Back to Journals » Neuropsychiatric Disease and Treatment » Volume 11
Emotional face recognition deficit in amnestic patients with mild cognitive impairment: behavioral and electrophysiological evidence
Authors Yang L, Zhao X, Wang L, Yu L, Song M, Wang X
Received 20 March 2015
Accepted for publication 18 May 2015
Published 5 August 2015 Volume 2015:11 Pages 1973—1987
DOI https://doi.org/10.2147/NDT.S85169
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Professor Wai Kwong Tang
Linlin Yang, Xiaochuan Zhao, Lan Wang, Lulu Yu, Mei Song, Xueyi Wang
Department of Mental Health, The First Hospital of Hebei Medical University, Hebei Medical University Institute of Mental Health, Shijiazhuang, People’s Republic of China
Abstract: Amnestic mild cognitive impairment (MCI) has been conceptualized as a transitional stage between healthy aging and Alzheimer’s disease. Thus, understanding emotional face recognition deficit in patients with amnestic MCI could be useful in determining progression of amnestic MCI. The purpose of this study was to investigate the features of emotional face processing in amnestic MCI by using event-related potentials (ERPs). Patients with amnestic MCI and healthy controls performed a face recognition task, giving old/new responses to previously studied and novel faces with different emotional messages as the stimulus material. Using the learning-recognition paradigm, the experiments were divided into two steps, ie, a learning phase and a test phase. ERPs were analyzed on electroencephalographic recordings. The behavior data indicated high emotion classification accuracy for patients with amnestic MCI and for healthy controls. The mean percentage of correct classifications was 81.19% for patients with amnestic MCI and 96.46% for controls. Our ERP data suggest that patients with amnestic MCI were still be able to undertake personalizing processing for negative faces, but not for neutral or positive faces, in the early frontal processing stage. In the early time window, no differences in frontal old/new effect were found between patients with amnestic MCI and normal controls. However, in the late time window, the three types of stimuli did not elicit any old/new parietal effects in patients with amnestic MCI, suggesting their recollection was impaired. This impairment may be closely associated with amnestic MCI disease. We conclude from our data that face recognition processing and emotional memory is impaired in patients with amnestic MCI. Such damage mainly occurred in the early coding stages. In addition, we found that patients with amnestic MCI had difficulty in post-processing of positive and neutral facial emotions.
Keywords: event-related potential, emotion, recognition memory, amnestic mild cognitive impairment, faces
Introduction
Amnestic mild cognitive impairment (MCI) frequently represents a transitional stage between normal aging and early Alzheimer’s disease (AD).1 Patients with amnestic MCI usually show normal cognitive ability and normal ability in activities of daily living. However, they are thought to be at high risk of developing AD.2,3 Therefore, it would be useful to find a way of determining which patients with amnestic MCI will go on develop AD by investigating aspects of memory that are impaired and those that remain relatively intact.
Previous studies had shown that information with an emotional message may boost memory.4 However, for patients with MCI, the amygdala, which is the key area of the brain for processing of emotional memory, may be damaged, as illustrated by significant elevation of neuritic plaques.5 Currently, it remains controversial as to whether amnestic MCI shows an emotional superiority effect. Some studies have demonstrated impairment of emotional face recognition in patients with amnestic MCI.6–9 However, others believe that the precedence effect of amnestic MCI memory encoding for emotional faces (beneficial effect) is damaged,10 and the most recent study11 showed that recognition memory for emotional faces is impaired in amnestic MCI.
Due to their high time and temporal resolution, event-related potentials (ERPs) are considered to be particularly useful in time process monitoring and processing of emotional faces. It is believed that three components of ERPs are related to earlier perception, ie, P100, N170, and P2. The P100 component is thought to reflect domain-general early visual processing and to be insensitive to face familiarity or correct recognition. The N170 component is considered to be an indicator of structural encoding of a face. In addition, a positive potential may emerge together with the N170 component in the frontocentral sites, such as the vertex positive potential (VPP).12,13 Previous reports demonstrated that the amplitude of N170 was enhanced for personally familiar faces when compared with unfamiliar faces and for emotional faces when compared with neutral faces. The P2 component directly follows N170, and is thought to reflect visual processing linked to perceptual grouping, and is also considered to reflect an early recognition process. Moreover, P2 has been shown to be sensitive to emotional significance.11 It was also described that ERPs were recorded while subjects performed a memory retrieval task that requires old/new judgments regarding visually presented old (previously studied) and new pictures.14 This so-called ERP old/new effect consists of temporally and spatially dissociable subcomponents that have been associated with specific retrieval mechanisms and are evoked by a variety of stimuli. Two subcomponents were used in the current study. The first subcomponent was an early frontal old/new effect (300–500 milliseconds) that has often been associated with familiarity. A second subcomponent, the parietal old/new effect (500–800 milliseconds), has been associated with recollection.15 Analysis of the ERP old/new effect in the early time window showed an early frontal old/new effect for negative faces but not for neutral faces. However, there is no difference in this regard between patients with amnestic MCI and healthy controls.11 It has also been argued that negative emotion may boost familiarity due to enhanced conceptual or perceptual fluency for emotionally negative information,16 and that emotional stimuli may be given priority for processing.17,18 Similar behavioral test results have also been reported by a number of other research groups.19–23
Facial expressions inform how people feel and their action tendency. Six basic categories of emotional facial expressions have been identified, ie, happiness, sadness, anger, disgust, fear, and surprise,24 Some involve signals of potential harm, whereas others convey signals of potential benefit. In prior behavioral research, happy facial expressions have been found to be more accurately and faster than other expressions.25 In recent years, studies of emotional memory in younger and older adults have suggested that older adults may have reduced memory advantages for negative information,26 and may in some instances demonstrate a positive bias, ie, a memory advantage for positive information.27–30 However, the underlying cognitive mechanisms are not fully understood. It has found that emotion-induced memory enhancement is abolished and positivity-related recognition bias is preserved in patients with amnestic MCI,10 but there have been no studies exploring the change in recognition memory of patients with amnestic MCI by performing ERP analysis.
