conditions may interfere with OCT findings, patients with a history of alcohol intake or a history of bilateral ON, whether simultaneous or sequential, and patients on Fingolimod for more than 6 months prior to recruitment, as it may cause macular oedema.
Methods
Clinical assessment
All patients had undergone thorough neurological, ophthalmological and disability evaluations using the expanded disability status scale (EDSS).13
Transorbital sonography (TOS)
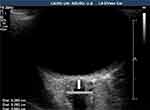
All examinations were performed at the Neurosonology Unit, Neurology Department, Cairo University Hospital, by a single experienced neurosonographer (certified by the European Society of Neurosonology and Cerebral Haemodynamics “ENSCH”), who was blinded to the subjects’ clinical data throughout the entire study. TOS was performed using a Philips IU22 system with a linear L9 probe at 13 MHz obtained from California, USA. Subjects were examined in the supine position with the upper part of the body and the head elevated 20º to 30º degrees to avoid any pressure on the eye. They were asked to keep their eyes in a mid-position and to suppress eye movements. For safety, the mechanical index was reduced to 0.2. The probe was placed on the temporal part of the closed upper eyelid using a thick layer of sonography gel. The anterior part of the optic nerve was depicted in an axial plane showing the papillae and the optic nerve in its longitudinal course. The measured parameters were the OND and the optic nerve sheath diameter (ONSD), which were measured 3 mm behind the posterior edge of the globe in a horizontal plane.14
The optic nerve was visualized as a hypoechogenic structure beyond the retina surrounded by hyperechoic subarachnoid space and hypoechoic dura mater (Figure 1). In patients with no history of ON, two readings were recorded for one eye, and their mean value was calculated. In patients with a history of ON, the examination was performed twice, and the mean value was calculated. The affected eye in MS-ON group and only one eye from MS N-ON and control groups were included in the statistical analysis.
OCT
Spectral-domain OCT was performed using the Optovue RTVue™ system (Optovue Inc., Fremont, CA, USA). Two different protocols were used in the two eyes: the 1st was the macular map protocol, which allows rapid macular scanning for measurement of the GCC thickness; the 2nd was the peripapillary RNFL protocol. All examinations were performed at the Ophthalmology Department, Cairo University Hospital, by a single experienced ophthalmologist.
The study protocols and data collection methods conformed to all local laws and were compliant with the principles of Declaration of Helsinki. The study was approved by the local ethical committee. Informed consent was provided by all patients upon enrolment in the study.
Statistical analysis
Data were coded and entered using the statistical package for the Social Sciences (SPSS) version 25 (IBM Corp., Armonk, NY, USA). Data was summarized using mean and standard deviation for quantitative variables and frequencies (number of cases) and relative frequencies (percentages) for categorical variables. Comparisons between groups were done using unpaired t test in normally distributed quantitative variables while non-parametric Mann-Whitney test was used for non-normally distributed quantitative variables.15 For comparing categorical data, Chi square (χ2) test was performed. Exact test was used instead when the expected frequency is less than 5.15 Correlations between quantitative variables were done using Spearman correlation coefficient.16 A receiver operating characteristic (ROC) curve was constructed, and the area under the curve (AUC) was analysed to detect the best cutoff value for the parameters for EDSS score detection. Logistic regression was done to detect independent predictors of EDSS>2.17 P-values less than 0.05 were considered as statistically significant. Graphs and tables were used to illustrate some information. Logistic regression was done to detect the independent factor reaching EDSS ˃2 as an outcome.
Results
Demographic and clinical characteristics
The patient group included 49 patients; 23 of these patients had a history of ON (MS-ON), and 26 had no history of ON (MS N-ON).Twenty-five patients had an EDSS score ≤2, and 24 patients had an EDSS score >2. An EDSS score of 2 was used as a cutoff value for early disability. The demographic and clinical characteristics of the patients in both groups are shown in Table 1.
 |
Table 1 Summary of demographic and clinical characteristics of the participants |
Results of TOS and OCT
As shown in Table 2, there was no significant difference between patients and controls in either the OND or ONSD. On the other hand, the RNFL and GCC were significantly thinner in the patient group than in the control group. The clinical and imaging results of the individual patients are summarised in Figure 2.
