Back to Journals » Neuropsychiatric Disease and Treatment » Volume 11
Efficacy and safety of intravenous nimodipine administration for treatment of hypertension in patients with intracerebral hemorrhage
Authors Li Y, Fang W, Tao L, Li M , Yang Y, Gao Y, Ge S, Gao L, Zhang B, Li Z, Zhou W, Wang B, Li L, Tian Q
Received 2 November 2014
Accepted for publication 12 December 2014
Published 19 May 2015 Volume 2015:11 Pages 1231—1238
DOI https://doi.org/10.2147/NDT.S76882
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 4
Editor who approved publication: Professor Wai Kwong Tang
Yuqian Li,1,* Wei Fang,1,* Lei Tao,2,* Min Li,1 Yanlong Yang,1 Yafei Gao,1 Shunnan Ge,1 Li Gao,1 Bin Zhang,1 Zhihong Li,1 Wei Zhou,1 Boliang Wang,3 Lihong Li1
1Department of Neurosurgery, 2Department of Anesthesiology, 3Department of Emergency, Tangdu Hospital, The Fourth Military Medical University, Xi’an, Shaanxi, People’s Republic of China
*These authors contributed equally to this study
Background: Nicardipine (NC) is the most commonly used antihypertensive drug in neurological patients with hypertension. Although nimodipine (NM) is widely used to treat cerebral vasospasm in patients with aneurysmal subarachnoid hemorrhage, trials exploring its antihypertensive effect after intravenous administration in subjects with intracerebral hemorrhage (ICH) are scarce.
Methods: A retrospective study was carried out to compare the safety and efficacy of NC and NM administered intravenously in patients with ICH. Therapeutic responses were assessed by achievement of goal blood pressure (BP); use of additional medications for BP control; proportion of time spent within goal; variability in BP; time to goal BP; number of dose adjustments; variability in ICH volume, Glasgow Coma Scale score, and intracranial pressure; and drug-related complications.
Results: A total of 87 patients were eligible for analysis (n=46 [NC]; n=41 [NM]), and baseline characteristics between groups were similar. Both agents were effective in achieving goal BP during infusion, with 93.5% and 87.8% patients in the NC and NM groups achieving goal, respectively. Fewer additional medications were needed to control BP in the NC group. BP variability was similar and no differences were observed in the mean time to goal BP and mean numbers of dose adjustments between both groups. Interestingly, intracranial pressure declined (P=0.048) during NC administration but increased (P=0.066) after NM treatment. Finally, the incidences of hematoma expansion, neurological deterioration, and adverse drug events were similar in both groups.
Conclusion: NM is effective and safe for BP control in patients with ICH.
Keywords: nicardipine, blood pressure, intravenous infusion
Introduction
Spontaneous, nontraumatic intracerebral hemorrhage (ICH) leads to high morbidity and mortality worldwide. Indeed, ICH accounts for 10%–17% of all strokes.1–4 In the United States, 37,000–52,400 people suffer from ICH each year.5 The median 1-month case lethality rate is 40%, and only 12% to 39% of patients achieve functional independence.6 Hypertension, the most common risk factor for ICH, is observed in 70% to 80% of patients with ICH.5 Furthermore, in patients with ICH, elevated blood pressure (BP) results in hematoma expansion and rebleeding at 3 months,7,8 which subsequently leads to poor outcomes. Previous studies have identified elevated BP, especially systolic BP (SBP) over 200 mmHg, as an important risk factor for bleeding in primary and recurrent ICH.5,9–12 These findings indicate the serious threat related to BP. The aim of antihypertensive therapy is to lower BP quickly but safely, avoiding complications. Since intravenous (IV) nicardipine (NC) is feasible and safe to treat hypertension in neurological patients with unfavorable outcome, it is recommended as a first-line agent in neurological intensive care unit (NICU) settings in the United States.13,14
Nimodipine (NM), an L-type calcium channel antagonist, is recommended in the prevention and treatment of cerebral vasospasm, and Class I data only exist for its use in aneurysmal subarachnoid hemorrhage.13 Treatment with NM reduces the severity of neurological deficits resulting from vasospasm in subarachnoid hemorrhage patients. However, the BP-lowering effects of IV NM should not be minimized, a clinical study has documented lowering BP in patients after acute stroke.15 Like other calcium channel blockers, such as NC, NM has antihypertensive effects – its main mechanism of action is vasodilatation, which causes decreased peripheral vascular resistance.16 Previous study has demonstrated that IV infusion of NM leads to BP lowering in patients with acute stroke.15 In our clinical work, we found that IV NM rapidly lowered BP. Despite the antihypertensive effect of NM administered intravenously, trials exploring the efficacy and safety of IV NM for treating hypertension in neurological patients with ICH are scarce, and no direct comparison between NC and NM delivered as continuous infusions in strictly ICH patients has been reported. We hypothesized that there were no differences in efficacy and safety profile between continuous infusions of NM and NC for management of hypertension in patients with ICH.
