Back to Journals » Neuropsychiatric Disease and Treatment » Volume 18
Effectiveness of Vortioxetine in Patients with Major Depressive Disorder in Real-World Clinical Practice: French Cohort Results from the Global RELIEVE Study
Authors Polosan M, Rabbani M, Christensen MC, Simonsen K, Ren H
Received 25 May 2022
Accepted for publication 16 August 2022
Published 31 August 2022 Volume 2022:18 Pages 1963—1974
DOI https://doi.org/10.2147/NDT.S374635
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Roger Pinder
Mircea Polosan,1 Marc Rabbani,2 Michael Cronquist Christensen,3 Kenneth Simonsen,3 Hongye Ren3
1Adult Psychiatry Department, Université Grenoble Alpes, Inserm U1216, Grenoble Institut de Neurosciences, CHU de Grenoble, Grenoble, France; 2Medical Affairs, Lundbeck SAS, Puteaux, France; 3Medical Affairs, H. Lundbeck A/S, Valby, Denmark
Correspondence: Marc Rabbani, Medical Affairs, Lundbeck SAS, Tour W, 102 Terrasse Boieldieu, Puteaux, 92800, France, Tel +33 67169528, Email [email protected]
Aim: Functional recovery is an important treatment goal in patients with major depressive disorder (MDD). The Real-Life Effectiveness of Vortioxetine in Depression (RELIEVE) study assessed the effectiveness of vortioxetine in patients with MDD receiving treatment in routine clinical care settings in France, Italy, Canada, and the United States. This paper presents the study data for the cohort enrolled in France.
Methods: RELIEVE was a 6-month, international, observational, prospective cohort study in outpatients initiating vortioxetine treatment for MDD at their physician’s discretion (NCT03555136). Patients were assessed at routine clinic visits at study entry (baseline) and after 12 and 24 weeks of vortioxetine treatment. The primary study outcome was patient functioning, assessed by the Sheehan Disability Scale (SDS). Secondary outcomes included depression severity (assessed by the Patient Health Questionnaire [PHQ-9]), cognitive symptoms (assessed by the Perceived Deficits Questionnaire-Depression [PDQ-D-5]), and cognitive performance (Digit Symbol Substitution Test [DSST]). Changes from baseline to week 24 were assessed using mixed models for repeated measures, adjusted for relevant confounders. Adverse events spontaneously reported by the patient or observed by the investigator were recorded.
Results: Data are available for 184 patients in France (mean age, 50.2 years; 65.2% female). Overall, 67.9% of patients had at least one comorbidity and 46.2% reported current anxiety symptoms at baseline. Adjusted least-squares mean (standard error) change in SDS score from baseline to week 24 was − 10.9 (0.6) points (P < 0.001). Respective changes for PHQ-9, PDQ-D-5 and DSST scores were − 9.3 (0.5), − 6.1 (0.4), and +6.9 (1.0) points (all P < 0.0001). Adverse events were reported by 29 patients (15.8%), most commonly nausea (11 patients, 6.0%).
Conclusion: Clinically relevant and sustained improvements in overall functioning, depressive symptoms, cognitive symptoms, and cognitive performance were observed in patients with MDD treated with vortioxetine for 6 months in routine clinical practice settings in France.
Keywords: major depressive disorder, vortioxetine, effectiveness, patient functioning, real-world evidence
Introduction
Major depressive disorder (MDD) affects approximately 10% of the French population annually,1 and accounted for 508,609 million years lived with disability in France in 2015.2 MDD is a heterogeneous condition associated with a spectrum of emotional, cognitive, and physical symptoms that impact significantly on psychosocial functioning.3,4 Functional impairment contributes significantly to the social and economic burden of MDD.5 Consequently, in patients with MDD, functional recovery is an important treatment goal.5–7
Vortioxetine is a novel antidepressant with a multimodal mechanism of action.8 It inhibits the serotonin (5-HT) transporter and modulates the activity of several 5-HT receptor subtypes, thus influencing a range of neurotransmitter systems relevant to the neurobiology of MDD.8,9 In addition to its direct effects on the 5-HT transporter, vortioxetine acts as a 5-HT1A receptor agonist, a 5-HT1B receptor partial agonist, and a 5-HT3, 5-HT7, and 5-HT1D receptor antagonist.9 Vortioxetine thereby directly and indirectly influences serotoninergic, noradrenergic, dopaminergic, cholinergic, histaminergic and glutamatergic neurotransmission in regions of the brain associated with depression, including the prefrontal cortex and hippocampus.9
In randomized controlled clinical trials, vortioxetine has been shown to have broad efficacy across the spectrum of symptoms experienced by patients with MDD.10–16 Results of a meta-analysis of data from nine international pivotal randomized, placebo-controlled studies demonstrated significant improvement in overall patient functioning in patients with MDD treated with vortioxetine,17 with greatest effects seen at a vortioxetine dosage of 20 mg/day. These studies enrolled patients from psychiatric inpatient and outpatient sites worldwide.
