Back to Journals » OncoTargets and Therapy » Volume 13
Emerging Targets of Immunotherapy in Gynecologic Cancer
Authors Cheng H , Zong L, Kong Y , Gu Y, Yang J, Xiang Y
Received 18 September 2020
Accepted for publication 31 October 2020
Published 18 November 2020 Volume 2020:13 Pages 11869—11882
DOI https://doi.org/10.2147/OTT.S282530
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Sanjay Singh
Hongyan Cheng,1 Liju Zong,1,2 Yujia Kong,1 Yu Gu,1 Junjun Yang,1 Yang Xiang1
1Department of Obstetrics and Gynecology, Peking Union Medical College Hospital, Chinese Academy of Medical Sciences and Peking Union Medical College, Beijing, People’s Republic of China; 2Department of Pathology, Peking Union Medical College Hospital, Chinese Academy of Medical Sciences and Peking Union Medical College, Beijing, People’s Republic of China
Correspondence: Yang Xiang
Department of Obstetrics and Gynecology, Peking Union Medical College Hospital, Chinese Academy of Medical Sciences and Peking Union Medical College, No. 1 Shuaifuyuan, Wangfujing, Dongcheng District, Beijing 100730, People’s Republic of China
Tel/Fax +86 1069154022
Email [email protected]
Abstract: Although programmed cell death protein 1/programmed death-ligand 1 (PD-1/PD-L1) and cytotoxic T lymphocyte antigen-4 (CTLA-4) have been successfully applied in the treatment of tumors, their efficiency is still not high enough. New immune targets need to be identified in order to seek alternative treatment strategies for patients with refractory tumors. Immune targets can be divided into stimulating and inhibiting molecules according to their function after receptor–ligand binding. We herein present a compendious summary of emerging immune targets in gynecologic tumors. These targets included coinhibitory molecules, such as T cell immunoglobulin-3 (TIM-3), T cell immunoglobulin and ITIM domain (TIGIT), lymphocyte activation gene-3 (LAG-3), V-type immunoglobulin domain-containing suppressor of T cell activation (VISTA), and B7-H3 and B7-H4, and co-stimulatory molecules, such as CD27, OX40, 4– 1BB, CD40, glucocorticoid-induced tumor necrosis factor receptor (GITR) and inducible co-stimulator (ICOS). In this review, the characteristics and preclinical/clinical progress of gynecological malignancies are briefly discussed. However, the potential mechanisms and interactions of immune targets need to be elucidated in further studies.
Keywords: immunotherapy, gynecologic neoplasms, T cell receptors, antigen presenting cells, molecular targeted therapy
Introduction
Immunotherapy has joined the ranks of surgery, radiotherapy, chemotherapy, and targeted therapy as an important pillar of cancer treatment.1 Inhibitors that target programmed cell death protein 1/programmed death-ligand 1 (PD-1/PD-L1) and cytotoxic T lymphocyte antigen-4 (CTLA-4) are now moving from second-line to first-line therapy, greatly changing the traditional strategy for advanced melanoma and non-small cell lung cancer.2,3 Numerous studies have been performed on the application of PD-1/PD-L1 and CTLA-4 inhibitors in advanced gynecologic cancer. However, the reported clinical benefits are still limited. Notably, because of the constant evolution and interaction of the immune signaling pathway and immune environment, it is unlikely to obtain antitumor effects using a single immune-targeted agent.1 Therefore, it is imperative to understand the tumor immunity pathway and seek new immune targets for gynecologic cancer. In this review, we describe the following emerging targets of immunotherapy in gynecologic cancer on the basis of development in preclinical/clinical studies and a limited number of review articles available. Figure 1 summarizes the targets associated with tumor immunity through a blockade of immune inhibitory checkpoints or stimulatory molecules.
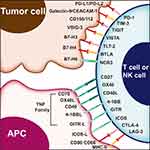 |
Figure 1 Immune targets in T cell, antigen presenting cells and tumor cell. |
Co-Inhibitory Molecules
PD-1
PD-1 is an important immune check point receptor which predominantly expressed on activated T cells, natural killer (NK) cells, and B cells.4,5 The two known ligands of PD-1 are PD-L1 (B7-H1) and PD-L2 (B7-DC). PD-L1 is constitutively expressed on a subset of macrophages and tumor cells, but may be rapidly upregulated by tumors in response to inflammatory mediators.6 PD-L2 is expressed on macrophages and dendritic cells, but its impact on T cell immunity still not clear.7 The ligation of PD-1/PD-L1 serves as a negative regulator of T cells and B cells immunity.
The expression of PD-L1 has been detected in 68.5% of ovarian cancer,8 96% of advanced cervical cancer,9 and 36.2% of endometrial cancer,10 In the KEYNOTE-028 Phase 1b study which was designed to evaluated the safety and efficacy of pembrolizumab (PD-1 inhibitor) in patients with PD-1 positive advanced solid tumors, the report overall response rate (ORR) is only 11.5% in 26 pre-treated ovarian cancer patients,11 17% in 24 advanced cervical cancer patients,12 and 13% in 24 locally advanced or metastatic endometrial cancer patients.13 The results from the JAVELIN Solid Tumor Trial of avelumab (PD-1 inhibitor) in 125 patients with refractory/recurrent ovarian cancer reported an ORR of 9.6%.14 In another Phase 2 KEYNOTE-158 study, the ORR of pembrolizumab in previously treated advanced cervical cancer is only 12.2%.15 A number of agents targeting both sides of the PD-1/PD-L1 interaction are currently used to treat gynecologic tumors, such as pembrolizumab, nivolumab, and avelumab. There are dozens of ongoing clinical trials of PD-1/PD-L1 in gynecologic tumors.
TIM-3
T cell immunoglobulin-3 (TIM-3) is a type I transmembrane protein that was first identified on interferon-γ (IFN-γ) secreting cells, including CD4+ T helper 1 (Th1) cells and CD8+ T cytotoxic 1 (Tc1) cells, and then later on Th17 cells, dendritic cells, monocytes, regulatory T cells (Tregs), NK cells, and tumor-infiltrating lymphocytes (TILs).16,17 TIM-3 plays an immunosuppressive role by binding to its ligands, galectin-9 and carcinoembryonic antigen-related cell adhesion molecule-1 (CEACAM-1), to promote calcium influx, cell aggregation, and apoptosis, and further inhibiting T cell activation and proliferation.18 CEACAM-1 is co-expressed with TIM-3 on T cells and their interaction is required for the inhibitory function of TIM-3.19
TIM-3 expression is considered one of the markers of T cell exhaustion.20 High expression of TIM-3 has been detected in TILs of ovarian cancer and cervical cancer. Yan et al21 showed that the proportion of TIM-3+CD4+T cells in ovarian cancer and cervical cancer tissues is significantly higher than that in patients’ peripheral blood and non-TILs. Tumor-derived TIM-3+ CD4 T cells exhibit an impaired capacity to produce IFN-γ and interleukin-2 (IL-2) but express higher levels of CD25 and CTLA-4 and lower levels of CD25, Foxp3 and CTLA-4 than those of their TIM-3− CD4+ T cell counterparts. Additionally, the expression of TIM-3 on CD4+ and CD8+ T cells in advanced ovarian cancer is significantly higher than that of early ovarian cancer. Meanwhile, the level of TIM-3+ CD4+ T cells in the peripheral blood of patients with recurrent ovarian cancer is remarkably higher than that of newly diagnosed ovarian cancer. TIM-3 expression in ovarian cancer tissues is associated with poor prognosis.22,23 Furthermore, the combination of anti-TIM-3 antibody and anti-CD137 antibody inhibits tumor growth in ovarian cancer tumor-bearing mice, with about 60% of tumor-bearing mice being able to achieve long-term survival.24 A study has showed that, in cervical cancer, the expression of TIM-3 in tumor metastatic lymph nodes is significantly higher than that in normal lymph nodes.25 In endometrial cancer, a study showed that tumor resident NK cells exhibits more co-inhibitory molecules such as TIM-3 and TIGIT compared to recruited ND cells, and that the expression of these molecules increases with the severity of the disease.26 Multiple Phase I clinical trials are currently testing anti-TIM-3 antibody alone or in combination with anti PD-1 therapy for the treatment of cervical and ovarian cancer (INCAGN02390) and advanced recurrent solid tumors (Table 1).
