Back to Journals » Cancer Management and Research » Volume 12
Efficiency and Tolerability of Induction and Consolidation Therapy with Arsenic Trioxide/Bortezomib/Ascorbic Acid/Dexamethasone (ABCD) Regimen Compared to Bortezomib/Dexamethasone (BD) Regimen in Newly Diagnosed Myeloma Patients
Authors Qian W, Wang L, Li P, Hu Y, Wang Q, Yi K, Wu M, Xu Y, Song J, Chen P, Zhang H, Ma J , Xie Y
Received 16 April 2019
Accepted for publication 5 November 2019
Published 20 January 2020 Volume 2020:12 Pages 431—441
DOI https://doi.org/10.2147/CMAR.S212455
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Kenan Onel
Wensi Qian, 1,* Li Wang, 2,* Pei Li, 3,* Yingwei Hu, 1 Qian Wang, 3 Ke Yi, 1 Min Wu, 1 Yu Xu, 1 Jin Song, 1 Pingping Chen, 1 Hongdi Zhang, 1 Jiexian Ma, 1, 4 Yanhui Xie 1
1Department of Hematology, Huadong Hospital Affiliated with Fudan University, Shanghai, People’s Republic of China; 2Department of Hematology, The Affiliated Yantai Yuhuangding Hospital of Qingdao University, Qingdao, People’s Republic of China; 3Department of Hematology, Huashan Hospital Affiliated with Fudan University, Shanghai, People’s Republic of China; 4Shanghai Key Laboratory of Clinical Geriatric Medicine, Huadong Hospital Affiliated with Fudan University, Shanghai, People’s Republic of China
*These authors contributed equally to this work
Correspondence: Yanhui Xie; Jiexian Ma
Department of Hematology, Huadong Hospital Affiliated with Fudan University, Ma 221 West Yan’an Road, Shanghai 200040, People’s Republic of China
Tel +86-21-62483180 Ext.80216;
+86-21-62483180 Ext.81303
Fax +86-21-62495490
Email [email protected] [email protected]
Purpose: This study was aimed at comparing the efficacy and tolerability of an arsenic trioxide/bortezomib/ascorbic acid/dexamethasone (ABCD) regimen with efficacy and tolerability of a bortezomib/dexamethasone (BD) regimen in patients with newly diagnosed myeloma.
Patients and Methods: Fifty-seven and sixty-four patients were treated with the ABCD and BD regimens, respectively. Eligible and agreeable patients received autologous hematopoietic stem cell transplantation followed by consolidation.
Results: The response rates (above VGPR) were 74.1% and 32.8% in the ABCD- and BD-treated groups, respectively (P = 0.000). Compared to BD regimen, ABCD regimen significantly improved PFS (P = 0.026) and OS (P = 0.000) in newly diagnosed patients. Patients with a high tumor burden, low or standard risk, and without auto-HSCT seemed to especially benefit compared to the same group with BD regimen. ABCD also showed better tolerability with lower bone marrow suppression (P = 0.026). Furthermore, complete response or near CR after induction therapy was a good prognostic factor for ABCD-associated OS and PFS.
Conclusion: ABCD is an effective and tolerable regimen compared with BD regimen in newly diagnosed myeloma patients. ABCD regimen could be an economical, effective, and tolerable choice in low- and standard-risk patients.
