Back to Journals » International Journal of General Medicine » Volume 11
Effectiveness of palivizumab immunoprophylaxis in preterm infants against respiratory syncytial virus disease in Qatar
Authors Abushahin A , Janahi I, Tuffaha A
Received 4 November 2017
Accepted for publication 9 December 2017
Published 31 January 2018 Volume 2018:11 Pages 41—46
DOI https://doi.org/10.2147/IJGM.S156078
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Scott Fraser
Ahmad Abushahin, Ibrahim Janahi, Amjad Tuffaha
Department of Pediatrics, Section of Pediatric Pulmonology, Hamad Medical Corporation, Doha, Qatar
Objective: The primary objective of this study was to document the hospitalization rate due to respiratory syncytial virus (RSV) and compliance with palivizumab use in preterm infants receiving palivizumab immunoprophylaxis during 2009–2012 RSV seasons.
Design: This is a descriptive single-center cohort study. Infants who were ≤35-week gestational age were eligible for enrollment if they received the first palivizumab dose between November 1 and March 31 (2009–2010, 2010–2011, 2011–2012). Primary endpoint was defined as RSV hospitalization of duration 24 hours or longer.
Results: The cumulative RSV hospitalization rate in the registry (2009–2012) was 1.9%. The compliance rate was 85.7%. It showed steady increase across the 3 successive RSV seasons, 2009–2010, 2010–2011, and 2011–2012 (57.7% vs 92.6%, 94.2%, respectively, p<0.05). The RSV hospitalization rate decreased from 3.7% for 2009–2010 to 1.3% for 2010–2011 and 1.7% for 2011–2012 RSV seasons. No deaths and no side effects linked to palivizumab injections were reported for any subjects enrolled.
Conclusion: Our study provides national data regarding characteristics, compliance rate, and hospitalization outcomes for preterm infants receiving palivizumab in Qatar. Our data is consistent with the previously reported efficacy and safety profile of palivizumab.
Keywords: palivizumab, preterm, bronchopulmonary dysplasia, bronchiolitis, passive immunization, immunoprophylaxis, RSV
Background
Respiratory syncytial virus (RSV) is the most common pathogen that causes lower respiratory tract infection in infants and young children.1 It has been reported that 70% of infants who are less than 1 year of age suffer from RSV bronchiolitis,2 and only 2% of those affected infants required hospitalization.3
Epidemiological studies demonstrated that the risk of RSV-related hospitalization is increased among preterm infants (10%–25%),2,3 infants with bronchopulmonary dysplasia (BPD) (15%–45%),2,3 and those with congenital heart disease (CHD) (15%–25%).4 Those infants are at high risk for severe RSV bronchiolitis and its associated increase in morbidity and mortality.3 Therefore, protection against RSV infection among those high-risk group infants is required.
Palivizumab is a humanized monoclonal antibody directed against RSV. Clinical studies showed that this passive immunization is effective in protecting high-risk infants from serious RSV disease.5–7 The US Food and Drug Administration approved palivizumab use for RSV immunoprophylaxis in 1998, to protect high-risk children from severe RSV infection.5
Published data demonstrated that palivizumab decreased the RSV hospitalization by 55% and by 45% in preterm infants6 and in children with CHD,7 respectively. However, the overall reported RSV-related hospitalization rate among infants and children receiving palivizumab was 1.5%–2.9%.8,9
Epidemiological data shows that RSV activities increase in winter and early spring and typically peak between November and March. However, this seasonal pattern may differ by region.10 In Qatar, RSV activities peak in winter season with increase in RSV-related hospitalization between November and February.11
Palivizumab was introduced in our institution in 2005 and is used to protect high-risk infants from severe RSV infection as per American Academy of Pediatrics recommendations for palivizumab immunoprophylaxis.12,13 To our knowledge, no data have been published determining palivizumab efficiency in our community. This observational study was carried out to determine RSV hospitalization rate in preterm infants immunized with palivizumab during 2009–2012 sequential RSV seasons and compare our results with published data from other international studies.