Real-world evidence in routine clinical practice settings is important to support and complement the findings of regulatory studies. However, the potential of antidepressant therapies to directly improve functional outcomes in patients with MDD in daily clinical practice has not been extensively evaluated. Vortioxetine was approved by the US Food and Drug Administration for the treatment of MDD in adults in 2013 and has since been licensed in more than 80 countries worldwide, including France. The global Real-Life Effectiveness of Vortioxetine in Depression (RELIEVE) study assessed the effectiveness and safety of vortioxetine for the treatment of MDD in routine care settings in Canada, France, Italy, and the United States. The overall study findings have been reported previously;18 this paper presents specific results for the patient cohort in France. Important differences exist between countries concerning routine practice for the management of patients with MDD and recommendations for vortioxetine use. In France, patients with chronic psychiatric disorders benefit from a national minimal insurance system that facilitates easy use of healthcare resources. Patients with MDD are predominantly treated in primary care settings and vortioxetine is recommended as a first-line treatment for MDD by the French National Authority for Health (Haute Autorité de Santé).19
Patients and Methods
Study Design and Participants
RELIEVE was an international, observational, prospective cohort study conducted from November 2017 to January 2021 (NCT03555136). The study design has been reported in detail previously.18 In brief, participants were outpatients aged ≥18 years with MDD who were experiencing a major depressive episode (MDE) and were initiating treatment with vortioxetine at their physician’s discretion according to the local approved label.20 Patients with schizophrenia, bipolar disorder, substance-use disorder, or any neurodegenerative disease significantly affecting cognitive function, and those considered at significant risk of suicide or who had attempted suicide within the last 6 months were excluded. As this was a real-world study, there were no exclusion criteria for concomitant medications. All necessary concomitant medications were allowed, and patients were permitted to receive other pharmacotherapy for MDD and/or other psychoactive medications during the study period.
Data were collected at routine clinic visits at study entry (baseline), week 12 (±4 weeks), and week 24 (±4 weeks). In response to the COVID-19 pandemic, a critical management plan was implemented that allowed patients to have remote follow-up visits and complete some patient-reported outcome assessments at home if needed.
The study was conducted in accordance with the Declaration of Helsinki and Good Clinical Practice guidelines. In France, ethics committee approval for this study was obtained from the Comité de Protection des Personnes Sud Est V (CHU Grenoble, France). Patients provided written informed consent for participation.
Study Outcomes
An overview of the assessment scales used in this study is provided in Supplementary Table S1. The primary study outcome was patient functioning, assessed using the Sheehan Disability Scale (SDS).21,22 This brief self-report measure assesses the impact of depressive symptoms over the previous 7 days in the following domains: work/school, social life/leisure, and family life/home responsibility. Patients rate the severity of impairment in each domain on a scale of 0–10, with higher values indicating greater impairment. Scores for the individual domains are combined to generate the total SDS score that ranges from 0 (unimpaired) to 30 (highly impaired). For the calculation of SDS total score in patients who were not working or studying due to reasons unrelated to MDD, an SDS work/school domain score was imputed based on the mean value of the other two SDS domain scores.23 A reduction (ie, improvement) in SDS total score of ≥4 points is considered meaningful for patients.22 Response was defined as SDS total score ≤12 points and remission (ie, no evidence of functional impairment) as SDS total score ≤6 points.22,23 The SDS also reports on the number of lost and underproductive work/school days during the preceding week.
Secondary outcomes were as follows: depressive symptoms (assessed using the 9-item Patient Health Questionnaire [PHQ-9] and the Clinical Global Impression [CGI] scale); cognitive symptoms (measured by the 5-item Perceived Deficits Questionnaire-Depression [PDQ-D-5]); cognitive performance (measured by the Digit Symbol Substitution Test [DSST]); sexual function (assessed using the Arizona Sexual Experiences Scale [ASEX]); and health-related quality of life (evaluated using the EuroQol 5-Dimension 5-Level questionnaire [EQ-5D-5L]). Healthcare resource utilization during the past 12 weeks was assessed at baseline. Mean change in healthcare resource utilization from baseline over the 12 weeks since the last visit was assessed at weeks 12 and 24.
Adverse events spontaneously reported by the patient or observed by the investigator were recorded according to local regulations and were summarized by lowest level Medical Dictionary for Regulatory Activities (Version 23.1) preferred terms.
Statistical Analysis
The full analysis set comprised all patients who initiated treatment with vortioxetine ≤7 days before the study baseline visit according to the local label and contributed to at least two data points during the study, one of which was the baseline assessment. Safety was assessed in all eligible patients who initiated treatment with vortioxetine for MDD. Except for SDS total score, missing data were not imputed.