Keywords: multiple myeloma, arsenic trioxide, bortezomib, overall survival, treatment response
Introduction
Multiple myeloma (MM) is a disease that typically requires multiple lines of therapy because many patients have a relatively long survival and relapse and gradually develop resistance to the treatment drugs.1,2 Many novel drugs are emerging for the treatment of MM, such as bortezomib, lenalidomide, pomalidomide, carfilzomib and daratumumab, clinicians tried to single administration or in combination of these drugs have improved responses and outcomes.3–5 Bortezomib and bortezomib-based therapies, including bortezomib plus dexamethasone, are now a cornerstone of treatment for both newly diagnosed and relapsed/refractory MM.6 A third agent added to the bortezomib and dexamethasone regimen has proven effective for the treatment of relapsed/refractory MM in a number of studies,7–10 and three-drug regimens are increasingly recommended for MM patients.11,12
Arsenic trioxide (ATO) is a promising antineoplastic chemotherapeutic agent, it has been approved to the treatment of acute promyeloid leukemia (APL), now it is tried in the treatment of MM. In preclinical studies, ATO induced apoptosis, reduced viability, and caused growth arrest in myeloma cell lines at concentrations low enough for safe use in patients.13–15 ATO exert its antitumor effects in part by generating reactive oxygen species (ROS).16 The cytotoxic effects of ATO in myeloma cell lines are markedly enhanced by the addition of ascorbic acid, as reported both in vitro and in vivo.17 intracellular glutathione (GSH) will neutralize the ROS generated by ATO, ascorbic acid could deplete GSH, which makes a contribution to the synergy of ATO and ascorbic acid. This supposition is supported by the findings of a small Phase I study in patients with stage III relapsed or refractory MM. The study showed ascorbic acid administration decreased intracellular GSH levels and increased the sensitivity of patients’ myeloma cells to ATO.18 Some in vitro studies showed that the sensitivity of myeloma cells to bortezomib is negatively associated with beta-catenin protein levels. After proteasome inhibition, ATO can reduce cytoplasmic beta-catenin accumulation and enhance the sensitivity of myeloma cells to bortezomib.19
Preclinical studies have also shown that ATO combined with bortezomib at low concentrations has synergistic antiproliferative and antimyeloma activity in xenograft animal models,17 suggesting the combination may have the potential to treat MM. Phase I/II trials of ATO and bortezomib have been conducted in heavy pretreatment, relapsed or refractory MM patients, while the addition of ATO has limited success in relapsed/refractory MM.20
In clinical trials conducted thus far, the combination of ATO, ascorbic acid, bortezomib, and dexamethasone for the treatment of MM has been evaluated in patients with relapsed MM and MM patients showing treatment resistance. The mechanism of ATO in MM treatment relies heavily on GSH level and the ROS system. Patients with relapsed MM and MM patients showing treatment resistance may show changes in levels of GSH and apoptotic regulators.21 However, all previous studies assessed the safety and tolerability of ATO/bortezomib/ascorbic acid (ABC) combination therapy in MM patients. Therefore, we conducted a retrospective study with an aim to evaluate the efficacy and safety of ATO/bortezomib/ascorbic acid/dexamethasone (ABCD) combination therapy in comparison with those of bortezomib/dexamethasone (BD) regimen for newly diagnosed MM patients at three medical centers in China.
Materials and Methods
Patients
Patients were enrolled from July 2012 to August 2018 from three medical centers. The inclusion criteria were as follows: a diagnosis of MM requiring treatment, age up to 85 years, Zubrod performance status score of <4, left ventricular ejection fraction of >40%, no uncontrolled arrhythmia or unstable cardiac disease, corrected QT interval less than 470 ms, no symptomatic pulmonary disease, adequate pulmonary function test, serum glutamic pyruvic transaminase level of <4× upper limit of normal, serum bilirubin level of <2× upper limit of normal, without chronic active hepatitis or cirrhosis. The remaining inclusion criterion was the absence of effusion or ascites of >1 L prior to drainage. Patients were excluded if they had a known allergy to any of the agents, had previously received arsenic therapy, were >85 years of age, or had uncontrolled diabetes, infections, or underlying illness. However, patients who had received local treatment for control of bone disease were considered eligible. Patients with neuropathy of grade ≥ 2 at the time of study entry were also excluded. Patients also need negative human immunodeficiency virus testing, a negative pregnancy test in a woman with child-bearing potential and the willingness of the patient or guardian to sign informed consent. The study was approved by the ethics committee of Huadong Hospital (approval number: 2018K115) and has been registered online (ChiCTR1800018811)
Treatment Plan
Patients newly diagnosed with MM at Huadong Hospital who satisfied the inclusion criteria of the trial received the ABCD regimen. All patients received ATO 0.16 mg/kg IV on days 1 to 3, 8–10, and 15–17 over 2 hrs; and AA 1000 mg IV on days 1 to 3, 8–10, and 15–17 over 30 mins. Bortezomib at a dose of 1.6 mg/m2 was administered subcutaneously on days 4, 11, and 18, and dexamethasone at a dose of 40 mg/day was administered IV on days 4–7,11–14, and 18–21. If the patient was aged more than 65 years or had diabetes, the dose of dexamethasone was decreased to 20 mg/day. The regimen was repeated every 28 days. Patients newly diagnosed with MM at the Huashan Hospital and Yuhuangding Hospital who satisfied the inclusion criteria of the trial received the BD regimen. Bortezomib was administered subcutaneously on days 4, 11, and 18 at a dose of 1.6 mg/m2, while dexamethasone was administered IV on days 4–7, 11–14, and 18–21 at a dose of 40 mg/day. The dexamethasone dosage was decreased for elderly (age above 65 years old) and diabetes patients. Patients received supportive care according to established departmental guidelines. Further, they received granulocyte colony stimulating factor (G-CSF) 5 μg/kg/day when the absolute neutrophil count (ANC) was less than 0.5 × 109/L for two consecutive days. Oral levofloxacin, acyclovir, and fluconazole were administered for the duration of neutropenia. Blood products were administered if the hemoglobin level was <6 g/dL and platelet count was <20 × 109/L.
