Back to Journals » Journal of Multidisciplinary Healthcare » Volume 14
Economic and Humanistic Burden Associated with Depression and Anxiety Among Adults with Non-Communicable Chronic Diseases (NCCDs) in the United States
Authors Armbrecht E, Shah R , Poorman GW, Luo L, Stephens JM, Li B , Pappadopulos E, Haider S, McIntyre RS
Received 10 October 2020
Accepted for publication 24 March 2021
Published 23 April 2021 Volume 2021:14 Pages 887—896
DOI https://doi.org/10.2147/JMDH.S280200
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Scott Fraser
Eric Armbrecht,1 Ruchit Shah,2 Gregory W Poorman,2 Linlin Luo,2 Jennifer M Stephens,2 Benjamin Li,3 Elizabeth Pappadopulos,4 Seema Haider,5 Roger S McIntyre6– 9
1Saint Louis University Center for Health Outcomes Research, Saint Louis University, Saint Louis, MO, USA; 2Pharmerit International, Bethesda, MD, USA; 3Pfizer Inc., New York, NY, USA; 4Viatris Inc., New York, NY, USA; 5Pfizer Inc., Groton, CT, USA; 6Mood Disorders Psychopharmacology Unit, University Health Network, University of Toronto, Toronto, ON, Canada; 7Department of Psychiatry, University of Toronto, Toronto, ON, Canada; 8Department of Pharmacology, University of Toronto, Toronto, ON, Canada; 9Brain and Cognition Discovery Foundation, Toronto, ON, Canada
Correspondence: Jennifer M Stephens
Pharmerit International, 4350 East West Hwy #1100, Bethesda, MD, 20814, USA
Tel +1-240-821-1290
Email [email protected]
Introduction: Patients with both major depressive disorder (MDD) and generalized anxiety disorder (GAD) in addition to one or multiple comorbid non-communicable chronic diseases (NCCDs) face unique challenges. However, few studies have characterized how the burden of co-occurring MDD and GAD differs from that of only MDD or only GAD among patients with NCCDs.
Methods: In this study, we used Medical Expenditures Panel Survey data from 2010– 2017 to understand how the economic and humanistic burden of co-occurring MDD and GAD differs from that of MDD or GAD alone among patients with NCCDs. We used generalized linear models to investigate this relationship and controlled for patient sociodemographics and clinical characteristics.
Results: Co-occurring MDD and GAD was associated with increases in mean annual per patient inpatient visits, office visits, emergency department visits, annual drug costs, and total medical costs. Among patients with 3+ NCCDs, MDD or GAD only was associated with lower odds ratios (ORs) of limitations in activities of daily living (ADLs; 0.532 and 0.508, respectively) and social (0.503, 0.526) and physical limitations (0.613, 0.613) compared to co-occurring MDD and GAD. Compared to patients with co-occurring MDD and GAD, having MDD only or GAD only was associated with significantly lower odds of cognitive limitations (0.659 and 0.461, respectively) in patients with 1– 2 NCCDs and patients with 3+ NCCDs (0.511, 0.416).
Discussion: Comorbid MDD and GAD was associated with higher economic burden, lower quality of life, and greater limitations in daily living compared to MDD or GAD alone. Health-related economic and humanistic burden increased with number of NCCDs.
