Back to Journals » Drug Design, Development and Therapy » Volume 8
Effect of administration of water enriched in O2 by injection or electrolysis on transcutaneous oxygen pressure in anesthetized pigs
Authors Charton A, Péronnet F, Doutreleau S, Lonsdorfer E, Klein A, Jimenez L, Geny B, Diemunsch P, Richard R
Received 15 April 2014
Accepted for publication 16 May 2014
Published 26 August 2014 Volume 2014:8 Pages 1161—1167
DOI https://doi.org/10.2147/DDDT.S66236
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Antoine Charton,1 François Péronnet,2 Stephane Doutreleau,3 Evelyne Lonsdorfer,3 Alexis Klein,4 Liliana Jimenez,4 Bernard Geny,3 Pierre Diemunsch,1 Ruddy Richard5
1Department of Anesthesia and Critical Care, and EA 3072, Hôpital de Hautepierre; University of Strasbourg, Strasbourg, France; 2Department of Kinesiology, Université de Montréal, Montreal, QC, Canada; 3CHRU of Strasbourg, Physiology and Functional Explorations Department, New Civil Hospital, Strasbourg, France and University of Strasbourg, Faculty of Medicine, Physiology Department, Strasbourg, France; 4Danone Research, Palaiseau, France; 5Department of Sport Medicine and Functional Explorations, CHU Clermont-Ferrand and INRA UMR 1019, CRNH-Auvergne, Clermont-Ferrand, France
Background: Oral administration of oxygenated water has been shown to improve blood oxygenation and could be an alternate way for oxygen (O2) supply. In this experiment, tissue oxygenation was compared in anesthetized pigs receiving a placebo or water enriched in O2 by injection or a new electrolytic process.
Methods: Forty-two pigs randomized in three groups received either mineral water as placebo or water enriched in O2 by injection or the electrolytic process (10 mL/kg in the stomach). Hemodynamic parameters, partial pressure of oxygen in the arterial blood (PaO2), skin blood flow, and tissue oxygenation (transcutaneous oxygen pressure, or TcPO2) were monitored during 90 minutes of general anesthesia. Absorption and tissue distribution of the three waters administered were assessed using dilution of deuterium oxide.
Results: Mean arterial pressure, heart rate, PaO2, arteriovenous oxygen difference, and water absorption from the gut were not significantly different among the three groups. The deuterium to protium ratio was also similar in the plasma, skin, and muscle at the end of the protocol. Skin blood flow decreased in the three groups. TcPO2 slowly decreased over the last 60 minutes of the experiment in the three groups, but when compared to the control group, the values remained significantly higher in animals that received the water enriched in O2 by electrolysis.
Conclusions: In this protocol, water enriched in O2 by electrolysis lessened the decline of peripheral tissue oxygenation. This observation is compatible with the claim that the electrolytic process generates water clathrates which trap O2 and facilitate O2 diffusion along pressure gradients. Potential applications of O2-enriched water include an alternate method of oxygen supply.
Keywords: transcutaneous oxygen partial pressure determination, tissue oxygenation, oxygenated water, water clathrate
Introduction
Adequate tissue oxygenation is a major issue in the management of anesthesia and intensive care in hypoxemic or shock state patients. It can also prevent surgical wound infection1 or postoperative nausea and vomiting.2 It is usually obtained by using mechanical ventilation and increased fraction of inspired oxygen (FIO2). Other procedures include intravenous administration of oxygen (O2),3 enteral administration of O2,4,5 and enteral administration of water enriched in O2 (O2 waters).6 The present experiment further explores this route of O2 supply and compares the effects of two types of O2 water to those of a placebo (100 mg versus [vs] 10 mg O2/L, respectively) administered intragastrically in anesthetized pigs. Enrichment in O2 was obtained by injection of O2 at high oxygen pressure (PO2) and by an electrolytic process7 (see Methods). It has been suggested that this process of enrichment in O2 generates water clathrates which are able to trap solutes such as O28–10 and can facilitate O2 diffusion along PO2 gradients. A previous study of this O2 water obtained by electrolytic process shows an improvement of mitochondrial respiration ex vivo, in oxidative saponin-skinned fibers isolated from soleus muscles of rats, for O2 concentration lower than 150 μmol/L (Zoll et al, unpublished data, 2012).