Methods
Study design
This was a descriptive single-institute cohort study conducted in the Department of Pediatrics, Hamad Medical Corporation in Qatar. Preterm infants hospitalized with RSV bronchiolitis in 3 successive RSV seasons (November 1 to March 31 from 2009 to 2012) were included in this observational cohort study. Data from November 1, 2011 to March 31, 2012 were collected prospectively, while data from November 1 to March 31 for each RSV season from 2009 to 2011 were collected from hospital records retrospectively and were used for purposes of comparison in relation to compliance and RSV hospitalization rates.
Subjects enrollment
In term of subject enrollment, preterm infants ≤35 weeks gestational age and <6 months old at enrollment, preterm infants ≤2 years old with BPD defined by oxygen supplementation on day 28 of age,14 and preterm infants ≤2 years old with hemodynamically significant CHD were eligible for enrollment if they received more than one dose of palivizumab during the RSV season between November 1 and March 31 from 2009 to 2012. Exclusion criteria included hospitalization for acute respiratory illness at the time of enrollment, chronic lung disease other than BPD, and presence of unstable neurological disorder.
Data collection
Data from each infant was collected and entered in standardized data forms at the following points: 1) first palivizumab immunization, 2) time of each subsequent palivizumab immunization, and 3) RSV hospitalization. Infants received palivizumab 15 mg/kg by intramuscular injection monthly for total of 3–5 doses over the RSV season, according to the starting month of the first dose. Patient characteristics (age at 1st dose, birth weight, gestational age, and multiple birth status), medical history (presence of BPD or CHD), family and environmental history (mother’s age, tobacco smoke exposure, number of sibling in the household, and childcare attendance for the preterm infant) were collected during the first visit for enrollment. Data for each palivizumab injection was collected, including the date and dose of each injection, the frequency of doses, the intervals between injections, the number of missed doses, and the side effects developed to the injections over the following 7 days (anaphylaxis, fever, erythema, swelling, rash, cough, wheezing, vomiting, and/or diarrhea). During the RSV season, parents/caregivers were contacted by telephone on a monthly basis to remind them of the next palivizumab prophylaxis appointment, to obtain information about any palivizumab injection-related side effects, and if any hospitalizations for respiratory events occurred in the previous month. Monitoring for RSV hospitalizations started from the first injection and continued till 30±5 days after the last dose of palivizumab. When a respiratory hospitalization was reported, data specific to respiratory illness including RSV-related lower respiratory tract infection was collected by reviewing individual hospital record to determine whether RSV testing was done, the type of isolated viruses, the days of hospital stay, intensive care unit admission, and the discharge diagnosis. The diagnosis of a lower respiratory tract infection and decision to obtain a virology test for isolation of RSV were at the discretion of the treating physician. An RSV hospitalization was defined according to Parnes et al15 as any hospitalization for ≥24 hours for which the infant had a positive RSV test. Multiplex real-time polymerase chain reaction assay using fast tract diagnostics-Respiratory 21 was the method of RSV virus detection in nasopharyngeal aspirates from hospitalized infants. Compliance with the immunization protocol was determined according to that reported by Parnes et al15 as well, by comparing the actual with the received number of doses based on the month the first dose was given. Infants who received the expected doses given at appropriate time interval (30±5 days) were considered compliant.
Ethical considerations
Our study was approved by Medical Research Center of Hamad Medical Corporation. A waiver on the necessity of signed informed consent was obtained because this was an observational and noninterventional study, and all patient data were anonymous. Privacy and confidentiality of patient data were maintained throughout the study period by excluding personal identifiers from the data collection forms.
Statistical analysis
Data (continuous and categorical data) were analyzed by standard descriptive methods. The results were expressed as absolute numbers and percentage of the entire cohort for qualitative variables, and as both mean and standard deviation for quantitative variables. Parametric (Student’s t-tests) and nonparametric (Mann–Whitney) tests were used as appropriate for comparison of means, and Pearson’s χ2 test and Fisher’s exact test were used for comparison of percentages. The Statistical Packages for Social Science (SPSS), Windows version No. 16 (Chicago, IL, USA) was used for data entry and data analysis.
