Back to Journals » Neuropsychiatric Disease and Treatment » Volume 16
Elevated Neutrophil-to-Lymphocyte Ratio Predicts Depression After Intracerebral Hemorrhage
Authors Gong X , Lu Z, Feng X, Yu C, Xue M, Yu L, Wang T, Cheng X, Lu J , Zhang M
Received 25 June 2020
Accepted for publication 31 August 2020
Published 21 September 2020 Volume 2020:16 Pages 2153—2159
DOI https://doi.org/10.2147/NDT.S269210
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Yuping Ning
Xiuqun Gong,1,* Zeyu Lu,2,* Xiwu Feng,3,* Chuanqing Yu,1 Min Xue,1 Liang Yu,1 Tao Wang,1 Xiaosi Cheng,1 Jun Lu,4,5 Mei Zhang1
1Department of Neurology, First Affiliated Hospital of Anhui University of Science and Technology, First People’s Hospital of Huainan, Huainan 232001, Anhui, People’s Republic of China; 2School of Medicine, Anhui University of Science and Technology, Huainan 232001, Anhui, People’s Republic of China; 3Department of Cardiothoracic Surgery, First Affiliated Hospital of Anhui University of Science and Technology, First People’s Hospital of Huainan, Huainan 232001, Anhui, People’s Republic of China; 4Department of Medical Laboratory, School of Medicine, Anhui University of Science and Technology, Huainan 232001, Anhui, People’s Republic of China; 5Department of Clinical Laboratory, First Affiliated Hospital of Anhui University of Science and Technology, First People’s Hospital of Huainan, Huainan 232001, Anhui, People’s Republic of China
*These authors contributed equally to this work
Correspondence: Jun Lu
Department of Medical Laboratory, School of Medicine, Anhui University of Science and Technology, 168 Taifeng Road, Huainan 232001, Anhui Province, People’s Republic of China
Tel +86- 13855453149
Email [email protected]
Mei Zhang
Department of Neurology, First Affiliated Hospital of Anhui University of Science and Technology, First People’s Hospital of Huainan, 203 Huaibin Road, Huainan 232007, Anhui Province, People’s Republic of China
Tel + 86- 13955466855
Email [email protected]
Purpose: Inflammation plays a critical role in the development of depression after intracerebral hemorrhage (ICH), while neutrophil-to-lymphocyte ratio (NLR) has been identified as a novel comprehensive inflammatory indicator in recent years. The aim of this study was to examine the association between NLR and depression after ICH.
Patients and Methods: From January 2016 to December 2018, ICH patients were prospectively enrolled. NLR was measured at admission. Depression at 3 months after ICH was diagnosed according to the Hamilton Depression Scale (HAMD).
Results: Of the 372 enrolled patients, 107 (28.8%) were diagnosed with depression at 3 months after ICH. Patients with depression had a higher NLR (6.15 vs 3.55, P < 0.001). Logistic regression analysis detected that after adjusting for major confounders, NLR remained independently associated with depression after ICH (OR = 2.25, 95% CI: 1.45– 3.49, P < 0.001). Moreover, NLR acted as the optimal variable for prediction, with the optimal predictive threshold of 4.53 in ROC analysis.
Conclusion: Elevated NLR is associated with depression at 3 months after ICH, suggesting that NLR may be a significant biomarker to predict depression after ICH.
Keywords: inflammation, neutrophil-to-lymphocyte ratio, depression, intracerebral hemorrhage
Introduction
Post-stroke depression (PSD) is one of the most frequently seen psychosomatic disorders that occurs in almost 20% intracerebral hemorrhage (ICH) survivors.1–3 Patients with PSD bear increased risk for cognitive impairment, stroke recurrence and death.4–6 Hence, it is vital to identify and understand the pathogenesis of PSD early.