 |
Figure 2 Clinical, TOS and OCT data of the patients group.Abbreviations: EDSS, expanded disability status scale; ON, optic nerve; RNFL, retinal nerve fibre layer. |
 |
Table 2 TOS and OCT results in patient and control groups |
Comparison between patients with EDSS scores ≤2 and >2
Patients with an EDSS score >2 had significantly a thinner OND and RNFL (p=0.014, p=0.010 respectively) than patients with an EDSS score ≤2 (Table 3).
 |
Table 3 Results of TOS and OCT in the N-ON eyes of patients with EDSS scores ≤2 and >2 |
Results of TOS and OCT in ON and N-ON eyes
The RNFL and GCC were significantly thinner in ON eyes than in N-ON eyes (p=0.029, p=0.017, respectively); however, the OND and ONSD showed no significant differences between these subgroups (Table 4).
 |
Table 4 Results of TOS and OCT in ON and N-ON eyes |
A receiver operating characteristic (ROC) curve was constructed, as shown in Figure 3, to determine the cutoff values for the RNFL and GCC thickness resulting in the highest sensitivity and specificity for the detection of patients with an EDSS score >2. According to the ROC curve, the cutoff value for the RNFL and GCC were 117.055 and 93.235 mm respectively meaning that patients with lesser values were more likely to have an EDSS score >2; this result indicates subtle progression with a sensitivity and specificity of 98% and 100%, respectively for RNFL; 85.7% and 91.8% respectively for GCC (Table 5).
 |
Figure 3 ROC curve for the sensitivity and specificity of the RNFL and GCC thickness in the detection of an EDSS score >2. |
 |
Table 5 Cutoff values for the RNFL and GCC for detecting an EDSS score >2 |
Statistical logistic regression was done to detect independent predictors of EDSS>2 among the demographic, clinical and imaging parameters. It had revealed that disease duration (p=0.023, OR=1.204, 95% CI=1.026–1.414), OND (p=0.044, OR=0.000, 95% CI=0.000–0.589) and RNFL thickness (p=0.026, OR=0.934, 95% CI=0.879–0.992) are independent predictors of EDSS>2 as shown in Table 6.
 |
Table 6 Logistic regression to detect independent predictors of EDSS>2 |
Discussion
Large numbers of biomarkers have been tested in the past few years for their ability to predict the response to therapy, disease course and progression, and outcome of MS. Axonal loss has been found to be a promising biomarker for MS. Axonal loss is the most important pathological factor that contributes to permanent disability in MS. Assessing axonal loss could be useful for monitoring disease evolution.18
RNFL thinning, as detected by OCT, has been established in many studies as a reliable biomarker of axonal loss, correlating adequately with brain atrophy measures.18–21
Numerous studies have found thinning of the RNFL not only in eyes with a previous episode of ON but also in the presumably asymptomatic contralateral eye of MS patients who had an episode of ON, as well as in MS patients who never had clinical acute ON.22–26 Moreover, several cross-sectional studies have reported these significant correlations between thinner RNFL and GCC values and higher EDSS scores in both RRMS and secondary progressive MS (SPMS) groups.27–29 Therefore, the presence of a significant correlation between both the RNFL thickness and the EDSS score supports the hypothesis that axonal damage occurs early in the disease course.
However, TOS can be used as an alternative tool to OCT in the evaluation of optic nerves, especially with increasing numbers of experienced neurosonographers worldwide over the past few years. TOS has mainly been used to evaluate intracranial hypertension and has shown a correlation between an increased ONSD and increased intracranial pressure.30–32
One of the main advantages of TOS over OCT is its low cost, high accessibility and reproducibility, with minimal inter- and intra-observer variability.30–33
TOS also offers a sensitive, user-friendly, and reliable technique for detecting the ONSD. Many studies have demonstrated the utility of TOS in evaluating ON, showing increased an OND in the acute phase of MS.34–38 TOS was able to detect a significant thickening of the OND and ONSD in acute ON, which is probably due to inflammation with a subsequent increase in perineural subarachnoid fluid early in the disease course.