After four cycles of chemotherapy, we evaluated the disease status of each patient. If patients achieved partial remission (PR) or very good partial remission (VGPR) or near-complete remission (nCR) or CR and agreed to receive autologous stem cell transplantation, autologous stem cell transplantation was performed with melphalan 200 mg/m2 and stem cell engraftment, followed by four cycles of chemotherapy every 3 months according to the previous regimen. If patients refused autologous stem cell transplantation or only achieve minor response (MR), another four cycles of MP regimen (prednisone 60 mg/(m2·d) days 1–4, melphalan 8 mg/(m2·d) days 1–4 every month) were administered, after which the patients received another four cycles of the ABCD or BD regimen (consistent with the previous regimen) every month. If the patients showed stable disease (SD) or progressive disease (PD) after four cycles of therapy or during the treatment, they were considered for exit this study and changing another therapy such as RVD regimen. The entire treatment plan is shown in Figure 1.
 |
Figure 1 The flowchart of treatment regimens. Regimen A stands for the ABCD regimen, regimen B stands for the BD regimen. |
If patients relapsed or showed disease progression after finishing the entire therapy, the patients will receive the previous regimen as ABCD regimen or BD regimen. After two cycles of chemotherapy, the treatment response was evaluated. If the response remained SD or PD, the patients would be changed to a lenalidomide/bortezomib/dexamethasone regimen and exit this study.
Disease Assessments
Disease assessments (skeletal survey, complete neurologic examination, Karnofsky performance status, beta2-microglobulin, C-reactive protein, serum and urine electrophoresis to quantify immunoglobulins, and immunofixation, bone marrow aspiration and biopsy) were performed within 14 days before day 1 of cycle 1 of treatment. Patients’ medical history was obtained, and physical examination and a complete neurologic were performed at baseline; furthermore, 12-lead electrocardiography and posteroanterior and lateral chest X-rays were performed. Bone marrow aspirate was evaluated and a biopsy was performed in addition to flow cytometry, chromosome analysis and fluorescence in situ hybridization (FISH) analysis. Clinical laboratory tests, including hematology; clinical chemistry (serum creatinine, blood urea nitrogen, uric acid, alkaline phosphatase, total bilirubin, lactate dehydrogenase, alanine transaminase and aspartate transaminase); electrolyte (calcium, magnesium, potassium, sodium, etc.) and glucose panel; total protein, amylase, and albumin tests; urinalysis; and women of child-bearing potential should take serum pregnancy, were performed on day 0 of each cycle. Treatment assessment staging was performed at entry, and restaging was performed every two cycles. Staging/restaging included bone marrow aspirate, serum protein electrophoresis, immunofixation, Ig quantitation, 24-h urine collection with urine protein electrophoresis/immunofixation electrophoresis, beta2-microglobulin, complete blood count, chemistry, lactate dehydrogenase (LDH). Staging and after four cycles of chemotherapy, restaging included biopsy with conventional cytogenetics and FISH analysis for common abnormalities, and radiological testing if indicated.
Response Criteria
Patient responses to treatment were monitored on day 0 of each cycle. Responses were evaluated according to criteria developed by Durie et al22. CR was defined as a negative immunofixation test for the original monoclonal protein (M-protein) from blood and urine, <5% plasma cells in bone marrow, the size or number of lytic bone lesions remain stable, and soft tissue plasmacytomas disappear for at least 6 weeks. Near CR (nCR) was defined as positive immunofixation but negative myeloma protein tests in serum and urine using densitometry on protein electrophoresis. Very good partial remission (VGPR) requires a ≥90% reduction in M‐protein. Partial response (PR) was defined as a ≥50% reduction in serum M-protein, ≥90% reduction in 24-h urinary light-chain excretion or levels <200 mg, no increase in size or number of lytic bone lesions, ≥50% reduction in the size of soft tissue plasmacytomas for at least 6 weeks. A minor response (MR) was defined as a 25% to 49% reduction in serum M-protein and the size of plasmacytomas, a 50% to 89% reduction in 24-h light-chain excretion (although still >200 mg/24 hrs) for at least 6 weeks, the size or number of lytic bone lesions remain stable. Progressive disease was defined as one or more of the following: >25% increase in plasma cells in a bone marrow aspirate or on trephine biopsy (an absolute increase of at least 10%), >25% increase in serum M-protein (confirmed absolute increase of >5 g/L), >25% increase in 24-hrs urinary light-chain excretion (confirmed absolute increase of >200 mg/24 hrs), an increase in the size of lytic bone lesions or soft tissue plasmacytomas, development of new bone lesions or plasmacytomas or hypercalcemia.
