Back to Journals » Infection and Drug Resistance » Volume 12
Endemic dissemination of different sequence types of carbapenem-resistant Klebsiella pneumoniae strains harboring blaNDM and 16S rRNA methylase genes in Kerman hospitals, Iran, from 2015 to 2017
Authors Kiaei S, Moradi M, Hosseini-Nave H, Ziasistani M , Kalantar-Neyestanaki D
Received 10 September 2018
Accepted for publication 19 November 2018
Published 21 December 2018 Volume 2019:12 Pages 45—54
DOI https://doi.org/10.2147/IDR.S186994
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Joachim Wink
Somayeh Kiaei,1,2 Mohammad Moradi,2 Hossein Hosseini-Nave,2 Mahsa Ziasistani,3 Davood Kalantar-Neyestanaki2
1Department of Microbiology and Virology, Student Research Committee, School of Medicine, Kerman University of Medical Sciences, Kerman, Iran; 2Department of Microbiology and Virology, School of Medicine, Kerman University of Medical Sciences, Kerman, Iran; 3Pathology and Stem Cell Research Center, School of Medicine, Kerman University of Medical Sciences, Kerman, Iran
Introduction: The emergence and spread of Klebsiella pneumoniae strains resistant to multiple antimicrobial agents are considered as a serious challenge for nosocomial infections.
Materials and methods: In this study, 175 nonrepetitive clinical isolates of K. pneumoniae were collected from hospitalized patients in Kerman, Iran. Extended-spectrum β-lactamases (ESBLs), AmpC, and carbapenemase-producing isolates were recognized by phenotypic methods. The resistance genes including efflux pumps oqxA/oqxB, 16S rRNA methylase, ESBL, AmpC, and carbapenemase were detected by PCR-sequencing method. Molecular typing was performed by enterobacterial repetitive intergenic consensus-PCR and multilocus sequence typing methods among blaNDM-positive isolates.
Results: Thirty-seven (21.14%) isolates along with sequence types (STs): ST43, ST268, ST340, ST392, ST147, and ST16 were harbored blaNDM. ST43 in 2015 and ST268 during 2016–2017 were the most frequent STs among New Delhi metallo-beta-lactamase (NDM)-positive isolates. We found the distribution of some isolates with blaNDM, blaCTX-M, blaSHV, blaOXA, blaTEM, blaCMY, rmtC, and oqxA/oqxB. Enterobacterial repetitive intergenic consensus-PCR represented seven clusters (A–G) plus four singletons among NDM-positive isolates. This study provides the first report of blaNDM-1-positve K. pneumoniae along with ST268 as well as the spread of nosocomial infections with six different STs harboring blaNDM-1 and other resistance genes in hospital settings especially neonatal intensive care unit.
Conclusion: The dissemination of various clones of NDM-producing K. pneumoniae can contribute to increase the rate of their spread in health care settings. Therefore, molecular typing and detection of resistance genes have an important role in preventing and controlling infection by limiting the dissemination of multidrug-resistant isolates.
Keywords: blaNDM, 16S rRNA methylase, MLST, ERIC-PCR
Introduction
Infections caused by multidrug-resistant bacteria have declared a substantial threat to public health worldwide.1 Carbapenems are the most important antibiotics used for the treatment of infections caused by extended-spectrum β-lactamases (ESBLs) and AmpC-producing Gram-negative bacteria.2 Several mechanisms including the loss of outer membrane proteins and carbapenemase such as KPC, GES, VIM, IMP, GIM, New Delhi metallo-beta-lactamase (NDM), and OXA-types are involved in resistance to carbapenems in Enterobacteriaceae.3 Carbapenemase-producing bacteria usually cause life-threatening infections and long-time hospitalization in health care settings.1 For the first time, the NDM has been identified in carbapenem-resistant Klebsiella pneumoniae in Sweden and then has been reported in other Gram-negative bacteria.4–7 In Iran, the first NDM-producing K. pneumoniae was identified in March 2011 from Tehran.1 NDM-producing K. pneumoniae are broadly considered as multidrug-resistant bacteria that have been commonly associated with additional resistance mechanisms such as AmpC, ESBLs, and methylation of 16S rRNA by armA, rmtA, rmtB, and rmtC.8 Several typing methods have been introduced and developed for epidemiological investigation of K. pneumoniae including enterobacterial repetitive intergenic consensus amplification (ERIC-PCR) and multilocus sequence typing (MLST).9,10 MLST is one of the best molecular typing methods for long-term and global epidemiological investigations, and ERIC-PCR is usually used for local outbreaks over a short period of time.10 In this study, we investigated the molecular epidemiology from NDM-1-producing clones among carbapenem-resistant K. pneumoniae isolates in Kerman hospitals, Iran, and we emphasized on the clonal relatedness of these isolates.