A recent study has demonstrated that TOS is a reliable method for measuring ON atrophy in MS. However, the author recommended further studies to compare TOS with visual evoked potentials (VEPs) and OCT to confirm its usefulness in routine clinical practice.9
Our aim was to study the potential role of the OND and ONSD determined using ultrasound as early markers of axonal loss and disability in RRMS patients compared to metrics determined by OCT as well-established axonal loss biomarkers. Our results showed no significant difference in the OND or ONSD between RRMS patients (with or without a past history of ON) and healthy controls. These results are contradictory to those of a previous study that detected OND atrophy in eyes with ON and, to a lesser extent, in the unaffected eyes of MS patients compared to those of controls.39
This difference could be attributed to the fact that the OND measurement Carraro et al used was more proximal to detect the maximum diameter of the nerve. However, in our study, we measured the OND 3 mm from the retinal plane, where the optic nerve is thinner and less myelinated; as such, we might not be able to distinguish small differences in nerve thickness between MS patients and controls. Our sample size might have also been insufficient to reveal statistically significant differences. Additionally, some of our patients in the MSN-ON group could have had an attack of silent or unreported ON. Another possibility is that our subjects might have had mild ON that would not have affected the OND or caused only minimal atrophy.39
However, we found that patients with an EDSS score >2 had significantly smaller ONDs than those with an EDSS score ≤2. Statistical logistic regression was done to detect independent predictors of EDSS>2 among the demographic, clinical and imaging parameters. It had revealed that disease duration OND (p=0.044, OR=0.000, 95% CI=0.000–0.589) is an independent predictors of achieving EDSS>2.
Collectively, our data may represent an indirect indicator for the potential role of the OND determined by sonography as a marker of detection of early disability in MS. To the best of our knowledge, there have been no published studies addressing the role of the sonographic OND as a potential marker of early disability in MS in comparison to OCT metrics.
A recent multicentre study of 59 patients with RRMS confirmed our results, as it showed that the thickness of the optic nerve measured with TOS was correlated with the EDSS score and the duration of the disease without interference from a previous history of ON; however, they did not compare the TOS and OCT results.9
Our study had some limitations. First, there was a lack of normative values for the OND. Second, the use of TOS and OCT can only detect structural lesions; therefore, it would have been more beneficial to combine these methods with VEPs for better correlation between structural lesions and functional impairment (VEPs were determined but not at the same time of our study). In conclusion, the OND, as determined by ultrasonography, could be used as a potential biomarker for the detection of early disability in RRMS patients. Further studies may be needed to confirm our findings and to support this hypothesis. The early detection of disability can facilitate early interventions and consequently better outcomes and prognoses.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Katsavos S, Anagnostouli M. Biomarkers in multiple sclerosis: an up-to-date overview. Mult Scler Int. 2013;2013:1–20. doi:10.1155/2013/340508
2. Singh S, Dallenga T, Winkler A, et al. Relationship of acute axonal damage, Wallerian degeneration, and clinical disability in multiple sclerosis. J Neuroinflammation. 2017;14(1):57. doi:10.1186/s12974-017-0831-8
3. Barkhof F, Calabresi PA, Miller DH, Reingold SC. Imaging outcomes for neuroprotection and repair in multiple sclerosis trials. Nat Rev Neurol. 2009;5(5):256. doi:10.1038/nrneurol.2009.41
4. Wollenweber FA, Schomburg R, Probst M, et al. Width of the third ventricle assessed by transcranial sonography can monitor brain atrophy in a time-and cost-effective manner-results from a longitudinal study on 500 subjects. Psychiatry Res. 2011;191(3):212–216. doi:10.1016/j.pscychresns.2010.09.010