OS was defined as the time from the day of diagnosis to death or time the patient was last known to be alive. PFS was the time from the day of diagnosis to progression or time the patient was last known to be still in progression free. All patients were observed until death or the date August 30, 2018. The primary endpoints were VGPR and time to grade 4 toxicity. The secondary endpoints were OS and PFS.
Safety was monitored after 30 days of the last dose of drug. Toxicities were graded according to National Cancer Institute Common Toxicity Criteria of Adverse Events (version3.0; Bethesda,MD). Engraftment was defined as an ANC of 0.5 × 109/L for two consecutive days.
Prognostic Factors
High-risk cytogenetics were defined as del 13 or hypodiploidy as determined by conventional cytogenetics and t(4;14), t(14;16) or del17p as determined by conventional cytogenetics or FISH.23
Statistical Analyses
Patient characteristics were summarized using the mean and standard deviation for numerical valued variables and frequencies with percentages for categorical variables. Differences of patient characteristics between two treatment groups were assessed using the t-test for numerical variables, and for categorical variables, we use Chi-square tests to assess. Kaplan–Meier (KM) method was used to estimate unadjusted probabilities of OS and PFS time. Unadjusted OS and PFS between subgroups were compared by Log-rank test. The joint effects and prognostic factors of patient covariates and treatment arms on OS and PFS were assessed by Cox regression models. SPSS 10.0 software (SPSS Inc.; Chicago, IL, USA) was used to perform all frequent statistical analyses.
Results
Patients and Treatment
A total of 121 patients were enrolled and treated, among which 57 and 64 patients received the ABCD and BD regimens, respectively, and 114 patients finished the entire therapy. Seven patients was changed to the RVD regimen because of poor response to the previous regimen or disease progression. Two of these patients were from the ABCD group because of SD after induction therapy, while five patients were from the BD group because of SD and progressive disease, respectively. Table 1 shows the patients’ baseline characteristics; the mean age in both groups was above 60 years, while gender, disease isotype, Durie-Salmon Stage at diagnosis, ISS stage at diagnosis, plasma blast in bone marrow, LDH level, albumin level, beta2-MG level, and metaphase cytogenetics, blood calcium level and renal function at diagnosis showed no significant differences between these two treatment groups. However, in the FISH analysis, the ABCD group showed a high incidence and significant difference in the incidence of t(4;14) compared with the BD group (P = 0.017). Twelve patients received auto-HSCT in the ABCD group and eight patients received auto-HSCT in the BD group (P = 0.266). All the details are presented in Table 1.
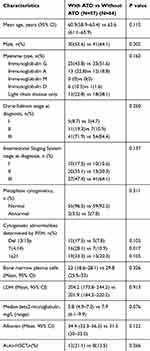 |
Table 1 Baseline Clinical Characteristics of Patients Receiving the Two Regimens |
High tumor burden patients could get greater treatment response with ABCD regimen
Fifty-seven patients received the ABCD regimen, and 64 patients received the BD regimen. All 57 patients who received the ABCD regimen finished induction therapy, and 55 patients finished the entire therapy in the ABCD group. Dexamethasone dose was reduced to 20 mg/d in 24 patients because of age or severe diabetes. All 64 patients who received the BD regimen finished induction therapy, and 57 of these patients finished the entire therapy in the BD group, while seven patients received the RVD regimen because of a poor response to BD regimen, with two of these patients showing progressive disease. The dexamethasone dose was reduced to 20 mg/d in 22 patients. Patient responses are summarized in Table 2. In the ABCD group, 13 patients achieved complete remission, two achieved near-complete remission, 28 achieved VGPR, 11 achieved PR, one achieved MR, and two showed SD. In the BD group, 14 patients achieved complete remission, seven achieved VGPR, 36 achieved PR, five showed SD, and two showed progressive disease. The clinical response in the ABCD and BD groups showed significant differences (P = 0.000). We analyzed the percentage of CR plus nCR patients in these two groups (31.3% vs 21.9%) and found no significant difference (P = 0.568). In contrast, the percentage of patients showing VGPR or a better response (CR + nCR + VGPR) in the ABCD group was higher than that in the BD group (74.1% vs 32.8%, P = 0.000). There was no significant difference in the clinical response rate above PR (93.1% vs 89.0%, P = 0.258).