In this study, we investigated whether recognition memory for positive emotional faces is impaired in patients with amnestic MCI, and if so, at what stage of memory recognition this impairment occurs, and whether it makes any difference if patients are tested for memory impairment using positive emotional faces or negative emotional faces. So far, all of these issues remain the subject of debate. We measured behavioral and ERP data in patients with amnestic MCI and in healthy controls to determine the time course of successful identification of emotional faces. We hypothesized that patients with amnestic MCI would show an early and late time window old/new effects deficit, and the changes in old/new effects in patients with amnestic MCI would be correlated with progression of amnestic MCI.
Materials and methods
Participants
All patients met Petersen’s criteria2 for amnestic MCI, which included the following: complaining of memory loss for more than 3 months; maintaining normal activities of daily living; presenting normal general cognitive function; showing objective impairment in one area of cognitive function as evidenced by scores >1.5 standard deviation of age-appropriate norms; and having a Clinical Dementia Rating score of 0.5.
The amnestic MCI and control groups were both required to meet the following conditions: no dementia; no significant changes on brain imaging, such as cerebral infarct, hydrocephalus, or leukoaraiosis; right-handedness; no history or evidence of psychiatric or neurological disease, cardiovascular disease, diabetes, thyroid disease, vitamin B12 deficiency, or substance abuse; normal vision or corrected normal vision; and being native Chinese.
The study was approved by the ethics committee at the First Hospital of Hebei Medical University. Before the experiments, all tested subjects were informed regarding the purpose of this study and signed consent forms.
The study included 24 patients with amnestic MCI and 24 age-matched healthy controls recruited as volunteers from Shijiazhuang City, People’s Republic of China between May 2012 and February 2014. Demographic data and results for neuropsychological assessments are shown in Table 1.
Stimulus materials
Three hundred pictures of faces were chosen from the China Faces Emotions Materials database for this study.31 One third of the selected pictures (n=100) were positive faces, one third were neutral faces, and one third were negative faces. The pictures selected were evenly divided between men and women. All pictures were of uniform size, background, and color, and were presented in the center of a 17-inch flat screen monitor with a gray background.
Three types of pictures matched in a pleasant degree of polarization (positive, 7.44±0.31; neutral, 5.03±0.22; negative, 2.45±0.28). The arousal of three types of pictures was consistent (positive, 5.54±0.47; neutral, 4.48±1.37; negative, 5.44±0.40).
Procedure and design
Using a learning-recognition paradigm, we divided our experiments into two steps, ie, a learning phase and a test phase. In the learning phase, subjects were presented with pictures depicting each of the three types of emotional face (there were 50 pictures for each type of emotion, with a total of 150 pictures presented). In response to presentation of each emotional picture, the test subjects were asked to type in a corresponding key. Each picture was presented four times in order to increase familiarity, and in a randomized order. The tests were started 40 minutes later after completion of the learning period, with recording of an electroencephalogram. Before the start of the learning phase, participants were asked to carefully observe and remember the presented faces. Forty minutes later, some novel faces were presented in order to test the ability to distinguish and memorize faces. During the learning stage, the subjects were required to focus on the point for 500 msec, after which a picture of a face was presented for 3 seconds and the subjects were required to make category judgments (positive, neutral, or negative) by pressing specific keys. Presentation of each picture was separated by a 1,000 msec blank screen. For the recognition phase, the three types of learned emotional faces and novel faces were mixed first, and then presented to the subjects, who made decisions regarding their familiarity with the faces.
The details of the testing procedure used in our study are as follows: at the beginning of each test, a “+” sign appears at the focus point for 500 msec, a picture of a face then appears for up to 2,000 msec, and the subject is asked to make the decision about the familiarity of the tested faces (learned or novel). A blank screen appears for 2,000 msec between each picture presentation. The guidance language used by the investigators was standardized after 12 practice trials before performing the recognition experiments.
Electrophysiological recording
An electroencephalogram was recorded continuously for each subject by means of a Neuroscan amplifier SynAmps2. The electrodes were placed according to the international 10/20 placement system using a 64-lead electrode cap. A unilateral reference electrode was placed in the mastoid of the left ear. Vertical and horizontal electro-oculographic activity was recorded with additional electrodes located above and below the left eye and outside the outer canthi of both eyes. Connections were also placed at the midpoint frontoparietal zone and at the frontal zone. Electrical activity was amplified with a bandpass filter from 0.01 to 100 Hz and at a sampling rate of 500 Hz. In offline analysis, the data were intercepted into single sweep recordings from 100 msec before each stimulus onset to 800 msec after the stimulus. The baseline was corrected by averaging the signal during the 100 msec preceding the onset of the stimulus. All epochs with ocular artifacts greater than 75 μV were automatically rejected by a regression method with a bandpass filter of 0.05–40 Hz. The resistance between each electrode and the scalp was less than 5 KΩ. Electroencephalographic data were analyzed with Scan 4.5 software. E-Prime software was used to collect the stimulus and behavioral data.