Materials and methods
Bacterial isolates
In this study, 175 nonduplicated isolates of K. pneumoniae were collected from hospitalized patients in four referral hospitals (Shafa, Afzalipoor, Bahonar, and Kashani) during February 2015 to November 2017 in Kerman, Iran. All the isolates were identified as K. pneumoniae by standard microbiological tests.11
Antibiotic susceptibility testing
Antibacterial susceptibility test of isolates to cefepime (30 µg), cefotaxime (30 µg), cefoxitin (30 µg), ceftazidime (30 µg), ceftizoxime (30 µg), cefpodoxime (10 µg), imipenem (10 µg), meropenem (10 µg), ertapenem (10 µg), gentamicin (10 µg), amikacin (30 µg), ciprofloxacin (5 µg), and norfloxacin (10 µg) (Mast Group Ltd., Bootle, UK) was determined by disk diffusion method on Müller–Hinton agar media (Laboratorios CONDA, Madrid, Spain) according to the Clinical and Laboratory Standards Institute (CLSI).12 Minimum inhibitory concentration (MIC) of isolates to cefotaxime, cefepime, and imipenem was determined by microbroth dilution method according to CLSI. To determine MIC of colistin and tigecycline by microbroth dilution method, we used the European Committee on Antimicrobial Susceptibility Testing recommendations (http://www.eucast.org/clinical-breakpoints). Escherichia coli ATCC 25922 and Pseudomonas aeruginosa ATCC 27853 were used as standard strains in antibacterial susceptibility testing.
Detection of ESBLs, AmpC, and carbapenemase-producing isolates
ESBLs and carbapenemase-producing isolates were determined according to CLSI recommendations by combination disk with clavulanate and Carba NP test, respectively.12 AmpC disk test was used to detect AmpC β-lactamase-producing isolates.13
Genomic DNA extraction
The genomic DNA was extracted using Exgene Clinic SV (GeneAll Biotechnology, Co., Ltd., Seoul, Republic of Korea; Kat: 106-152) according to the manufacturer’s guidelines.
Detection of resistance genes by PCR sequencing
Antibiotic resistance genes including ESBLs (blaTEM, blaSHV, blaCTX-M, blaOXA-1, and blaPER), caebapenemase (blaKPC, blaGES, blaOXA-48, blaIMP, blaVIM, blaNDM, blaSPM, blaSIM, blaGIM, and blaAIM), efflux pump (oxqA/B), 16S rRNA methylase (rmtA, rmtB, rmtC, and armA), and mcr-1 (colistin resistance gene) were detected by PCR. The primers used for amplification of resistance genes are listed in Table 1. The AmpC β-lactamase genes including blaCMY, blaFOX, blaACC, blaACT, blaDHA, blaEBC, and blaCIT were detected by using multiplex PCR as previously described, and furthermore, PCR products were confirmed by sequencing (Bioneer Corporation, Daejeon, Republic of Korea).29
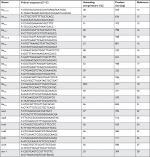  | Table 1 Sequence of primers used in this study for the detection of resistance genes in PCR method |
MLST of NDM-producing isolates
MLST of isolates was performed using seven conserved housekeeping genes (gapA, infB, mdh, pgi, phoE, rpoB, and tonB) according to protocols available at the MLST Pasteur website (http://bigsdb.pasteur.fr/klebsiella/klebsiella.html) for NDM-producing isolates. Products of the above genes in MLST were sequenced by Bioneer, Co. Sequences of each housekeeping gene in both directions were analyzed by Sequence Scanner Software v.2.0 (Applied Biosystems by Thermo Fisher Scientific, Waltham, MA, USA) and assembled by Lasergene 6 software (DNASTAR). The sequence types (STs) of each isolate were determined based on the seven studied loci described at http://bigsdb.pasteur.fr/klebsiella/klebsiella.html.
Molecular typing of blaNDM-positive isolates by ERIC-PCR
ERIC-PCR using ERIC2 primer (5′-AAGTAAGTGACTGGGGTGAGC-3′) was used for molecular typing of NDM-positive isolates.30 The results of ERIC-PCR were analyzed in http://insilico.ehu.eus/dice_upgma/ using the Dice similarity coefficient. Clusters were defined as DNA patterns sharing ≥80% similarity.
