Back to Journals » Journal of Pain Research » Volume 13
Early Blockade of EphA4 Pathway Reduces Trigeminal Neuropathic Pain
Authors Kim MJ, Son JY, Ju JS, Ahn DK
Received 10 February 2020
Accepted for publication 29 April 2020
Published 22 May 2020 Volume 2020:13 Pages 1173—1183
DOI https://doi.org/10.2147/JPR.S249185
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Michael Schatman
Min-Ji Kim,* Jo-Young Son,* Jin-Sook Ju, Dong-Kuk Ahn
Department of Oral Physiology, School of Dentistry, Kyungpook National University, Daegu, Korea
*These authors contributed equally to this work
Correspondence: Dong-Kuk Ahn
Department of Oral Physiology, School of Dentistry, Kyungpook National University, 2177 Dalgubeol-daero, Chung-gu, Daegu 41940, Korea
Tel +82-53-660-6840
Fax +82-53-421-4077
Email [email protected]
Background: Although the Eph receptor plays an important role in the development of neuropathic pain following nerve injury, there has been no evidence of the participation of the ephrin A4 receptor (EphA4) in the development of trigeminal neuropathic pain. The present study investigated the role of EphA4 in central nociceptive processing in rats with inferior alveolar nerve injury.
Materials and Methods: Male Sprague-Dawley rats were used in all our experiments. A rat model for trigeminal neuropathic pain was produced using malpositioned dental implants. The left mandibular second molar was extracted under anesthesia, followed by the placement of a miniature dental implant to injure the inferior alveolar nerve.
Results: Our current findings show that nerve injury induced by malpositioned dental implants evokes significant mechanical allodynia and up-regulation of EphA4 expression in the ipsilateral trigeminal subnucleus caudalis. Although daily treatment with EphA4-Fc, an EphA4 antagonist, did not produce prolonged anti-allodynic effects after the chronic neuropathic pain had been already established, an early treatment protocol with repeated EphA4-Fc administration significantly attenuated mechanical allodynia before initiation of chronic neuropathic pain. Finally, we confirmed the participation of the central EphA4 pathway in the development of trigeminal neuropathic pain by reducing EphA4 expression using EphA4 siRNA. This suppression of EphA4 produced significantly prolonged anti-allodynic effects.
Conclusion: These results suggest that early blockade of central EphA4 signaling provides a new therapeutic target for the treatment of trigeminal neuropathic pain.
Keywords: neuropathic pain, ephrin, EphA4, siRNA, allodynia, trigeminal
Introduction
It has been well established that ephrin (Eph) receptors, which comprise one of the largest receptor tyrosine kinase families, participate in the regulation of synaptic formation through neuron–neuron and/or neuron–astrocyte interactions in the central nervous system.1–3 Multiple Eph receptors and ephrins have been found to be upregulated at injury sites after nerve injury.4 In particular, the ephrin A4 receptor (EphA4) was expressed in the reactive astrocytes in mice and human brain tissue after spinal cord hemisection5,6 and traumatic brain injury,7 respectively. Moreover, deleting or blocking EphA4 in mice or rats with spinal cord injury promoted axonal regeneration and functional recovery.6,8,9 This suggests that EphA4 plays an important role in axonal reorganization and synaptic plasticity after neuronal damage, and targeting EphA4 could be a promising means of promoting functional recovery following neural injury.
Recent studies have demonstrated that Eph receptor-mediated cross-talk is involved in the central nociceptive processing associated with sensory abnormalities in persistent pain states. Mechanical allodynia in rats with bone cancer is significantly alleviated by the intrathecal administration of an EphB1 receptor antagonist.10 Furthermore, intrathecal application of EphB-receptor antagonists inhibits the induction and maintenance of nerve injury-induced thermal hyperalgesia and mechanical allodynia,11,12 as well as formalin-induced inflammatory pain.12,13 However, there has been no evidence of the participation of EphA4 in the development of trigeminal neuropathic pain.