Keywords: economic burden, humanistic burden, depression, anxiety, non-communicable diseases
Introduction
Major depressive disorder (MDD) and generalized anxiety disorder (GAD) frequently co-occur, which complicates disease diagnosis and treatment, increases health expenditures, and negatively impacts quality of life.1 There is a high prevalence of comorbidity between the two disorders, and an estimated 62% of patients with GAD have an MDD episode in their lifetime.2 Treating this patient group requires a tailored approach, as patients with comorbid MDD and GAD have high rates of misdiagnosis and a higher percentage of treatment resistance than patients with either condition alone.3 Comorbid MDD and GAD is associated with greater health care resource use (HCRU), elevated medical expenditures, and lower quality of life than either condition alone.4–7
Comorbid non-communicable chronic diseases (NCCDs) pose a challenge in MDD and GAD care. Approximately 77% and 93% of individuals with MDD or GAD, respectively, have at least one concomitant chronic disease, representing 12.4 million and 6.3 million adults in the United States (US).8,9 A causal relationship has been shown not only between NCCDs increasing the likelihood of developing MDD or GAD, but also MDD or GAD increasing the likelihood of developing NCCDs.10,11 Patients with MDD and/or GAD and comorbid NCCDs utilize more health care resources but have poorer treatment outcomes than patients with isolated conditions.12–16 The presence of MDD and/or GAD can complicate the course of NCCDs by causing increased inflammation, unhealthy lifestyles, and poor adherence to treatment.12 Comorbid medical and mental conditions increase symptom burden, decrease length and quality of life, and elevate medical costs.13–16
MDD, GAD, and NCCDs are closely linked and affect many people in the US. Although previous research has evaluated the impact of NCCDs on MDD and GAD separately, no study has described the incremental impact of multiple NCCD comorbidities on patients with MDD, GAD, or both MDD and GAD. Furthermore, research has shown that treating comorbid MDD and GAD is more challenging than managing either condition alone.16 In this study, we used Medical Expenditures Panel Survey (MEPS) data to determine how the economic and humanistic burden of co-occurring MDD and GAD differs from that of only MDD or only GAD among patients with NCCDs.
Materials and Methods
Overview/Data Source
This retrospective, cross-sectional study used data from the 2010–2017 Medical Expenditure Panel Survey (MEPS). MEPS is a nationally representative survey of US civilian noninstitutionalized adults.17 MEPS employs a survey design to collect health status, health care utilization, health insurance, and clinical, demographic, and economic characteristics of participants.18 Survey respondents’ answers are validated by a treating physician and linked to claims codes. Using MEPS weighting schema, estimates generalizable to US non-institutionalized adults may be obtained. This study queried the MEPS Household Component survey.18 Due to the deidentified nature of this publicly available dataset, institutional review board approval was not needed for this study.
Study Population
The study population was derived from US adults (aged ≥ 19) in the MEPS database within the years 2010–2017. Patients were identified using ICD-9-CM/ICD-10-CM and Clinical Classifications Software (CCS) diagnosis codes.
Patients having MDD (ICD-9-CM: 311/ICD-10-CM: F32) and/or GAD (CSS: 651/ICD-10-CM: F41) present in all three rounds of MEPS data collection for the given year were included. Subjects were required to have completed all rounds of the MEPS survey. Exclusion criteria were any history of cancer; adjustment disorder (CCS: 650/ICD-10-CM: F43); personality disorders (CCS: 658/ICD-10-CM: F60); schizophrenia and psychotic disorders (CCS: 659/ICD-10-CM: F20, F23); substance-related disorders (CCS: 660, 661/ICD-10-CM: F19); delirium, dementia, and amnestic disorders, including Alzheimer’s disease (CCS: 653/ICD-10-CM: F03, F04, F05, G30); bipolar disorder (ICD-9-CM: 296/ICD-10-CM: F31); or Parkinson’s disease (CCS: 079/ICD-10-CM: G20).
NCCDs of interest were cardiovascular disease (CVD), pain, diabetes, high cholesterol, pulmonary disorders, and obesity (Table 1). NCCDs of interest were selected based on the AHRQ list of priority conditions.19 Based on the number of NCCDs, patients with MDD, GAD, or co-occurring MDD and GAD were stratified into three cohorts:
- Patients with 0 NCCDs
- Patients with 1–2 NCCDs
- Patients with 3 or more (3+) NCCDs
 |
Table 1 List of NCCDs Assessed |
Groupings of number of NCCD risk factors were created after exploratory analyses to evenly distribute patients across NCCD risk factor cohorts.
Independent Variables
Independent variables were the presence of mental conditions (MDD, GAD, comorbid MDD and GAD).
Outcome Variables
Direct and indirect economic burden were assessed. Direct economic burden was measured using annual inpatient, outpatient, emergency department (ED), and office visits; mean inpatient length of stay; and annual medical costs (reported in 2018 US dollars). Indirect economic burden was measured using productivity loss, defined as number of work days missed per year.