Neuropsychological tests
All study subjects underwent a series of neuropsychological tests one day before implementation of the ERP test. The main tests used were the Mini-Mental State Examination32 and the Beijing version of the Montreal Cognitive Assessment.33 Assessment of daily activity was done using the activities of daily living.34 The Center for Epidemiologic Studies Depression Scale35 was used to identify depression.
Data analyses
Behavioral data
In the learning phase, we analyzed how often the subjects classified the different types of emotional pictures correctly. In the subsequent recognition phase, we analyzed four indicators as follows: the percentage of subjects correctly recognizing pictures of happy, neutral, and angry faces (hits); the percentage of erroneously recognized faces (false positives); discrimination accuracy, calculated as (Pr = p [hits] − p [false positives]); and recognition bias, ie, the probability of saying “yes” to an item when uncertain, calculated as (Br = p [false positives]/p [1− Pr]).36
ERP data
ERP data were analyzed from 800 msec after the stimulus onset. The baseline was taken for the time window of 100 msec before the stimulus onset. As in previous studies, ERP data were analyzed on the basis of the early components (P100, N170/VPP, P200), which were quantified by mean amplitude, and the late old/new effect (early frontal old/new effect, parietal old/new effect). The range of peak value and latency were selected based on the total averaged figure.
Three-factor repeated measures analysis of variance was performed for the peak latency of each early component (P100, N170/VPP, P200). The three factors were group (healthy control, amnestic MCI), emotion (positive, neutral, negative), scalp position (P100 at 90–140 msec for O1 and O2; N170 at 160–240 msec for PO8 and PO7; VPP at 160–240 msec for the frontal zone and the central zone; P2 at 240–380 msec for PO8 and PO7). The average amplitude of the early components (P100, N170/VPP, P200) were similarly analyzed, with the corresponding three factors being groups (normal, amnestic MCI), emotion (positive, neutral, negative), and scalp position (left [P07] and right [P08]). The average amplitude detection range was at 80–130 msec for P1, 130–200 msec for N170/VPP, and 240–380 msec for P2.
ERP old/new effect
The ERP old/new effect was analyzed using data collected from 12 factors (F5, FZ, F6, C5, CZ, C6, P5, PZ, P6, CB1, OZ, CB2). The time window for analysis was 300–500 msec for the early frontal old/new effect and 500–800 msec for the parietal old/new effect. Five-factor repeated-measures analysis of variance was performed for the ERP old/new effect from two recorded brain areas. These five factors were: factors region (frontal [F5, FZ, F6] versus central [C5, CZ, C6] versus parietal [P5, CZ, P6] versus occipital [CB1, OZ, CB2], hemisphere (left [F5, C5, P5, CB1] versus midline [FZ, CZ, PZ, OZ] versus right [F6, C6, P6, CB2]), old/new response, emotion, and between-subject factor group. The P-value for analysis of variance was corrected using the Greenhouse–Geisser method.
Results
Behavioral data
Study session
The accuracy of emotion classification was high in both groups. The mean (± standard deviation) percentage of correct classifications was 81.19%±12.7% for the amnestic MCI group and 96.46%±8.3% for the control group. The percentage of correct classifications was higher for healthy controls than for patients with amnestic MCI (F=5.26, P=0.028). There were no differences in or interactions between the emotional categories.
Test session
Parameters of recognition performance are displayed in Table 2. In this study, the average error rate was 30.56% and the error rate for all subjects was 19.55%–43.55%. The error rates during the test are listed in Table 3.
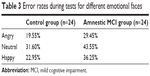  | Table 3 Error rates during tests for different emotional faces |
ERP data
In this analysis, we only included electroencephalographic data from tests with correct rejections (CR), ie, the “old” response to faces we researched in this study (hits) and the “new” response to novel faces (CR). The total mean ERPs evoked by hits and CR for negative, positive, and neutral faces are presented in Figures 1–3, respectively.
P100 component
Four-factor repeated-measures analysis of variance was performed for the amplitude and peak latency of P100. The four factors were group (healthy control, amnestic MCI), emotion (positive, neutral, negative), scalp position (01, 02), and old/new effect (hits, CR). The results are summarized in Table 4. The data indicate that there were no significant differences between the two groups of subjects (amplitude [F=0.437, P=0.512], latency [F=0.187, P=0.667]), or between different scalp positions (amplitude [F=0.103, P=0.749] and latency [F=0.836, P=0.365]). Similar results were found for old/new effect (amplitude [F=0.275, P=0.603] and latency [F=0.320, P=0.547]), and emotional effect (amplitude [F=1.024, P=0.317] and latency [F=2.085, P=0.130]). No significant difference was found with regard to interaction (amplitude [F=0.254, P=0.776] and latency [F=1.181, P=0.311]).