Results
In this study, 175 nonduplicated isolates of K. pneumoniae were recovered from hospitalized patients in four referral hospitals in Kerman, Iran. The isolates were collected from different specimens including burning wounds 9 (5.1%), urine 126 (72%), blood 21 (12%), bronchoalveolar lavage 16 (9.1%), and cerebrospinal fluid 3 (1.7%).
Antimicrobial susceptibility testing
The rate of resistance to antibiotics was the following: cefpodoxime 83 (47.4%), cefotaxime 80 (45.7), ceftizoxime 78 (44.6%), ceftazidime 68 (38.9%), cefoxitin 64 (36.6%), cefepime 71 (40.5%), imipenem 45 (25.7%), meropenem 33 (18.9%), ertapenem 30 (17.1%), amikacin 68 (38.9%), gentamicin 59 (33.7%), norfloxacin 33 (18.9%), and ciprofloxacin 31 (17.7%). The ranges of MIC to imipenem, cefepime, and cefotaxime were 4–128 µg/mL, 16–2,048 µg/mL, and 8–2,048 µg/mL, respectively. MIC to colistin was increased in seven (4%) isolates with range 2–16 µg/mL and among other isolates were ≤0.5 µg/mL. All isolates were sensitive to tigecycline with MIC ≤0.5 µg/mL. The MIC results of the clinical isolates are shown in Table 2.
Phenotypic confirmatory tests
Among the 175 K. pneumoniae isolates, 72 (41.1%) strains produced ESBLs, 12 (6.8%) isolates produced AmpC, and 8 (4.5%) isolates produced both ESBLs and AmpC β-lactamase. Out of 175 K. pneumoniae isolates, 37 (21.1%) isolates were considered as positive carbapenemases with Carba NP test.
PCR amplification of antibiotic resistance genes
Based on the PCR assays, the prevalence of ESBL genes was as follows: blaCTX-M 46.28% (n=81), blaSHV 41.1% (n=72), blaTEM 38.9% (n=68), and blaOXA-1 21.7% (n=38). The only carbapenemase gene found in isolates was blaNDM-1 21.14% (n=37). The major AmpC β-lactamase genes found were blaCMY 2.85% (n=5), followed by blaFOX 1.1% (n=2) and blaACC, blaACT 0.6% (n=1). The efflux pump genes including oqxA/oqxB were detected in 36.6% (n=64) and 19.4% (n=34) of isolates. Aminoglycoside-resistant genes (16S rRNA methylase) including rmtC and armA were observed in 5.7% (n=10) and 1.1% (n=2) of isolates, respectively. The rest of the antibiotic resistance genes (blaEBC, blaCIT, blaVIM, blaIMP, blaGIM, blaAIM, blaSPM, blaSIM, blaGES, blaKPC, blaOXA-48, blaPER, blaDHA, rmtA, rmtB, and mcr-1) were negative.
Some sequences of the antibiotic resistance genes including blaNDM, blaTEM, blaCTX-M, blaOXA-1, blaSHV, armA, and rmtC were submitted to the GenBank under accession numbers MG515599, MG515594, MG515597, MG515600, MG515593, MG515596, and MG515592, respectively.
Molecular typing of NDM-producing isolates
In this study, we described the first NDM-producing K. pneumoniae isolates belonging to the ST268 (n=14), which was the major ST. The other STs were as follows: ST43 (n=9), ST340 (n=7), ST392 (n=5), ST147 (n=1), and ST16 (n=1).
According to the eBURST results, ST268 is triple-locus variants of ST16 reporting NDM-producing K. pneumoniae previously. In this study, in comparison with other STs, most isolates of K. pneumoniae ST268 carrying rmtC gene were associated with neonatal intensive care unit (NICU), whereas one of K. pneumoniae ST43 isolate coproducing armA and blaNDM genes was associated with surgical unit (Table 3).
Table 3 showed distribution and genetic characterization of 37 NDM-producing K. pneumoniae strains. ERIC-PCR findings showed that the 37 NDM-producing strains were divided into 7 clusters A to G (11 strains in clusters A, 2 strains in clusters B, E, G, 7 strains in cluster C, 5 strains in cluster D, 4 strains in cluster F, and 3 strains were selected to represent sporadic strains) (Figure 1). ST43 was divided into three clusters (A, C, E), ST268 divided into four clusters (C, D, E, F), ST340 divided into four clusters (A, C, F, G), ST392 divided into three clusters (A, B, G), ST16 was subdivided into one cluster, and ST147 showed as one singleton (Figure 1 and Table 3).