Humanistic burden was assessed using measurements of health-related quality of life (HRQOL) and health limitations. HRQOL was measured using the 12-Item Short Form Health Survey Version 2 (SF-12v2) physical component summary (PCS) and mental component summary (MCS). Health limitations were measured using self-reported (yes/no) answers to questions in the following domains: functional limitations, limitations in activities of daily living (ADLs), social/recreational limitations, and cognitive limitations.
Covariates
Sociodemographic data obtained included age, sex, census region, race, employment status, family income, and marital status.
Statistical Analysis
Outcomes variables were described across each cohort (0 NCCDs, 1–2 NCCDs, 3+ NCCDs) using median and interquartile range (IQR) for continuous variables and frequency and percentages for categorical variables. Bivariate comparisons between cohorts used Chi-square tests for categorical variables and the Kruskal–Wallis test for continuous non-normal variables (ie, age).
After adjusting for significant covariates, multivariate generalized linear models (GLMs) estimated the incremental change in each outcome associated with the presence of MDD and/or GAD. Zero-inflated Poisson GLMs with logarithmic link were used for ED, inpatient, and outpatient visits. GLMs with a Poisson distribution and logarithmic link were used for office-based visits and number of work days missed. GLMs with a gamma distribution and logarithmic link were used to estimate annual medical costs.
Multivariate logistic regression models estimated the odds ratios (ORs) of health limitations associated with MDD and/or GAD. Multivariate linear regression models estimated change in the PCS and MCS scores associated with MDD and/or GAD.
AHRQ-recommended weighting and sampling methods were used.18 Analyses were performed using SAS 9.4 and Stata 14.
Results
Patient Selection
5638 survey respondents were included, which represented a weighted sample of 11,417,939 persons (Figure 1). The weighted (unweighted) sample size for each cohort was 5,352,245 (2714) patients for MDD; 4,581,767 (2188) for GAD; and 1,483,927 (781) for co-occurring MDD and GAD (Table 2).
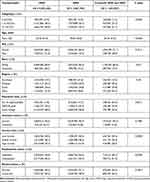 |
Table 2 Weighted Sociodemographic Characteristics of Patients with MDD Only, GAD Only, and Co-Occurring MDD and GAD |
Patient/Sample Characteristics
Patients with co-occurring MDD and GAD were less likely to be employed and had lower household incomes compared to those with MDD or GAD alone. Other demographic characteristics were not significantly different between the three cohorts (Table 2). Mean (SD) patient age was 52.39 (0.47), 68.2% of patients were female, 82.0% of patients were white, and 58.5% of patients had some college education.
Outcomes
Number of NCCDs in Patients with MDD and/or GAD
In the entire study cohort, over 86% of patients had at least one NCCD (n = 9,864,473) and over 45% had 3+ NCCDs (n = 5,232,616). The cohort of patients with co-occurring MDD and GAD had the highest proportion of individuals with 3+ NCCDs (51.7%), compared to the cohorts with MDD (49.8%) or GAD (39.3%) only (Table 2).
Economic Burden of NCCDs in Patients with MDD and/or GAD
Increases in adjusted HCRU and expenditures were observed in patients with co-occurring MDD and GAD, and the burden increased as the number of NCCDs increased. Compared with GAD only, co-occurring MDD and GAD was associated with a significant (P ≤.05) increase in mean annual per patient inpatient visits among patients with 1–2 NCCDs (Table 3). Mean annual office-based visits and costs, regardless of number of NCCDs, were highest in the co-occurring MDD and GAD cohort (Table 3; Figure 2A), compared to those with either condition only. Compared with GAD only, co-occurring MDD and GAD was associated with a significant increase in mean annual per patient ED visits among patients with 0 NCCDs (p=0.109; Table 3).
 |
Table 3 Adjusted Mean (SE) Difference in HCRU and Costs Among Patients with MDD Only, GAD Only, or Co-Occurring MDD and GAD |
Co-occurring MDD and GAD was associated with the highest annual drug costs ($6851) among patients with 3+ NCCDs, followed by MDD ($5129) and GAD ($4120; Table 3). Compared with GAD only, co-occurring MDD and GAD was associated with the highest incremental annual total costs among patients with 0 and 3+ NCCDs, compared to those with either condition only (Table 3).