The purpose of the present experiment was to verify in vivo the ability of these O2 waters to improve O2 diffusion to the peripheral tissues, by monitoring the transcutaneous oxygen pressure (TcPO2), which is a convenient way to assess tissue oxygenation during anesthesia.11–14 We hypothesized that in a condition of hemodynamic and respiratory stability, TcPO2 will be higher with the water enriched in O2 by electrolysis.
Methods
Animals
The experiments, which were conducted on three groups of 14 male pigs (Large White, 6 months old, 22.5±0.8 kg; no significant difference between the three groups) were carried out at Institut de recherche contre les cancers de l’appareil digestif – European Institute of Telesurgery (IRCAD-EITS, University of Strasbourg, France). The protocol was conducted in accordance with the American Physiological Society’s Guiding Principles in the Care and Use of Animals and was approved by the local Institutional Animal Ethics Authority (authorization number 67-147).
Study protocol
The animals were housed at the research facility, at constant temperature (21°C) and with a 12:12 day/night cycle, with food and water ad libitum for 3 days before the experiment. Following premedication with intramuscular ketamine (20 mg/kg) and azaperone (2 mg/kg), a 20-gauge catheter was placed in an ear vein for normal saline infusion at 5 mL/kg/h. The animals were intubated (Portex Blue Line 6 mm; Smith Medical, Kent, UK) under laryngoscopy after injection of propofol (2 mg/kg) and pancuronium (0.2 mg/kg). A 20G arterial catheter (Leader-cath, Vygon, Ecouen, France) was then placed in the femoral artery by Seldinger technique, and a venous catheter was inserted in an internal jugular vein and advanced into the right ventricle under continuous pressure monitoring. Anesthesia was maintained using isoflurane up to a maximum of 2.5% end-tidal concentration, with 21% oxygen delivered using a ventilator (Aisys Carestation; GE Healthcare, Little Chalfont, UK). Tidal volume was set at 10 mL/kg, with a 4 cm H2O positive end expiratory pressure (PEEP), and end tidal PCO2 was kept constant between 36 and 40 mmHg by adjusting the respiratory rate between 14 and 18 cycles/minute. Body temperature was maintained with an isolation blanket. For the administration of waters, an orogastric tube was also put in place, and the gastric content was aspirated before the beginning of the procedure.
Measurement
Heart rate, ECG, continuous arterial pressure, esophageal temperature (esophageal probe, Odam Physiogard SM 785™; Schiller, Baar, Switzerland), and oxygen consumption (VO2) (CPX, SensorMedics, Yorba Linda, CA, USA) were monitored throughout the experiment. Skin perfusion was explored from phase shift of a laser light beamed by a fiber optic on moving red blood cells (Doppler flowmetry, Periflux PF4; Perimed AB, Stockholm, Sweden). The laser Doppler flowmeter probe was placed on the internal portion of the right thigh. Although precise quantifications cannot be made, it has been shown that changes in skin microcirculation can be monitored with this technique during sedation or anesthesia.15,16
Measurement of TcPO2 (Tina TCM4 series, Radiometer, Denmark) was performed on the internal portion of the right thigh, close to the Doppler flowmeter probe. For this purpose, following two calibrations, the electrode (heated at 43°C) was placed on the skin, which had been shaved and cleaned with alcohol. A stabilization period of 20 minutes (TcPO2 changes <2 mmHg within 5 minutes) was allowed before recording basal preingestion values.
Water administered
Three types of water were used: remineralized water used as placebo and waters enriched in O2 by injection and electrolysis. The waters were prepared from demineralized water which was remineralized with Na+ (200 mg/L), SO42− (250 mg/L), and PO42− (240 mg/L). The water ingested as placebo was enriched in O2 by injection at a level of 10 mg O2/L (ie, close to the value at equilibrium with atmospheric O2 at sea level at the temperature of ingestion [~8 mg/L at 5°C–10°C]).17 The two other waters were enriched at 100 mg O2/L (PO2 ~1,220–1,350 mmHg) by injection or the electrolytic process. In this process, the remineralized water was pumped between two electrodes separated by a membrane permeable to electrical charges but not to gases and the water enriched in O2, which was recovered on the negative electrode (pH =7.1–7.2, conductivity =750–770 μS/cm, 4,375–5,000 μmol O2/L) was used to prepare the final product, which was kept in glass bottles with a narrow neck. Upon opening, and keeping the bottle unagitated at 20°C, the half-life of the decrease in O2 content was 5.5 days. Accordingly, only negligible loss of O2 should be expected within the short delay (<1 minute) between opening of the bottle and administration of the water. It is worth mentioning that Nestle et al18 also examined the disappearance of O2 from an O2 water with an initial content of ~100 mg/L using nuclear magnetic resonance relaxometry. The half-life measured was also large (~100 minutes), although lower than that measured in the present experiment, probably because the water was poured in drinking beakers with a much larger and more ventilated surface for O2 to escape than that in a bottleneck.