 |  |  |
Table 1 An Overview of Clinical Trials in Gynecologic Cancer and/or Solid Cancer for Immune Target Agents |
TIGIT
T cell immunoglobulin and immunoglobulin and immunoreceptor tyrosine-based inhibitory motif (ITIM) domain (TIGIT) is a T cell immunoreceptor, which was first identified as an immune checkpoint molecule of T cells and NK cells in 2009.27 TIGIT can be detected on memory T cells, Tregs, and natural killer T (NKT) cells. TIGIT competes with immunoactivator CD226 or DNAX accessory molecule-1 (DNAM-1) for the same set of ligands: CD155 [poliovirus receptor (PVR)] and CD122 [Nectin-2 or poliovirus receptor-related 2(PVRL2)].28 TIGIT binds with high affinity to PVR and weakly interacts with PVRL2.29 Engagement of TIGIT and CD155 dampens T cell responses and shifts the cytokine production from IL-12 to IL-10. However, the TIGIT-CD112 interaction is not very strong, and its role in cancer remains unknown.30
The expression of TIGIT and its ligands has been found in multiple gynecologic cancer tissues. In a study by Oshima et al,31 high expression of Nectin-2, one of the ligands of TIGIT, was found on the surface of the ovarian cancer cell line OV-90 as well as in 48.4% of ovarian cancer tissue samples using gene expression profile analysis and immunohistochemistry. Moreover, it was observed that the anti-Nectin-2 antibody suppresses the in vitro proliferation of OV-90 cells, and that the anti-Nectin-2 monoclonal antibody (mAb) exhibits anti-tumor effects against OV-90 cells in mouse therapeutic models through an antibody-dependent cellular cytotoxicity mechanism. In a study by Textor et al32 the authors found that the expression of CD155 in cervical cancer tissues is higher than that in intraepithelial neoplasia and normal cervical tissues. The expression of CD155 might enhance tumor cell motility and, hence, aid the progressive growth of tumor cells. In patients with endometrial cancer, lymph node invasion is associated with a high expression of inhibitory molecules, such as TIGIT and TIM-3. Exhaustion hallmarks such as TIM-3 and TIGIT are associated with advanced diseases.26 Notably, TIGIT antagonists are still in early phase study; however, at least three agents (AB154, BGB-A1217, and MK7684) are being investigated in human solid tumors (Table 1).
LAG-3
Lymphocyte activation gene-3 (LAG-3) was first reported by Triebel et al33 in 1990. Structurally resembling the CD4 co-receptor, LAG-3 binds main histocompatibility complex class II (MHC II) with a high affinity. Hence, MHC II is considered a typical ligand of LAG-3. Recently, in a study by Wang et al,34 it was found that fibrinogen-like protein 1 (FGL1) plays a more important role than that of MHC II as a ligand of LAG-3. Notably, LAG-3 expression was detected on the surface of T cells and NK cells, which is induced by T cell activation. Moreover, the blockade of LAG-3 improves cytotoxic T lymphocyte (CTL) proliferation and effector function.35,36
Studies have shown that LAG-3 plays an important role in the development of ovarian and cervical cancer. Tu et al37 analyzed the Oncomine and PrognoScan databases and found that LAG-3, PD-1, CTLA-4, and TIM-3 may potentially serve as prognostic factors and therapeutic targets in ovarian cancer. In cervical cancer, the level of LAG-3 in metastatic tumor-draining lymph nodes is significantly higher than that in normal lymph nodes.25 On the other hand, in epithelial ovarian cancer, tumor-derived CD8+ T cells demonstrated impaired effector function and enriched co-expression of LAG-3 and PD-1. The expression of LAG-3 and PD-1 is upregulated by IL-10, IL-6, and tumor-derived antigen presenting cells (APCs). Dual blockade of LAG-3 and PD-1 efficiently augments proliferation and cytokine production by CD8+ T cells.38 Multiple early phase clinical trials investigating anti-LAG-3 agents alone or in combination with anti-PD-1 in gynecologic cancer are currently underway (Table 1).
VISTA
V-type immunoglobulin domain-containing suppressor of T cell activation (VISTA) is also known as C10orf54, B7-H5, or precursor of platelet receptor Gi24. The extracellular Ig domain of VISTA is homologous to the B7 family ligands PD-L1 and PD-L2.39 VISTA is predominantly expressed in myeloid cells, monocytes, macrophages and dendritic cells. Within T lymphocytes, VISTA is most highly expressed on naïve CD4+ and FoxP3+ Tregs.39,40 Notably, VISTA functions as both a ligand and a receptor separately, but these roles are not mutually exclusive.41 A recent study has shown that V-set and immunoglobin domain containing 3 (VSIG-3) is a ligand of VISTA and that the VSIG-3/VISTA pathway significantly reduces the production of cytokines and chemokines by human T cells.42 The ligand function of VISTA was first described by Wang et al,39 who found that VISTA Ig suppresses the production of IL-2 and IFN-γ by stimulated T cells. Hence, it appears that VISTA negatively regulates T cell responses by suppressing early T cell receptor activation and arresting cell division, but with minimal direct impact on apoptosis. However, the receptor of VISTA has not yet been identified.
VISTA is highly expressed in human ovarian and endometrial cancer, but absent or low in normal ovarian epithelial specimens and normal endometrial specimens. In a study by Mulati et al,43 it was found that VISTA in tumor cells suppresses T cell proliferation and cytokine production in vitro, and that silencing the expression of VISTA in human endometrial cancer cells and ovarian cancer cells restores T cell proliferation and cytokine secretion. It was also found that anti-VISTA antibody prolongs the survival of tumor-bearing mice. Immunohistochemistry revealed that no difference in the expression of VISTA between primary and metastatic sites in ovarian cancer. Likewise, in a study by Liao et al, the authors analyzed 65 ovarian cancer specimens and concluded that high VISTA expression on tumor or immune cells is significantly associated with advanced tumor stage and the presence of lymph node metastasis.44 In contrast, Zong et al45 assessed 146 ovarian cancer samples and found that 51.4% of the samples was exhibited VISTA expression and that VISTA was expressed in 28.8%, 35.6%, and 4.1% of tumor cells, immune cells, and endothelial cells, respectively. Notably, VISTA expression in tumor cells is associated with prolonged overall survival. In a study by Kuang et al on cervical cancer,46 the authors found that a total of 32 out of 104 cases (30.76%) exhibited VISTA expression in tumor cells, with only 11 out of the 104 cases (10.57%) being defined as having high VISTA expression. Survival analysis indicated that high expression of VISTA in immune and vascular endothelial cells is significantly associated with advanced tumor stage and lymph node metastasis. Additionally, a recently published study has shown that VISTA is widely overexpressed in 98.2% of all gestational trophoblastic neoplasia (GTN) tissues but negative in normal adult and fetal tissues.47
To date, only two phase I clinical trials of anti-VISTA agents (Table 1) have been performed: one for CA-170, an oral small-molecule dual antagonist that selectively targets PD-1 and VISTA (NCT02812875), and the other for JNJ-61,610,588, an ant-VISTA mAb developed by Jassen Company (NCT02671955). In the phase I clinical study regarding JNJ-61,610,588 (NCT02671955), 12 patients were recruited, one of whom developed dose-limiting adverse effects associated with cytokine release syndrome. Therefore, the company decided to terminate the clinical trial.