Humanistic Burden of NCCDs in Patients with MDD and/or GAD
Co-occurring MDD and GAD was associated with lower HRQOL and higher activity limitations, compared to patients with either condition only. The humanistic burden also increased as the number of NCCDs increased. Across all three cohorts, patients with co-occurring MDD and GAD had significantly lower SF-12v2 MCS scores (0 NCCD: 37.48; 1–2 NCCDs: 38.80; 3+ NCCDs: 38.33) compared to those with MDD only (0 NCCD: 41.82; 1–2 NCCDs: 42.24; 3+ NCDs: 41.83) or GAD only (0 NCCD: 44.57; 1–2 NCCDs: 44.07; 3+ NCCDs: 44.00; Figure 2B). Co-occurring MDD and GAD was associated with significantly lower SF-12v2 PCS scores (37.65) among patients with 3+ NCCDs compared to those with MDD (40.01) or GAD (39.47) only (Figure 2C). Co-occurring MDD and GAD was associated with significantly lower EQ-5D scores (0.80, 0.65) compared to those with either MDD (0.84, 0.76) or GAD (0.86, 0.75) only among patients with 1–2 NCCDs and 3+ NCCDs, respectively (Figure 2D).
Among patients with 3+ NCCDs, patients with MDD only or GAD only had significantly lower odds of reporting ADL limitations (adjusted ORs: 0.532 and 0.508, respectively), physical limitations (0.613, 0.613), and social limitations (0.503, 0.526) compared to those with co-occurring MDD and GAD (Figure 3). Having either MDD or GAD only was associated with significantly lower odds of reporting cognitive limitations (0.659 times and 0.461 times, respectively) among patients with 1–2 NCCDs and patients with 3+ NCCDs (0.511 times, 0.416 times) compared to those with co-occurring MDD and GAD (Figure 3).
Discussion
The results of this study suggest that comorbid MDD and GAD was associated with higher economic burden, lower HRQOL, and higher activity limitations compared to MDD or GAD alone. This burden increased with greater numbers of NCCDs. The study further highlights different aspects of economic and humanistic burden impacted by MDD and/or GAD among patients with NCCDs.
In the current study, over 80% of included patients with MDD and/or GAD had at least one NCCD and over 40% had 3+ NCCDs. In the most vulnerable group of individuals, those with both MDD and GAD, over half (51.7%) had multiple NCCD comorbidities. Globally, the incidence and prevalence of not only NCCDs, but also mood disorders including MDD, is increasing, and patients presenting with multiple NCCDs comorbid to MDD and/or GAD may become the norm rather than an exception.9,20 The current analysis describes individuals with multiple chronic NCCDs in addition to MDD and GAD as having higher medical costs, lower HRQOL, and higher activity limitations. Policy makers and health care providers should use these data to direct resource allocation and improve approaches to care.
Individuals with comorbid MDD and GAD had greater HCRU and total medical costs, and this increase was greatest in patients with multiple NCCDs. Substantially higher individual and societal costs in patients with comorbid mental and medical conditions have been established in the literature.21 In the current MEPS dataset, the number of office-based visits was higher in patients with both MDD and GAD. This is consistent with literature describing elevated rates of office-based visits in patients with MDD or GAD: Luber et al showed that patients with depression visit their primary care physician twice as frequently as those without depression.22 To our knowledge, our study is the first to describe patients with both MDD and GAD as also having a greater number of office-based visits than those with either MDD or GAD alone. Literature describing elevated health care expenditures in patients with both mental and medical disorders frequently describes increased medical spending, rather than mental health services spending, as the main driver of higher costs. For example, Melek and Norris found that the presence of comorbid MDD or GAD significantly increased total health care expenditures in patients with common chronic conditions, and 80% of the increased cost was due to higher medical spending in the patients with comorbid MDD.23 Also, a claims-based study of patients with diabetes found that mental health service costs accounted for less than 15% of the elevated spending in patients with comorbid depression and diabetes.24 In our MEPS analysis, the greatest increase in total health care expenditures was seen in patients with multiple NCCDs. Although a breakdown of costs attributable to medical versus mental health services spending was not available, the magnitude of the increase of costs in the patients with multiple NCCDs implies that the NCCDs are a driver of the elevated cost burden.