Patients and methods
Patients
A total of 87 patients were enrolled in this study performed between June 2013 and March 2014 in the NICU of Fourth Military Medical University Tangdu Hospital. As it was a retrospective study, neither informed consent nor approval of the ethics committee was needed. Admission data collected from each eligible patient included race; age; sex; weight; heart rate (HR); history of smoking, alcohol consumption, diabetes and hypertension; intracranial pressure (ICP); Glasgow Coma Scale (GCS) score; SBP; and intracerebral hemorrhage (ICH) volume. The inclusion and exclusion criteria used in this study are summarized in Table 1.
General care
All patients received the standard intensive care management. Critically ill patients with GCS scores <9 were mechanically ventilated. If necessary, patients were treated with midazolam (0.2 mg/kg/h, increased as needed) for sedation and fentanyl (2 μg/kg/h, increased as needed) for analgesia. Besides, blood glucose values were kept between 6–10 mmol/L and the core temperature between 36.0°C and 37.0°C. Blood gases were tested at least once a day to avoid hypercarbia, hypoxemia or electrolyte imbalance. During a period of infusion, cerebral perfusion pressure (CPP) was kept at ≥70 mmHg. Central venous pressure (CVP) was maintained between 4 and 12 cm H2O to avoid hypervolemia or hypovolemia. Mannitol (20%), at a dose of 0.25 to 1 g/kg, was given as an IV bolus in patients with ICP >20 mmHg. Surgery was considered if the ICP remained at >20 mmHg for 30 minutes.
Methods
A retrospective study was conducted with patients showing SBP >140 mmHg – the goal for BP was to reduce and maintain SBP between 120 and 140 mmHg. Patients were administered either NC (n=46) or NM (n=41), intravenously from admission. The following antihypertensive management protocol was used (Figure 1): NC was administered as a continuous infusion with an hourly starting dose of 2 mg/h; if the BP goal was not achieved, the dose was increased by 2 mg/h every 10 minutes as needed, up to a maximum of 15 mg/h. The use was limited when side effects appeared before the maximum dose was reached. Likewise, NM was administered at a starting dose of 1 mg/h, and the dose was increased by 0.5 mg/h every 10 minutes, up to a maximum of 4 mg/h. Once the BP fell below 140 mmHg, IV drug amounts were gradually reduced, and if there was any rebound effect, we returned and maintained the drug to control BP. Nitroprusside was administered if the SBP was still greater than 140 mmHg 30 minutes after the maximum dose had been given. If side effects associated with both drugs limited their use during infusion, antihypertensive medications were discontinued. No patients in either group had oral antihypertensive medications during this period.
  | Figure 1 Antihypertensive management protocol. |
Statistical analysis
The Kolmogorov–Smirnov test was used to assess the distribution of continuous variables. Normally distributed data were reported as mean or mean percentage. The comparison of variables was carried out by independent-samples t-test or paired-samples t-test. Categorical variables were compared using the chi-square test. P<0.05 was considered statistically significant.