Least-squares (LS) mean changes in assessment scores from baseline at weeks 12 and 24 were estimated by linear mixed models for repeated measures, adjusted for clinically relevant baseline variables: age, sex, educational level, duration of current depressive episode, presence of somatic and psychiatric comorbidities, and baseline depression severity as measured by PHQ-9 score as a continuous variable. Change in SDS and PHQ-9 total scores from baseline at weeks 12 and 24 were also analyzed according to vortioxetine treatment line (first, second, and third or later line). Changes in measures of work productivity and healthcare resource utilization from baseline at weeks 12 and 24 were analyzed by paired t test (two-sided).
The proportion of patients reporting sexual dysfunction was analyzed at baseline, week 12 and week 24. As proposed by the authors of the ASEX scale,24 sexual dysfunction was defined as ASEX total score ≥19, any individual ASEX item score ≥5, or a score ≥4 for any three ASEX items.
Statistical analyses were performed using R version 3.6.1,25 with significance set at P < 0.05.
Results
Study Population
Data were available for a total of 184 patients participating at 33 sites in France between November 2017 and January 2021. Approximately two-thirds (65.2%) of the study participants in France were female, mean age was 50.2 years (14.1% were aged >65 years), and 67.9% had a least one comorbidity (Table 1). The most frequently reported comorbid medical conditions were sleep disorders (reported in 27.7% of patients), cardiovascular disease (13.0%), diabetes (6.5%), chronic fatigue (6.5%), and chronic pain (6.5%). Half of all patients for whom baseline body-mass index data were available were overweight or obese (50.3%). In all, 85 patients (46.2%) reported current anxiety symptoms, and over half of these (51.8%) reported that they had experienced anxiety for >5 years; type of anxiety was unspecified in two-thirds of patients with current anxiety symptoms. In the overall cohort of patients in France, 9.2% had a diagnosis of generalized anxiety disorder, 6.0% had a diagnosis of post-traumatic stress disorder, and 3.3% had a diagnosis of panic disorder (Table 1).
 |
Table 1 Baseline Patient Demographic Characteristics (Full Analysis Set) |
At baseline, mean duration of MDD was 9.1 years, and 75.0% of patients reported at least one previous MDE (Table 2). In most patients, the duration of the current MDE was ≤14 weeks (72.8%). Patients had moderate-to-severe depressive and cognitive symptoms and severe functional impairment at baseline, as evidenced by mean PHQ-9, PDQ-D-5 and SDS total scores. Data concerning healthcare resource utilization in the 12 weeks before baseline were available for all patients. Patients reported a mean (standard deviation [SD]) of 3.7 (4.0) healthcare visits during this time (median, 3 visits; range, 0–41), most commonly visits to general practitioners (mean [SD], 1.8 [2.2] visits [median, 1 visit; range, 0–13]) and psychiatrists (mean [SD], 1.5 [2.1] visits [median, 1 visit; range, 0–12]). Use of psychotherapy or counseling services was infrequent (mean [SD], 0.2 [1.2] visits in the 12 weeks before baseline [median, 0 visits; range, 0–12]).
 |
Table 2 Patient Clinical Characteristics at Baseline (Full Analysis Set) |
One hundred and eight patients (58.7%) were initiating vortioxetine as first-line treatment for their current MDE (Table 2). Of the 76 patients who were receiving vortioxetine as second- or later-line treatment for the current MDE, 65 (85.5%) were switching to vortioxetine due to lack of effectiveness of their prior antidepressant therapy and 6 (7.6%) were switching due to lack of tolerability. Data on previous antidepressant treatment for the current MDE prior to initiation of vortioxetine were available for 52 patients. The most common previous antidepressants reported were paroxetine (received by 12 patients [23.1% of those with available data]), venlafaxine (10 patients [19.2%]), escitalopram (8 patients [15.4%]), fluoxetine (8 patients [15.4%]), and amitriptyline (6 patients [11.5%]).
In the overall cohort, 16 patients (8.7%) were receiving another antidepressant in addition to vortioxetine at baseline, most commonly mirtazapine and venlafaxine (each received by four patients [2.2% of the total patient cohort]). Just over half of all patients (103 patients [56.0%]) were receiving at least one other psychotropic medication at baseline. The most common concomitant psychotropic medications reported were anxiolytics (received by 85 patients [46.2%]) and hypnotics (received by 37 patients [20.1%]). This is consistent with the proportion of patients reporting current anxiety symptoms and sleep disorders at baseline. Concomitant antipsychotic use was reported for nine patients (4.9%).