Our study findings show that having comorbid MDD and GAD was associated with poorer mental and physical quality of life and greater activity limitations relative to patients with either disease alone. It is expected that individuals with mental health diagnoses perceive poorer mental health. However, the current study also shows that, in patients with multiple NCCDs, having comorbid MDD and GAD was associated with lower physical health scores relative to those with either disease alone. Similarly, Zhou et al demonstrated that, among patients diagnosed with MDD, the presence of comorbid GAD decreased physical HRQOL.25 This effect was not seen in patients with no or few NCCDs in our analysis. One explanation is that patients with multiple NCCDs received medical attention for their physical rather than mental symptoms. In a study from the National Comorbidity Survey Replication, 60% of adults with a mental health condition did not receive care for their condition.26 An alternative explanation is that patients with MDD, GAD, and multiple NCCDs had impaired symptom management. NCCDs often require lifestyle modifications and self-care in order to improve physical health, but comorbid mental conditions may hamper patients’ ability and motivation to adhere to treatment.9,27
Numerous studies have shown adverse economic and clinical outcomes in patients with comorbid MDD and GAD in patient populations such as oncology, elderly, and COPD patients.4–7 The current analysis is a valuable addition to this growing body of literature because it compares patients with comorbid MDD and GAD to patient groups having only MDD or GAD to view the effect of having both MDD and GAD on outcomes. Our study also describes the additive effects of NCCDs within this context. We found that not only does comorbid MDD and GAD result in worse economic and clinical outcomes, this effect is greater in patients with more NCCD comorbidities. This helps build understanding of a growing cohort of patients in America, namely patients with high number of NCCDs in addition to mental health diagnoses.
The “collaborative care” model is a systematic approach to treatment of MDD and GAD in primary care and is one way in which health care delivery systems have attempted to improve treatment for patients with comorbid NCCDs.28 Collaborative care models integrate care managers, consultant psychiatrists, and primary care physicians to proactively manage not only mental disorders but also NCCDs. Over 30 randomized controlled trials have shown an improvement in quality of care and patient outcomes using collaborative care.29,30 For example, in 2010, Katon et al randomized patients to receive either collaborative care or usual care in individuals with poorly controlled diabetes, coronary heart disease, or both, and coexisting depression.31 Katon et al showed an improvement in outcomes with collaborative care. The magnitude of improvement in disease-specific outcome measures compared favorably with improvements described in trials that focused on a single intervention.
There are certain limitations in our study that may impact the interpretation of the results. The analysis did not control for intracluster correlations in family income or census region variables. This study was cross-sectional in design and therefore it is not possible to establish causal inferences between MDD and/or GAD and health care costs. In addition, many of the outcomes are based on self-reported data, which may be subject to several biases (eg, recall, social desirability) typical of self-reported data. Furthermore, it is possible that MDD and/or GAD prevalence was underestimated because only 3-digit ICD-9-CM/ICD-10-CM codes were present in the MEPS data; hence some patients with MDD and/or GAD might not have been captured in the study. Overall, the limitations of the study do not negate the benefits of the versatile and comprehensive MEPS data source.
Conclusions
Comorbid MDD and GAD was associated with higher economic burden, lower HRQOL, and greater activity limitations as compared to MDD or GAD alone. Moreover, the economic and humanistic burden increased with an increase in the number of NCCDs. The presence of at least one NCCD was high (>80%) among patients with MDD and/or GAD. This study’s findings are generalizable to a sample of noninstitutionalized US adults. Coordination of care for these patients is a key metric to which policy makers should pay close attention.
Acknowledgments
Medical writing support was provided by Catherine Mirvis and Rachel Shah of Pharmerit International and was funded by Pfizer (legacy Upjohn, a division of Pfizer, now part of Viatris Inc.).
Funding
The study was sponsored by Pfizer (legacy Upjohn, a division of Pfizer, now part of Viatris Inc.).