 |
Table 2 Treatment Response (After Four Cycles) and Analysis of Survival Associated with These Two Regimens |
We also analyzed which group of patients benefited from the ABCD regimen compared with BD regimen by assessing patients in different subgroups, and we found that patients in DSS stage III showed an even better clinical response with the ABCD regimen, and the percentage of CR plus nCR and VGPR was higher and significantly different with the ABCD regimen among these patients (80.5% vs 29.6%, P = 0.000, Table 3). Responses above VGPR (CR + nCR + VGPR) were higher with the ABCD regimen in the low- and standard-risk groups (73.3% vs 34.8%, P = 0.001) as well as in the high-risk group (77.8% vs 27.8%, P = 0.001, Table 3)
 |
Table 3 Treatment Response to the Two Regimens Stratified by the DSS Stage and Risk |
The ABCD Regimen Was Associated with a Longer OS and PFS in Newly Diagnosed Myeloma Patients, and Especially Prominent in High Tumor Burden, Low and Standard Risk Categories,and no Auto-HSCT patients
In the final analysis (September 2018), the ABCD group contained 57 patients, of which 12 died before the end point and did not reach the median OS. The 5-year survival rate in the ABCD group was 64.9%. Twenty-two patients showed disease progression before the end point; the median PFS was 36 months, and the 3-year PFS was 65.8%. The BD group contained 64 patients, of which 41 died before the end point. The median OS was 40.0 (33.7–46.3) months, and the 5-year survival in the BD group was 19.9% (Table 2). Forty patients showed disease progression before the end point; the median PFS was 20 (13.8–26.2) months; and the 3-year PFS was 25.7%. The ABCD regimen was associated with a significantly longer OS (Figure 2A, P = 0.000) and longer PFS (Figure 2B, P = 0.026) than the BD regimen.
We also analyzed subgroup OS and PFS according to the patients’ characteristics in these two regimens. In these assessments, we analyzed age (above 65 years vs under 65 years of age), sex (male vs female), ISS phage (low tumor burden vs high tumor burden), risk stratification (low/standard risk vs high risk) as the history of auto-stem cell transplantation to identify the group of patients who could benefit from the ABCD regimen. It should be noted that patient numbers were small for these analyses, and the results should be interpreted in this context. We found that patients with a high tumor burden at diagnosis (ISS III), those in the low and standard risk categories, and those who did not receive auto-HSCT showed a longer OS and PFS with the ABCD regimen compared with the BD regimen (P = 0.001, 0.001, and 0.001 for OS and P = 0.022, 0.018, and 0.05 for PFS, Table 4), while age under 65 y was associated with a longer OS (P = 0.026) but not PFS (P = 0.46). The summary and details are presented in Table 4.
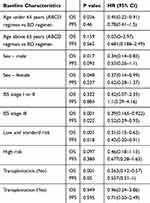 |
Table 4 Comparison of OS and PFS Associated with the Two Regimens Stratified by Baseline Characteristics |
Safety and Tolerability of ABCD regimen
The ABCD combination therapy was well tolerated by most patients. The adverse effects and their frequencies are listed in Table 5. All adverse effects can be handled and controlled by proper treatment, and none of the patients died of the adverse effects. The BD group showed a higher incidence of neutropenia or thrombocytopenia than the ABCD group (n = 2 vs n = 10, P = 0.026), while the ABCD group showed a higher incidence of cardiac complications such as cardiac arrhythmia (n = 8 vs n = 0, P = 0.002) and limb edema (n = 5 vs n = 0, P = 0.016) than the BD group. The incidence of other adverse effects such as neuropathy, diarrhea, constipation, infection, and electrolyte imbalance did not show a significant difference.
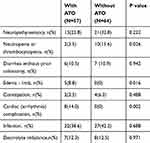 |
Table 5 Comparison of Adverse Effects Associated with the Two Regimens |
Prognostic Factors for Patients Receiving the ABCD Regimen
We performed a multivariate analysis to evaluate the prognostic factors for the OS and PFS independently in the ABCD group. The prognostic influence of several factors (including age, sex, myeloma type, Durie-Salmon Stage at diagnosis, ISS stage, cytogenetics, bone marrow plasma cell, LDH, auto-HSCT, DSS stage, and treatment response) was analyzed. Treatment response CR or nCR after four cycles of chemotherapy was predictive of better OS in the ABCD group (HR = 0.196(0.072–0.535), P = 0.001), which was also the predictive of better PFS in the ABCD group (HR = 0.31(0.13–0.71), P = 0.006). Female patients, ISS stage of I&II stage as well as auto-transplantation were also independent prognostic factor for better OS in the ABCD group (p=0.02, 0.003, 0.019, respectively) while not a prognostic factor for longer PFS. All the details are provided in Table 6.