Mean (SD) vortioxetine dosage at baseline was 10.3 (3.7) mg/day. In all, 113 patients (61.4%) initiated treatment with vortioxetine 10 mg/day; vortioxetine starting dosage was 5 mg/day in 35 patients (19.0%), 15 mg/day in 26 patients (14.1%), and 20 mg/day in 10 patients (5.4%).
Effectiveness
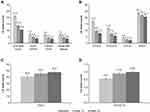
Clinically meaningful improvement in overall functioning, assessed using the SDS, was seen over the 24 weeks of vortioxetine treatment (Figure 1). The adjusted LS mean SDS total score decreased from 21.4 at baseline to 13.8 after 12 weeks of vortioxetine treatment and to 10.5 after 24 weeks. This corresponds to a mean (standard error [SE]) change from baseline of −7.6 (0.6) points at week 12 and −10.9 (0.6) points at week 24 (both differences, P < 0.0001) (Table 3). Statistically significant reductions from baseline were also observed for all adjusted SDS domain scores at weeks 12 and 24 (P < 0.001 for all SDS domains at both time points). In all, 79/174 patients (45.4%) met the criteria for SDS response (ie, SDS total score ≤12 points) at week 12 and 102/160 (63.8%) at week 24; SDS total score was ≤12 points in 14/184 patients (7.6%) at study baseline. Three patients (1.6%) had no evidence of functional impairment (ie, SDS total score ≤6 points) at baseline. SDS remission was achieved by 48/174 patients (27.6%) after 12 weeks of vortioxetine treatment and 65/160 patients (40.6%) after 24 weeks.
 |
Table 3 Adjusted Least-Squares Mean Change (SE) from Baseline for Primary and Secondary Study Endpoints After 12 and 24 Weeks of Vortioxetine Treatment (Full Analysis Set) |
In the working population (ie, patients in full/part-time work or education), reductions in absenteeism and presenteeism were also observed after 24 weeks of vortioxetine treatment. At week 24, the absolute mean (SD) reduction from baseline in absenteeism (work/school days lost) was 1.7 (3.1) days/week (paired t test, P < 0.0001). The corresponding reduction from baseline for presenteeism (work/school days underproductive) was 3.3 (3.1) days/week (paired t test, P < 0.0001). At week 24, the mean (SD) reduction in healthcare resource utilization from baseline during the preceding 12 weeks was 1.7 (3.1) visits (paired t test, P < 0.0001).
Clinically relevant improvements in all measures of depression severity, cognitive function, sexual function, and health-related quality of life were also observed after 12 and 24 weeks of vortioxetine treatment (Figure 1). At 24 weeks, patients had only mild depressive and cognitive symptoms and improved cognitive performance, sexual function and health-related quality of life as evidenced by mean assessment scores. For all secondary outcomes, adjusted LS mean changes from baseline were statistically significant at both week 12 and week 24 (all P < 0.0001) (Table 3).
Statistically significant and sustained improvements in functioning and depression severity were observed over the 24 weeks of treatment, irrespective of vortioxetine treatment line (Figure 2). However, numerically greater improvements in adjusted LS mean SDS and PHQ-9 total scores from baseline were seen when vortioxetine was used as first-line treatment for the current MDE compared with subsequent treatment lines (Table 4). At week 24, the LS mean (SE) change in SDS total score from baseline was −12.3 (0.8) points in patients receiving vortioxetine as first-line treatment (P < 0.0001), −9.1 (1.0) points in those receiving vortioxetine as second-line treatment (P < 0.0001), and −6.7 (2.1) points in patients receiving vortioxetine as third- or later-line treatment (P = 0.005). Corresponding LS mean (SE) changes in PHQ-9 total score from baseline were −10.2 (0.6) points (P < 0.0001), −9.0 (0.9) points (P < 0.0001), and −4.7 (1.7) points (P = 0.015), respectively.
 |
Table 4 Adjusted Least-Squares Mean (SE) Change from Baseline for SDS and PHQ-9 Total Score After 12 and 24 Weeks of Vortioxetine Treatment According to Treatment-Line (Full Analysis Set) |
Safety
Adverse events were reported by 29 patients (15.8%), most commonly nausea (11 patients [6.0%]), headache (4 patients [2.2%]) and pruritus (3 patients [1.6%]). No other adverse events were reported by >1% of patients. No sexual adverse effects were reported; indeed, statistically significant improvement in sexual function was observed over the 6 months of vortioxetine treatment (LS mean [SE] change in ASEX score from baseline to week 24, −3.4 [0.4] points; P < 0.001). The proportion of patients with sexual dysfunction (ie, ASEX total score ≥19, any individual ASEX item score ≥5, or a score ≥4 for any three ASEX items) decreased from 86.1% at baseline to 73.8% after 12 weeks of vortioxetine treatment and 64.2% after 24 weeks. Only three patients (ie, 1.6% of all patients enrolled in France) discontinued treatment with vortioxetine due to tolerability issues, all before the week 12 visit. There were no reports of suicide or suicidal ideation in this patient cohort.