Disclosure
Dr Eric Armbrecht reports personal fees from Pfizer and grants from the State of Missouri Department of Health and Senior Services during the conduct of the study as well as grants from the Centers for Disease Control and Prevention, outside the submitted work. Ruchit Shah, Gregory Poorman, and Jennifer M Stephens are employees of Pharmerit International, which received financial support from Pfizer in connection with this study; Linlin Luo was an employee of Pharmerit International when the study was conducted. Benjamin Li is an employee of Pfizer and owns Pfizer stock. Elizabeth Pappadopulos and Seema Haider were employees of Pfizer when the study was conducted (legacy Upjohn, a division of Pfizer, now part of Viatris Inc.). Dr Roger S McIntyre reports grants from CIHR/GACD/Chinese National Natural Research Foundation; and personal fees from Lundbeck, Janssen, Purdue, Pfizer, Otsuka, Allergan, Takeda, Neurocrine, Sunovion, Minerva, Intra-Cellular, Abbvie, and Eisai. Dr. Roger S McIntyre is a shareholder in The 420 Company and CEO of Champignon. The authors report no other conflicts of interest in this work.
References
1. Egede LE, Walker RJ, Bishu K, Dismuke CE. Trends in costs of depression in adults with diabetes in the United States: Medical Expenditure Panel Survey, 2004–2011. J Gen Intern Med. 2016;31(6):615–622. doi:10.1007/s11606-016-3650-1
2. Judd L, Kessler R, PauIus M, Zeller P, Wittchen HU, Kunovac J. Comorbidity as a fundamental feature of generalized anxiety disorders: results from the National Comorbidity Study (NCS). Acta Psychiatr Scand. 1998;98(s393):6–11. doi:10.1111/j.1600-0447.1998.tb05960.x
3. Coplan JD, Aaronson CJ, Panthangi V, Kim Y. Treating comorbid anxiety and depression: psychosocial and pharmacological approaches. World J Psychiatr. 2015;5(4):366. doi:10.5498/wjp.v5.i4.366
4. Arrieta O, Angulo LP, Nunez-Valencia C, et al. Association of depression and anxiety on quality of life, treatment adherence, and prognosis in patients with advanced non-small cell lung cancer. Ann Surg Oncol. 2013;20(6):1941–1948. doi:10.1245/s10434-012-2793-5
5. Katon WJ, Lin E, Russo J, Unutzer J. Increased medical costs of a population-based sample of depressed elderly patients. Arch Gen Psychiatry. 2003;60(9):897–903. doi:10.1001/archpsyc.60.9.897
6. Xu W, Collet JP, Shapiro S, et al. Independent effect of depression and anxiety on chronic obstructive pulmonary disease exacerbations and hospitalizations. Am J Respir Crit Care Med. 2008;178(9):913–920. doi:10.1164/rccm.200804-619OC
7. Dalal AA, Shah M, Lunacsek O, Hanania NA. Clinical and economic burden of depression/anxiety in chronic obstructive pulmonary disease patients within a managed care population. COPD. 2011;8(4):293–299. doi:10.3109/15412555.2011.586659
8. Anxiety and Depression Association of America. Facts & Statistics. Anxiety and Depression Association of America; 2018.