5. Popescu DP, Choo-Smith LP, Flueraru C, et al. Optical coherence tomography: fundamental principles, instrumental designs and biomedical applications. Biophys Rev. 2011;3(3):155. doi:10.1007/s12551-011-0054-7
6. Winges KM, Werner JS, Harvey DJ, et al. Baseline retinal nerve fiber layer thickness and macular volume quantified by OCT in the North American phase 3 fingolimod trial for relapsing-remitting multiple sclerosis. J Neuroophthalmol. 2013;33:322–329. doi:10.1097/WNO.0b013e31829c51f7
7. Dinkin M, Paul F. Higher macular volume in patients with MS receiving fingolimod: positive outcome or side effect? Neurology. 2013;80:128–129. doi:10.1212/WNL.0b013e31827ccf4a
8. Kim NR, Lee ES, Seong GJ, et al. Comparing the ganglion cell complex and retinal nerve fibre layer measurements by Fourier domain OCT to detect glaucoma in high myopia. Br J Ophthalmol. 2011;95:1115–1121. doi:10.1136/bjo.2010.182493
9. Candeliere Merlicco A, Gabaldón Torres L, Villaverde González R, Fernández Romero I, Aparicio Castro E, Lastres Arias MC. Transorbital ultrasonography for measuring optic nerve atrophy in multiple sclerosis. Acta Neurol Scand. 2018;138(5):388–393. doi:10.1111/ane.12976
10. Cruz-Herranz A, Balk LJ, Oberwahrenbrock T, et al. The APOSTEL recommendations for reporting quantitative optical coherence tomography studies. Neurology. 2016;86(24):2303–2309. doi:10.1212/WNL.0000000000002774
11. Perez-Sanchez S, Eichau-Madueno S, Rus-Hidalgo M, Dominguez-Mayoral A, NavarroMascarell G, Izquierdo G. Optic nerve ultrasonography in multiples sclerosis. Mult Scler J. 2016;22:452.
12. Polman CH, Reingold SC, Banwell B, et al. Diagnostic criteria for multiple sclerosis: 2010 revisions to the McDonald criteria. Ann Neurol. 2011;69(2):292–302. doi:10.1002/ana.22366
13. Kurtzke J. Rating neurologic impairment in multiple sclerosis: an expanded disability status scale (EDSS). Neurology. 1983;33:1444–1452. doi:10.1212/wnl.33.11.1444
14. Kishk NA, Ebraheim AM, Ashour AS, Badr NM, Eshra MA. Optic nerve sonographic examination to predict raised intracranial pressure in idiopathic intracranial hypertension: the cut-off points. Neuroradiol J. 2018;31(5):490–495. doi:10.1177/1971400918789385
15. Chan YH. Biostatistics 102: quantitative data–parametric & non-parametric tests. Blood Press. 2003a;140(24.08):79.
16. Chan YH. Biostatistics 104: correlational analysis. Singapore Med J. 2003b;44(12):614–619.
17. Chan YH. Biostatistics 202: logistic regression analysis. Singapore Med J. 2004;45(4):149–153.
18. Toledo J, Sepulcre J, Salinas-Alaman A, et al. Retinal nerve fiber layer atrophy is associated with physical and cognitive disability in multiple sclerosis. Mult Scler J. 2008;14(7):906–912. doi:10.1177/1352458508090221
19. Khalil DH, Labib DM. Correlation between spectral-domain optical coherence tomography parameters and neurological functional disability in multiple sclerosis. J Egypt Ophthalmol Soc. 2015;108(4):243. doi:10.4103/2090-0686.174686
20. Petzold A, de Boer JF, Schippling S, et al. Optical coherence tomography in multiple sclerosis: a systematic review and meta-analysis. Lancet Neurol. 2010;9(9):921–932. doi:10.1016/S1474-4422(10)70168-X
21. El Ayoubi NK, Ghassan S, Said M, Allam J, Darwish H, Khoury SJ. Retinal measures correlate with cognitive and physical disability in early multiple sclerosis. J Neurol. 2016;263(11):2287–2295. doi:10.1007/s00415-016-8271-4
22. Fisher JB, Jacobs DA, Markowitz CE, et al. Relation of visual function to retinal nerve fiber layer thickness in multiple sclerosis. Ophthalmology. 2006;113(2):324–332. doi:10.1016/j.ophtha.2005.10.040
23. Frohman EM, Dwyer MG, Frohman T, et al. Relationship of optic nerve and brain conventional and non-conventional MRI measures and retinal nerve fiber layer thickness, as assessed by OCT and GDx: a pilot study. J Neurol Sci. 2009;282(1):96–105. doi:10.1016/j.jns.2009.04.010
24. Henderson APD, Trip SA, Schlottmann PG, et al. An investigation of the retinal nerve fibre layer in progressive multiple sclerosis. J Neurol Neurosurg Psychiatry. 2008;79(3):339.