B7-H3 and B7-H4
B7-H3 (CD276) and B7-H4 (B7x or B7S1) are members of the immune regulatory ligand of the B7 family. The potential receptors of B7-H3 and B7-H4 are considered to be triggering receptors expressed on myeloid cells (TREM)-like transcript 2 (TLT-2, TREML2)48 and the B and T lymphocyte attenuator,49 respectively. Although their receptors have yet to be definitively identified, they are believed to be involved in both co-stimulatory and co-inhibitory pathways.50,51 The mRNA of these two ligands can be found in most peripheral tissues, but the protein level of these two ligands is not constitutively expressed. B7-H3 and B7-H4 have been found to be overexpressed in various tumors, including lung, breast, pancreatic, and ovarian cancer.50,52
It was also found that the expression of B7-H3 in high-grade tumors and type II endometrial cancer is higher than that in low-grade tumors and endometrioid adenocarcinoma. In addition, the expression of B7-H3 in endometrial tumor cells is correlated with TILs and negatively associated with the overall survival of patients.53 In patients with GTN, B7-H3 is highly expressed in almost all types of tumors but is not correlated with the clinical outcome.47 Notably, B7-H4 cytoplasmic and membranous expression has been detected in serous, endometrioid, and clear cell carcinoma, and in all types of metastatic serous and endometrioid ovarian carcinoma, whereas normal tissue is consistently negative for B7-H4.54
Except for individual expression, B7-H3 and B7-H4 have also been found to be co-expressed in multiple gynecologic tumors. In a study by Zang et al,55 it was found that B7-H3 and B7-H4 were expressed in 93% and 100% of ovarian tumors in 103 samples of ovarian borderline tumors and ovarian cancer. Han et al56 found that both B7-H3 and B7-H4 are highly expressed in cervical cancer and are associated with poor prognosis and that their expression favors an immunosuppressive microenvironment by promoting the production of IL-10 and transforming growth factor-beta (TGF-β), thereby resulting in the progression of cervical carcinogenesis. However, all the ongoing clinical trials of B7-H3 and B7-H4 are still in the early stage (Table 1).
Co-Stimulatory Molecules
TNF Receptor Superfamily
CD27
The tumor necrosis factor (TNF) receptor superfamily contains death receptors, and receptors that bind TNF receptor-associated factors (Traf).57 CD27 is a Traf-linked receptor, that is expressed exclusively on lymphocytes. Even naïve CD4+ and CD8+ T cells express low levels of CD27.51,58 Notably, the interaction between CD27 and its ligand, CD70, may promote the differentiation of CD8+ T cells to effector CTLs, and contribute to T cell expansion and B cell differentiation through T-B cell and/or B-B cell interactions.59
Studies of CD27/CD70 in gynecologic tumors have only been reported in ovarian cancer. Aggarwal et al60 and Kochet al61 found that CD70 is highly expressed in ovarian cancer tissues, with no expression whatsoever in normal ovarian tissues. In addition, it has been found that CD70 is overexpressed at the mRNA and protein levels in the drug-resistant ovarian cancer cell line A2780. After CD70 antibody co-cultivation, the proliferation of cisplatin-resistant cells was found to decrease significantly. In a mouse model, mice with overexpressed CD70 exhibited a stronger anti-tumor response to CD8+ T cells than that exhibited by wild type mice.62
Recently, the American Society of Clinical Oncology (ASCO) reported the results of a phase 1/2 clinical trial of Varlilumab, a CD27 agonist mAb, combined with a PD-1 inhibitor in advanced ovarian cancer (NCT02335918). In total, 66 patients were included in that trial, 49 of whom were assessable. Among these, five cases (10%) achieved partial response and 19 cases (39%) achieved stable disease. The results suggested that Varlilumab combined with a PD-1 inhibitor exhibits a significant anti-tumor effect in advanced ovarian cancer, which may have prognostic benefits for such patients. Several clinical trials are currently underway investing the efficacy of Varlilumab combined with ONT-10, a cancer vaccine, for patients with advanced ovarian and breast cancer (Table 1).
OX40
OX40 (CD134), a member of the TNF superfamily, is a 50 kDa glycoprotein that has a cytoplasmic tail, a transmembrane domain, and an extracellular region.63 OX40 is mainly detected on active effector CD4+ T cell and NKT cells, especially in Tregs.64 One known ligand of OX40 is OX40L (CD252), which is expressed on active APCs, including dendritic cells, B cells, and macrophages.64,65 Tregs can inhibit the secretion of immunosuppressive cytokines (e.g. IL-10 and TGF-β) of effector T cells. Notably, OX40/OX40L ligation can counteract the negative regulation of Tregs and impair their suppressive ability.66
In a study conducted by Ramser et al,67 the authors evaluated 47 samples of high-grade serous ovarian cancer and found that high expression of OX40 in ovarian cancer may be associated with good chemosensitivity and prognosis. In a mouse ovarian cancer model, treatment using a PD-1 inhibitor or OX40 antibody alone had no clear effect on tumor control. However, combining a PD-1 inhibitor and OX40 antibody had a significant inhibitory effect on tumor growth, and 60% of the mice became tumor-free after 90 days of tumor inoculation.68 Notably, OX40/OX40L expression on the RNA level has only been reported in one study. Zhao et al69 investigated samples from 30 cases of cervical cancer and 20 cases of normal cervical tissues and found that the expression level of OX40/OX40L mRNA in the microenvironment of cervical cancer is remarkably lower than that in normal cervical tissues and that the mRNA level in high-grade squamous cell carcinoma (SCC) is slightly lower than that in low grade SCC. Notably, a Phase II clinical umbrella trial has been performed on an OX40 agonist mAb for ovarian cancer (NCT03267589), as well as multiple clinical trials of advanced solid tumors (Table 1).
4-1BB
4–1BB, also known as CD137, is a member of the TNF receptor family that is predominantly expressed on active T cells and APCs.70 4–1BB is not constitutively expressed on resting T cells and is induced within 24 h after activation, peaking several days after the initial stimulation. Similar to OX40L, the 4–1BB ligand can be expressed in APCs and induced again hours or days after activation. Thus, the expressions of both OX40/4-1BB and OX40L/4-1BBL can parallel each other under certain conditions on activated T cells and APCs, suggesting that some similarities may exist in their roles.71
In ovarian cancer, 4–1BB has been investigated in combination with other immune checkpoint agents. In a study by Wei et al,72 it was found that combined CD137 co-stimulation and PD-1 co-inhibition molecular therapy can induce synergistic antitumor immune responses in a mouse ovarian cancer model and that therapeutic efficacy can be enhanced by cisplatin treatment.72 Another study suggested that combined anti-TIM-3 and CD137 mAb significantly inhibits the growth of murine ovarian cancer with 60% of the mice becoming tumor-free 90 days after tumor inoculation.24 Currently, no clinical trial exist targeting 4–1BB specifically for gynecologic tumors. Table 1 summarizes the ongoing clinical trials in advanced solid tumors.