  | Table 4 Amplitude and latency of p100 component |
N170/VPP component
Four-factor repeated-measures analysis of variance was performed for the amplitude and peak latency of N170. The four factors were group (healthy control, amnestic MCI), emotion (positive, neutral, negative), scalp position (P07 and P08), and old/new effect (hits, CR). Compared with the healthy control group, the peak latency of N170 for the amnestic MCI group was clearly prolonged (t=5.57, P<0.01). For the control group, at the P08 recording position, the amplitude of hits for negative faces was larger than the amplitude for CR (t=5.61, P<0.01). Similar results were observed for positive faces (t=3.11, P<0.05), but no difference was observed for neutral faces. For the control group, the amplitude of N170 in the hits condition was enhanced for both positive faces (t=12.36, P<0.001) and negative faces (t=15.26, P<0.001). The difference was significant compared with neutral faces. For the amnestic MCI group, the amplitude of N170 did not change for old/new effect. However, for the presentation of novel faces, the difference was significant for the different emotional faces. Compared with neutral faces, the amplitude of N170 was decreased for negative faces in both recording electrodes for P08 (t=6.01, P<0.01) and P07 (t=7.73, P<0.01, Table 5).
  | Table 5 Amplitude and latency of N170 component |
Four-factor repeated-measures analysis of variance was also performed for the amplitude of VPP. The four factors were group (healthy control, amnestic MCI), emotion (positive, neutral, negative), scalp position (frontal zone, central zone), and old/new effect (hits, CR). The results indicated that the amplitude of negative hits in the amnestic MCI group was increased (t=13.30, P<0.001) when compared with the CR for negative faces in the healthy control group. No significant differences were observed with the rest of the dimensions (Table 6).
P2 component
In the amnestic MCI group, the tested subjects showed strong effects of recognition with negative faces, but no effects with neutral faces or positive faces. In this study, four-factor repeated-measures analysis of variance was performed for P2 amplitude. The four factors were group (healthy control, amnestic MCI), emotion (positive, neutral, negative), scalp position (P07 and P08), and old/new effect (hits, CR). In the control group, the P2 amplitude in the hits situation was significantly larger than that of the CR with negative faces (t=6.09, P<0.01), and was similar with positive faces (t=7.33, P<0.01). However, for hits in the control group, the P2 amplitude for recognition of negative faces was much higher than that for recognition of neutral faces (t=5.17, P<0.01) and positive faces (t=3.36, P<0.05), and the difference was significant. In the amnestic MCI group, at the P08 recording electrode, the peak hits for negative faces was more positively shifted than the CR peak for negative faces (t=6.60, P<0.01), but no statistically significant differences were observed for P2 peak latency (F<0.81, P>0.05, Table 7). This result suggests that when the tested subjects tried to recognize negative faces, they could distinguish easily between studied faces and unstudied faces. The participants retained the negative face recognition effect, and had a more accurate memory for negative faces. P2 is believed to be more sensitive to emotional information,37–39 and our results suggest that negative emotional information improves face information processing during the learning period.
  | Table 7 Amplitude and latency of P2 component |
ERP old/new effect
In the control group, the average amplitude of the negative face hits was greater than the average CR amplitude in the early time window. This effect appeared in F5 (t=6.65, P<0.01), the frontal zone (t=7.07, P<0.01), and the central zone (t=5.33, P<0.01). The result was similar for recognition of positive faces at recording electrodes in the frontal zone (t=3.17, P<0.05) and central zone (t=13.80, P<0.001). In the control group, when compared with neutral face recognition, the peak for angry face hits was more positively shifted. This emotional effect was found only in the frontal zone (t=6.01, P<0.01) and central zone (t=6.82, P<0.01). In the amnestic MCI group, the average hits amplitude was larger than the average CR amplitude. This emotional effect occurred in the frontal zone (t=5.14, P<0.01) and the central zone (t=3.78, P<0.05). Compared with the control group, the amplitude of hits for angry faces was more positively offset than that for neutral faces. This emotional effect was found in the frontal zone (t=6.90, P<0.01) and central zone (t=12.41, P<0.001, Table 8).
  | Table 8 Old/new effect in early time window |
In the control group, the hits amplitude for positive faces was greater than the CR amplitude in the late time window. The main electrodes showing this phenomenon were at the central zone (t=14.69, P<0.001) and parietal zone (t=15.28, P<0.001). Similar results were also obtained with negative faces at the main electrodes of the central zone (t=7.02, P<0.01) and parietal zone (t=5.33, P<0.01). In the control group, the hits amplitude for negative faces was greater than that for either positive faces (t=6.38, P<0.01) or neutral faces (t=4.72, P<0.05). However, in this late time window, no significant difference of old/new effect was observed in the amnestic MCI group (Table 9).
  | Table 9 Old/new effect in late time window |
Discussion
In the current study, we investigated the time course of emotional face recognition in patients with amnestic MCI and in healthy controls by measuring behavioral and ERP data. Our main findings are summarized here. The accuracy of face recognition memory was lower in patients with amnestic MCI than in healthy controls, which supports the presence of damaged episodic memory in amnestic MCI.2,11 Both patients with amnestic MCI and healthy controls showed an emotional preference for face memory. Compared with emotional faces (positive and negative), the hits rate was relatively low and the false positive rate was relatively high for neutral faces. The accuracy of face memory is clearly reduced for a neutral face. This finding indicates that emotional messages can improve episodic memory in amnestic MCI, which is supported by similar studies.11,40,41
P100 reflects visual processing, a positive going wave in the occipital lobe with its peak normally observed in the 100 milliseconds range post-stimulus onset.42–44 P1 is one of the components of P100, and can be modulated by attention. In this study, we did not find a difference in P1 amplitude and latency between patients with amnestic MCI and healthy controls, indicating that early visual processing during face recognition was not damaged in patients with amnestic MCI. Schefter et al found that the amplitude of P100 for hits was increased compared with that for CR, independent of emotion or group.11 However, we did not find similar results in our study. The difference between our current study and the previous one may reflect the fact that Schefter et al reused old studied faces in their test phase. The learning procedure and test producer are also different, such as test face presentation time when test session runs. All these factors could contribute to this discrepancy. However, this would not affect the conclusion of our study, since it is independent of emotion and group.