Results
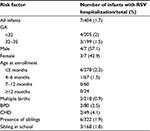
During the RSV season from November 1, 2011 to March 31, 2012, 429 preterm infants were enrolled. Demographics, medical history, and family history are presented in Table 1. Approximately, 52.4% (n=225) infants were born at <32 weeks of gestational age (GA) and weighed 1268.9±354 g, and those born between 32 and 35 weeks represent 47.6% (n=204) of the cohort and weighed 1,945±342 g. Of all subjects, 64.8% (n=278) and 15.6% (n=67) were ≤3 months of age and between 4 and 6 months, respectively, at the time of the first dose. Among all enrolled preterm infants during this season, multiple births were present in 50.8% (n=218), BPD in 18.6% (n=80), and CHD in 11.5%, (n=49). Of other characteristics among those preterm infants, a childcare center attendance was observed in 1.9% (n=8), having a sibling in school was seen in 41.8% (n=168), and exposure to tobacco smoke was observed in 32.4% (n=139). There was no statistically significant difference when these variables were analyzed according to GA subgroups (GA <32 and ≥32). Table 2 shows the RSV-related respiratory disease characteristics of hospitalized infants for the period December 1 to March 31, 2011–2012. Overall, 94.2% (404/429) were compliant with the palivizumab doses protocol. The compliance was 91.1% and 97.5% among the subset of infants whose GA was <32 weeks and those whose GA was 32–35 weeks, respectively (p=0.04). Of the 404 subjects (compliant group), 1.7% (n=7) had confirmed RSV-related hospitalization. Among preterm infants <32 weeks and those between 32 and 35 weeks, the hospitalization rate was 2% (n=4/205) and 1.5% (n=3/199), respectively (Table 2). For subjects hospitalized due to RSV, mean age in months at time of first palivizumab dose was 1.5±2 and 2.6±2 at the time of hospital admission. In comparison with infants between 32 and 35 weeks gestation, preterm subjects <32 weeks were older at the time of admission (1.8±0.8 vs 3.2±2.6, respectively) and had longer hospital duration of stay (13±7.5 vs 4.6±1.2, respectively). Only one subject out of seven subjects required admission to the pediatric intensive care unit (Table 2). The highest percentage of RSV-related admissions occurred between November 1 and December 1 – for the RSV season 2011–2012 (71.4%, n=5/7), which was within the first palivizumab injection interval for this season. For the season 2010–2011, the 3 RSV hospitalizations occurred within the 1st injection interval (November 1 to December 1). For the 2009–2010 season, 1 RSV hospitalization was within the 1st interval and 2 RSV hospitalizations were within the 2nd injection interval (December 1 to January 1). Table 3 shows the characteristics of infants hospitalized for RSV during the December 1 to March 31, 2011–2012 season; the hospitalization rate was higher among infants with CHD (4.1%), followed by infants with BPD (2.5%). Table 4 shows the characteristics of preterm subjects who received at least one dose of palivizumab in each of the 3 successive RSV seasons from 2009 to 2012. Of the entire cohort, 85.7% (n=740/826) were compliant with the palivizumab injection protocol and 1.9% (n=14/740) had confirmed hospitalization due to RSV in successive RSV seasons during the period 2009 to 2012. The 2009–2010 season had a lower percentage of compliant subjects compared to the 2010–2011 and 2011–2012 seasons (57.7% vs 92.6%, 94.2%, respectively, p<0.05). The RSV hospitalization rate decreased from 3.7% for 2009–2010 to 1.3% for 2010–2011 and 1.7% for the 2011–2012 season. This means that there was at least a 54% reduction in hospitalization due to RSV. No deaths and no side effects linked to palivizumab injections were reported for any of the subjects enrolled.