CD40
CD40 is a type I transmembrane protein that is predominately expressed on B cells and is also expressed on dendritic cells, monocytes, platelets, and macrophages, as well as on hematopoietic cells, such as myofibroblasts, fibroblasts, and epithelial and endothelial cells.73,74 Notably, the ligand of CD40 is CD154, which is also known as CD40L. The CD40 and CD40L combination plays an essential role in the initiation and progression of humoral immune responses. It also triggers B-cell intercellular adhesion, sustained proliferation, expansion, differentiation, and antibody isotype switching in vitro, and is required for germinal center formation and progression as well as affinity maturation, processes that are essential for the generation of memory B cells and long-lived plasma cells in vivo.75
Several studies have shown that neoplastic growth utilizes the CD40/CD40-L pathway to sustain the proliferative capacity and survival and that it also establishes an immunosuppressive tumor microenvironment. Generally, high expression of CD40 has been found in gynecologic tumors. In a study by Wang et al,76 the authors concluded that low expression of CD40 mRNA in ovarian cancer tissues is associated with a poor prognosis. Melichar et al77 found that the expression of CD40 was elevated in five out of eight ovarian cancer cell lines, and that CD40 transcripts have been detected in all eight cell lines. Notably, IFN-γ can enhance the expression of CD40. In ovarian cancer tissues, CD40 expression has been detected in 60.3% of paraffin-embedded samples and 73.3% of fresh samples, but not in normal ovarian tissues. Moreover, CD40 expression has been found to be significantly associated with advanced tumor stage. An in vitro experiment revealed that adenovirus-mediated CD40 ligand therapy induces apoptotic effects on ovarian cancer cells derived from fresh surgical specimens, in comparison to a null adenovirus vector and phosphate-buffered saline.78 More interestingly, another study has shown that soluble CD40-L sensitizes epithelial ovarian cancer cells to cisplatin treatment.79
In cervical tissues, CD40 is overexpressed in human papillomavirus (HPV) infected lesions and advanced squamous carcinoma but is very low in normal cervical epithelia. Moreover, CD40 stimulation in cervical cancer cell lines results in the activation of the nuclear factor-κB and mitogen-activated protein kinase signaling pathways and enhanced killing by specific CTLs.80 However, the activation of the CD40 pathway has revealed a dual function for macrophages in human endometrial cancer cell survival and invasion. CD40-activated macrophage type 1 greatly enhances cytotoxicity and reduces tumor growth, whereas CD40-activated macrophage type 2 increases cell invasion and survival. Therefore, the function of CD40 in tumor-associated macrophages might limit the therapeutic development of CD40 agonists in endometrial cancer.81 ADC-1013 is an anti-CD40 agonist mAb that was first used in human solid tumors. The results showed that the intratumoral injection of ADC-1013 is well tolerated at a clinical dose (NCT02379741).82
GITR
Glucocorticoid-induced tumor necrosis factor receptor (GITR), also known as TNFRSF18, is a type II transmembrane receptor that is predominately expressed on active B cells, NK cells, and T cells, especially Tregs.83 As a co-stimulator, GITR is involved in T cell proliferation, activation, and cytokine production. It also plays a key role in the maintenance of immune tolerance in Tregs.
Notably, GITR has been reported in ovarian and cervical cancer. In a study by Lu et al84 the authors showed that combined anti-PD-1/GITR mAb treatment remarkably inhibited peritoneal ID8 tumor growth, with 20% of the mice becoming tumor-free 90 days after tumor injection. On the other hand, treatment with either anti-PD-1 or anti-GITR mAb alone exhibited minor antitumor effects. Combined treatment with anti-PD-1/GITR mAb and chemotherapeutic drugs (cisplatin, paclitaxel) further increased the antitumor efficacy, with 80% of mice with murine ID8 ovarian cancer achieving tumor-free long-term survival. Padovani et al85 investigated 49 paraffin-embedded cervical tissue samples to detect the expression of GITR and HPV. They observed that 76.9% of all the samples with high GITR expression were HPV-positive regardless of the histopathological findings and that the expression of GITR increased in low-grade intraepithelial neoplasia, high-grade intraepithelial neoplasia, and cervical cancer tissues successively, accounting for 33.3%, 61.5%, and 85.7% in all samples, respectively. These results suggest that GITR may be involved in the progression of HPV-induced cervical cancer. Another study has shown that treatment with the adenovirus vector T cell vaccine AD-P14 and GITR mAb alone resulted in only 10% and 30% cure rates in mouse models of cervical cancer, whereas the combinationof AD-P14 and GITR mAb resulted in 100% complete response and long-term tumor-free survival in tumor-bearing mice.86
The 2018 annual ASCO meeting released the results of a phase I trial of MK-1248, a GITR mAb, alone or in combination with a PD-1 inhibitor in 37 patients with advanced solid tumors (NCT02553499). Notably, one patient achieved complete remission, and two exhibited partial remission. MK-1248 showed good tolerance with regard to the treatment dose, and no treatment-related deaths occurred. Table 1 summarizes the clinical trials performed on GITR agonist mAb targeting solid tumors.
ICOS
Inducible co-stimulator (ICOS), also known as CD278, is a member of the CD28 receptor superfamily that and exhibits significant homology to the co-stimulatory molecule CD28 and the immune-attenuator CTLA-4.87 ICOS is mainly expressed on activated CD4 and CD8 T cells.88 In a previous study, it has been shown that the in vitro binding of ICOS with its receptor induces the anti-inflammatory cytokine IL-10,89 whereas blocking ICOS signaling leads to poor T cell differentiation and decreases IL-4 production.90
The ligand of ICOS, B7-H2 has been found to be highly expressed the mRNA and protein levels in cisplatin-resistant ovarian cancer cell lines.61 Using immunohistochemical analysis of ovarian cancer tissues, Conrad et al91 confirmed that the accumulation of ICOS+ Tregs is more significant than the accumulation of ICOS− Tregs in the tumor microenvironment. The authors also found that tumor plasmacytoid dendritic cells expressing high levels of the ICOS ligand (ICOS-L) may co-stimulate the expression of ICOS+ Tregs, which can be blocked by anti-ICOS-L antibodies. Survival analysis revealed that ICOS+, but not ICOS− Tregs are a predictor of poor clinical outcomes and that ICOS+ Tregs are a better predictor of disease progression than total Foxp3+ Tregs. Although only a few studies have been reported, the results suggest that targeting ICOS/ICOS-L may exert anti-tumor effects in ovarian cancer.
Several mAbs have been screened and developed for ICOS, with encouraging results with animal models. Similar to TIM-3, ICOS is unlikely to be used as a single treatment. This is because neither of them can independently induce a cytotoxic immune response. Several clinical trials of ICOS agonist mAbs, including JTX-2011 and GSK3359609, combined with PD-1 or CTLA-4 inhibitors for advanced solid tumors are currently underway (Table 1).
B7-H6
B7-H6 is a newly identified B7 family member, which encodes a type I transmembrane protein with a predicted molecular mass of 51 kDa. B7-H6 binds to its receptor on NK cells, natural cytotoxicity receptor 3 (NCR3) (also known as NKp30), and then triggers the antitumor NK cell cytotoxicity and leads to the cytokine secretion.92 The expression of B7-H6 is found on the surface of several types of tumor cells, including melanoma, neuroblastoma and primary blood or bone marrow cells derived from different types of hematological malignancies.93
In gynecologic cancer, B7-H6 has been reported in ovarian cancer and cervical cancer. In the study by Zhou et al,94 positive B7-H6 staining was predominantly observed on the membrane and in the cytoplasm of the ovarian cancer cells, and B7-H6 expression was significantly correlated with distant metastasis status and advanced tumor stage. Another study showed that B7-H6 is detectable as a surface/cytosolic molecule in ovarian cancer cells and as a soluble molecule in the peritoneal fluid. The expression of the activating receptor NKp30 in tumor-associated NK cells is substantially reduced as compared to autologous peripheral blood NK cells. The impaired expression of this receptor is associated with the presence of this soluble ligand B7-H6.95 It has been reported that the expression of B7-H6 is positively correlated with disease stage in the development of cervical cancer, and the expression level is higher in cervical adenocarcinoma than that in SCC.96 The studies on B7-H6 in gynecologic tumors are all in the preclinical stage, and there is no relevant clinical trial ongoing at present.