25. Pueyo V, Ara JR, Almarcegui C, et al. Sub‐clinical atrophy of the retinal nerve fibre layer in multiple sclerosis. Acta Ophthalmol (Copenh). 2010;88(7):748–752. doi:10.1111/j.1755-3768.2009.01527.x
26. Siger M, Dzięgielewski K, Jasek L, et al. Optical coherence tomography in multiple sclerosis. J Neurol. 2008;255(10):1555–1560. doi:10.1007/s00415-008-0985-5
27. Abalo-Lojo JM, Limeres CC, Gómez MA, et al. Retinal nerve fiber layer thickness, brain atrophy, and disability in multiple sclerosis patients. J Neuroophthalmol. 2008;34(1):23–28. doi:10.1097/WNO.0000000000000057
28. Saidha S, Al‐Louzi O, Ratchford JN, et al. Optical coherence tomography reflects brain atrophy in multiple sclerosis: a four‐year study. Ann Neurol. 2015;78(5):801–813. doi:10.1002/ana.24487
29. Siepman TA, Bettink-Remeijer MW, Hintzen RQ. Retinal nerve fiber layer thickness in subgroups of multiple sclerosis, measured by optical coherence tomography and scanning laser polarimetry. J Neurol. 2010;257(10):1654–1660. doi:10.1007/s00415-010-5589-1
30. Bäuerle J, Lochner P, Kaps M, Nedelmann M. Intra-and interobsever reliability of sonographic assessment of the optic nerve sheath diameter in healthy adults. J Neuroimaging. 2012;22:
31. Rajajee V, Fletcher JJ, Rochlen LR, Jacobs TL. Comparison of accuracy of optic nerve ultrasound for the detection of intracranial hypertension in the setting of acutely fluctuating vs stable intracranial pressure: post-hoc analysis of data from a prospective, blinded single center study. Crit Care. 2012;16:R79. doi:10.1186/CC11336
32. Geeraerts T, Merceron S, Benhamou D, Vigué B, Duranteau J. Non-invasive assessment of intracranial pressure using ocular sonography in neurocritical care patients. Intensive Care Med. 2008;34:
33. Ballantyne SA, O’Neill G, Hamilton R, Hollman AS. Observer variation in the sonographic measurement of optic nerve sheath diameter in normal adults. Eur J Ultrasound. 2002;15:
34. Dees C, Buimer R, Dick AD, Atta HR. Ultrasonographic investigation of optic neuritis. Eye (Lond). 1995;9(Pt 4):
35. Dehghani A, Giti M, Akhlaghi MR, Karami M, Salehi F. Ultrasonography in distinguishing optic neuritis from nonarteritic anterior ischemic optic neuropathy. Adv Biomed Res. 2012;1:3. doi:10.4103/2277-9175.94425
36. Karami M, Janghorbani M, Dehgha A, Riahinejad M. Orbital Doppler evaluation of blood flow velocities in optic neuritis. Korean J Ophthalmol. 2011;23:
37. Lochner P, Cantello R, Brigo F, et al. Transorbital sonography in acute optic neuritis: a case-control study. Am J Neuroradiol. 2014;35:
38. Lochner P, Leone MA, Coppo L, et al. B-mode transorbital ultrasononography for the diagnosis of acute optic neuritis: a systematic review. Clin Neurophysiol. 2016;127:
39. Carraro N, Servillo G, Sarra VM, Bignamini A, Pizzolato G, Zorzon M. Ultrasound findings of the optic nerve and its arterial venous system in multiple sclerosis patients with and without optic neuritis vs. healthy controls. Perspect Med. 2012;1(1–12):381–384. doi:10.1016/j.permed.2012.04.008
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.