Discussion
Several studies in the literature have demonstrated decrease in RSV-related hospitalization in infants and young children who received palivizumab following its approval.6,7 Conversely, very few studies showed that the risk for RSV hospitalization was not altered by palivizumab prophylaxis.16,17 This study is the first comprehensive registry that generates national data from the state of Qatar on the palivizumab prophylaxis practices, compliance, and effectiveness in reducing the RSV-related hospitalization rate. The characteristics of infants included in our study reveal that the use of palivizumab was in accordance with the American Academy of Pediatrics guidelines.12,13 Through the three successive seasons of our study, there was a constant increase observed in the number of infants who received palivizumab prophylaxis. This increase may reflect increase alertness for serious RSV infection among high-risk infants as well as its associated significant morbidities and mortality. The cumulative RSV hospitalization rate in our registry (2009–2012) was 1.9%, which is lower than the 4.8% reported in the IMpact-RSV study,7 and this is within the range reported by other palivizumab outcomes clinical studies (1.5%–2.9%).8,9 In other studies, the reported rate of RSV hospitalization among nonimmunized preterm infants was 13%–13.5%;18,19 this rate was reduced to 1.1% among preterm infants who received palivizumab.19 However, the low RSV hospitalization rate reported in our registry supports the benefits of RSV passive immunization in infants at risk for severe RSV infection. This study reveals that peak incidence of RSV hospitalization was in November and December; this reflects an increase in the incidence of RSV hospitalization in the 1st and 2nd injection intervals. A similar observation was reported in other studies.15 Low serum levels of palivizumab early in the schedule regimen of prophylaxis may explain this observation. This may mean adjusting our injection schedule to be initiated early during the peak of RSV epidemics in our community.
Compliance with the palivizumab injections schedule is an important factor when assessing hospitalization for RSV infection. The overall compliance reported in our registry was 85.7%, and it showed a steady increase across the 3 consecutive RSV seasons (57.8%–94.2%; p<0.005). Our attempts to increase awareness about the risk of RSV respiratory infection in high-risk infants, using leaflets and the reminder telephone calls for injections time possibly can explain the observed increased compliance. The observed increase in the compliance rate across the 3 RSV seasons studied was associated with a decline in RSV hospitalization rate from 3.7% for 2009–2010 (57.7% compliance rate) to 1.3% for 2010–2011 (92.6% compliance rate) and 1.7% for the 2011–2012 season (94.2% compliance rate). This means that there was a reduction in the RSV hospitalization rate by at least 54%, which is consistent with the reported efficacy of palivizumab passive immunization.20 Furthermore, palivizumab was well tolerated, and no side effects were reported in this study, which is consistent with that reported by others on palivizumab safety profiles.6
Conclusion
Our study provides national data regarding characteristics, compliance, and RSV hospitalization rates among a high-risk group of infants receiving palivizumab in Qatar. It highlights the benefits of RSV passive immunization in infants at risk for severe RSV infection. One of the limitations in our study is the lack of a control group for comparison to demonstration palivizumab effectiveness. However, our data support the previously published data on the efficacy and safety of palivizumab immunoprophylaxis among high-risk infants for severe RSV disease.
Disclosure
The authors report no conflicts of interest in this work.