Inflammation may play an important role in the development of PSD.7 Higher levels of tumor necrosis factor-α (TNF-α), interleukin (IL)-6 and IL-18 have been shown to be associated with PSD.8,9 In another study, TNF-α and IL-1β genes polymorphism were reported to be correlated with increased risk for PSD.10 A recent review also indicated that inflammation may promote the occurrence of depression after intracerebral hemorrhage through a series of signaling pathways.11
The neutrophil-to-lymphocyte ratio (NLR) has been reported as a credible, simple and convenient marker of inflammation recently, which is a more potent indicator than neutrophils or lymphocytes themselves.12,13 Previous studies have been conducted to examine the relationship between NLR and depression after ischemic stroke.14 However, the association of NLR and depression after ICH has not been clarified. Thus, this study aimed to investigate the predictive value of NLR in depression at 3 months after ICH.
Patients and Methods
Study Population
Patients with ICH were enrolled consecutively from Huainan First People’s Hospital from January 2016 to December 2018. Patients were included if they: (1) were diagnosed with ICH verified by CT scans within 24 h from symptom onset; (2) aged ≥18 years. The patients were excluded if they: (1) had secondary hemorrhage as a result of tumor, trauma, vascular malformation, aneurysm, hemorrhagic transformation of cerebral infarct and blood coagulation abnormalities; (2) had concurrent disease that might affect the value of NLR, such as active infection (fever, cough, or diarrhea), chronic inflammatory disease or malignancy; (3) had severe cognitive impairment, a history of major depression or other psychiatric disorders; (4) with severe aphasia so that could not complete the psychological measurement; (5) had severe heart, renal or liver diseases. This study was conducted in accordance with the Declaration of Helsinki and approved by the ethic committee of Huainan First People’s Hospital. Informed consent was obtained from each patient.
Clinical Data
Demographic characteristics, clinical variables and vascular risk factors were all collected from medical records. Stroke severity was assessed by the National Institutes of Health Stroke Scale (NIHSS) score.15 The extent of coma was determined by Glasgow Coma Scale (GCS) score. Fasting venous blood was collected in the next morning for routine blood test and NLR was defined as the ratio of neutrophil counts to lymphocyte counts.
Assessment of Outcomes
All patients were followed up through telephone interviews or clinical visits at 3 months after ICH, unless they died or were lost. The functional outcomes were assessed using modified Rankin Scale (mRS) score and Barthel Index (BI) score. Cognitive functions were measured through Mini-Mental State Examination (MMSE). The 17-item Hamilton Depression Scale (HAMD) was used to assess depression.16 Patients with a HAMD score of >7 were diagnosed with depression after ICH.14
Statistical Analysis
Continuous variables were presented as mean ± SD or median (interquartile range). Categorical variables were presented as numbers (percentages). Independent t-test or Mann–Whitney U-test was applied for continuous variables. Chi-square test or Fisher exact test were employed for categorical data. Multivariable regression analyses were performed using 3 models to recognize the predictive factors of PSD, among which, model 1 was adjusted for age and sex; model 2 was adjusted for age, sex and risk factors (history of smoking, history of drinking, hypertension, diabetes, hyperlipidemia, atrial fibrillation); whereas model 3 was further adjusted for variables with P < 0.05 upon univariate analysis. Besides, the association was indicated as odds ratio (OR) with 95% confidence interval (CI). In addition, the predictive accuracy of NLR in depression after ICH was assessed through the receiver operating characteristic (ROC) curve and area under the curve (AUC). DeLong test was employed to compare the AUC between groups. Results were considered as statistically significant if P < 0.05. The R software package (version 3.0) was utilized for all statistical analyses for DeLong test, while SPSS 22.0 (IBM, New York, USA) was used for other statistical analyses.
Results
A total of 372 ICH patients were enrolled in this study, including 232 (62.4%) males and 140 (37.6%) females. The median patient age was 66 (56–75) years. Among them, 107 (28.8%) patients were eventually diagnosed with depression at 3 months after ICH.