The waters, which were prepared by Danone Research (Palaiseau, France), were administered through the orogastric tube. Each animal received 10 mL/kg of water.
Experimental procedure
The animals were studied for 90 minutes following water administration (T0). At each measurement point blood samples were simultaneously drawn from the femoral artery and the right ventricle. The arteriovenous oxygen difference (avDO2 mL O2/dL) was calculated from arterial and venous O2 contents (in mL/dL of blood) computed as [(1.34× [Hb] × SaO2) + (0.003× PO2)], where [Hb] is the hemoglobin concentration in g/dL and SaO2 the saturation of hemoglobin.
At the end of experiments the gastric content was aspirated through the orogastric tube, ~3 g samples of skin and gracilis muscle were quickly dissected in the vicinity of the TcPO2 electrode, and the animal was killed by an intravenous injection of potassium chloride.
Water absorption and distribution
Absorption and distribution in the body water pool of the waters administered were assessed using D2O in eight animals in each group. For this purpose, the waters were labeled by adding 10 mL of D2O/L in order to obtain a final deuterium to protium ratio (D/H) ~10,000 ppm (actual values: 9,276±135 ppm). The D/H ratio was then measured 1) in plasma (separated from the blood by centrifugation) at selected intervals following water administration, 2) in the gastric content recovered at the end of experiment, and 3) in fluids recovered from the skin and muscle samples after homogenization and centrifugation. The D/H ratio was measured by mass spectrometry in water purified from the various fluids by reverse lyophilization; ie, the sample is freeze-dried and the water removed in the process is recovered in a cold trap.
Statistical analysis
Data are presented as mean ± SEM. After testing for normality and homogeneity of variance, data were compared using parametric (one-way or two-way analysis of variance) or nonparametric tests (Kruskal–Wallis). For each parameter, except blood gas, the reference mean during the basal period is the average of the values recorded over successive 2.5-minute intervals during the steady state period of 20 minutes. A P value <0.05 was regarded as statistically significant. Statistical analysis was performed by GraphPad Prism 5.0.
Results
As shown in Table 1, heart rate, mean arterial pressure, PaO2, avDO2, and VO2 were stable throughout the experiment and not significantly different in the three groups. The temperature was also similar in the three groups and the small decrease did not reach statistical significance. Skin blood flow significantly decreased during the experiment in the three groups between T0 and T90 (P<0.05) (Table 1). Figure 1 shows changes in TcPO2 vs the mean of basal preingestion. From minute 0 to 30 following water administration, TcPO2 was stable and not significantly different between the three groups. Beginning at minute 30, TcPO2 slowly but significantly decreased until the end of experiment in the three groups. However, when compared to the control group, the decrease in TcPO2 was significantly lower in animals that received the water enriched in O2 by the electrolytic process (two-way analysis of variance: main effect of time [P<0.001] and water [P<0.001]) (Figure 1).
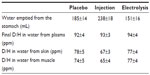
No significant difference was observed between the three groups for plasma D/H observed in the basal period (149±1, 157±2, and 149±1 ppm before administration of the placebo and the waters enriched by injection and electrolysis, respectively), and for the progressive increase in plasma D/H over background values, following administration of waters labeled with D2O (Figure 2). Based on the actual D/H and volume of O2 water administered in the stomach, and on the D/H and volume of the fluid aspirated from the stomach at the end of experiment, the volume of water which was emptied in the small intestine was computed and was found not significantly different in the three groups (Table 2). The D/H ratio in water purified from the skin and skeletal muscle at the end of experiment were also not significantly different, respectively, in the three groups, but the values were ~91%–92%, those simultaneously observed in plasma (Table 2).