Discussion
The development of tumor immunotherapy research is encouraging, and translating such emerging immune targeting studies into clinical practice is one of the most important focuses in current research. However, many difficulties and challenges exist. For example, although PD-1/PD-L1 and CTLA-4 have been widely used successfully as target immune checkpoint inhibitors, the overall response rate reported with gynecologic cancer is not satisfactory.
T cell exhaustion and the upregulation of coinhibitory immune checkpoints can lead to immune tolerance or resistance.97 Generally, single-target inhibitors cannot successfully induce cytotoxic immune responses, and combined treatment probably enhances the therapeutic efficacy, although at the same time it increases the adverse effects. Therefore, it is crucial to balance the relationship between the two counterparts. In a recently published phase II randomized clinical trial, it has been reported that the objective remission rate of Nivolumab alone was 12% in patients with recurrent and persistent ovarian cancer, whereas the rate was 31.4% when Nivolumab was combined with Ipilimumab, with tolerable toxic side effects.98 Combining of other targeted drugs also helps counteract the adverse effect of immune-targeted therapy and enhance drug efficacy. For example, combining the anti-angiogenic drug Apatinib with the PD-1 inhibitor Camrelizumab can enhance the antitumor effects and reduce the incidence of unique reactive hemangioma. Several studies have shown that prophylactic TNF blocking improves the antitumor efficacy of dual immunotherapy with CTLA-4 and PD-1 and alleviates immune-related colitis and hepatitis.99,100 Fan et al101 showed that anti-CTLA-4 in combination with ICOS-L expressing tumor vaccines led to tumor rejection in more than 80% of the mice, a fourfold increase in efficacy, and demonstrated that the antitumor responses enhanced by CTLA-4 blockade can be greatly improved by targeting the ICOS/ICOS-L pathway vaccine.
The sequence of drug combinations is another a noteworthy issue, especially when using a combination of co-inhibitory and co-stimulatory molecules. In a previous study, it has been shown that concurrent PD-1 and OX40 blockade did not increase the antitumor effects of OX40 but rather upregulate the T cell inhibitory receptors and increased serum cytokines. This, in turn, accelerated the depletion of T cells in tumors and significantly attenuated the therapeutic effect of anti-OX40 co-stimulation. However, sequentially delayed PD-1 blockade augments the antitumor effects of anti-OX40, which maintains the proliferation of T cells and reduces T cell exhaustion.102
Due to the complexity of the interaction and regulation of the immune system, the development of pharmacodynamics and prognostic biomarkers for immune-targeted molecules is currently facing unique challenges. According to the clinical experience with PD-1 inhibitors, the effects of drugs can be predicted by detecting the expression of ligands in tumor tissues or TILs, microsatellite instability, tumor mutational burden, and mismatch repair deficiency.103 In addition, circulating biomarkers and gut microbiota play a vital role in the dynamic monitoring of tumor immune status due to minimum invasion.104
Conclusion
Although scientific research and clinical trials focused on immune targets are numerous, studies on gynecologic tumors are still in the early stages. Currently, targeted therapy for ovarian cancer is becoming increasingly recognized. The HPV vaccine aimed at preventing cervical cancer is becoming popularized, and endocrine therapy for endometrial cancer has also been standardized. Immunotherapy, with its inherent specificity, adaptability, and memory response, can provide salvage treatment for patients with refractory and recurrent disease. The expression patterns of these emerging immune targets are closely associated with the treatment responses, and the relevant research is mostly in the early experimental stages. Potential mechanisms and interactions of immune targets need to be further elucidated.
Acknowledgments
This study was supported by the National Natural Science Foundation of China (Nos. 81672586, 81971475, and 81972451) and the Chinese Academy of Medical Sciences Initiative for Innovative Medicine (No. CAMS-2017-I2M-1-002).
Disclosure
The authors declare no conflict of interest.
References
1. Sharma P, Allison JP. The future of immune checkpoint therapy. Science. 2015;348(6230):56–61. doi:10.1126/science.aaa8172
2. Reck M, Rodriguez-Abreu D, Robinson AG, et al. Pembrolizumab versus chemotherapy for PD-L1–positive non–small-cell lung cancer. N Engl J Med. 2016;375(19):1823–1833. doi:10.1056/NEJMoa1606774
3. Larkin J, Chiarion-Sileni V, Gonzalez R, et al. Combined nivolumab and ipilimumab or monotherapy in untreated melanoma. N Engl J Med. 2015;373(1):23–34. doi:10.1056/NEJMoa1504030
4. Agata Y, Kawasaki A, Nishimura H, et al. Expression of the PD-1 antigen on the surface of stimulated mouse T and B lymphocytes. Int Immunol. 1996;8(5):765–772. doi:10.1093/intimm/8.5.765
5. Pesce S, Greppi M, Tabellini G, et al. Identification of a subset of human natural killer cells expressing high levels of programmed death 1: a phenotypic and functional characterization. J Allergy Clin Immunol. 2017;139(1):335–346. doi:10.1016/j.jaci.2016.04.025
6. Dong H, Strome SE, Salomao DR, et al. Tumor-associated B7-H1 promotes T-cell apoptosis: a potential mechanism of immune evasion. Nat Med. 2002;8(8):793–800. doi:10.1038/nm730
7. Ishida M, Iwai Y, Tanaka Y, et al. Differential expression of PD-L1 and PD-L2, ligands for an inhibitory receptor PD-1, in the cells of lymphohematopoietic tissues. Immunol Lett. 2002;84(1):57–62. doi:10.1016/S0165-2478(02)00142-6
8. Hamanishi J, Mandai M, Iwasaki M, et al. Programmed cell death 1 ligand 1 and tumor-infiltrating CD8+ T lymphocytes are prognostic factors of human ovarian cancer. Proc Natl Acad Sci U S A. 2007;104(9):3360–3365. doi:10.1073/pnas.0611533104
9. Enwere EK, Kornaga EN, Dean M, et al. Expression of PD-L1 and presence of CD8-positive T cells in pre-treatment specimens of locally advanced cervical cancer. Mod Pathol. 2017;30(4):577–586. doi:10.1038/modpathol.2016.221
10. Gulec UK, Bagir EK, Paydas S, Guzel AB, Gumurdulu D, Vardar MA. Programmed death-1 (PD-1) and programmed death-ligand 1 (PD-L1) expressions in type 2 endometrial cancer. Arch Gynecol Obstet. 2019;300(2):377–382.