Discussion
To our knowledge, this is the first study to assess the effectiveness of vortioxetine for the treatment of MDD in real-world clinical practice in France. Data are available for a large number of patients (n=174; ie, 25% of the overall RELIEVE study population). Clinically meaningful and sustained improvements in overall functioning, depressive symptoms, cognitive symptoms and performance, and health-related quality of life were observed in patients with MDD treated with vortioxetine in routine practice settings over a period of 6 months. At weeks 12 and 24, the observed improvement in functioning as assessed by LS mean change in SDS total score from baseline (approximately 8 and 11 points, respectively) was higher than the threshold considered meaningful for patients (ie, ≥4 points).22 Almost two-thirds of all patients were considered to be SDS responders and just over 40% had achieved SDS remission after 24 weeks of vortioxetine treatment. Broad improvement was observed across all aspects of patient functioning, including work/school life, social life, and family/home life. Patients also reported missing significantly fewer days of work during treatment with vortioxetine and were more productive while at work compared with before initiating vortioxetine treatment.
Treatment with vortioxetine was well tolerated in daily clinical practice in France, with reported adverse effects consistent with its known tolerability profile.8 In this patient cohort, statistically significant improvement in ASEX score from baseline was observed over the 6 months of vortioxetine treatment, while the proportion of patients considered to have sexual dysfunction decreased from 86.1% at baseline to 64.2% at 24 weeks. This is encouraging, as sexual dysfunction is common in patients with depression,26 and sexual side effects are one of the main reasons for poor adherence and treatment discontinuation with some antidepressants.27,28
The greatest improvement in functioning and depression severity was seen in patients who were receiving vortioxetine as first-line treatment for the current MDE. However, clinically meaningful and statistically significant improvements in SDS and PHQ-9 scores were also observed in patients receiving vortioxetine as a second- or later-line treatment. Our findings are consistent with those of other studies in patients with MDD treated with vortioxetine in routine clinical care settings in other countries.29–35 Of note, approximately 40% of patients in the French cohort had received prior antidepressant therapy, which might be expected to have a negative impact on subsequent treatment outcomes.36
It is recognized that data are limited concerning the prevalence and clinical characteristics of MDD in the general population in France.1 The main strength of this study is that it was conducted in a heterogeneous patient population representative of that likely to be encountered in daily clinical practice in France, many of whom had clinically relevant comorbidities with the potential to affect treatment outcomes, including patient functioning. Indeed, over two-thirds of patients in this cohort had a least one comorbid medical condition. This cohort also included a high proportion of patients with concurrent anxiety symptoms (46%). Although the impact of vortioxetine on anxiety was not assessed in the present study, other studies have shown vortioxetine to be effective for the treatment of concurrent anxiety symptoms in patients with MDD,11,30 including those with comorbid anxiety disorders.37–39
The use of patient-reported outcome measures to assess treatment effectiveness in this study is in keeping with the increased awareness of the importance of understanding patient perspectives and addressing patient needs in MDD.40 Patients’ perceptions of symptoms and treatment outcomes in MDD have been shown to differ from those of their healthcare providers.41–43 As such, clinician-rated scales may not fully capture a patient’s subjective experience of the effects of antidepressant treatment.
Study limitations include the lack of a placebo or active-comparator arm and the potential for selection bias, as patients who do not feel that their condition is being adequately treated may be more likely to enroll in this type of study. Due to the relatively infrequent study visits (ie, at baseline and after 3 and 6 months of treatment), collection of data concerning use of concomitant medication was limited, precluding assessment of any changes in usage across the study period. It should also be noted that the last 10 months of the study period coincided with the COVID-19 pandemic. A critical management plan was implemented in response to this that allowed patients to have remote follow-up visits and complete some patient-reported outcome assessments at home if needed. However, this is not expected to have significantly impacted on our findings as only a very small number of patients failed to complete their visits or completed their visits remotely (22 patients in the entire global RELIEVE study population [ie, <3% of patients included in the full analysis set for the overall study]).18 In France, the COVID-19 pandemic has had significant negative psychological effects both in the general population and in individuals with pre-existing mental health conditions, such as depression,44–46 which may potentially have impacted on the study findings. Additional studies may be of interest to further explore the potential benefits of vortioxetine in patients with MDD in France, including studies in patients with comorbid conditions and comparative studies versus other antidepressant therapies.
Conclusions
In summary, clinically meaningful and sustained improvements in patient functioning, work productivity, depressive symptoms, cognitive symptoms, cognitive performance, and health-related quality of life were observed over the 6-month treatment period in a broad and representative population of patients with MDD receiving treatment with vortioxetine under conditions of routine clinical practice in France.