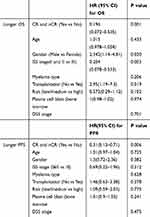 |
Table 6 Longer OS and PFS, as Prognostic Factors, Associated with the ABCD Regimen |
Discussion
An important objective of this trial was to evaluate the feasibility and safety of induction and consolidation therapy with our ABCD regimen in comparison with the standard BD regimen. This study has shown that the strategy is feasible (96% completed the planned sequence), with favorable tolerability and no treatment-related mortality. In comparison with the BD regimen, the ABCD regimen showed a lower incidence of bone marrow suppression and a slightly higher incidence of limb edema and cardiac arrhythmia, which were acceptable and controllable.
The major goal of this trial was to explore the efficiency of this regimen in comparison with the standard BD regimen. Importantly, we noted high response rates. The ABCD regimen yielded VGPR or better response rates of 74% after four cycles of chemotherapy, while only 32.8% of the patients in the BD group attained these response levels. Patients with a high tumor burden at diagnosis especially derived benefits from this regimen in the induction period. The ABCD regimen also yielded a longer OS and PFS than the BD regimen. Patients with low and standard risk and a high tumor burden at diagnosis as well as those who did not receive auto-HSCT could achieve longer OS and PFS with the ABCD regimen. For high-risk patients, the ABCD regimen showed better treatment response than the BD regimen, but the OS and PFS did not show any significant difference between these two groups. For these patients, some reported study recommend lenalidomide/bortezomib/dexamethasone regimen and carfilzomib/lenalidomide/dexamethasone (CRd) regimen.5,24–26 As reported, the 3-year OS and PFS of the RVD regimen was 100% and 77%;27 for CRd regimen, at a median follow up of 33.5 months, OS was 100% and PFS was 66%. In our study, the ABCD regimen had a 3-year OS of 78% and 3-year PFS was 65.8%. While the financial cost of the RVD regimen and the CRd regimen was much higher than that of the ABCD regimen, and both regimens were associated with a higher incidence of a second tumor and stem cell impairment.26,28,29
Previous studies have explored the ABCD regimen in relapsed/refractory myeloma, but none showed any response improvement. Many in vitro studies reported that ATO and bortezomib show synergistic effects in inducing myeloma cell apoptosis.17 Therefore, they may also have some mechanisms in common, and thus, when patients show resistance to bortezomib treatment, they may also show resistance to ATO, which is why the ABCD regimen did not yield a good response in these relapsed/refractory myeloma patients. However, in newly diagnosed patients, the ABCD regimen seems to be promising and shows a better outcome than the standard BD regimen, especially for low- and standard-risk patients and patients who refused or are ineligible for auto-HSCT, ABCD could be a safe, effective and economical choice for such patients.
Patients showing a better response (those who achieved CR and nCR after induction therapy) to the treatment seemed to have longer OS and PFS in this study. There is another very interesting phenomenon, females become an independent prognostic factor in ABCD regimen for better OS. Previous study showed physiological levels of androgens can induce oxidative stress (manifested as lower glutathione levels) that could result partially from increased mitochondrial activity.30 In human kidney, CBS (precursor of glutathione) activity is 35% higher in women than in men.31 As we know, the mechanism of ascorbic acid and arsenic was just dependent on glutathione and ROS activity, whether there was any correlation, we do not know now, but the mechanism of this regimen needs to be further studied and the effect of this regimen needs to be validated in an open, multicenter, randomized clinical trial in the future.
Abbreviations
ABCD, arsenic/bortezomib/ascorbic acid/dexamethasone; BD, bortezomib/dexamethasone; RVD, lenalidomide//bortezomib/dexamethasone; CRd, carfilzomib/lenalidomide/dexamethasone; OS, overall survival; PFS, progression-free survival; CR, complete remission; nCR, near-complete remission; VGPR, very good partial remission; PR, partial remission; SD, stable disease; ISS, International Staging System; DSS, Durie-Salmon Staging; HSCT, hematologic stem cell transplantation; ATO, arsenic trioxide; DLT, dose-limiting toxicity; GSH, intracellular glutathione; ROS, reactive oxygen specie.
Ethics Approval and Consent
This work approved by the ethics committee of Huadong Hospital (approval number: 2018K115), all the participant has signed informed consent after the enrollment. All of the methods were performed in accordance with the Declaration of Helsinki and the relevant guidelines. All the participants and the contributors of this work have signed informed consent for publication.
Data Sharing Statement
All the data will be available from: http://www.chictr.org/showproj.aspx?proj=31726 after the paper publication. Individual participant data that underlie the results reported in this article as well as study protocol will be available. Study protocol will be available for anyone who wishes to access the data, but individual data will be available only for researchers who provide a methodologically sound proposal. Proposals should be directed to [email protected] or [email protected]; to gain access, requestors will need to sign a data access agreement.