Discussion
Results from the present experiment show that over the last hour of anesthesia, when compared to the placebo and the water enriched in O2 by injection, TcPO2 was maintained at a higher level with the water enriched in O2 by the electrolytic process. When compared to the placebo, the progressive reduction in TcPO2 observed between minute 30 and 90 was slightly smaller with both waters enriched in O2, and the difference was significant for the water enriched in O2 by electrolysis. This was not associated with any significant difference in water absorption and distribution. Indeed, no significant difference was observed for the amount of water emptied from the stomach, and the appearance of D2O in plasma was fast and similar in the three groups, suggesting that in contrast to what was observed for CO2,19 the presence of O2 in the stomach does not delay gastric emptying. In addition, the kinetics of plasma D/H was not significantly different in the three groups, with a large increase within the first ~20 minutes followed by a more progressive rise until the end of experiment. As for D2O appearance in muscle and skin, it was also remarkably similar in the three groups but slightly lagged behind D2O appearance in plasma. This result suggests that water ingested is well distributed to various body compartments. Due to technical reasons, there is a paucity of data on the kinetics of distribution of labeled water ingested or administered in the blood20 and we are not aware of any data concerning the skin. As for the skeletal muscle, Schwartz21 showed that in mice, the D/H in this tissue with a low perfusion at rest was slow to equilibrate with that in plasma and tends to plateau at about 90% of the plasma value for at least 30 minutes. Our data are well in line with this observation, and suggest that the perfusion was small in both skeletal muscle and skin.
During general anesthesia, the skin is a tissue of choice to monitor because it is readily accessible, quickly vasoconstricts under shock states, and is the last to be reperfused in shock and resuscitation states.14 Skin oxygenation can be monitored using TcPO2, which might better reflect PaO2 than SaO2.22 Indeed, TcPO2 is closely related to PaO2 during anesthesia23 and accurately reflects local ischemic suffering and global shock.14,24 It is also a sensitive and reliable index in the diagnosis of peripheral arterial disease.25 The location and temperature of the electrode might affect the value of TcPO2 recorded.22,26,27 For these reasons, we consistently positioned the electrode on the internal portion of the upper thigh and the electrode was heated at 43°C, which allows for a better diffusion of O2, probably by shifting to the right the HbO2 dissociation curve.28
During the first 30 minutes following administration of water, TcPO2 remained fairly stable. In contrast, over the subsequent 60 minutes, TcPO2 values consistently decreased in the three groups. This was not related to a reduction in PaO2. Indeed, in spite of the low FiO2 value used (21% as found in atmosphere versus the higher values commonly used in general anesthesia [30%–60%]), PaO2 was stable over the duration of experiment (Table 1). The progressive reduction in TcPO2 was, thus, probably due to vasoconstriction, and this was confirmed by the reduction in skin blood flow observed in the three groups. The reasons for this progressive reduction in skin blood flow are difficult to pinpoint. Ingestion of water and stomach distension can induce skin vasoconstriction.29,30 Although the amount of water administered was small (~225±80 mL) and a large portion was transferred to the small intestine and absorbed, this mechanism cannot be excluded. Another hypothesis is that the significant decrease in skin blood flow is due to the low body temperature of the pigs documented at the start of the experiment and at all time points (Table 1). This hypothermia is due to temperature regulation in the research facility, to ingestion of water at ambient temperature, and probably to the lack of efficacy of the isolation blanket. This experimental condition could be a limitation of this study, but it should be noted that hypothermia was comparable in the three groups, and that hypothermia is a condition often observed during general anesthesia.
The better tissue oxygenation observed with the water enriched by the electrolytic process cannot be accounted for by the low amount of O2 which was supplied in the small amount of O2 waters administered: 225 mL of O2 waters with 100 mg of O2/L provide only ~16 mL of O2 vs an average VO2 slightly larger than 100 mL/min in the 22.5 kg animals studied (~5 mL/kg/min; see Table 1). In addition, the same amount of O2 was supplied by the waters enriched in O2 by injection and the electrolytic process, and a better tissue oxygenation was only observed with the water enriched in O2 by the electrolytic process.