Results
Patient characteristics
A total of 87 Chinese patients were included in this study; of these, 46 patients received NC, with 32 patients within 6 hours of ICH symptom onset, while 41 individuals received NM, with 33 patients within 6 hours of symptom onset (Figure 2). Baseline characteristics were similar, with no significant differences observed between the groups initially. Overall, 61% and 54% were female in the NC and NM groups, respectively (P=0.497). The mean age was 62±9 years in the NC group and 60±8 years for NM patients (P=0.684). In addition, there was no statistical difference between both groups in baseline mean SBP (176±17 mmHg [NC] vs 172±19 mmHg [NM]) (P=0.399), mean HR (88±25 bpm [NC] vs 91±25 bpm [NM]) (P=0.820), or mean weight (62±10 kg [NC] vs 65±11 kg [NM]) (P=0.523). Further, the mean ICH volume was similar between both groups, with 29±11 mL obtained in NC subjects and 31±10 mL in the NM group (P=0.352). Moreover, no statistically significant difference was observed in mean GCS scores (9±3 [NC] vs 9±2 [NM]) (P=0.417) or ICP (18±8 mmHg [NC] vs 14±6 mmHg [NM]) (P=0.186) at admission (Table 2).
  | Figure 2 Study protocol diagram. |
Efficacy and safety
Electrocardiographic data were recorded for all patients treated with the present antihypertensive protocol after admission to the NICU. All patients had noninvasive monitoring devices to measure BP every 5 minutes, and all the values of ICP were acquired by ventricular ICP monitors. The hemodynamic indices recorded over time for both groups are shown in Figure 3 and Table 3. Interestingly, mean SBP significantly decreased after treatment with both drugs: administration of NC resulted in a significant reduction, from 176±17 mmHg at admission to 136±4 mmHg after 1 hour of infusion (P<0.001); likewise, mean SBP decreased from 172±19 mmHg to 138±8 mmHg after NM administration (P<0.001). Importantly, most patients treated with either agent achieved the SBP goal of 140 mmHg within 1 hour, which was steadily maintained during infusion. As for ICP, a decline from 18±8 mmHg, at admission, to 16±8 mmHg (P=0.048) was recorded during IV administration of NC, but this parameter slightly increased, from 14±6 mmHg to 16±5 mmHg, after NM treatment (P=0.066). Of note, HR was reduced in both groups, although neither treatment resulted in a statistically significant change (P=0.121 [NC]; P=0.135 [NM]).
  | Figure 3 Blood pressure recordings over time for the nicardipine and nimodipine groups. |
In the NC group, 93.5% (43/46) patients achieved goal BP, while 6.5% (3/46) patients failed and received nitroprusside for BP control; similarly, 87.8% (36/41) NM patients achieved goal BP, with 12.2% (5/41) individuals failing and receiving nitroprusside. Therefore, the proportions of patients achieving the BP target or requiring additional antihypertensive agent were not statistically different between the two groups (P=0.361).
BP variability between both groups is shown in Figure 4. Patients in the NC group spent 87%±6% of the time at goal BP values, corresponding to 85%±7% obtained for NM individuals. Although this percentage was slightly higher in the NC group, the differences between both groups were not statistically significant (P=0.139). Of note, hypotension (SBP <90 mmHg) was not recorded during treatment with either agent.
  | Figure 4 Mean percentage of time at goal BP values. |
In addition, the mean time to goal BP in the NC group was 43±18 minutes, showing no significant difference with the value obtained for NM patients (48±16 minutes) (P=0.868). Analysis of time to goal SBP demonstrated that both NC and NM were able to help reach the BP goal within the first hour of administration (Figure 5).
  | Figure 5 Mean time to goal BP. |
An intention-to-treat analysis showed that the mean number of dose adjustments required to maintain the goal BP was slightly lower in the NC group (8±2 times) than in NM patients (9±2 times) during IV infusion, although the difference was not statistically significant (P=0.910) (Figure 6).