N170 is a negative wave that can be recorded in the temporal occipital region. The N170 component is thought to be related to face structural encoding.45,46 In the current study, we found that N170 was delayed in patients with amnestic MCI when compared with healthy controls. The increase in N170 latency in patients with amnestic MCI indicates that the face structure encoding process may be slowed down by this disease. Similar results have been reported elsewhere.11,47,48 In addition, with regard to N170 amplitude, the healthy controls showed a recognition effect with negative faces and positive faces, but not with neutral faces. However, the patients with amnestic MCI did not show a face recognition effect, suggesting that emotional face recognition is impaired in patients with amnestic MCI in term of face structural encoding. Further analysis demonstrated that an emotion effect was present for hits but not for CR. However, previous studies of N170 have reported both a face familiarity effect49,50 and a face emotion effect.51,52 The interaction of these two face recognition effects over the right hemisphere has been reported. Hits can represent this interaction, yet CR does not share the characteristic of face familiarity. A candidate process driving this interaction effect could be attention because facial emotion can serve as a cue to attract attention to the face in order further to assist the process of face recognition.51,53,54
P2 showed a strong signal in the temporal and occipital sites, which is considered to reflect visual processing associated with perceptual grouping.55 P2 is believed to be more sensitive to emotional information.53–55 With regard to face recognition, P2 may reflect an early identification process.49,56,57 Like N170, in healthy controls P2 showed a memory recognition effect for negative and positive faces, but not for neutral faces. Thus, our results strongly support the conclusion of previous studies, ie, that an emotional message can improve memory processing.49,57 It has been suggested that within the context of face memory, P2 may reflect an early recognition process involved in personalized face recognition.56 For patients with amnestic MCI, P2 demonstrated a memory recognition effect only with negative faces, but not with neutral or positive faces. Therefore, our results imply that in the early frontal processing stage, patients with amnestic MCI are still able to make personalized processing for negative faces, but not for neutral faces or positive faces. This indicates that emotional recognition and memory processing is impaired in patients with amnestic MCI and that these impairments in the early recognition process are selective for neutral and positive emotional faces. In other words, the advantageous negative emotional effect in the early recognition process is preserved in patients with amnestic MCI.
In addition, hits show an emotional effect on P2, since emotional stimuli can produce automatic visual attention.56–58 Schefter et al11 suggest that the recognition effect of hits with negative emotional faces might result from a one-way or reciprocal influence in the processing stage.
ERP old/new effect
ERPs are sensitive to neural activity underlying recognition. Measuring ERPs during the recognition test, a study/test paradigm has been a fruitful research strategy when examining the effects of emotion on recognition memory. Generally correctly recognized “old” items elicit more positive moving waveforms than correctly rejected “new” items. This so-called “ERP old/new effect” consists of several temporally and spatially dissociable subcomponents.58–61
The first subcomponent is an early frontal old/new effect (300–500 milliseconds) that has often been associated with familiarity.62–64 It has been suggested that the early frontal old/new effect may reflect implicit conceptual memory.65–67 In young adults, the early frontal old/new effect is largely free from emotional stimuli,68–72 but an early frontal old/new effect can be elicited by emotional stimuli in elderly people; neutral image cannot be drawn.69
In the early time window, an old/new effect could be observed with both negative and positive faces in the two tested groups, but not for neutral faces. For patients with amnestic MCI, no difference in frontal old/new effect was found in comparison with normal controls.
The second subcomponent is a parietal old/new effect (500–800 milliseconds), which is related to recollection.14,64,73,74 Among the young subjects, compared with neutral stimuli, emotional stimuli would enhance this effect.68–72,75,76 It has been pointed out that when tested subjects fail a recollection task, the old/new effect became weakened or absent.77
In the present study, emotional stimuli (positive faces and negative faces) led to a parietal old/new effect in the control group. However, neutral stimuli did not lead to the same effects, which is consistent with previous studies suggesting that non-emotional information has no parietal old/new effect.78
In the current study, we found that three types of stimuli (emotional and neutral faces) did not elicit any parietal old/new effect in patients with amnestic MCI. Since the parietal old/new effect is an indicator of recollection,58–61 our results suggest impairment of recollection in patients with amnestic MCI. Previous behavioral data support the idea that recollection is impaired in patients with amnestic MCI.19–21,23,79,80 This impairment may be closely associated with amnestic MCI, and the data strongly suggest that patients with amnestic MCI have difficulties in the late stage of emotional processing. Moreover, at this stage, the advantageous effect of negative face recognition even disappeared. This is not supported by a previous report11 in which patients with amnestic MCI demonstrated a parietal old/new effect with negative faces. However, in that study, the old pictures were repeatedly presented in the learning stage, and the difference between that study and ours does suggest that a parietal old/new effect could be induced with intense stimuli in patients with amnestic MCI.