Data Sharing Statement
The data supporting the findings of this study are available within the manuscript. The corresponding author may be contacted for further data sharing.
Acknowledgments
The authors would like to thank the following investigators at the French study sites: Jerome Attal, Jean-Luc Bartoli, Michel Benoit, Stephane Billard, Eric Boissicat, Daniel Bonneau, Fabrice Boulet, Patrick Bourgoin, Karim Boutayeb, Jean Pierre Clement, Jean Michel Dorey, Fabrice Duval, Joel Gailledreau, Benedicte Gohier, Cyrille Guillaumont, Geraldine Hernandez, Dominique Januel, Xavier Laqueille, Philippe Leclercq, Charles Lousqui, Philippe Marmor, David Modavi, Faycal Mouaffak, Christian Netillard, Eric Neuman, Joel Pon, Philippe Remaud, Norbert Rigaud, and Anne Sauvaget. Medical writing assistance was provided by Jennifer Coward of Piper Medical Communications, funded by H. Lundbeck A/S.
Author Contributions
All authors made substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data; took part in drafting the article or revising it critically for important intellectual content; agreed to submit to the current journal; gave final approval of the version to be published; and agree to be accountable for all aspects of the work.
Funding
The RELIEVE study was funded by H. Lundbeck A/S, whose personnel contributed to the data analysis, review of the data, and review of the manuscript.
Disclosure
M.P. has received honoraria for lecturing from Janssen, LivaNova, Lundbeck, Medtronic, and the Movement Disorder Society, and research support from Boston Scientific. M.R. was an employee of Lundbeck SAS at the time this study was conducted. M.C.C. is an employee of H. Lundbeck A/S. K.S. and H.R. were employees of H. Lundbeck A/S at the time this study was conducted. The authors report no other conflicts of interest in this work.
References
1. Fond G, Lancon C, Auquier P, Boyer L. Prevalence of major depression in France in the general population and in specific populations from 2000 to 2018: A systematic review of the literature [Prévalence de la dépression majeure en France en population générale et en populations spécifiques de 2000 à 2018: une revue systématique de la littérature]. Presse Med. 2019;48(4):365–375. French. doi:10.1016/j.lpm.2018.12.004
2. World Health Organization. Depression and other common mental disorders: global health estimates. Geneva: World Health Organization; 2017. Available from: https://apps.who.int/iris/bitstream/handle/10665/254610/WHO-MSD-MER-2017.2-eng.pdf.
3. Fried EI, Nesse RM. The impact of individual depressive symptoms on impairment of psychosocial functioning. PLoS One. 2014;9(2):e90311. doi:10.1371/journal.pone.0090311
4. IsHak WW, James DM, Mirocha J, et al. Patient-reported functioning in major depressive disorder. Ther Adv Chronic Dis. 2016;7(3):160–169. doi:10.1177/2040622316639769
5. Sheehan DV, Nakagome K, Asami Y, Pappadopulos EA, Boucher M. Restoring function in major depressive disorder: a systematic review. J Affect Disord. 2017;215:299–313. doi:10.1016/j.jad.2017.02.029
6. Habert J, Katzman MA, Oluboka OJ, et al. Functional recovery in major depressive disorder: focus on early optimized treatment. Prim Care Companion CNS Disord. 2016;18(5). doi:10.4088/PCC.15r01926
7. Oluboka OJ, Katzman MA, Habert J, et al. Functional recovery in major depressive disorder: providing early optimal treatment for the individual patient. Int J Neuropsychopharmacol. 2018;21(2):128–144. doi:10.1093/ijnp/pyx081
8. Gonda X, Sharma SR, Tarazi FI. Vortioxetine: a novel antidepressant for the treatment of major depressive disorder. Expert Opin Drug Discov. 2019;14(1):81–89. doi:10.1080/17460441.2019.1546691
9. Sanchez C, Asin KE, Artigas F. Vortioxetine, a novel antidepressant with multimodal activity: review of preclinical and clinical data. Pharmacol Ther. 2015;145:43–57. doi:10.1016/j.pharmthera.2014.07.001