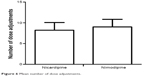  | Figure 6 Mean number of dose adjustments. |
A total of seven of 32 (21.9%) patients with ICH within 6 hours of symptom onset in the NC group underwent hematoma expansion (more than 33% increase in hematoma size), as measured by follow-up cerebral computed tomography (CT) at 24 hours; six of 33 (18.2%) such patients were found in the NM group (P=0.710). Finally, 13 of all 46 (28.3%) patients in the NC group suffered from neurological deterioration (GCS score decrease ≥2, from baseline) during treatment, as did eleven of all 41 (26.8%) patients in the NM group (P=0.881).
Discussion
To the best of our knowledge, no study comparing the clinical antihypertensive effects of NC and NM treatment in patients with ICH has been previously reported. Therefore, the purpose of this retrospective study was to assess the efficacy and safety of IV NM for treating hypertension in neurological patients with ICH. Theoretically and clinically, NC is efficient and safe for controlling hypertension in neurological patients and is recommended as a preferred agent in guidelines.13,14 Interestingly, Ahmed et al16 reported that IV NM treatment resulted in a statistically significant decrease in SBP, in comparison with placebo, for patients with ischemic stroke. In the NICU, we also found that IV administration of NM lowered BP promptly. However, trials exploring the antihypertensive effect of IV NM are scarce. The efficacy and safety of IV NM administration was assessed by comparing its clinical antihypertensive outcomes to those of NC.
In the treatment of hypertension in patients with ICH, the appropriate control of BP remains controversial. For instance, the guidelines for the management of ICH from the American Heart Association/American Stroke Association recommend a goal BP of less than 160/90 mmHg.8 However, a new recommendation from the Acute Cerebral Hemorrhage Trial (INTERACT) suggests that in patients with SBP of 150 to 220 mmHg, acute lowering of systolic BP to 140 mmHg is probably safe. INTERACT was undertaken with 404 patients, mostly Chinese, with ICH within 6 hours of onset, and the results showed a reduced hematoma growth, with no excess of neurological deterioration or other adverse events after controlling SBP to <140 mmHg.17,18 Elsewhere, SBP between 140 and 150 mmHg within 12 hours of ICH was reported to be associated with higher mortality.19,20 Consequently, according to INTERACT, we chose to reduce and maintain SBP between 120 and 140 mmHg in the present study. In the whole process, CPP was maintained above 70 mmHg.21
Specifically, five patients in the NM group failed to achieve the goal BP. We found that the pretreatment SBP for all five patients reached values >200 mmHg. To verify these findings, we next selected eight other patients with SBP >200 mmHg upon admission who received IV NM to control SBP, and we found that only one patient achieved goal. When NC was chosen instead of NM to control SBP, the patients were more likely to reach the SBP target, and only one patient failed to achieve the goal. Despite the small sample size, we recommend that NM could be beneficial with SBP <200 mmHg. However, more comprehensive and rigorous studies are needed to further explore the best applicable scope of NM in treating hypertension.
Patients spent the majority of time at goal, and no differences were found in BP variability between both groups. Indeed, patients in both groups spent greater percentage of time above goal parameters in this study. Both drugs rarely caused hypotension symptoms: when the BP goal was reached, drug doses were gradually reduced or discontinued, and hypotension (SBP <90 mmHg) did not occur during the course of our treatment.