In the present study, we investigated the hypothesis that changes in EphA4 expression play an important role in the modulation of neuropathic pain following nerve injury. We examined the changes in EphA4 expression in the ipsilateral trigeminal subnucleus caudalis of rats surgically inflicted with inferior alveolar nerve injury. We also evaluated the anti-nociceptive effects of blocking the EphA4 pathway in rats with trigeminal neuropathic pain. Finally, we confirmed the participation of EphA4 signaling in the development of trigeminal neuropathic pain after reducing EphA4 expression using EphA4 siRNA.
Materials and Methods
Animals
Male Sprague-Dawley rats, weighing 200–230 g, were used in all experiments. The animals were maintained in a temperature-controlled room (23 ± 1 °C) on a 12:12-hour light-dark cycle. Food and water were freely available. All procedures involving the use of animals were approved by the Institutional Care and Use Committee of the School of Dentistry, Kyungpook National University (No. 20180108). Animal experiments were also carried out in accordance with the ethical guidelines for the evaluation of experimental pain in conscious animals established by the International Association for the Study of Pain. All experiments were performed in a blind manner.
Animal Model for Trigeminal Neuropathic Pain
Rats were anesthetized with a mixed solution of ketamine (40 mg/kg, intraperitoneal (ip)) and xylazine (4 mg/kg, ip). Under anesthesia, the left mandibular second molar was extracted, and a miniature dental implant (diameter, 1 mm; length, 4 mm; donated by Megagen, Gyengsan, Korea) was placed to intentionally injure the inferior alveolar nerve, as described previously.14,15 The rats constituting the sham group were operated on without dental implant placement. For the control group, we used naïve rats that did not receive any operation. Only data from the rats with inferior alveolar nerve injury caused by the malpositioned dental implant were used in the final analyses. Approximately 90% of rats with inferior alveolar nerve injury experienced neuropathic pain.
Intracisternal Catheterization
For intracisternal administration in the rat model, the individual rats were anesthetized with a mixed solution of ketamine (40 mg/kg, ip) and xylazine (4 mg/kg, ip). Each anesthetized rat was mounted on a stereotaxic frame and a polyethylene tube (PE 10) was implanted, as described previously.16–19 The polyethylene tube was inserted through a tiny hole made in the atlantooccipital membrane and dura using a 27-gauge needle. The tip of the cannula was placed dorsal to the obex level. The anterior portion of the polyethylene tube (length, 8 cm) was advanced subcutaneously to the top of the skull and secured in place using a stainless steel screw and dental acrylic resin. All intracisternal treatments were administered as 4 μL microinjections delivered slowly, and 10 µL saline was injected to flush the cannula after each drug microinjection with the aid of a Hamilton microsyringe connected to the catheter. A recovery period of 72 hours was allowed before commencing the experiment.15,20 Any animals showing motor dysfunction or malpositioning of the catheter after intracisternal catheterization were excluded from further analysis.
Evaluation of Mechanical Allodynia
Mechanical allodynia was measured at 2 days before and 1, 3, 5, 7, 10, 12, 15, 18, 21, 25, 30, 35, and 40 days after the placement of the malpositioned dental implant, as described previously.14,21 For behavioral observation, each animal was placed in a transparent plastic cage (width, 25 cm; height, 18 cm; length, 35 cm) in a dimly lit testing room for 30 minutes before behavioral measurements to allow the animals to adapt to the test environment. The behavioral withdrawal response was evaluated after each 4-second application of air-puff pressure delivered ipsilateral to the nerve injury site for 10 times at 10-second intervals at the same pressure, before increasing the pressure intensity in the subsequent trial, as described in previous studies.21–23 The intensity and intervals of the air-puff pressure were controlled using a pneumatic pump module (BH2 System, Harvard Apparatus, MA). After inducing injury to the inferior alveolar nerve, we searched for the most sensitive area via air-puff stimulation, as previously described.24,25 The most sensitive areas included the lower jaw and the mouth angle area of the facial region. We applied the air-puff stimulation protocol to the most sensitive area through a 10 cm long 26-gauge metal tube located 1 cm from the skin at a 90° angle. The air-puff thresholds were determined as the air-puff pressure at which each rat responded in 50% of the trials. Allodynia was defined as the intensity of air puffs when the rats attempted to escape or exhibited aggressive behavioral responses to stimulation, such as face withdrawal responses or biting. The cut-off pressure for the air-puff threshold determination was 40 psi, as described previously.26–28 The naïve animals did not respond to a pressure lower than 40 psi.