Discussion
During the past decades, carbapenem resistance among K. pneumoniae is typically caused by the emergence of transmissible carbapenemases, such as blaKPC and blaNDM.5 NDM specially comprises of the most rapidly growing group of metallo-beta-lactamases.5 They have been increasingly detected in different countries, suggesting a worldwide dissemination.1 Here, we reported the distribution of nosocomial infections caused by NDM-producing K. pneumoniae, especially in NICU from four referral hospitals in Kerman, Iran.
According to the previous studies, our findings showed that the most effective antibiotics against isolates were colistin and tigecycline.31,32 However, most isolates exhibited a high resistance level to other antimicrobial agents including extended spectrum cephalosporins, carbapenems, quinolones, and aminoglycosides. Similar to our findings, in the study in Egypt, all carbapenem-resistant K. pneumoniae isolates were sensitive to colistin and tigecycline.32
In this study, eleven (17.46%) isolates indicated positive results for AmpC disk test. In our study, non-AmpC-producing isolates might be associated with other resistance mechanisms. Shi et al reported that cefoxitin resistance could be related to the change of cellular permeability to antibiotics, resulting from the loss or deficiency of outer membrane proteins.33,34
In our hospital settings, we found the emergence and establishment of NDM-producing K. pneumoniae along with rmtC and armA. Sporadic dissemination of NDM-1 in Iran was first described in 2013, which was resistant to the majority of antibiotics except for colistin.1 In the current findings, we detected four clinical isolates being resistant to colistin, although one of them only harbored blaNDM-1 gene. This study also focused on epidemiological investigation of MLST and ERIC-PCR in the NDM-positive K. pneumoniae. To the best of our knowledge, the obvious report of the ST prevalence has not been yet accounted for NDM-producing K. pneumoniae in Iran. However, we reported the prevalence of different STs of NDM-producing K. pneumoniae among hospitalized patients during 3 years from March 2015 to November 2017 in Kerman.
According to this study, NDM-producing STs including ST16, ST147, and ST340 were found in India and Korea.35,36 On the contrary, ST147 was recently observed in NDM-positive K. pneumoniae in Iraq.37 Our data showed a dissemination of a novel ST namely 268, which has not been reported in NDM-1-producing K. pneumoniae, during February 2016 to November 2017. The ST268 has been established as a major threat to NICUs from two referral hospitals after detecting the following STs including ST43, ST340, ST392, and ST14.
In general, the epidemiological trend of NDM-producing isolates in our hospital might be divided into three stages. From March 2015 to December 2015, the following STs ST43, ST340, ST392, and ST147 were found. From the beginning of February 2016 up till November 2017, the ST268 has been mainly investigated. Based on these findings, we supposed the ST268 was a successful clone to have recently been established in our hospital settings in 2017. This ST has been found in hypermucoviscous K. pneumonia, which was associated with invasive liver abscess syndrome in eastern Asia.38 On the contrary, ST268 was recognized in capsule serotype K20 K. pneumoniae isolates, relating to primary meningitis in Taiwan.39 Furthermore, these isolates were significantly associated with the virulence factors such as rmpA, rmpA2, and aerobactin.39 Importantly, nine of ten rmtC-positive isolates in our current study belonged to K. pneumoniae, representing ST268, which often exhibited the most predominant ST of carbapenem-resistant K. pneumoniae isolates in NICU. Coproduction of 16S rRNA methylase resistance genes (rmtC, armA) among carbapenem-resistant K. pneumoniae with ST14 and ST340 was reported by Poirel et al.40
In our findings, we observed another major ST, namely ST43, identified during 2015. This ST was able to carry virulence factors causing bacteremia.41 In this study, most isolates belonging to ST43 have been detected from blood samples (Table 3). Recently, ST147 has been associated with blaCMY-4 gene and blaOXA-48 described in Tunisia.42 In our study, ST147 was associated with blaNDM, blaCTX-M, and blaSHV, although this ST has been recognized as a serious threat to public health worldwide.36 ST16 was the other type represented by one isolate from Afzalipoor Hospital. In this study, ST16 has been associated with blaNDM, blaCTX-M, and blaSHV. This ST has been recently reported in Italy, coproducing NDM-1 and OXA-232, recovering from blood and urine samples of a hospitalized patient with urosepsis.43 However, Lester et al34 and Hammerum et al44 in New Zealand and Denmark showed that K. pneumoniae ST16 was recognized at several occasions, disseminating ESBLs and NDM-5 carbapenemase genes.