Intracranial hematoma expands in the acute phase, and increased hematoma is one of the main causes of neurological deterioration.22 Brott et al23 showed that the growth of intracranial hematoma occurred in 26% of patients within 4 hours after the first symptoms, and another 12% had such growth within the following 20 hours. The time between onset of symptoms and treatment may be an important predictor of hemorrhage growth. Since hemorrhage expansion most likely happened during a time window, timely therapeutic intervention may lower the incidence of hematoma expansion and subsequent mortality and disability.22,24 Furthermore, INTERACT and another trial, the Antihypertensive Treatment in Acute Cerebral Hemorrhage (ATACH) trial,25 have confirmed that early rapid BP lowering is feasible and safe in patients with ICH. In this retrospective study, a significant decrease in SBP was obtained after treatment, with both drugs (P<0.001), and most patients achieved the SBP goal within 1 hour. All patients underwent follow up, by CT scan, 24 hours after admission. A total seven of 32 (21.9%) patients with ICH within 6 hours of symptom onset underwent hematoma expansion during NC infusion, and six of 33 (18.2%) such patients were recorded after NM treatment. Specifically, hematoma expansion in this trial was defined as a 33% increase in hematoma volume at 24 hours in comparison with baseline, as measured by image analysis on a CT. Neurological status was evaluated by GCS score, and neurological deterioration was defined by a decline in GCS by at least 2 points. In all, 13 of the 46 (28.3%) patients in the NC group suffered neurological deterioration, and so did eleven of the 41 (26.8%) patients in the NM group. The incidences of hematoma expansion and neurological deterioration described herein were lower compared with previous studies.23,26 Indeed, IV administration of NC and NM did not increase the rates of hematoma expansion and neurological deterioration.
Elevated BP leads to high perfusion pressure, which may increase ICP, while reducing BP results in autoregulatory vasodilatation of cerebral vessels, which may also increase ICP.27,28 In the present study, ICP declined (P=0.048) during IV administration of NC but increased (P=0.066) after NM treatment. Guidelines for the management of spontaneous ICH from the American Heart Association/American Stroke Association and guidelines for the management of subarachnoid hemorrhage recommended IV NC as the preferred antihypertensive agent not affecting ICP. Nishiyama et al29 found that NC decreased ICP, although it was still higher than normal, in patients with acute cerebral hemorrhage. Schmidt et al30 reported an ICP increase of 3 (0–10) mmHg (P<0.05) under treatment with NM, in patients with normal pressure hydrocephalus. Kotwica and Persson31 found the same trend, with IV infusion of NM leading to elevation of ICP in a rat model. However, Aslan et al32 found lower ICP in NM-treated individuals compared with controls (without NM) (P<0.001), in patients with severe head trauma. Our results, along with those of Nishiyama et al and Schmidt et al29,30 suggest that NC and NM lead to reduction and elevation of ICP, respectively, when administered intravenously. Cerebral vasodilatation caused by NM may lead to raised ICP. Furthermore, elevated ICP did not lead to neurological deterioration in the NM group.
There are some limitations in our study. The first is the goal BP (SBP <140 mmHg), which is lower than that in the guidelines by the American Heart Association/American Stroke Association and other studies.10,11,33 Secondly, the ICH volume in our study was less than 60 mL, and the range of GCS scores was 3–15, so GCS scores of 3 or 4 were not included in our study which may not be complete and accurate when assessing therapeutic response of both agents after infusion. Finally, the sample size (87 patients) was small. A larger, prospective, and randomized study is required to further explore the full effects of NM.
Conclusion
This retrospective study confirms that NM is effective and safe in maintaining BP within goal, in patients with ICH in the NICU setting. Although NC individuals had better maintenance of BP, a higher percentage of patients achieving the BP goal, higher percentage of time spent within goal, less time to goal BP, fewer dose adjustments, and fewer additional medications for BP control, the differences were not statistically significant for these clinical outcomes between both groups.
Acknowledgment
The authors thank professor Qian Yang for his thorough proofreading and valuable comments.
Author contributions
BLW, LHL, WF, LT, and YQL initially designed the study. YQL, WF, LT, ML, YLY, YFG, SNG, LG, BZ, ZHL, and WZ participated in collecting the clinical information. YQL, WF, BLW, ML, YLY, YFG, SNG, and LT analyzed the clinical data and performed the statistical analysis. YQL, WF, LT, LHL, and BLW drafted or contributed to the writing of the manuscript. ML, YLY, YFG, SNG, LG, BZ, ZHL, and WZ carried out critical revision of the manuscript. All authors read and approved the final manuscript.
Disclosure
The authors report no conflicts of interest in this work.