Western Blotting
The rats were sacrificed under anesthesia, and the ipsilateral dorsal parts of the caudal medulla were removed and frozen rapidly in liquid nitrogen. The samples were sonicated with Biorupture (Cosmo Bio., Tokyo, Japan) in a lysis buffer containing a cocktail of protease and a phosphatase inhibitor cocktail (Thermo Scientific, Rockford, IL). The protein concentrations in the samples were measured using a fluorometer (Invitrogen, Carlsbad, CA). Total proteins (30 μg) were separated on a 10% NuPAGE Novex Bis-Tris gel (Invitrogen) and transferred to a polyvinylidene fluoride membrane using the iBlot Dry blotting system (Invitrogen). The membranes were blocked with 5% non-fat milk in Tris-buffered saline containing 0.1% Tween 20 for one hour at room temperature and then incubated with rabbit anti-EphA4 antibody (1:2000; Santa Cruz Biotechnology, Santa Cruz, CA) at 4°C overnight. The mouse anti-glyceraldehyde 3-phosphate dehydrogenase (GAPDH) antibody (1:10,000; Santa Cruz Biotechnology) was used as the loading control. The blots were incubated with goat anti-rabbit or anti-mouse horseradish peroxidase-conjugated IgG for two hours at room temperature. The blots were developed using enhanced chemiluminescence kits (Millipore, Billerica, MA), followed by exposure to an Amersham Imager 600 (GE Healthcare, Little Chalfont, UK). The bands were captured with the image analysis system and the Image J analysis system (NIH, Bethesda, MD) was used to quantify the protein bands of interest.
Intracisternal Administration of EphA4 siRNA
An siRNA molecule against EphA4 and another for a non-targeting control were designed and purchased from Ambion (Carlsbad, CA) with the following sequences: sense, 5′-GCCGUAGACGGAGUAAGUAtt-3′ and antisense, 5′-UACUUACUCCGUCUACGGCtg-3′. Catalog numbers were 4457308 for the siRNA construct directed against EphA4 and 4459405 for the in vivo negative control. The siRNA solution was mixed with a complexation buffer and then diluted with Invivofectamine 3.0 Reagent (Invitrogen) before being incubated for 30 minutes at 50°C. The EphA4 siRNA or negative control solutions at doses mentioned below were administered intracisternally to the rats immediately after inflicting inferior alveolar nerve injury.
Experimental Protocols
Changes in Behavioral Response and EphA4 Expression After Inferior Alveolar Nerve Injury
In several previous studies, inferior alveolar nerve injury was shown to produce prolonged nociceptive behavior.14,21 The present study examined ipsilateral mechanical allodynia after inferior alveolar nerve injury (n=8 per group). To determine the changes in EphA4 expression in rats with inferior alveolar nerve injury, we examined EphA4 expression in the ipsilateral trigeminal subnucleus caudalis by Western blot analysis (n = 6 per group) on postoperative day (POD) 1, 3, and 5 because the trigeminal subnucleus caudalis (medullary dorsal horn) receives nociceptive information from the orofacial area, including skin, tooth pulp, and muscle.29
Effects of a Single Treatment with EphA4-Fc on Mechanical Allodynia
To block EphA4, EphA4-Fc (0.1, 1, or 10 μg/10 μL), an EphA4 antagonist, was administered intracisternally through the implanted PE10 tube on POD 3. EphA4-Fc, a soluble chimeric fusion protein of the EphA4 receptor, has been shown to block the activation of the endogenous EphA4 receptor by acting as a soluble decoy receptor, which prevents the activation of membrane-bound EphA4 study.30 Changes in the air-puff threshold were determined at 10, 30, 60, 90, 120, 180, 360 minutes, and 24 hours after the intracisternal administration of EphA4-Fc or vehicle (n=8 per group). EphA4-Fc was purchased from R&D systems (Minneapolis, MN) and dissolved in sterile PBS.