Conclusion
Our results show that in patients with amnestic MCI, face recognition was impaired for negative faces in terms of face structural encoding. However, emotional stimuli can enhance episodic memory in patients with amnestic MCI. The impairment of emotional face recognition in patients with amnestic MCI mainly occurred during the stage of structural encoding and in the early recognition stage when the stimuli were individualized. In addition, we found difficulties in the late stage of emotional processing in patients with amnestic MCI.
Acknowledgment
This study was supported by the National Natural Science Foundation of China (81271489), the Hebei Province Natural Science Foundation of China (H2014206280), and the Major Projects of Hebei Science and Technology Support Program (09276103D).
Disclosure
The authors report no conflicts of interest in this work.
References
Petersen RC, Smith GE, Waring SC, Ivnik RJ, Tangalos EG, Kokmen E. Mild cognitive impairment: clinical characterization and outcome. Arch Neurol. 1999;56:303–308. | ||
Petersen RC. Mild cognitive impairment as a diagnostic entity. J Intern Med. 2004;256:183–194. | ||
Winblad B, Palmer K, Kivipelto M, et al. Mild cognitive impairment – beyond controversies, towards a consensus: report of the International Working Group on Mild Cognitive Impairment. J Intern Med. 2004;256:240–246. | ||
Kensinger EA, Corkin S. Memory enhancement for emotional words: are emotional words more vividly remembered than neutral words? Mem Cognit. 2003;31:1169–1180. | ||
Markesbery WR, Schmitt FA, Kryscio RJ, Davis DG, Smith CD, Wekstein DR. Neuropathologic substrate of mild cognitive impairment. Arch Neurol. 2006;63:38–46. | ||
Fujie S, Namiki C, Nishi H, et al. The role of the uncinate fasciculus in memory and emotional recognition in amnestic mild cognitive impairment. Dement Geriatr Cogn Disord. 2008;26:432–439. | ||
Spoletini I, Marra C, Di Iulio F, et al. Facial emotion recognition deficit in amnestic mild cognitive impairment and Alzheimer disease. Am J Geriatr Psychiatry. 2008;16:389–398. | ||
Teng E, Lu PH, Cummings JL. Deficits in facial emotion processing in mild cognitive impairment. Dement Geriatr Cogn Disord. 2007;23:271–279. | ||
Weiss EM, Kohler CG, Vonbank J, et al. Impairment in emotion recognition abilities in patients with mild cognitive impairment, early and moderate Alzheimer disease compared with healthy comparison subjects. Am J Geriatr Psychiatry. 2008;16:974–980. | ||
Werheid K, Gruno M, Kathmann N, Fischer H, Almkvist O, Winblad B. Biased recognition of positive faces in aging and amnestic mild cognitive impairment. Psychol Aging. 2010;25:1–15. | ||
Schefter M, Werheid K, Almkvist O, Lönnqvist-Akenine U, Kathmann N, Winblad B. Recognition memory for emotional faces in amnestic mild cognitive impairment: an event-related potential study. Neuropsychol Dev Cogn B Aging Neuropsychol Cogn. 2013;20:49–79. | ||
Bötzel K, Grüsser OJ. Electric brain potentials – evoked by pictures of faces and non-faces – a search for face-specific EEG-potentials. Exp Brain Res. 1989;77:349–360. | ||
Joyce C, Rossion B. The face-sensitive N170 and VPP components manifest the same brain processes: the effect of reference electrode site. Clin Neurophysiol. 2005;116:2613–2631. | ||
Wilding EL. In what way does the parietal ERP old/new effect index recollection? Int J Psychophysiol. 2000;35:81–87. | ||
Schefter M, Knorr S, Kathmann N, Werheid K. Age difference on ERP old/new effects for emotional and neutral faces. Int J Psychophysiol. 2012;85:257–269. | ||
Ochsner KN. Are affective events richly recollected or simply familiar? The experience and process of recognizing feelings past. J Exp Psychol Gen. 2000;129:242–261. | ||
Burton LA, Rabin L, Wyatt G, Frohlich J, Bernstein Vardy S, Dimitri D. Priming effects for affective vs neutral faces. Brain Cogn. 2005;59:322–329. | ||
LaBar KS, Torpey DC, Cook CA, et al. Emotional enhancement of perceptual priming is preserved in aging and early-stage Alzheimer’s disease. Neuropsychologia. 2005;43:1824–1837. | ||
Ally BA, Gold CA, Budson AE. An evaluation of recollection and familiarity in Alzheimer’s disease and mild cognitive impairment using receiver operating characteristics. Brain Cogn. 2009;69:504–513. | ||
Anderson ND, Ebert PL, Jennings JM, Grady CL, Cabeza R, Graham SJ. Recollection- and familiarity-based memory in healthy aging and amnestic mild cognitive impairment. Neuropsychology. 2008;22:177–187. | ||
Hudon C, Belleville S, Gauthier S. The assessment of recognition memory using the remember/know procedure in amnestic mild cognitive impairment and probable Alzheimer’s disease. Brain Cogn. 2009;70:171–179. | ||
O’Connor MK, Ally BA. Using stimulus form change to understand memorial familiarity for pictures and words in patients with mild cognitive impairment and Alzheimer’s disease. Neuropsychologia. 2010;48:2068–2074. | ||