9. Barnett K, Mercer SW, Norbury M, Watt G, Wyke S, Guthrie B. Epidemiology of multimorbidity and implications for health care, research, and medical education: a cross-sectional study. Lancet. 2012;380(9836):37–43. doi:10.1016/S0140-6736(12)60240-2
10. Golden SH, Lazo M, Carnethon M, et al. Examining a bidirectional association between depressive symptoms and diabetes. JAMA. 2008;299(23):2751–2759. doi:10.1001/jama.299.23.2751
11. Renn BN, Feliciano L, Segal DL. The bidirectional relationship of depression and diabetes: a systematic review. Clin Psychol Rev. 2011;31(8):1239–1246. doi:10.1016/j.cpr.2011.08.001
12. Lin EH, Katon W, Von Korff M, et al. Relationship of depression and diabetes self-care, medication adherence, and preventive care. Diabetes Care. 2004;27(9):2154–2160. doi:10.2337/diacare.27.9.2154
13. Dickerson F, Brown CH, Fang L, et al. Quality of life in individuals with serious mental illness and type 2 diabetes. Psychosomatics. 2008;49(2):109–114. doi:10.1176/appi.psy.49.2.109
14. Egede LE. Major depression in individuals with chronic medical disorders: prevalence, correlates and association with health resource utilization, lost productivity and functional disability. Gen Hosp Psychiatry. 2007;29(5):409–416. doi:10.1016/j.genhosppsych.2007.06.002
15. Katon WJ. Clinical and health services relationships between major depression, depressive symptoms, and general medical illness. Biol Psychiatry. 2003;54(3):216–226. doi:10.1016/S0006-3223(03)00273-7
16. Stein MB, Cox BJ, Afifi TO, Belik S-L, Sareen J. Does co-morbid depressive illness magnify the impact of chronic physical illness? A population-based perspective. Psychol Med. 2006;36(5):587–596. doi:10.1017/S0033291706007239
17. Medical Expenditure Panel Survey background [Internet]; 2018. Available from: https://meps.ahrq.gov/mepsweb/about_meps/survey_back.jsp. Accessed December 18, 2020.
18. Machlin S, Yu W, Zodet M. Computing Standard Errors for MEPS Estimates. Rockville, MD: Agency for Healthcare Research and Quality; 2005.
19. AHRQ. MEPS topics: priority conditions; 2020. Available from: https://meps.ahrq.gov/mepsweb/data_stats/MEPS_topics.jsp?topicid=41Z-1.
20. Patel V, Chatterji S. Integrating mental health in care for noncommunicable diseases: an imperative for person-centered care. Health Aff. 2015;34(9):1498–1505. doi:10.1377/hlthaff.2015.0791
21. Talbott J. Individual and societal effects of mental disorders on earnings in the United States: results from the National Comorbidity Survey Replication. In: Year Book of Psychiatry & Applied Mental Health. Vol. 2009; 2009:140–142.
22. Luber MP, Hollenberg JP, Williams-Russo P, et al. Diagnosis, treatment, comorbidity, and resource utilization of depressed patients in a general medical practice. Int J Psychiatry Med. 2000;30(1):1–13. doi:10.2190/YTRY-E86M-G1VC-LC79
23. Melek S, Norris D Chronic conditions and comorbid psychological disorders.
24. Simon GE, Katon WJ, Lin EH, et al. Diabetes complications and depression as predictors of health service costs. Gen Hosp Psychiatry. 2005;27(5):344–351. doi:10.1016/j.genhosppsych.2005.04.008
25. Zhou Y, Cao Z, Yang M, et al. Comorbid generalized anxiety disorder and its association with quality of life in patients with major depressive disorder. Sci Rep. 2017;7(1):1–8. doi:10.1038/s41598-016-0028-x
26. Wang PS, Lane M, Olfson M, Pincus HA, Wells KB, Kessler RC. Twelve-month use of mental health services in the United States: results from the National Comorbidity Survey Replication. Arch Gen Psychiatry. 2005;62(6):629–640. doi:10.1001/archpsyc.62.6.629
27. DiMatteo MR, Lepper HS, Croghan TW. Depression is a risk factor for noncompliance with medical treatment: meta-analysis of the effects of anxiety and depression on patient adherence. Arch Intern Med. 2000;160(14):2101–2107. doi:10.1001/archinte.160.14.2101
28. Eghaneyan BH, Sanchez K, Mitschke DB. Implementation of a collaborative care model for the treatment of depression and anxiety in a community health center: results from a qualitative case study. J Multidiscip Healthc. 2014;7:503. doi:10.2147/JMDH.S69821
29. Williams JW
30. Gilbody S, Bower P, Fletcher J, Richards D, Sutton AJ. Collaborative care for depression: a cumulative meta-analysis and review of longer-term outcomes. Arch Intern Med. 2006;166(21):2314–2321. doi:10.1001/archinte.166.21.2314
31. Katon W, Lin EH, Von Korff M, et al. Integrating depression and chronic disease care among patients with diabetes and/or coronary heart disease: the design of the TEAMcare study. Contemp Clin Trials. 2010;31(4):312–322. doi:10.1016/j.cct.2010.03.009
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.