References
Broderick JP, Brott T, Tomsick T, Miller R, Huster G. Intracerebral hemorrhage more than twice as common as subarachnoid hemorrhage. J Neurosurg. 1993;78(2):188–191. | ||
Sacco RL, Boden-Albala B, Gan R, et al. Stroke incidence among white, black, and Hispanic residents of an urban community: the Northern Manhattan Stroke Study. Am J Epidemiol. 1998;147(3):259–268. | ||
Kolominsky-Rabas PL, Sarti C, Heuschmann PU, et al. A prospective community-based study of stroke in Germany – the Erlangen Stroke Project (ESPro): incidence and case fatality at 1, 3, and 12 months. Stroke. 1998;29(12):2501–2506. | ||
Weimar C, Weber C, Wagner M, et al; German Stroke Data Bank Collaborators. Management patterns and health care use after intracerebral hemorrhage. a cost-of-illness study from a societal perspective in Germany. Cerebrovasc Dis. 2003;15(1–2):29–36. | ||
Qureshi AI, Tuhrim S, Broderick JP, Batjer HH, Hondo H, Hanley DF. Spontaneous intracerebral hemorrhage. N Engl J Med. 2001;344(19):1450–1460. | ||
van Asch CJ, Luitse MJ, Rinkel GJ, van der Tweel I, Algra A, Klijn CJ. Incidence, case fatality, and functional outcome of intracerebral haemorrhage over time, according to age, sex, and ethnic origin: a systematic review and meta-analysis. Lancet Neurol. 2010;9(2):167–176. | ||
Shah QA, Ezzeddine MA, Qureshi AI. Acute hypertension in intracerebral hemorrhage: pathophysiology and treatment. J Neurol Sci. 2007;261(1–2):74–79. | ||
Morgenstern LB, Hemphill JC, Anderson C, et al; American Heart Association Stroke Council and Council on Cardiovascular Nursing. Guidelines for the management of spontaneous intracerebral hemorrhage: a guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke. 2010;41(9):2108–2129. | ||
Rasool AHG, Rahman ARA, Choudhury SR, Singh RB. Blood pressure in acute intracerebral haemorrhage. J Hum Hypertens. 2004;18(3):187–192. | ||
Ohwaki K, Yano E, Nagashima H, Hirata M, Nakagomi T, Tamura A. Blood pressure management in acute intracerebral hemorrhage: relationship between elevated blood pressure and hematoma enlargement. Stroke. 2004;35(6):1364–1367. | ||
Qureshi AI, Mohammad YM, Yahia AM, et al. A prospective multicenter study to evaluate the feasibility and safety of aggressive antihypertensive treatment in patients with acute intracerebral hemorrhage. J Intensive Care Med. 2005;20(1):34–42. | ||
Maruishi M, Shima T, Okada Y, Nishida M, Yamane K. Involvement of fluctuating high blood pressure in the enlargement of spontaneous intracerebral hematoma. Neurol Med Chir (Tokyo). 2001;41(6):300–304; discussion 304–305. | ||
Bederson JB, Connolly ES Jr, Batjer HH, et al; American Heart Association. Guidelines for the management of aneurysmal subarachnoid hemorrhage: a statement for healthcare professionals from a special writing group of the Stroke Council, American Heart Association. Stroke. 2009;40(3):994–1025. | ||
Mayer SA, Kurtz P, Wyman A, et al; STAT Investigators. Clinical practices, complications, and mortality in neurological patients with acute severe hypertension: the Studying the Treatment of Acute hyperTension registry. Crit Care Med. 2011;39(10):2330–2336. | ||
Ahmed N, Näsman P, Wahlgren NG. Effect of intravenous nimodipine on blood pressure and outcome after acute stroke. Stroke. 2000;31(6):1250–1255. | ||
Langley MS, Sorkin EM. Nimodipine. A review of its pharmacodynamic and pharmacokinetic properties, and therapeutic potential in cerebrovascular disease. Drugs. 1989;37(5):669–699. | ||