Our findings showed that ST340 has different ERIC-PCR patterns, that is a single-locus variant of ST11, which was found in Sweden and the UK.45,46 Additionally, NDM-producing isolates from ST340 detected in March 2015 were compared with isolates collected with the same ST in November 2017 to check for ERIC-PCR profile variations within NDM-positive K. pneumoniae. As shown in Figure 1, ERIC-PCR pattern from ST340 displayed identical ERIC-PCR profiles among NDM-producing isolates with the other STs (43, 268, 392, and 16) in different clusters. Similar to this study, Richter et al in Italy showed that no ERIC-PCR profile variation was found between carbapenemase-producing K. pneumoniae strains from STs 258 and 37.47
Lascols et al in India showed a diverse range of clones harboring carbapenemase-producing K. pneumoniae strains representing STs 147 and 340.36,40,48,49 The prevalence of blaNDM-1 is frequently associated with promiscuous plasmids related to a broad host range of clinical variants harboring blaNDM-1, hence our findings hypothesized that nosocomial acquisition of blaNDM by both outward sources including patients who have traveled to Iran specially neighboring countries and also have acquired a broad spectrum of different resistance genes.48 We detected K. pneumoniae ST392, which has been previously associated with the dissemination of blaNDM-1, blaKPC, and blaOXA-48 genes.50,51 However, in this study, ST392 was detected among blaNDM- and blaOXA-positive isolates, recovering from one hospital with different ERIC-PCR patterns. In this study, some STs were observed to have different ERIC-PCR pattern types; therefore, our molecular typing results revealed that ERIC-PCR and MLST provided measures of genetic diversity, while they were not similar methods. These findings showed that ERIC-PCR displayed pattern discriminations for same STs; however, ERIC-PCR has provided a potential molecular typing method to distinguish greater ranges of genetic changes among NDM-positive K. pneumoniae in our hospital settings.52 In this study, up to November 2017, 37 patients with at least one NDM-positive K. pneumoniae isolates were identified. Most isolates were recovered at Afzalipoor Hospital from NICU (Table 3). Interestingly, the most NDM-positive K. pneumoniae isolates belonged to the two STs; 268 and 340 displayed various ERIC-PCR patterns within different clusters. On the contrary, in different hospitals, K. pneumoniae isolates with similar STs revealed diverse ERIC-PCR patterns during 2015–2017. It was suggested that these isolates might be affected by coacquisition of some antibiotic resistance plasmids; therefore, it might affect the results of the ERIC-PCR patterns. Moreover, the molecular typing techniques such as ERIC-PCR and pulsed-field gel electrophoresis were used to identify alterations in short-term, while no obvious difference was observed in STs during 3 years, since MSLT is considered to evaluate the alternations in the most conserved genes, showing long-term variations.52
Conclusion
In this study, the molecular characterization and epidemiological investigations revealed the dissemination of different clones of NDM-producing K. pneumoniae in our hospital settings. Due to the highly resistant nature of bacterial strains carrying the blaNDM, there are very limited antibiotics to combat these bacteria. In this study, MLST differentiated the 37 representative NDM-positive K. pneumoniae strains into 7 STs. The STs included ST268 (n=14), ST43 (n=9), ST340 (n=7), ST392 (n=5), and single isolates representing STs 147 and 16. To the best of our knowledge, the clinical isolates of K. pneumoniae representing ST268 have not been reported among NDM-producing K. pneumoniae. Distribution of blaNDM is obviously related to the promiscuity of many plasmids resulting in a wide range of Gram-negative bacteria containing diverse blaNDM harboring plasmids. However, our study suggests that dominant clones (STs 43 and 268) have had a potential role to monitor and continue a long-term survival of blaNDM-1 dissemination. In addition, our data highlight that the potential for local and neighboring countries such as Pakistan, India, and Iraq has been reported as the endemic dissemination of blaNDM-producing K. pneumoniae clones and plasmid-mediated resistance. Therefore, dissemination of different clones with blaNDM among carbapenem-resistant K. pneumoniae might have resulted in more frequent opportunities for the emergence of blaNDM-positive among other Gram-negative bacteria and it highlighted the need to ongoing epidemiological surveillance and comprehensive infection control guidelines. We also suggested the intrinsic genetic factors, causing a spread and establishment of ST268, as a new NDM-producing K. pneumoniae clone identified to increase our knowledge about it.
Ethical statement
The K. pneumoniae strains were originally taken as part of routine hospital procedure, and then specifically recovered for this work. This study was approved by ethical numbers: IR:KMU.REC.1395.436 and IR.KMU.REC.1395.806 in ethical committee of Kerman University of Medical Sciences.
Acknowledgments
We thank Student Research Committee Kerman University of Medical Sciences for supporting in financial aid for MLST. This research was supported by the research council and student research committee of Kerman University of Medical Sciences, Kerman, Iran, grant numbers: 95000056 and 95000522.