11. Varga A, Piha-Paul S, Ott PA, et al. Pembrolizumab in patients with programmed death ligand 1–positive advanced ovarian cancer: analysis of KEYNOTE-028. Gynecol Oncol. 2019;152(2):243–250. doi:10.1016/j.ygyno.2018.11.017
12. Frenel J-S, Le Tourneau C, O’Neil B, et al. Safety and efficacy of pembrolizumab in advanced, programmed death ligand 1–positive cervical cancer: results from the phase Ib KEYNOTE-028 trial. J clin oncol. 2017;35(36):4035–4041. doi:10.1200/JCO.2017.74.5471
13. Ott PA, Bang Y-J, Berton-Rigaud D, et al. Safety and antitumor activity of pembrolizumab in advanced programmed death ligand 1–positive endometrial cancer: results from the KEYNOTE-028 Study. J clin oncol. 2017;35(22):2535–2541. doi:10.1200/JCO.2017.72.5952
14. Disis ML, Taylor MH, Kelly K, et al. Efficacy and safety of avelumab for patients with recurrent or refractory ovarian cancer: phase 1b results from the JAVELIN solid tumor trial. JAMA oncol. 2019;5(3):393–401. doi:10.1001/jamaoncol.2018.6258
15. Chung HC, Ros W, Delord J-P, et al. Efficacy and safety of pembrolizumab in previously treated advanced cervical cancer: results from the phase II KEYNOTE-158 Study. J clin oncol. 2019;37(17):1470–1478. doi:10.1200/JCO.18.01265
16. Monney L, Sabatos CA, Gaglia JL, et al. Th1-specific cell surface protein Tim-3 regulates macrophage activation and severity of an autoimmune disease. Nature. 2002;415(6871):536–541. doi:10.1038/415536a
17. Xu Y, Zhang H, Huang Y, Rui X, Zheng F. Role of TIM-3 in ovarian cancer. Clin Transl Oncol. 2017;(9(9):1079–1083.
18. Zhu C, Anderson AC, Schubart A, et al. The tim-3 ligand galectin-9 negatively regulates T helper type 1 immunity. Nat Immunol. 2005;6(12):1245–1252. doi:10.1038/ni1271
19. Huang YH, Zhu C, Kondo Y, et al. CEACAM1 regulates TIM-3-mediated tolerance and exhaustion. Nature. 2015;517(7534):386–390.
20. Zhu C, Anderson AC, Schubart A, et al. The tim-3 ligand galectin-9 negatively regulates T helper type 1 immunity. Nat Immunol. 2005;6(12):1245–1252.
21. Yan J, Zhang Y, Zhang JP, Liang J, Li L, Zheng L. Tim-3 expression defines regulatory T cells in human tumors. PLoS One. 2013;8:e58006.
22. Fucikova J, Rakova J, Hensler M, et al. TIM-3 dictates functional orientation of the immune infiltrate in ovarian cancer. Clin Cancer Res. 2019;25(15):4820–4831.
23. Wu J, Liu C, Qian S, Hou H. The expression of tim-3 in peripheral blood of ovarian cancer. DNA Cell Biol. 2013;32(11):648–653. doi:10.1089/dna.2013.2116
24. Guo Z, Cheng D, Xia Z, et al. Combined TIM-3 blockade and CD137 activation affords the long-term protection in a murine model of ovarian cancer. J Transl Med. 2013;11(1):215. doi:10.1186/1479-5876-11-215
25. Heeren AM, Rotman J, Stam AGM, et al. Efficacy of PD-1 blockade in cervical cancer is related to a CD8+FoxP3+CD25+ T-cell subset with operational effector functions despite high immune checkpoint levels. J Immunother Cancer. 2019;7(1):1–14. doi:10.1186/s40425-019-0526-z
26. Degos C, Heinemann M, Barrou J, et al. Endometrial tumor microenvironment alters human NK cell recruitment, and resident NK cell phenotype and function. Front Immunol. 2019;10:877.
27. Yu X, Harden K, Gonzalez LC, et al. The surface protein TIGIT suppresses T cell activation by promoting the generation of mature immunoregulatory dendritic cells. Nat Immunol. 2009;10(1):48–57. doi:10.1038/ni.1674
28. Zhang B, Zhao W, Li H, et al. Immunoreceptor TIGIT inhibits the cytotoxicity of human cytokine-induced killer cells by interacting with CD155. Cancer Immunol Immunother. 2016;65(3):305–314.
29. Stanietsky N, Simic H, Arapovic J, et al. The interaction of TIGIT with PVR and PVRL2 inhibits human NK cell cytotoxicity. Proc Natl Acad Sci U S A. 2019;106(42):17858–17863.
30. Dougall WC, Kurtulus S, Smyth MJ, Anderson AC. TIGIT and CD96: new checkpoint receptor targets for cancer immunotherapy. Immunol Rev. 2017;276(1):112–120.
31. Oshima T, Sato S, Kato J, et al. Nectin-2 is a potential target for antibody therapy of breast and ovarian cancers. Mol Cancer. 2013;12(1):60. doi:10.1186/1476-4598-12-60
32. Textor S, Durst M, Jansen L, et al. Activating NK cell receptor ligands are differentially expressed during progression to cervical cancer. Int J Cancer. 2008;123(10):2343–2353. doi:10.1002/ijc.23733
33. Triebel F, Jitsukawa S, Baixeras E. LAG-3, a novel lymphocyte activation gene closely related to CD4. J Exp Med. 1990;171(5):1393–1405. doi:10.1084/jem.171.5.1393
34. Wang J, Sanmamed MF, Datar I, et al. Fibrinogen-like protein 1 is a major immune inhibitory ligand of LAG-3. Cell. 2019;176(1–2):334–347.e312. doi:10.1016/j.cell.2018.11.010
35. Grosso JF, Kelleher CC, Harris TJ, et al. LAG-3 regulates CD8+ T cell accumulation and effector function in murine self- and tumor-tolerance systems. J Clin Invest. 2007;117(11):3383–3392. doi:10.1172/JCI31184
36. Lui Y, Davis SJ. LAG-3: a very singular immune checkpoint. Nat Immunol. 2018;19(12):1278–1279. doi:10.1038/s41590-018-0257-1
37. Tu L, Guan R, Yang H, et al. Assessment of the expression of the immune checkpoint molecules PD-1, CTLA4, TIM-3 and LAG-3 across different cancers in relation to treatment response, tumor-infiltrating immune cells and survival. Int J Cancer. 2019.
38. Matsuzaki J, Gnjatic S, Mhawech-Fauceglia P, et al. Tumor-infiltrating NY-ESO-1–specific CD8 + T cells are negatively regulated by LAG-3 and PD-1 in human ovarian cancer. Proc Natl Acad Sci U S A. 2010;107(17):7875–7880. doi:10.1073/pnas.1003345107
39. Wang L, Rubinstein R, Lines JL, et al. VISTA, a novel mouse Ig superfamily ligand that negatively regulates T cell responses. J Exp Med. 2011;208(3):577–592. doi:10.1084/jem.20100619
40. ElTanbouly MA, Croteau W, Noelle RJ, Lines JL. VISTA: a novel immunotherapy target for normalizing innate and adaptive immunity. Semin Immunol. 2019;42:101308. doi:10.1016/j.smim.2019.101308
41. Nowak EC, Lines JL, Varn FS, et al. Immunoregulatory functions of VISTA. Immunol Rev. 2017;276(1):66–79. doi:10.1111/imr.12525
42. Wang J, Wu G, Manick B, et al. VSIG-3 as a ligand of VISTA inhibits human T-cell function. Immunology. 2019;156(1):74–85. doi:10.1111/imm.13001
43. Mulati K, Hamanishi J. VISTA expressed in tumour cells regulates T cell function. Jan. 2019;120(1):115–127.
44. Liao H, Zhu H, Liu S, Wang H. Expression of V-domain immunoglobulin suppressor of T cell activation is associated with the advanced stage and presence of lymph node metastasis in ovarian cancer. Oncol Lett. 2018;16(3):3465–3472.