Westerberg CE, Paller KA, Weintraub S, et al. When memory does not fail: familiarity-based recognition in mild cognitive impairment and Alzheimer’s disease. Neuropsychology. 2006;20:193–205. | ||
Ekman P. Strong evidence for universals in facial expressions: a reply to Russell’s mistaken critique. Psychol Bull. 1994;115:268–287. | ||
Calvo MG, Beltrán D. Recognition advantage of happy faces: tracing the neurocognitive processes. Neuropsychologia. 2013;51:2051–2061. | ||
Spaniol J, Voss A, Grady CL. Aging and emotional memory: cognitive mechanisms underlying the positivity effect. Psychol Aging. 2008;23:859–872. | ||
Charles ST, Mather M, Carstensen LL. Aging and emotional memory: the forgettable nature of negative images for older adults. J Exp Psychol Gen. 2003;132:310–324. | ||
Knight BG, Maines ML, Robinson GS. The effects of sad mood on memory in older adults: a test of the mood congruence effect. Psychol Aging. 2002;17:653–661. | ||
Leigland LA, Schulz LE, Janowsky JS. Age related changes in emotional memory. Neurobiol Aging. 2004;25:1117–1124. | ||
Thomas RC, Hasher L. The influence of emotional valence on age differences in early processing and memory. Psychol Aging. 2006;21:821–825. | ||
Wang Y, Luo YJ. | ||
Folstein MF, Folstein SE, McHugh PR. “Mini-mental state”. A practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res. 1975;12:189–198. | ||
Nasreddine ZS, Phillips NA, Bédirian V, et al. The Montreal Cognitive Assessment, MoCA: a brief screening tool for mild cognitive impairment. J Am Geriatr Soc. 2005;53:695–699. | ||
Lawton MP, Brody EM. Assessment of older people: self-maintaining and instrumental activities of daily living. Gerontologist. 1969;9:179–186. | ||
Radloff LS. The CES-D scale: a self-report depression scale for research in the general population. Appl Psychol Meas. 1977;1:385–401. | ||
Snodgrass JG, Corwin J. Pragmatics of measuring recognition memory: applications to dementia and amnesia. J Exp Psychol Gen. 1988;117:34–50. | ||
Balconi M, Pozzoli U. Face-selective processing and the effect of pleasant and unpleasant emotional expressions on ERP correlates. Int J Psychophysiol. 2003;49:67–74. | ||
Sato W, Kochiyama T, Yoshikawa S, Matsumura M. Emotional expression boosts early visual processing of the face: ERP recording and its decomposition by independent component analysis. Neuroreport. 2001;12:709–714. | ||
Schupp HT, Junghöfer M, Weike AI, Hamm AO. The selective processing of briefly presented affective pictures: an ERP analysis. Psychophysiology. 2004;41:441–449. | ||
Brueckner K, Moritz S. Emotional valence and semantic relatedness differentially influence false recognition in mild cognitive impairment, Alzheimer’s disease, and healthy elderly. J Int Neuropsychol Soc. 2009;15:268–276. | ||
Döhnel K, Sommer M, Ibach B, Rothmayr C, Meinhardt J, Hajak G. Neural correlates of emotional working memory in patients with mild cognitive impairment. Neuropsychologia. 2008;46:37–48. | ||
Cheng PJ, Pai MC. Dissociation between recognition of familiar scenes and of faces in patients with very mild Alzheimer disease: an event-related potential study. Clin Neurophysiol. 2010;121:1519–1525. | ||
Herzmann G, Sommer W. Effects of previous experience and associated knowledge on retrieval processes of faces: an ERP investigation of newly learned faces. Brain Res. 2010;1356:54–72. | ||
Marzi T, Viggiano MP. Deep and shallow encoding effects on face recognition: an ERP study. Int J Psychophysiol. 2010;78:239–250. | ||
Bentin S, Allison T, Puce A, Perez E, McCarthy G. Electrophysiological studies of face perception in humans. J Cogn Neurosci. 1996;8:551–565. | ||
Eimer M. Event-related brain potentials distinguish processing stages involved in face perception and recognition. Clin Neurophysiol. 2000;111:694–705. | ||
Jacques C, d’Arripe O, Rossion B. The time course of the inversion effect during individual face discrimination. J Vis. 2007;7:3. | ||
Rossion B, Gauthier I. How does the brain process upright and inverted faces? Behav Cogn Neurosci Rev. 2002;1:63–75. | ||
Caharel S, Poiroux S, Bernard C, Thibaut F, Lalonde R, Rebai M. ERPs associated with familiarity and degree of familiarity during face recognition. Int J Neurosci. 2002;112:1499–1512. | ||
Caharel S, Fiori N, Bernard C, Lalonde R, Rebaï M. The effects of inversion and eye displacements of familiar and unknown faces on early and late-stage ERPs. Int J Psychophysiol. 2006;62:141–151. | ||
Batty M, Taylor MJ. Early processing of the six basic facial emotional expressions. Brain Res Cogn Brain Res. 2003;17:613–620. | ||
Frühholz S, Jellinghaus A, Herrmann M. Time course of implicit processing and explicit processing of emotional faces and emotional words. Biol Psychol. 2011;87:265–674. | ||