Although it is difficult to speculate on the basis of the limited data from the present pilot experiment, the better maintenance of skin oxygenation as tracked by TcPO2 with the water enriched in O2 by the electrolytic process is consistent with the claim that this enrichment process could stabilize O2 in solution, and favors its diffusion along PO2 gradients.7 This phenomenon can be due to the fact that the electrolytic process generates supramolecular water structures, or water clathrates,9 which can trap O2 molecules and could modify the local pressure/content relationship for O27. This has been previously described with hemoglobin-based oxygen carriers, which should reduce the intracapillary resistance of O2 transport, enhancing the supply of O2 to the tissue.31,32 The possibility that water clathrates can increase O2 delivery and correct hypoxemia has been investigated by Spears.33 Unfortunately, in this experiment, intravenous administration of water clathrates was associated with transient episodes of fall in oxygen saturation, probably due to pulmonary capillary occlusion because of formation of large bubbles (>50 μm) generated by defects in the delivery system. In the study by Spears,33 O2 clathrate hydrates were produced at high pressures and low temperatures, but Ozeki and Otsuka have recently shown that these water superstructures can also be generated using a magnetic field8 and they could also be produced by the electrolytic process7 used in the present experiment.
Conclusion
In this protocol, O2 water produced by electrolytic process lessens the decline of peripheral tissue oxygenation. Potential applications of O2 water could be an alternate way of oxygen supply, for treatment of chronic or acute hypoxia. In the field of intensive care medicine or anesthesia, the possibility to administer intravenously O2 fixed in clathrates in saline solution could lead to other indications similar to those advocated for lipidic oxygen-containing microparticles,3 like rescue of hypoxemic patients.
Acknowledgments
Danone Research provided the three types of waters used in this study, and also provided financial support for this work. An abstract of this work was presented at the 2012 ASA Annual Meeting in Washington (October 13–17, 2012; A410).
Disclosure
François Péronnet and Ruddy Richard are occasional consultants for Danone Research. Alexis Klein and Liliana Jimenez are employees of Danone Research. The authors report no further conflicts of interest in this work.
References
Greif R, Akça O, Horn EP, Kurz A, Sessler DI; Outcomes Research Group. Supplemental perioperative oxygen to reduce the incidence of surgical-wound infection. N Engl J Med. 2000;342(3):161–167. | |
Greif R, Laciny S, Rapf B, Hickle RS, Sessler DI. Supplemental oxygen reduces the incidence of postoperative nausea and vomiting. Anesthesiology. 1999;91(5):1246–1252. | |
Kheir JN, Scharp LA, Borden MA, et al. Oxygen gas-filled microparticles provide intravenous oxygen delivery. Sci Transl Med. 2012;4(140):140ra88. | |
Gelman SI. The effect of enteral oxygen administration on the hepatic circulation during halothane anaesthesia: experimental investigations. Br J Anaesth. 1975;47(12):1253–1259. | |
Gelman SI. The effect of enteral oxygen administration on the hepatic circulation during halothane anaesthesia: clinical observations. Br J Anaesth. 1975;47(12):1261–1264. | |
Forth W, Adam O. Uptake of oxygen from the intestine – experiments with rabbits. Eur J Med Res. 2001;6(11):488–492. | |
Lascoste C, Brunner S, Jimenez L, Klein A, inventors and assignees. Method for enriching water with oxygen by an electrolytic process, oxygen enriched water or beverages and uses thereof. United States patent US 2011/0064824 (A1). March 17, 2011. | |
Ozeki S, Otsuka I. Transient oxygen clathrate-like hydrate and water networks induced by magnetic fields. J Phys Chem B. 2006;110:20067–20072. | |
Chaplin MF. A proposal for the structuring of water. Biophys Chem. 2000;83:211–221 | |
Wu HF, Chin CC, Liu BM, et al. Self-assembly formation of the magic ion of (H2O)20O+: observation of nanoscale cages of oxygenated water clusters induced from iron nanoparticles. Rapid Commun Mass Spectrom. 2011;25:410–414. | |