45. Zong L, Zhou Y, Zhang M, Chen J, Xiang Y. VISTA expression is associated with a favorable prognosis in patients with high-grade serous ovarian cancer. Cancer Immunol Immunother. 2020;69(1):33–42. doi:10.1007/s00262-019-02434-5
46. Kuang L. Potential value of V-domain Ig suppressor of T-cell activation for assessing progn osis in cervical cancer and as a target for therapy. Int J Clin Exp Pathol. 2020;13(1):26–37.
47. Zong L, Zhang M, Wang W, et al. PD-L1, B7-H3 and VISTA are highly expressed in gestational trophoblastic neoplasia. Histopathology. 2019;75(3):421–430. doi:10.1111/his.13882
48. Hashiguchi M, Kobori H, Ritprajak P, Kamimura Y, Kozono H, Azuma M. Triggering receptor expressed on myeloid cell-like transcript 2 (TLT-2) is a counter-receptor for B7-H3 and enhances T cell responses. Proc Natl Acad Sci U S A. 2008;105(30):10495–10500. doi:10.1073/pnas.0802423105
49. Carreno BM, Collins M. BTLA: a new inhibitory receptor with a B7-like ligand. Trends Immunol. 2003;24(10):524–527. doi:10.1016/j.it.2003.08.005
50. Fauci JM, Straughn JM, Ferrone S, Buchsbaum DJ. A review of B7-H3 and B7-H4 immune molecules and their role in ovarian cancer. Gynecol Oncol. 2012;127(2):420–425. doi:10.1016/j.ygyno.2012.08.017
51. Burugu S, Dancsok AR, Nielsen TO. Emerging targets in cancer immunotherapy. Semin Cancer Biol. 2018;52:39–52. doi:10.1016/j.semcancer.2017.10.001
52. Wang J-Y, Wang W-P. B7-H4, a promising target for immunotherapy. Cell Immunol. 2020;347:104008. doi:10.1016/j.cellimm.2019.104008
53. Brunner A, Hinterholzer S, Riss P, Heinze G, Brustmann H. Immunoexpression of B7-H3 in endometrial cancer: relation to tumor T-cell infiltration and prognosis. Gynecol Oncol. 2012;124(1):105–111.
54. Tringler B, Liu W, Corral L, et al. B7-H4 overexpression in ovarian tumors. Gynecol Oncol. 2006;100(1):44–52. doi:10.1016/j.ygyno.2005.08.060
55. Zang X, Sullivan PS, Soslow RA, et al. Tumor associated endothelial expression of B7-H3 predicts survival in ovarian carcinomas. Mod Pathol. 2010;23(8):1104–1112. doi:10.1038/modpathol.2010.95
56. Han S, Wang Y, Shi X, et al. Negative roles of B7-H3 and B7-H4 in the microenvironment of cervical cancer. Exp Cell Res. 2018;371(1):222–230. doi:10.1016/j.yexcr.2018.08.014
57. Arch RH, Gedrich RW, Thompson CB. Tumor necrosis factor receptor-associated factors (TRAFs)---a family of adapter proteins that regulates life and death. Genes Dev. 1998;12(18):2821–2830. doi:10.1101/gad.12.18.2821
58. Lens SMA, Tesselaar K, van Oers MHJ, van Lier RAW. Control of lymphocyte function through CD27–CD70 interactions. Semin Immunol. 1998;10(6):491–499. doi:10.1006/smim.1998.0154
59. Hendriks J, Gravestein LA, Tesselaar K, van Lier RAW, Schumacher TNM, Borst J. CD27 is required for generation and long-term maintenance of T cell immunity. Nat Immunol. 2000;1(5):433–440. doi:10.1038/80877
60. Koch M, Krieger ML, Stolting D, et al. Overcoming chemotherapy resistance of ovarian cancer cells by liposomal cisplatin: molecular mechanisms unveiled by gene expression profiling. Biochem Pharmacol. 2013;85(8):1077–1090. doi:10.1016/j.bcp.2013.01.028
61. Aggarwal S, He T, FitzHugh W, et al. Immune modulator CD70 as a potential cisplatin resistance predictive marker in ovarian cancer. Gynecol Oncol. 2009;115(3):430–437. doi:10.1016/j.ygyno.2009.08.031
62. Keller AM, Schildknecht A, Xiao Y, Broek M, Borst J. Expression of costimulatory ligand CD70 on steady-state dendritic cells breaks CD8+ T cell tolerance and permits effective immunity. Immunity. 2008;29(6):934–946. doi:10.1016/j.immuni.2008.10.009
63. Aspeslagh S, Postel-Vinay S, Rusakiewicz S, Soria J-C, Zitvogel L, Marabelle A. Rationale for anti-OX40 cancer immunotherapy. Eur J Cancer. 2016;52:50–66. doi:10.1016/j.ejca.2015.08.021
64. Jensen SM, Maston LD, Gough MJ, et al. Signaling through OX40 enhances antitumor immunity. Semin Oncol. 2010;37(5):524–532. doi:10.1053/j.seminoncol.2010.09.013
65. Ohshima Y, Tanaka Y, Tozawa H, Takahashi Y, Maliszewski C, Delespesse G. Expression and function of OX40 ligand on human dendritic cells. J Immunol. 1997;159(8):3838–3848.
66. Kroemer A, Xiao X, Vu MD, et al. OX40 controls functionally different T cell subsets and their resistance to depletion therapy. J Immunol. 2007;179(8):5584–5591. doi:10.4049/jimmunol.179.8.5584
67. Ramser M, Eichelberger S, Däster S, Weixler B, Kraljević M. High OX40 expression in recurrent ovarian carcinoma is indicative for response to repeated chemotherapy. BMC Cancer. 2018;18(1):425. doi:10.1186/s12885-018-4339-0
68. Guo Z, Wang X, Cheng D, et al. PD-1 blockade and OX40 triggering synergistically protects against tumor growth in a murine model of ovarian cancer. PLoS One. 2014;9(2):e89350. doi:10.1371/journal.pone.0089350
69. Zhao M, Li Y, Wei X, et al. Negative immune factors might predominate local tumor immune status and promote carcinogenesis in cervical carcinoma. Virol J. 2017;14(1):5. doi:10.1186/s12985-016-0670-8
70. Gramaglia I, Cooper D, Miner KT, Kwon BS, Croft M. Co‐stimulation of antigen‐specific CD4 T cells by 4‐1BB ligand. Eur J Immunol. 2000;30(2):392–402. doi:10.1002/1521-4141(200002)30:2<392::AID-IMMU392>3.0.CO;2-H
71. Croft M. Costimulation of T cells by OX40, 4-1BB, and CD27. Cytokine Growth Factor Rev. 2003;14(3–4):265–273. doi:10.1016/S1359-6101(03)00025-X
72. Wei H, Zhao L, Hellstrom I, Hellstrom KE, Guo Y. Dual targeting of CD137 co-stimulatory and PD-1 co-inhibitory molecules for ovarian cancer immunotherapy. OncoImmunology. 2014;3(4):e28248. doi:10.4161/onci.28248
73. van Kooten C, Banchereau J. Functions of CD40 on B cells, dendritic cells and other cells. Curr Opin Immunol. 1997;9(3):330–337. doi:10.1016/S0952-7915(97)80078-7
74. Bourgeois C. A role for CD40 expression on CD8+ T cells in the generation of CD8+ T cell memory. Science. 2002;297(5589):2060–2063. doi:10.1126/science.1072615
75. Elgueta R, Benson MJ, Vries VCD, Wasiuk A, Guo Y, Noelle RJ. Molecular mechanism and function of CD40/CD40L engagement in the immune system . Immunol Rev. 2009;229(1):152–172.