Compared with subjects without depression after ICH, those with depression had higher NIHSS score (P < 0.001), mRS score (P < 0.001), total white blood cells (WBC) (P = 0.006), absolute neutrophil count (ANC) (P < 0.001), NLR (P < 0.001) and higher proportions of basal ganglia lesions (P = 0.036); besides, the levels of GCS score (P < 0.001), BI score (P < 0.001) and absolute lymphocyte count (ALC) (P < 0.001) were lower (Table 1).
 |
Table 1 Comparisons of Baseline Characteristics According to the Presence or Absence of Depression After ICH |
Table 2 shows ascending tertiles of NLR was associated with older age (P = 0.030), male sex (P = 0.002), lower education years (P = 0.048) and smoking (P = 0.038), also with higher NIHSS score (P = 0.001), mRS score (P < 0.001) and lower GCS score (P < 0.001) and BI score (P < 0.001). The numbers of patients with depression after ICH were 15 (14%), 30 (28%) and 62 (58%) in Q1, Q2 and Q3, respectively. The proportion of subjects with depression in the highest tertile was higher than those in the two lower tertiles (χ2 = 45.37, P < 0.001).
 |
Table 2 Comparisons of Baseline Characteristics According to NLR Tertiles |
In the unadjusted multivariable logistic regression analysis, NLR was independently associated with depression after ICH (OR 1.50, 95% CI 1.35–1.66, P < 0.001). The association remained significant after adjusting for the potential confounders (OR 2.25, 95% CI 1.45–3.49, P < 0.001) (Table 3).
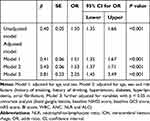 |
Table 3 Logistic Regression Model of NLR and Depression After ICH at 3 Months |
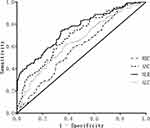
ROC curve analysis showed that NLR was the best discriminating variable, with the best predictive threshold of 4.53 (sensitivity 73.8%, specificity 66.0%, positive likelihood ratio 2.17, and negative likelihood ratio 0.40). DeLong test indicated that the AUC of NLR was evidently greater than those of the other groups (Z = −4.76, P <0.001, compared with WBC; Z = −2.34, P = 0.02, compared with ANC; Z = −4.19, P <0.001, compared with ALC) (Figure 1).
Discussion
This study observed a 2.25-fold higher risk of depression after ICH among individuals with the highest tertiles of NLR compared to those with the lowest NLR tertiles after adjusting for major confounders. To the best of our knowledge, this is the first study to investigate the association between NLR and depression after ICH.
In the current study, we observed that the incidence of depression at 3 months after ICH was 28.8%, which was almost consistent with that of previous report.2 In addition, our study found that patients with depression after ICH had a more severe condition and a poorer prognosis, which were in agreement with previous studies on the risk factors of PSD.17,18 The frontal lobe and basal ganglia are the core brain areas of the emotional network, and any damage in these brain areas will result in depressive symptoms.19 A similar result was found in our study, that patients with depression were more likely to have basal ganglia lesions. However, there was no significant difference of frontal lobe lesion between patients with and without depression after ICH, which might be ascribed to a small number of injuries at frontal lobe in our research. Therefore, it needs to be further verified in a larger sample size.
The association between NLR and depression after ICH could be explained by the following mechanisms. Firstly, the biological mechanism. Numerous studies have shown that NLR may play an important role in the development of ICH and depression. Once ICH occurs, blood components, such as leukocytes, red blood cells (RBCs) and macrophages, immediately infiltrate into the surrounding brain tissues to activate the inflammatory cells. Notably, numerous animal studies on ICH demonstrate the presence of neutrophil infiltration into the hematoma.20,21 Afterwards, the neutrophils can induce neurotoxicity through a multitude of inflammatory signaling pathways, including the release of cytokines, free radicals, chemokines and other toxic chemicals, ultimately exacerbating the ICH-induced brain injury.22 In contrast, lymphocyte was shown to decrease inflammation in ICH patients.23 Moreover, many studies have shown a close relationship between NLR and depression. Demircan and Aydin et al found that NLR levels were significantly higher in depressive patients.24,25 Inflammation induces the dysfunction of synaptic plasticity by mediating alterations in neurotransmitter, especially the synthesis and metabolism of 5-hydroxytryptamine and glutamine, and eventually leads to depression.26 Therefore, an elevated NLR may represent high levels of inflammation which probably promotes the occurrence of depression after ICH. The second one is the psychological mechanism. Stroke is a serious and highly disabling disease, which is a heavy blow to the mental health of patients. The decline in self-care ability of daily living and the lack of working capacity have rendered unavoidable serious psychological defect in stroke patients, eventually leading to depression.