Rafferty TD, Marrero O, Nardi D, Schachter EN, Mentelos R, Ngeow YF. Transcutaneous PO2 as a trend indicator of arterial PO2 in normal anesthesized adults. Anesth Analg. 1982;61:252–255. | |
Barker SJ, Tremper KK, Gamel DM. A clinical comparison of transcutaneous PO2 and pulse oximetry in the operating room. Anesth Analg. 1986;65:805–808. | |
Yang W, Hafez T, Thompson CS, et al. Direct measurement of hepatic tissue hypoxia by using a novel tcpO2/pCO2 monitoring system in comparison with near-infrared spectroscopy. Liver Int. 2003;23:163–170. | |
Yu M, Morita SY, Daniel SR, Chapital A, Waxman K, Severino R. Transcutaneous pressure of oxygen: a noninvasive and early detector of peripheral shock and outcome. Shock.2006;26:450–456. | |
Lamblin V, Favory R, Boulo M, Mathieu D. Microcirculatory alterations induced by sedation in intensive care patients. Effects of midazolam alone and in association with sufentanil. Crit Care. 2006;10:R176. | |
Lecoq JP, Brichant JF, Lamy ML, Joris JL. Norepinephrine and ephedrine do not counteract the increase in cutaneous microcirculation induced by spinal anesthesia. Br J Anaesth. 2010;105:214–219. | |
Lide DF, Frederikse HPR. CRC Handbook of Chemistry and Physics: Solubility of Selected Gases in Ware. 77th ed, CRC Press, New York, NY, 1997. | |
Nestle N, Baumann T, Niessner R. Oxygen determination in oxygen-supersaturated drinking waters by NMR relaxometry. Water Res. 2003;37(14):3361–3366. | |
Ridout F, Gould S, Nunes C, Hindmarch I. The effects of carbon dioxide in champagne on psychometric performance and blood-alcohol concentration. Alcohol Alcohol. 2003;38(4):381–385. | |
Péronnet F, Mignault D, du Souich P, et al. Pharmacokinetic analysis of absorption, distribution and disappearance of ingested water labeled with D2O in humans. Eur J Appl Physiol. 2012;112(6):2213–2222. | |
Schwartz PL. Exchange of blood water in normal tissues and mammary carcinomas in mice. Am J Physiol. 1966;211(1):173–176. | |
Nishiyama T, Nakamura S, Yamashita K. Effects of the electrode temperature of a new monitor, TCM4, on the measurement of transcutaneous oxygen and carbon dioxide tension. J Anesth. 2006;20(4):331–334. | |
McDowell JW. Correlation of arterial and transcutaneous po2s following wet and dry calibration techniques: a comparison study. Respir Care. 1981;26(7):649–652. | |
Tremper KK, Shoemaker WC. Transcutaneous oxygen monitoring of critically ill adults, with and without low flow shock. Crit Care Med. 1981;9(10):706–709. | |
Abraham P, Picquet J, Vielle B, et al. Transcutaneous oxygen pressure measurements on the buttocks during exercise to detect proximal arterial ischemia: comparison with arteriography. Circulation. 2003;107(14):1896–1900. | |
Nishiyama T, Nakamura S, Yamashita K. Comparison of the transcutaneous oxygen and carbon dioxide tension in different electrode locations during general anaesthesia. Eur J Anaesthesiol. 2006;23(12):1049–1054. | |
Takiwaki H, Nakanishi H, Shono Y, Arase S. The influence of cutaneous factors on the transcutaneous pO2 and pCO2 at various body sites. Br J Dermatol. 1991;125(3):243–247. | |
Tremper KK, Waxman K, Shoemaker WC. Effects of hypoxia and shock on transcutaneous PO2 values in dogs. Crit Care Med. 1979;7(12):526–531. | |
Vacca G, Mary DA, Battaglia A, Grossini E, Molinari C. The effect of distension of the stomach on peripheral blood flow in anaesthetized pigs. Exp Physiol. 1996;81(3):385–396. | |
Molinari C, Battaglia A, Grossini E, et al. Activation of the renin-angiotensin system contributes to the peripheral vasoconstriction reflexly caused by stomach distension in anaesthetized pigs. Exp Physiol. 2003;88(3):359–367. | |
Roy TK, Popel AS. Theoretical predictions of end-capillary PO2 in muscles of athletic and nonathletic animals at VO2max. Am J Physiol. 1996;271(2 Pt 2):H721–H737. | |
Page TC, Light WR, McKay CB, Hellums JD. Oxygen transport by erythrocyte/hemoglobin solution mixtures in an in vitro capillary as a model of hemoglobin-based oxygen carrier performance. Microvasc Res. 1998;55(1):54–64. | |
Spears JR. Advances in the management of respiratory failure. Aqueous preparations of oxygen. ASAIO J. 1996;42:196–198. |
 © 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.