Author Contributions
Wensi Qian, Min Wu, Qian Wang, Yingwei Hu and Li Wang collected patients’ data. Jiexian Ma analyzed the data and wrote the manuscript. Pei Li, Yu Xu, Jin Song, Pingping Chen, Hongdi Zhang and Ke Yi took part in the treatment of patients. Yanhui Xie and Jiexian Ma conducted this study and revised the manuscript. All authors made substantial contributions to conception and design, acquisition of data or analysis and interpretation of data; took part in drafting the article or revising it critically for important intellectual content; gave final approval of the version to be published; and agree to be accountable for all aspects of the work.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Kumar SK, Therneau TM, Gertz MA, et al. Clinical course of patients with relapsed multiple myeloma. Mayo Clin Proc. 2004;79:867–874. doi:10.4065/79.7.867
2. Moreau P. The future of therapy for relapsed/refractory multiple myeloma: emerging agents and novel treatment strategies. Semin Hematol. 2012;49(Suppl 1):S33–S46. doi:10.1053/j.seminhematol.2012.05.004
3. Gozzetti A, Candi V, Papini G, Bocchia M. Therapeutic advancements in multiple myeloma. Front Oncol. 2014;4:241. doi:10.3389/fonc.2014.00241
4. Richardson PG, Hideshima T, Mitsiades C, Anderson KC. The emerging role of novel therapies for the treatment of relapsed myeloma. J Natl Compr Cancer Netw. 2007;5:149–162. doi:10.6004/jnccn.2007.0015
5. Korde N, Roschewski M, Zingone A, et al. Treatment with carfilzomib-lenalidomide-dexamethasone with lenalidomide extension in patients with smoldering or newly diagnosed multiple myeloma. JAMA Oncol. 2015;1(6):746. doi:10.1001/jamaoncol.2015.2010
6. Moreau P, Richardson PG, Cavo M, et al. Proteasome inhibitors in multiple myeloma: 10 years later. Blood. 2012;120:947–959. doi:10.1182/blood-2012-04-403733
7. Ciolli S, Leoni F, Gigli F, Rigacci L, Bosi A. Low dose Velcade, thalidomide and dexamethasone (LD-VTD): an effective regimen for relapsed and refractory multiple myeloma patients. Leuk Lymphoma. 2006;47:171–173. doi:10.1080/10428190500272721
8. Dimopoulos MA, Beksac M, Benboubker L, et al. Phase II study of bortezomib-dexamethasone alone or with added cyclophosphamide or lenalidomide for sub-optimal response as second-line treatment for patients with multiple myeloma. Haematologica. 2013b;98:1264–1272. doi:10.3324/haematol.2013.084376
9. Garderet L, Iacobelli S, Moreau P, et al. Superiority of the triple combination of bortezomib-thalidomide-dexamethasone over the dual combination of thalidomide-dexamethasone in patients with multiple myeloma progressing or relapsing after autologous transplantation: the MMVAR/IFM 2005-04 randomized PhaseIII TRIAL from the chronic leukemia working Party of the European Group for blood and marrow transplantation. J Clin Oncol. 2012;30:2475–2482. doi:10.1200/JCO.2011.37.4918
10. Jakubowiak A, Offidani M, Pégourie B, et al. Randomized Phase 2 study: elotuzumab plus bortezomib/dexamethasone vs bortezomib/dexamethasone for relapsed/refractory MM. Blood. 2016;127:2833–2840. doi:10.1182/blood-2016-01-694604
11. Ludwig H, Sonneveld P, Davies F, et al. European perspective on multiple myeloma treatment strategies in 2014. Oncologist. 2014b;19:829–844. doi:10.1634/theoncologist.2014-0042
12. Vincent RS. Multiple myeloma: 2014 update on diagnosis, risk-stratification, and management. Am J Hematol. 2014;89:999–1009.
13. Hideshima T, Chauhan D, Richardson P. NF kappa B as a therapeutic target in multiple myeloma. J Biol Chem. 2002;277:16639–16647. doi:10.1074/jbc.M200360200
14. Park WH, Seol JG, Kim ES. Arsenic trioxide-mediated growth inhibition in MC/CAR myeloma cells via cell cycle arrest in association with induction of cyclin dependent kinase inhibitor, p21, and apoptosis. Cancer Res. 2000;60:3065–3071.