10. Thase ME, Mahableshwarkar AR, Dragheim M, Loft H, Vieta E. A meta-analysis of randomized, placebo-controlled trials of vortioxetine for the treatment of major depressive disorder in adults. Eur Neuropsychopharmacol. 2016;26(6):979–993. doi:10.1016/j.euroneuro.2016.03.007
11. Baldwin DS, Florea I, Jacobsen PL, Zhong W, Nomikos GG. A meta-analysis of the efficacy of vortioxetine in patients with major depressive disorder (MDD) and high levels of anxiety symptoms. J Affect Disord. 2016;206:140–150. doi:10.1016/j.jad.2016.07.015
12. McIntyre RS, Harrison J, Loft H, Jacobson W, Olsen CK. The effects of vortioxetine on cognitive function in patients with major depressive disorder: a meta-analysis of three randomized controlled trials. Int J Neuropsychopharmacol. 2016;19(10):pyw055. doi:10.1093/ijnp/pyw055
13. Christensen MC, Florea I, Lindsten A, Baldwin DS. Efficacy of vortioxetine on the physical symptoms of major depressive disorder. J Psychopharmacol. 2018;32(10):1086–1097. doi:10.1177/0269881118788826
14. Christensen MC, McIntyre RS, Florea I, Loft H, Fagiolini A. Vortioxetine 20 mg/day in patients with major depressive disorder: updated analysis of efficacy, safety, and optimal timing of dose adjustment. CNS Spectr. 2021;Oct 18:1–26. doi:10.1017/S1092852921000936
15. Iovieno N, Papakostas GI, Feeney A, et al. Vortioxetine versus placebo for major depressive disorder: a comprehensive analysis of the clinical trial dataset. J Clin Psychiatry. 2021;82(4):20r13682. doi:10.4088/JCP.20r13682
16. McIntyre RS, Loft H, Christensen MC. Efficacy of vortioxetine on anhedonia: results from a pooled analysis of short-term studies in patients with major depressive disorder. Neuropsychiatr Dis Treat. 2021;17:575–585. doi:10.2147/NDT.S296451
17. Florea I, Loft H, Danchenko N, et al. The effect of vortioxetine on overall patient functioning in patients with major depressive disorder. Brain Behav. 2017;7(3):e00622. doi:10.1002/brb3.622
18. Mattingly GW, Ren H, Christensen MC, et al. Effectiveness of vortioxetine in patients with major depressive disorder in real-world clinical practice: results of the RELIEVE study. Front Psychiatry. 2022;13:824831. doi:10.3389/fpsyt.2022.824831
19. Haute Autorité de Santé. Adult major depressive disorder: management in the primary care setting [Épisode dépressif caractérisé de l’adulte: prise en charge en premier recours]. Saint-Denis La Plaine: HAS; 2017. Available from: https://www.has-sante.fr/jcms/c_1739917/fr/episode-depressif-caracterise-de-l-adulte-prise-en-charge-en-premier-recours.
20. European Medicines Agency. Brintellix (vortioxetine): summary of product characteristics. Available from: https://www.ema.europa.eu/en/documents/product-information/brintellix-epar-product-information_en.pdf.
21. Sheehan DV, Harnett-Sheehan K, Raj BA. The measurement of disability. Int Clin Psychopharmacol. 1996;11(Suppl 3):89–95. doi:10.1097/00004850-199606003-00015
22. Sheehan KH, Sheehan DV. Assessing treatment effects in clinical trials with the discan metric of the Sheehan Disability Scale. Int Clin Psychopharmacol. 2008;23(2):70–83. doi:10.1097/YIC.0b013e3282f2b4d6
23. Sheehan DV, Harnett‐Sheehan K, Spann ME, Thompson HF, Prakash A. Assessing remission in major depressive disorder and generalized anxiety disorder clinical trials with the discan metric of the Sheehan Disability Scale. Int Clin Psychopharmacol. 2011;26(2):75–83. doi:10.1097/YIC.0b013e328341bb5f
24. McGahuey CA, Gelenberg AJ, Laukes CA, et al. The Arizona Sexual Experience Scale (ASEX): reliability and validity. J Sex Marital Ther. 2000;26(1):25–40. doi:10.1080/009262300278623
25. R Core Team. R: A Language and Environment for Statistical Computing. Vienna, Austria: R Foundation for Statistical Computing; 2016. Available from http://www.R-project.org/.