Leleu A, Caharel S, Carré J, et al. Perceptual interactions between visual processing of facial familiarity and emotional expression: an event-related potentials study during task-switching. Neurosci Lett. 2010;482:106–111. | ||
Vuilleumier P, Schwartz S. Emotional facial expressions capture attention. Neurology. 2001;56:153–158. | ||
Schendan HE, Kutas M. Neurophysiological evidence for transfer appropriate processing of memory: processing versus feature similarity. Psychon Bull Rev. 2007;14:612–619. | ||
Halit H, de Haan M, Johnson MH. Modulation of event-related potentials by prototypical and atypical faces. Neuroreport. 2000;11:1871–1875. | ||
Wiese H, Schweinberger SR, Hansen K. The age of the beholder: ERP evidence of an own-age bias in face memory. Neuropsychologia. 2008;46:2973–2985. | ||
Curran T, Hancock J. The FN400 indexes familiarity-based recognition of faces. Neuroimage. 2007;36:464–471. | ||
Friedman D, Johnson R Jr. Event-related potential (ERP) studies of memory encoding and retrieval: a selective review. Microsc Res Tech. 2000;51:6–28. | ||
Mecklinger A. Interfacing mind and brain: a neurocognitive model of recognition memory. Psychophysiology. 2000;37:565–582. | ||
Rugg MD, Curran T. Event-related potentials and recognition memory. Trends Cogn Sci. 2007;11:251–257. | ||
Curran T. Brain potentials of recollection and familiarity. Mem Cognit. 2000;28:923–938. | ||
Nessler D, Mecklinger A, Penney TB. Event related brain potentials and illusory memories: the effects of differential encoding. Brain Res Cogn Brain Res. 2001;10:283–301. | ||
Woodruff CC, Hayama HR, Rugg MD. Electrophysiological dissociation of the neural correlates of recollection and familiarity. Brain Res. 2006;1100:125–135. | ||
Paller KA, Voss JL, Boehm SG. Validating neural correlates of familiarity. Trends Cogn Sci. 2007;11:243–250. | ||
Voss JL, Paller KA. Neural correlates of conceptual implicit memory and their contamination of putative neural correlates of explicit memory. Learn Mem. 2007;14:259–267. | ||
Voss JL, Lucas HD, Paller KA. Conceptual priming and familiarity: different expressions of memory during recognition testing with distinct neurophysiological correlates. J Cogn Neurosci. 2010;22:2638–2651. | ||
Johansson M, Mecklinger A, Treese AC. Recognition memory for emotional and neutral faces: an event-related potential study. J Cogn Neurosci. 2004;16:1840–1853. | ||
Langeslag SJ, Van Strien JW. Age differences in the emotional modulation of ERP old/new effects. Int J Psychophysiol. 2008;70:105–114. | ||
Maratos EJ, Rugg MD. Electrophysiological correlates of the retrieval of emotional and non-emotional context. J Cogn Neurosci. 2001;13:877–891. | ||
Maratos EJ, Allan K, Rugg MD. Recognition memory for emotionally negative and neutral words: an ERP study. Neuropsychologia. 2000;38:1452–1465. | ||
Windmann S, Kutas M. Electrophysiological correlates of emotion-induced recognition bias. J Cogn Neurosci. 2001;13:577–592. | ||
Paller KA, Kutas M. Brain potentials during memory retrieval provide neurophysiological support for the distinction between conscious recollection and priming. J Cogn Neurosci. 1992;4:375–392. | ||
Trott CT, Friedman D, Ritter W, Fabiani M, Snodgrass JG. Episodic priming and memory for temporal source: event-related potentials reveal age-related differences in prefrontal functioning. Psychol Aging. 1999;14:390–413. | ||
Dietrich DE, Waller C, Johannes S, Wieringa BM, Emrich HM, Münte TF. Differential effects of emotional content on event-related potentials in word recognition memory. Neuropsychobiology. 2001;43:96–101. | ||
Inaba M, Nomura M, Ohira H. Neural evidence of effects of emotional valence on word recognition. Int J Psychophysiol. 2005;57:165–173. | ||
Tendolkar I, Schoenfeld A, Golz G, et al. Neural correlates of recognition memory with and without recollection in patients with Alzheimer’s disease and healthy controls. Neurosci Lett. 1999;263:45–48. | ||
Ally BA, McKeever JD, Waring JD, Budson AE. Preserved frontal memorial processing for pictures in patients with mild cognitive impairment. Neuropsychologia. 2009;47:2044–2055. | ||
Algarabel S, Escudero J, Mazón JF, et al. Familiarity-based recognition in the young, healthy elderly, mild cognitive impaired and Alzheimer’s patients. Neuropsychologia. 2009;47:2056–2064. | ||
Wolk DA, Signoff ED, Dekosky ST. Recollection and familiarity in amnestic mild cognitive impairment: a global decline in recognition memory. Neuropsychologia. 2008;46:1965–1978. |
 © 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.







 [Standardization and assessment of college students facial expression of emotion]. Chinese Journal of Clinical Psychology. 2005;13:396–398. Chinese.
[Standardization and assessment of college students facial expression of emotion]. Chinese Journal of Clinical Psychology. 2005;13:396–398. Chinese.