15. Perkin SC, Kim CN, Fang G, Bhalla KN. Arsenic induces apoptosis of multidrug-resistant human myeloid leukemia cells that express Bcr-Abl or overexpress MDR, MRP, Bcl-2, or Bcl-x(L). Blood. 2000;95:1014–1022. doi:10.1182/blood.V95.3.1014.003k04_1014_1022
16. Jing Y, Dai J, Chalmers-Redman RM, Tatton WG, Waxman S. Arsenic trioxide selectively induces acute promyelocytic leukemia cell apoptosis via a hydrogen peroxide-dependent pathway. Blood. 1999;94:2102–2111. doi:10.1182/blood.V94.6.2102
17. Campbell RA, Chen H, Zhu D. Ascorbic acid overcomes drug resistance in myeloma and significantly increases the anti-myeloma effects of both arsenic trioxide and melphalan in vitro and in vivo. Blood. 2004;104:2470.
18. Bahlis NJ, Cafferty-Grad J, Jordan-McMurrel I. Feasibility and correlates of arsenic trioxide combined with ascorbic acid-mediated depletion of intracellular glutathione for the treatment of relapsed/refractory multiple myeloma. Clin Cancer Res. 2002;8:3658–3668.
19. Zhou L, Hou J, Fu W, Wang D, Yuan Z, Jiang H. Arsenic trioxide and 2-methoxyestradiol reduce beta-catenin accumulation after proteasome inhibition and enhance the sensitivity of myeloma cells to Bortezomib. Leuk Res. 2008;32(11):1674–1683. doi:10.1016/j.leukres.2008.03.039
20. Hussein MA, Saleh M, Ravandi F, Mason J, Rifkin RM, Ellison R. Phase 2 study of arsenic trioxide in patients with relapsed or refractory multiple myeloma. Br J Haematol. 2004;125:470–476. doi:10.1111/bjh.2004.125.issue-4
21. Matulis SM, Morales AA, Yehiayan L, Lee KP, Cai Y, Boise LH. Alterations in glutathione levels and apoptotic regulators are associated with acquisition of arsenic trioxide resistance in multiple myeloma. PLoS One. 2012;7:e52662. doi:10.1371/journal.pone.0052662
22. Durie BG, Harousseau JL, Miguel JS, et al. International uniform response criteria for multiple myeloma. Leukemia. 2006;20(9):1467–1473. doi:10.1038/sj.leu.2404284
23. Badros AZ. In the age of novel therapies, what defines high-risk multiple myeloma? J Natl Compr Canc Netw. 2010;8(Suppl 1):S28–S34. doi:10.6004/jnccn.2010.0114
24. Nooka AK, Kaufman JL, Muppidi S, et al. Consolidation and maintenance therapy with lenalidomide, bortezomib and dexamethasone (RVD) in high-risk myeloma patients. Leukemia. 2014;28:690–693. doi:10.1038/leu.2013.335
25. Richardson PG, Xie W, Jagannath S, et al. A phase II trial of lenalidomide, bortezomib and dexamethasone in patients with relapsed and relapsed/refractory myeloma. Blood. 2014;123(10):1461–1469. doi:10.1182/blood-2013-07-517276
26. Chaudhry M, Steiner R, Claussen C, et al. Carfilzomib-based combination regimens are highly effective frontline therapies for multiple myeloma and Waldenstrom’s macroglobulinemia. Leuk Lymphoma. 2019;60(4):964–970. doi:10.1080/10428194.2018.1508668
27. Roussel M, Lauwers-Cances V, Robillard N, et al. Front-line transplantation program with lenalidomide, bortezomib, and dexamethasone combination as induction and consolidation followed by lenalidomide maintenance in patients with multiple myeloma: a phase II study by the Intergroupe Francophone du Myélome. J Clin Oncol. 2014. 32:2712–2717.
28. Palumbo A, Bringhen S, Kumar SK, et al. Second primary malignancies with lenalidomide therapy for newly diagnosed myeloma: a meta-analysis of individual patient data. Lancet Oncol. 2014;15(3):333–342. doi:10.1016/S1470-2045(13)70609-0
29. Popat U, Saliba R, Thandi R, et al. Impairment of filgrastim-induced stem cell mobilization after prior lenalidomide in patients with multiple myeloma. Biol Blood Marrow Transplant. 2009;15(6):718–723. doi:10.1016/j.bbmt.2009.02.011
30. Ripple MO, Henry WF, Rago RP, Wilding G. Prooxidant-antioxidant shift induced by androgen treatment of human prostate carcinoma cells. J Natl Cancer Inst. 1997;89:40–48. doi:10.1093/jnci/89.1.40
31. Vitvitsky V, Prudova A, Stabler S, Dayal S, Lentz SR, Banerjee R. Testosterone regulation of renal cystathionine â-synthase: implications for sex-dependent differences in plasma homocysteine levels. Am J Physiol Renal. 2007;293:F594–600. doi:10.1152/ajprenal.00171.2007
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.