Anderson CS, Huang Y, Arima H, et al; INTERACT Investigators. Effects of early intensive blood pressure-lowering treatment on the growth of hematoma and perihematomal edema in acute intracerebral hemorrhage: the Intensive Blood Pressure Reduction in Acute Cerebral Haemorrhage Trial (INTERACT). Stroke. 2010;41(2):307–312. | ||
Anderson CS, Huang Y, Wang JG, et al; INTERACT Investigators. Intensive blood pressure reduction in acute cerebral haemorrhage trial (INTERACT): a randomised pilot trial. Lancet Neurol. 2008;7(5):391–399. | ||
Zhang Y, Reilly KH, Tong W, et al. Blood pressure and clinical outcome among patients with acute stroke in Inner Mongolia, China. J Hypertens. 2008;26(7):1446–1452. | ||
Willmot M, Leonardi-Bee J, Bath PM. High blood pressure in acute stroke and subsequent outcome: a systematic review. Hypertension. 2004;43(1):18–24. | ||
Broderick J, Connolly S, Feldmann E, et al; American Heart Association; American Stroke Association Stroke Council; High Blood Pressure Research Council; Quality of Care and Outcomes in Research Interdisciplinary Working Group. Guidelines for the management of spontaneous intracerebral hemorrhage in adults: 2007 update: a guideline from the American Heart Association/American Stroke Association Stroke Council, High Blood Pressure Research Council, and the Quality of Care and Outcomes in Research Interdisciplinary Working Group. Stroke. 2007;38(6):2001–2023. | ||
Kazui S, Naritomi H, Yamamoto H, Sawada T, Yamaguchi T. Enlargement of spontaneous intracerebral hemorrhage. Incidence and time course. Stroke. 1996;27(10):1783–1787. | ||
Brott T, Broderick J, Kothari R, et al. Early hemorrhage growth in patients with intracerebral hemorrhage. Stroke. 1997;28(1):1–5. | ||
Qureshi AI, Ezzeddine MA, Nasar A, et al. Prevalence of elevated blood pressure in 563,704 adult patients with stroke presenting to the ED in the United States. Am J Emerg Med. 2007;25(1):32–38. | ||
Qureshi AI. Antihypertensive Treatment of Acute Cerebral Hemorrhage (ATACH): rationale and design. Neurocrit Care. 2007;6(1):56–66. | ||
Mayer SA, Sacco RL, Shi T, Mohr JP. Neurologic deterioration in noncomatose patients with supratentorial intracerebral hemorrhage. Neurology. 1994;44(8):1379–1384. | ||
Yang GY, Betz AL, Chenevert TL, Brunberg JA, Hoff JT. Experimental intracerebral hemorrhage: relationship between brain edema, blood flow, and blood-brain barrier permeability in rats. J Neurosurg. 1994;81(1):93–102. | ||
Qureshi AI, Harris-Lane P, Kirmani JF, et al. Treatment of acute hypertension in patients with intracerebral hemorrhage using American Heart Association guidelines. Crit Care Med. 2006;34(7):1975–1980. | ||
Nishiyama T, Yokoyama T, Matsukawa T, Hanaoka K. Continuous nicardipine infusion to control blood pressure after evacuation of acute cerebral hemorrhage. Can J Anaesth. 2000;47(12):1196–1201. | ||
Schmidt JF, Albeck M, Gjerris F. The effect of nimodipine on ICP and CBF in patients with normal-pressure hydrocephalus. Acta Neurochir (Wien). 1990;102(1–2):11–13. | ||
Kotwica Z, Persson L. [Effect of intravenous infusion of nimodipine on intracranial pressure. Experimental study of ICP measurement conducted in a rat model]. Neurol Neurochir Pol. 1989;23(3):227–230. Polish. | ||
Aslan A, Gurelik M, Cemek M, Goksel HM, Buyukokuroglu ME. Nimodipine can improve cerebral metabolism and outcome in patients with severe head trauma. Pharmacol Res. 2009;59(2):120–124. | ||
Wallin JD, Fletcher E, Ram CV, et al. Intravenous nicardipine for the treatment of severe hypertension. A double-blind, placebo-controlled multicenter trial. Arch Intern Med. 1989;149(12):2662–2669. |
 © 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.