26. Laforgue ÉJ, Busnel G, Lauzeille D, et al. Evolution of sexual functioning of men through treated and untreated depression. Encephale. 2022;48(4):383–389. doi:10.1016/j.encep.2021.06.008
27. Baldwin DS, Manson C, Nowak M. Impact of antidepressant drugs on sexual function and satisfaction. CNS Drugs. 2015;29(11):905–913. doi:10.1007/s40263-015-0294-3
28. Atmaca M. Selective serotonin reuptake inhibitor-induced sexual dysfunction: current management perspectives. Neuropsychiatr Dis Treat. 2020;16:1043–1050. doi:10.2147/NDT.S185757
29. Chin CN, Zain A, Hemrungrojn S, et al. Results of a real-world study on vortioxetine in patients with major depressive disorder in South East Asia (REVIDA). Curr Med Res Opin. 2018;34(11):1975–1984. doi:10.1080/03007995.2018.1477746
30. Chokka P, Bougie J, Proulx J, Tvistholm AH, Ettrup A. Long-term functioning outcomes are predicted by cognitive symptoms in working patients with major depressive disorder treated with vortioxetine: results from the AtWoRC study. CNS Spectr. 2019;24(6):616–627. doi:10.1017/S1092852919000786
31. Chokka P, Tvistholm AH, Bougie J, Clerzius G, Ettrup A. Improvements in workplace productivity in working patients with major depressive disorder: results from the AtWoRC study. J Occup Environ Med. 2020;62(3):e94–101. doi:10.1097/JOM.0000000000001805
32. Fagiolini A, Florea I, Loft H, Christensen MC. Effectiveness of vortioxetine on emotional blunting in patients with major depressive disorder with inadequate response to SSRI/SNRI treatment. J Affect Disord. 2021;283:472–479. doi:10.1016/j.jad.2020.11.106
33. Yang YK, Chen CS, Tsai CF, et al. A Taiwanese study on real-world evidence with vortioxetine in patients with major depression in Asia (TREVIDA). Curr Med Res Opin. 2021;37(12):2163–2173. doi:10.1080/03007995.2021.1980869
34. Bose R, Hamdani SU, Minhas FA, Herr KJ. A comparison of real-world effectiveness of vortioxetine along the treatment algorithm for major depressive disorder. Curr Med Res Opin. 2022;38(5):661–671. doi:10.1080/03007995.2022.2031146
35. Papalexi E, Galanopoulos A, Kontis D, et al. Real-world effectiveness of vortioxetine in outpatients with major depressive disorder: functioning and dose effects. BMC Psychiatry. 2022;22(1):548. doi:10.1186/s12888-022-04109-5
36. Rush AJ, Trivedi MH, Wisniewski SR, et al. Acute and longer-term outcomes in depressed outpatients requiring one or several treatment steps: a STAR*D report. Am J Psychiatry. 2006;163(11):1905–1917. doi:10.1176/ajp.2006.163.11.1905
37. Bidzan L, Mahableshwarkar AR, Jacobsen P, Yan M, Sheehan DV. Vortioxetine (Lu AA21004) in generalized anxiety disorder: results of an 8-week, multinational, randomized, double-blind, placebo-controlled clinical trial. Eur Neuropsychopharmacol. 2012;22(12):847–857. doi:10.1016/j.euroneuro.2012.07.012
38. Liebowitz MR, Careri J, Blatt K, et al. Vortioxetine versus placebo in major depressive disorder comorbid with social anxiety disorder. Depress Anxiety. 2017;34(12):1164–1172. doi:10.1002/da.22702
39. Christensen MC, Schmidt S, Grande I. Effectiveness of vortioxetine in patients with major depressive disorder comorbid with generalized anxiety disorder: results of the open-label RECONNECT study. J Psychopharmacol. 2022;36(5):566–577. doi:10.1177/02698811221090627
40. IsHak WW, Mirocha J, Pi S, et al. Patient-reported outcomes before and after treatment of major depressive disorder. Dialogues Clin Neurosci. 2014;16(2):171–183. doi:10.31887/DCNS.2014.16.2/rcohen
41. Demyttenaere K, Donneau AF, Albert A, Ansseau M, Constant E, van Heeringen K. What is important in being cured from depression? Discordance between physicians and patients. J Affect Disord. 2015;174:390–396. doi:10.1016/j.jad.2014.12.004
42. Baune BT, Christensen MC. Differences in perceptions of major depressive disorder symptoms and treatment priorities between patients and health care providers across the acute, post-acute, and remission phases of depression. Front Psychiatry. 2019;10:335. doi:10.3389/fpsyt.2019.00335
43. Christensen MC, Wong CMJ, Baune BT. Symptoms of major depressive disorder and their impact on psychosocial functioning in the different phases of the disease: do the perspectives of patients and healthcare providers differ? Front Psychiatry. 2020;11:280. doi:10.3389/fpsyt.2020.00280
44. Andersen AJ, Mary-Krause M, Bustamante JJH, Héron M, El Aarbaoui T, Melchior M. Symptoms of anxiety/depression during the COVID-19 pandemic and associated lockdown in the community: longitudinal data from the TEMPO cohort in France. BMC Psychiatry. 2021;21(1):381. doi:10.1186/s12888-021-03383-z
45. Martinelli N, Gil S, Chevalère J, et al. The impact of the COVID-19 pandemic on vulnerable people suffering from depression: two studies on adults in France. Int J Environ Res Public Health. 2021;18(4):3250. doi:10.3390/ijerph18063250
46. Mary-Krause M, Herranz Bustamante JJ, Héron M, Andersen AJ, El Aarbaoui T, Melchior M. Impact of COVID-19-like symptoms on occurrence of anxiety/depression during lockdown among the French general population. PLoS One. 2021;16(7):e0255158. doi:10.1371/journal.pone.0255158
 © 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.