76. Wang P, Zhang Z, Ma Y, et al. Prognostic values of GMPS, PR, CD40, and p21 in ovarian cancer. PeerJ. 2019;7:e6301. doi:10.7717/peerj.6301
77. Melichar B, Patenia R, Gallardo S, Melicharova K, Hu W, Freedman RS. Expression of CD40 and growth-inhibitory activity of CD40 ligand in ovarian cancer cell lines. Gynecol Oncol. 2007;104(3):707–713. doi:10.1016/j.ygyno.2006.10.056
78. Jiang E, He X, Chen X, et al. Expression of CD40 in ovarian cancer and adenovirus-mediated CD40 ligand therapy on ovarian cancer in vitro. Tumori. 2008;94(3):356–361. doi:10.1177/030089160809400312
79. Qin L, Qiu H, Zhang M, et al. Soluble CD40 ligands sensitize the epithelial ovarian cancer cells to cisplatin treatment. Biomed Pharmacother. 2016;79:166–175. doi:10.1016/j.biopha.2016.01.006
80. Hill SC, Youde SJ, Man S, et al. Activation of CD40 in cervical carcinoma cells facilitates CTL responses and augments chemotherapy-induced apoptosis. J Immunol. 2005;174(1):41–50. doi:10.4049/jimmunol.174.1.41
81. Dumas G, Dufresne M, Asselin É, Girouard J, Carrier C, Reyes-Moreno C. CD40 pathway activation reveals dual function for macrophages in human endometrial cancer cell survival and invasion. Cancer Immunol Immunother. 2013;62(2):273–283. doi:10.1007/s00262-012-1333-2
82. Irenaeus SMM, Nielsen D, Ellmark P, et al. First-in-human study with intratumoral administration of a CD40 agonistic antibody, ADC-1013, in advanced solid malignancies. Int J Cancer. 2019;145(5):1189–1199. doi:10.1002/ijc.32141
83. Watts TH. TNF/TNFR family members in costimulation of T cell responses. Annu Rev Immunol. 2005;23(1):23–68. doi:10.1146/annurev.immunol.23.021704.115839
84. Lu L, Xu X, Zhang B, Zhang R, Ji H, Wang X. Combined PD-1 blockade and GITR triggering induce a potent antitumor immunity in murine cancer models and synergizes with chemotherapeutic drugs. J Transl Med. 2014;12(1):36. doi:10.1186/1479-5876-12-36
85. Padovani CTJ, Bonin CM, Tozetti IA, Ferreira AMT, Costa IPD. Glucocorticoid-induced tumor necrosis factor receptor expression in patients with cervical human papillomavirus infection. Rev Soc Bras Med Trop. 2013;46(3):288–292. doi:10.1590/0037-8682-0029-2013
86. Hoffmann C, Stanke J, Kaufmann AM, Loddenkemper C, Schneider A, Cichon G. Combining T-cell vaccination and application of agonistic anti-GITR mAb (DTA-1) induces complete eradication of HPV oncogene expressing tumors in mice. J Immunother. 2010;33(2):136–145. doi:10.1097/CJI.0b013e3181badc46
87. Hutloff A, Dittrich AM, Beier KC, Eljaschewitsch B, Kraft R. ICOS is an inducible T-cell co-stimulator structurally and functionally related to CD28. Nature. 1999;397(6716):263–266. doi:10.1038/16717
88. Watanabe M, Hara Y, Tanabe K, Toma H, Abe R. A distinct role for ICOS-mediated co-stimulatory signaling in CD4+ and CD8+ T cell subsets. Int Immunol. 2005;17(3):269–278. doi:10.1093/intimm/dxh206
89. Witsch EJ, Peiser M, Hutloff A, et al. ICOS and CD28 reversely regulate IL-10 on re-activation of human effector T? Cells with mature dendritic cells. Eur J Immunol. 2002;32(9):2680–2686. doi:10.1002/1521-4141(200209)32:9<2680::AID-IMMU2680>3.0.CO;2-6
90. Dong C, Juedes AE, Temann U-A, Shresta S, Allison JP. ICOS co-stimulatory receptor is essential for T-cell activation and function. Nature. 2001;409(6816):97–101. doi:10.1038/35051100
91. Conrad C, Gregorio J, Wang Y-H, et al. Plasmacytoid dendritic cells promote immunosuppression in ovarian cancer via ICOS costimulation of Foxp3+ T-regulatory cells. Cancer Res. 2012;72(20):5240–5249. doi:10.1158/0008-5472.CAN-12-2271
92. Chen Y, Mo J, Jia X, He Y. The B7 family member B7-H6: a new bane of tumor. Patholo Oncol Res. 2018;24(4):717–721.
93. Schlecker E, Fiegler N, Arnold A, et al. Metalloprotease-mediated tumor cell shedding of B7-H6, the Ligand of the natural killer cell-activating receptor NKp30. Cancer Res. 2014;74(13):3429–3440. doi:10.1158/0008-5472.CAN-13-3017
94. Zhou Y, Xu Y, Chen L, Xu B, Wu C, Jiang J. B7-H6 expression correlates with cancer progression and patient’s survival in human ovarian cancer. Int J Clin Exp Pathol. 2015;8(8):9428–9433.
95. Pesce S, Tabellini G, Cantoni C, et al. B7-H6-mediated downregulation of NKp30 in NK cells contributes to ovarian carcinoma immune escape. Oncoimmunology. 2005;174(1):e1001224. doi:10.1080/2162402X.2014.1001224
96. Gutierrez-Silerio GY, Franco-Topete RA, Haramati J, et al. Positive staining of the immunoligand B7-H6 in abnormal/transformed keratinocytes consistently accompanies the progression of cervical cancer. BMC Immunol. 2020;21(1):9.
97. Wang Q, Wu X. Primary and acquired resistance to PD-1/PD-L1 blockade in cancer treatment. Int Immunopharmacol. 2017;46:210–219.
98. Zamarin D, Burger RA, Sill MW, et al. Randomized phase II trial of nivolumab versus nivolumab and ipilimumab for recurrent or persistent ovarian cancer: an NRG Oncology Study. J clin oncol. 2020;38(16):1814–1823.
99. Perez-Ruiz E, Minute L, Otano I, et al. Prophylactic TNF blockade uncouples efficacy and toxicity in dual CTLA-4 and PD-1 immunotherapy. Nature. 2019;569(7756):428–432.
100. Li W, Wei Z, Yang X, et al. Salvage therapy of reactive capillary hemangiomas: apatinib alleviates the unique adverse events induced by camrelizumab in non-small cell lung cancer. J Cancer Res Ther. 2019;15(7):1624–1628. doi:10.4103/jcrt.JCRT_997_19
101. Fan X, Quezada SA, Sepulveda MA, Sharma P, Allison JP. Engagement of the ICOS pathway markedly enhances efficacy of CTLA-4 blockade in cancer immunotherapy. J Exp Med. 2014;211(4):715–725. doi:10.1084/jem.20130590
102. Messenheimer DJ, Jensen SM, Afentoulis ME, et al. Timing of PD-1 blockade is critical to effective combination immunotherapy with anti-OX40. Clin Cancer Res. 2017;23(20):6165–6177. doi:10.1158/1078-0432.CCR-16-2677
103. Wu X, Gu Z, Chen Y, et al. Application of PD-1 BLOCKADE IN CANCER IMMUNotherapy. Comput Struct Biotechnol J. 2019;17:661–674. doi:10.1016/j.csbj.2019.03.006
104. Yi M, Yu S, Qin S, et al. Gut microbiome modulates efficacy of immune checkpoint inhibitors. J Hematol Oncol. 2018;11(1):47.
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