Author contributions
All authors made substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data; took part in drafting the article or revising it critically for important intellectual content; gave final approval of the version to be published; and agree to be accountable for all aspects of the work.
Disclosure
The authors report no conflicts of interest in this work.
References
Shahcheraghi F, Nobari S, Rahmati Ghezelgeh F, et al. First report of New Delhi metallo-beta-lactamase-1-producing Klebsiella pneumoniae in Iran. Microb Drug Resist. 2013;19(1):30–36. | ||
Yan J, Pu S, Jia X, et al. Multidrug resistance mechanisms of carbapenem resistant Klebsiella pneumoniae strains isolated in chongqing, China. Ann Lab Med. 2017;37(5):398–407. | ||
Nordmann P, Naas T, Poirel L. Global spread of carbapenemase-producing enterobacteriaceae. Emerg Infect Dis. 2011;17(10):1791–1798. | ||
Dortet L, Poirel L, Nordmann P. Worldwide dissemination of the NDM-type carbapenemases in Gram-negative bacteria. Biomed Res Int. 2014;2014:249856. | ||
Yong D, Toleman MA, Giske CG, et al. Characterization of a new metallo-beta-lactamase gene, bla(NDM-1), and a novel erythromycin esterase gene carried on a unique genetic structure in Klebsiella pneumoniae sequence type 14 from India. Antimicrob Agents Chemother. 2009;53(12):5046–5054. | ||
Zheng R, Zhang Q, Guo Y, et al. Outbreak of plasmid-mediated NDM-1-producing Klebsiella pneumoniae ST105 among neonatal patients in Yunnan, China. Ann Clin Microbiol Antimicrob. 2016;15:10. | ||
Nordmann P. Carbapenemase-producing Enterobacteriaceae: overview of a major public health challenge. Med Mal Infect. 2014;44(2):51–56. | ||
Kocsis E, Gužvinec M, Butić I, et al. blaNDM-1 Carriage on IncR plasmid in enterobacteriaceae strains. Microb Drug Resist. 2016;22(2):123–128. | ||
Brisse S, Verhoef J. Phylogenetic diversity of Klebsiella pneumoniae and Klebsiella oxytoca clinical isolates revealed by randomly amplified polymorphic DNA, gyrA and parC genes sequencing and automated ribotyping. Int J Syst Evol Microbiol. 2001;51(Pt 3):915–924. | ||
Diancourt L, Passet V, Verhoef J, Grimont PA, Brisse S. Multilocus sequence typing of Klebsiella pneumoniae nosocomial isolates multilocus sequence typing of Klebsiella pneumoniae nosocomial isolates. J Clin Microbiol. 2005;43:4178–4182. | ||
Mahon CR, Lehman DC, Manuselis G. Text Book of Diagnostic Microbiology. 3rd ed. Saunders: Philadelphia, PA; 2007. | ||
CLSI. Performance Standards for Antimicrobial Susceptibility Testing. CLSI Supplement M100. 27th ed. Wayne, PA: Clinical and Laboratory Standards Institute; 2017. | ||
Black JA, Moland ES, Thomson KS. AmpC disk test for detection of plasmid-mediated AmpC beta-lactamases in Enterobacteriaceae lacking chromosomal AmpC beta-lactamases. J Clin Microbiol. 2005;43(7):3110–3113. | ||
Monstein HJ, Östholm-Balkhed A, Nilsson MV, et al. Multiplex PCR amplification assay for the detection of blaSHV, blaTEM and blaCTX-M genes in Enterobacteriaceae. APMIS. 2007;115(12):1400–1408. | ||
Feizabadi MM, Delfani S, Raji N, et al. Distribution of bla(TEM), bla(SHV), bla(CTX-M) genes among clinical isolates of Klebsiella pneumoniae at Labbafinejad Hospital, Tehran, Iran. Microb Drug Resist. 2010;16(1):49–53. | ||
Poirel L, Bonnin RA, Nordmann P. Analysis of the resistome of a multidrug-resistant NDM-1-producing Escherichia coli strain by high-throughput genome sequencing. Antimicrob Agents Chemother. 2011;55(9):11:AAC–4229. | ||
Khorvash F, Yazdani MR, Soudi AA, Shabani S, Tavahen N. Prevalence of acquired carbapenemase genes in Klebsiella Pneumoniae by multiplex PCR in Isfahan. Adv Biomed Res. 2017;6:41. | ||
Poirel L, Walsh TR, Cuvillier V, Nordmann P. Multiplex PCR for detection of acquired carbapenemase genes. Diagn Microbiol Infect Dis. 2011;70(1):119–123. | ||