References
Moore HC, de KN, Keil AD, et al. Use of data linkage to investigate the aetiology of acute lower respiratory infection hospitalisations in children. J Paediatr Child Health. 2012;48(6):520–528. | ||
Glezen WA, Taber LL, Frank AL, Kasel JA. Risk of primary infection and reinfection with respiratory syncytial virus. Am J Dis Child. 1996;140:543–546. | ||
Prober CG, Wang EE. Reducing the morbidity of lower respiratory tract infections caused by respiratory syncytial virus: still no answer. Pediatrics. 1997;99(3):472–475. | ||
Simoes EA, Sondheimer HM, Top FH Jr, et al. Respiratory syncytial virus immune globulin for prophylaxis against respiratory syncytial virus disease in infants and children with congenital heart disease. The Cardiac Study Group. J Pediatr. 1998;133(4):492–499. | ||
Prevention of respiratory syncytial virus infections: indications for the use of palivizumab and and update on the use of RSV-IGIV. American Academy of Pediatrics Committee on Infectious Diseases and Committee on Fetus and Newborn. Pediatrics. 1998;102(5):1211–1216. | ||
The IMpact-RSV Study Group. Palivizumab, a humanized respiratory syncytial virus monoclonal antibody, reduces hospitalization from respiratory syncytial virus infection in high-risk infants. Pediatrics. 1998;102(3):531–537. | ||
Feltes TF, Cabalka AK, Meissner HC, et al. Palivizumab prophylaxis reduces hospitalization due to respiratory syncytial virus in young children with hemodynamically significant congenital heart disease. J Pediatr. 2003;143(4):532–540. | ||
Romero JR. Palivizumab prophylaxis of respiratory syncytial virus disease from 1998 to 2002: results from four years of palivizumab usage. Pediatr Infect Dis J. 2003;22(2):S46–S54. | ||
Paes B, Mitchell I, Li A, Harimoto T, Lanctot KL. Respiratory-related hospitalizations following prophylaxis in the Canadian registry for palivizumab (2005–2012) compared to other international registries. Clin Dev Immunol. 2013;2013:917068. | ||
Centers for Disease Control and Prevention. Respiratory syncytial virus activity – United States, July 2007–December 2008. MMWR Morb Mortal Wkly Rep. 2008;57(50);1355–1358. | ||
Wahab AA,·Dawod ST,·Raman HM. Clinical characteristics of respiratory syncytial virus infection in hospitalized healthy infants and young children in Qatar. J Trop Pediatr. 2001;47(6):363–366. | ||
American Academy of Pediatrics Committee on Infectious Diseases; American Academy of Pediatrics Bronchiolitis Guidelines Committee. Updated guidance for palivizumab prophylaxis among infants and young children at increased risk of hospitalization for respiratory syncytial virus infection. Pediatrics. 2014;134(2):415–420. | ||
Committee on Infectious Diseases. From the American Academy of Pediatrics: Policy statements – Modified recommendations for use of palivizumab for prevention of respiratory syncytial virus infections. Pediatrics. 2009;124(6):1694–1701. | ||
Jobe AH, Bancalari E. NICHD/NHLBI/ORD workshop summary: bronchopulmonardysplasia. Am J Respir Crit Care Med. 2001;163:1723–1729. | ||
Parnes C, Guillermin J, Habersang R; Palivizumab Outcomes Registry Study Group. Palivizumab prophylaxis of respiratory syncytial virus disease in 2000–2001: results from the palivizumab outcomes registry. Pediatr Pulmonol. 2003;35(6):484–489. | ||
Henckel E, Luthander J, Berggren E, et al. Palivizumab prophylaxis and hospitalization for respiratory syncytial virus disease in the Stockholm infant population, 1999 through 2002. Pediatr Infect Dis J. 2004;23(1):27–31. | ||
Duppenthaler A, Gorgievski-Hrisoho M, Aebi C. Regional impact of prophylaxis with the monoclonal antibody palivizumab on hospitalisations for respiratory syncytial virus in infants. Swiss Med Wkly. 2001;131(11–12):146–151. | ||
Carbonell-Estrany X, Quero J. Hospitalization rates for respiratory syncytial virus infection in premature infants born during two consecutive seasons. Pediatr Infect Dis J. 2001;20(9):874–879. | ||
Grimaldi M, Gouyon B, Sagot P, Quantin C, Huet F, Gouyon JB. Palivizumab efficacy in preterm infants with gestational age < or = 30 weeks without bronchopulmonary dysplasia. Pediatr Pulmonol. 2007;42(3):189–192. | ||
Andabaka T, Nickerson JW, Rojas-Reyes MX, Rueda JD, Bacic Vrca V, Barsic B. Monoclonal antibody for reducing the risk of respiratory syncytial virus infection in children. Cochrane Database Syst Rev. 2013;(4):CD006602. |
 © 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.