Some limitations should be noted in this study. Firstly, the NLR level was detected only once on admission, but not dynamically monitored during the course of disease. The combination of baseline and dynamic changes of NLR could provide better prognostic information. Secondly, the connections between NLR and some other inflammatory mediators had not been investigated. Finally, several studies report that some risk factors are related to PSD, including economic status and social support.27,28 However, the influence of these factors was not examined in our research.
Conclusions
To sum up, a higher NLR is independently associated with depression at 3-month after ICH, which suggests that NLR on admission can serve as a significant biomarker of systemic inflammation to predict the occurrence of depression after spontaneous ICH. Further studies regarding the immune mediators will contribute to determining the therapeutic strategies.
Disclosure
All authors declare that they have no conflicts of interest for this work.
References
1. Stern-Nezer S, Eyngorn I, Mlynash M, et al. Depression one year after hemorrhagic stroke is associated with late worsening of outcomes. Neuro Rehabilitation. 2017;41:179–187. doi:10.3233/NRE-171470
2. Koivunen RJ, Harno H, Tatlisumak T, et al. Depression, anxiety, and cognitive functioning after intracerebral hemorrhage. Acta Neurol Scand. 2015;132:179–184. doi:10.1111/ane.12367
3. Christensen MC, Mayer SA, Ferran JM, et al. Depressed mood after intracerebral hemorrhage: the fast trial. Cerebrovasc Dis. 2009;27:353–360. doi:10.1159/000202012
4. Tu J, Wang LX, Wen HF, et al. The association of different types of cerebral infarction with post-stroke depression and cognitive impairment. Medicine. 2018;97:e10919. doi:10.1097/MD.0000000000010919
5. Sibolt G, Curtze S, Melkas S, et al. Post-stroke depression and depression-executive dysfunction syndrome are associated with recurrence of ischaemic stroke. Cerebrovasc Dis. 2013;36:336–343.
6. Bartoli F, Di Brita C, Crocamo C, et al. Early post-stroke depression and mortality: meta-analysis and meta-regression. Front Psychiatry. 2018;9:530. doi:10.3389/fpsyt.2018.00530
7. Fang M, Zhong L, Jin X, et al. Effect of inflammation on the process of stroke rehabilitation and poststroke depression. Front Psychiatry. 2019;10:184. doi:10.3389/fpsyt.2019.00184
8. Meng G, Ma X, Li L, et al. Predictors of early-onset post-ischemic stroke depression: A cross-sectional study. BMC Neurol. 2017;17:199. doi:10.1186/s12883-017-0980-5
9. Kang HJ, Bae KY, Kim SW, et al. Effects of interleukin-6, interleukin-18, and statin use, evaluated at acute stroke, on post-stroke depression during 1-year follow-up. Psychoneuroendocrinology. 2016;72:156–160. doi:10.1016/j.psyneuen.2016.07.001
10. Kim JM, Kang HJ, Kim JW, et al. Associations of tumor necrosis factor-α and interleukin-1β levels and polymorphisms with post-stroke depression. Am J Geriatr Psychiatry. 2017;25:1300–1308. doi:10.1016/j.jagp.2017.07.012
11. Wu Y, Wang L, Hu K, et al. Mechanisms and therapeutic targets of depression after intracerebral hemorrhage. Front Psychiatry. 2018;9:682.