Mirsalehian A, Feizabadi M, Nakhjavani FA, et al. Detection of VEB-1, OXA-10 and PER-1 genotypes in extended-spectrum beta-lactamase-producing Pseudomonas aeruginosa strains isolated from burn patients. Burns. 2010;36(1):70–74. | ||
Giske CG, Fröding I, Hasan CM, et al. Diverse sequence types of Klebsiella pneumoniae contribute to the dissemination of blaNDM-1 in India, Sweden, and the United Kingdom. Antimicrob Agents Chemother. 2012;56(5):2735–2738. | ||
Labuschagne CJ, Weldhagen GF, Ehlers MM, Dove MG. Emergence of class 1 integron-associated GES-5 and GES-5-like extended-spectrum beta-lactamases in clinical isolates of Pseudomonas aeruginosa in South Africa. Int J Antimicrob Agents. 2008;31(6):527–530. | ||
Rodríguez-Martínez JM, Díaz de Alba P, Briales A, et al. Contribution of OqxAB efflux pumps to quinolone resistance in extended-spectrum-β-lactamase-producing Klebsiella pneumoniae. J Antimicrob Chemother. 2013;68(1):68–73. | ||
Rojas LJ, Wright MS, de La Cadena E, et al. Initial assessment of the molecular epidemiology of bla NDM-1 in Colombia. Antimicrob Agents Chemother. 2016;60(7):4346–4350. | ||
Fritsche TR, Castanheira M, Miller GH, Jones RN, Armstrong ES. Detection of methyltransferases conferring high-level resistance to aminoglycosides in enterobacteriaceae from Europe, North America, and Latin America. Antimicrob Agents Chemother. 2008;52(5):1843–1845. | ||
Guo Y, Zhou H, Qin L, et al. Frequency, antimicrobial resistance and genetic diversity of Klebsiella pneumoniae in food samples. PLoS One. 2016;11(4): e0153561. | ||
Hidalgo L, Hopkins KL, Gutierrez B, et al. Association of the novel aminoglycoside resistance determinant RmtF with NDM carbapenemase in Enterobacteriaceae isolated in India and the UK. J Antimicrob Chemother. 2013;68(7):1543–1550. | ||
Kim JY, Park YJ, Kwon HJ, Han K, Kang MW, Woo GJ. Occurrence and mechanisms of amikacin resistance and its association with beta-lactamases in Pseudomonas aeruginosa: a Korean nationwide study. J Antimicrob Chemother. 2008;62(3):479–483. | ||
Liu YY, Wang Y, Walsh TR, et al. Emergence of plasmid-mediated colistin resistance mechanism MCR-1 in animals and human beings in China: a microbiological and molecular biological study. Lancet Infect Dis. 2016;16(2):161–168. | ||
Pérez-Pérez FJ, Hanson ND. Detection of plasmid-mediated AmpC beta-lactamase genes in clinical isolates by using multiplex PCR. J Clin Microbiol. 2002;40(6):2153–2162. | ||
Meacham KJ, Zhang L, Foxman B, Bauer RJ, Marrs CF. Evaluation of genotyping large numbers of Escherichia coli isolates by enterobacterial repetitive intergenic consensus-PCR. J Clin Microbiol. 2003;41(11):5224–5226. | ||
Samonis G, Maraki S, Karageorgopoulos DE, Vouloumanou EK, Falagas ME. Synergy of fosfomycin with carbapenems, colistin, netilmicin, and tigecycline against multidrug-resistant Klebsiella pneumoniae, Escherichia coli, and Pseudomonas aeruginosa clinical isolates. Eur J Clin Microbiol Infect Dis. 2012;31(5):695–701. | ||
Gamal D, Fernández-Martínez M, Salem D, et al. Carbapenem-resistant Klebsiella pneumoniae isolates from Egypt containing blaNDM-1 on IncR plasmids and its association with rmtF. Int J Infect Dis. 2016;43:17–20. | ||
Shi W, Li K, Ji Y, et al. Carbapenem and cefoxitin resistance of Klebsiella pneumoniae strains associated with porin OmpK36 loss and DHA-1 β-lactamase production. Braz J Microbiol. 2013;44(2):435–442. | ||
Lester CH, Olsen SS, Jakobsen L, et al. Emergence of extended-spectrum β-lactamase (ESBL)-producing Klebsiella pneumoniae in Danish hospitals; this is in part explained by spread of two CTX-M-15 clones with multilocus sequence types 15 and 16 in Zealand. Int J Antimicrob Agents. 2011;38(2):180–182. | ||