12. Xue P, Kanai M, Mori Y, et al. Neutrophil-to-lymphocyte ratio for predicting palliative chemotherapy outcomes in advanced pancreatic cancer patients. Cancer Med. 2014;3:406–415. doi:10.1002/cam4.204
13. Wang X, Zhang G, Jiang X, et al. Neutrophil to lymphocyte ratio in relation to risk of all-cause mortality and cardiovascular events among patients undergoing angiography or cardiac revascularization: A meta-analysis of observational studies. Atherosclerosis. 2014;234:206–213. doi:10.1016/j.atherosclerosis.2014.03.003
14. Chen H, Luan X, Zhao K, et al. The association between neutrophil-to-lymphocyte ratio and post-stroke depression. Clin Chim Acta. 2018;486:298–302. doi:10.1016/j.cca.2018.08.026
15. Brott T, Adams HP
16. Hamilton M. A rating scale for depression. J Neurol Neurosurg Psychiatry. 1960;23:56–62. doi:10.1136/jnnp.23.1.56
17. Ayerbe L, Ayis S, Rudd AG, et al. Natural history, predictors, and associations of depression 5 years after stroke: the south london stroke register. Stroke. 2011;42:1907–1911. doi:10.1161/STROKEAHA.110.605808
18. Tsai CS, Wu CL, Hung TH, et al. Incidence and risk factors of poststroke depression in patients with acute ischemic stroke: A 1-year prospective study in taiwan. Biomed J. 2016;39:195–200. doi:10.1016/j.bj.2015.10.004
19. Jastorff J, Huang YA, Giese MA, et al. Common neural correlates of emotion perception in humans. Hum Brain Mapp. 2015;36:4184–4201.
20. Gong C, Hoff JT, Keep RF. Acute inflammatory reaction following experimental intracerebral hemorrhage in rat. Brain Res. 2000;871:57–65. doi:10.1016/S0006-8993(00)02427-6
21. Wang J, Tsirka SE. Neuroprotection by inhibition of matrix metalloproteinases in a mouse model of intracerebral haemorrhage. Brain. 2005;128:1622–1633. doi:10.1093/brain/awh489
22. Chen S, Yang Q, Chen G, et al. An update on inflammation in the acute phase of intracerebral hemorrhage. Transl Stroke Res. 2015;6:4–8. doi:10.1007/s12975-014-0384-4
23. Morotti A, Marini S, Jessel MJ, et al. Lymphopenia, infectious complications, and outcome in spontaneous intracerebral hemorrhage. Neurocrit Care. 2017;26:160–166. doi:10.1007/s12028-016-0367-2
24. Demircan F, Gözel N, Kılınç F, et al. The impact of red blood cell distribution width and neutrophil/lymphocyte ratio on the diagnosis of major depressive disorder. Neurol Therapy. 2016;5:27–33. doi:10.1007/s40120-015-0039-8
25. Aydin Sunbul E, Sunbul M, Yanartas O, et al. Increased neutrophil/lymphocyte ratio in patients with depression is correlated with the severity of depression and cardiovascular risk factors. Psychiatry Investig. 2016;13:121–126. doi:10.4306/pi.2016.13.1.121
26. Müller N, Schwarz MJ. The immune-mediated alteration of serotonin and glutamate: towards an integrated view of depression. Mol Psychiatry. 2007;12:988–1000.
27. Park EY, Kim JH. An analysis of depressive symptoms in stroke survivors: verification of a moderating effect of demographic characteristics. BMC Psychiatry. 2017;17:132. doi:10.1186/s12888-017-1292-4
28. Volz M, Möbus J, Letsch C, et al. The influence of early depressive symptoms, social support and decreasing self-efficacy on depression 6 months post-stroke. J Affect Disord. 2016;206:252–255. doi:10.1016/j.jad.2016.07.041
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.