Kim SY, Rhee JY, Shin SY, Ko KS. Characteristics of community-onset NDM-1-producing Klebsiella pneumoniae isolates. J Med Microbiol. 2014;63(Pt 1):86–89. | ||
Lee C-R, Lee JH, Park KS, Kim YB, Jeong BC, Lee SH. Global dissemination of carbapenemase-producing Klebsiella pneumoniae: epidemiology, genetic context, treatment options, and detection methods. Front Microbiol. 2016;7(DM01):1–30. | ||
Gharout-Sait A, Alsharapy SA, Brasme L, et al. Enterobacteriaceae isolates carrying the New Delhi metallo-β-lactamase gene in Yemen. J Med Microbiol. 2014;63(Pt 10):1316–1323. | ||
Hirai Y, Asahata-Tago S, Ainoda Y, et al. First case of liver abscess with endogenous endophthalmitis due to non- K1/K2 serotype hypermucoviscous Klebsiella pneumoniae clone ST268. J Gastrointest Dig Syst. 2016;6(1):383. | ||
Ku YH, Chuang YC, Chen CC, et al. Klebsiella pneumoniae isolates from meningitis: epidemiology, virulence and antibiotic resistance. Sci Rep. 2017;7(1):6634. | ||
Poirel L, Dortet L, Bernabeu S, Nordmann P. Genetic features of blaNDM-1-positive Enterobacteriaceae. Antimicrob Agents Chemother. 2011;55(11):5403–5407. | ||
Holt KE, Wertheim H, Zadoks RN, et al. Genomic analysis of diversity, population structure, virulence, and antimicrobial resistance in Klebsiella pneumoniae, an urgent threat to public health. Proc Natl Acad Sci U S A. 2015;112(27):E3574–E3581. | ||
Grami R, Mansour W, Ben Haj Khalifa A, et al. Emergence of ST147 Klebsiella pneumoniae-producing OXA-204 carbapenemase in a University Hospital, Tunisia. Microb Drug Resist. 2016;22(2):137–140. | ||
Avolio M, Vignaroli C, Crapis M, Camporese A. Co-production of NDM-1 and OXA-232 by ST16 Klebsiella pneumoniae, Italy, 2016. Future Microbiol. 2017;12:1119–1122. | ||
Hammerum AM, Hansen F, Olesen B, et al. Investigation of a possible outbreak of NDM-5-producing ST16 Klebsiella pneumoniae among patients in Denmark with no history of recent travel using whole-genome sequencing. J Glob Antimicrob Resist. 2015;3(3):219–221. | ||
Woodford N, Turton JF, Livermore DM. Multiresistant Gram-negative bacteria: the role of high-risk clones in the dissemination of antibiotic resistance. FEMS Microbiol Rev. 2011;35(5):736–755. | ||
Giske CG, Fröding I, Hasan CM, et al. Diverse sequence types of Klebsiella pneumoniae contribute to the dissemination of blaNDM-1 in India, Sweden, and the United Kingdom. Antimicrob Agents Chemother. 2012;56(5):2735–2738. | ||
Richter SN, Frasson I, Franchin E, et al. KPC-mediated resistance in Klebsiella pneumoniae in two hospitals in Padua, Italy, June 2009-December 2011: massive spreading of a KPC-3-encoding plasmid and involvement of non-intensive care units. Gut Pathog. 2012;4(1):7. | ||
Sartor AL, Raza MW, Abbasi SA, et al. Molecular epidemiology of NDM-1-producing Enterobacteriaceae and Acinetobacter baumannii isolates from Pakistan. Antimicrob Agents Chemother. 2014;58(9):5589–5593. | ||
Lascols C, Hackel M, Marshall SH, et al. Increasing prevalence and dissemination of NDM-1 metallo-β-lactamase in India: data from the SMART study (2009). J Antimicrob Chemother. 2011;66(9):1992–1997. | ||
Rojas LJ, Wright MS, de La Cadena E, et al. Initial assessment of the molecular epidemiology of blaNDM-1 in Colombia. Antimicrob Agents Chemother. 2016;60(7):4346–4350. | ||
Bocanegra-Ibarias P, Garza-González E, Morfín-Otero R, et al. Molecular and microbiological report of a hospital outbreak of NDM-1-carrying Enterobacteriaceae in Mexico. PLoS One. 2017;12(6):e0179651. | ||
Kidd TJ, Grimwood K, Ramsay KA, Rainey PB, Bell SC. Comparison of three molecular techniques for typing Pseudomonas aeruginosa isolates in sputum samples from patients with cystic fibrosis. J Clin Microbiol. 2011;49(1):263–268. |
 © 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.