Prolonged Anti-Allodynic Effects of Early or Late Repeated Treatments with EphA4-Fc
To determine whether repeated treatments with EphA4-Fc exerted a prolonged anti-allodynic effect, we investigated the anti-allodynic effects after daily treatment with EphA4-Fc (0.1 or 1 μg/10 μL). A previous study demonstrated that central hypersensitivity was established by POD 3 after nerve injury.31 This time point is very important to identify whether chronic neuropathic pain will develop after nerve injury. To examine the effects of daily treatment with EphA4-Fc prior to the establishment of chronic neuropathic pain, daily treatment with EphA4-Fc or vehicle (n = 8 per group) was initiated immediately after inferior alveolar nerve injury (POD 0) and repeated on the following two days (POD 1 and 2, early treatment protocol). After daily treatment with EphA4-Fc, the changes in air-puff thresholds were evaluated. To investigate the long-term anti-nociceptive effects of EphA4-Fc, we measured the air-puff thresholds once a day until POD 40, including 24 hours after daily treatment with EphA4-Fc. On the other hand, to examine the prolonged anti-allodynic effects of EphA4-Fc after chronic neuropathic pain was already established, either EphA4-Fc or vehicle (n = 8 per group) was administered intracisternally for three days starting on POD 3 (late treatment protocol). We also measured the changes in the air-puff threshold after daily treatment with EphA4-Fc and measured air-puff thresholds once a day until POD 40, including 24 hours after daily treatment with EphA4-Fc.
Effects of Intracisternal Administration of EphA4 siRNA on Mechanical Allodynia
EphA4 siRNA (0.1, 0.5, 1 nmol/10 μL) or a negative control solution (10 μL) was administered intracisternally immediately after inferior alveolar nerve injury. Subsequently, the changes in the air-puff threshold were evaluated for 9 days (n = 9 per group). To confirm the effects of EphA4 siRNA and the negative control, changes in EphA4 expression in the ipsilateral medullary dorsal horn were evaluated by Western blot analysis on POD 3 (n = 6 per group).
Data Analysis
Statistical analysis of the behavioral data was performed by repeated-measures analysis of variance (ANOVA) followed by Holm-Sidak post hoc analysis. Data from the Western blot were analyzed by one-way ANOVA followed by Holm-Sidak post hoc analysis or unpaired Student’s t-test with one-tailed analysis. In all statistical comparisons, P < 0.05 was considered significant. All data are presented as the mean ± SEM.
Results
Changes in Air-Puff Thresholds and EphA4 Expression After Inferior Alveolar Nerve Injury
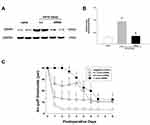
We found that the rats with a malpositioned dental implant manifested obvious nocifensive behavioral changes. Inferior alveolar nerve injury resulted in a decrease in the air-puff threshold as compared with sham surgery (F(2,21) = 1335.3, P < 0.05; Figure 1A). However, the sham-operated rats did not show an altered air-puff threshold compared with the naïve group. Ipsilateral mechanical allodynia presented on POD 1 and persisted until 35 days after surgery. Western blot analysis showed that EphA4 expression increased significantly in the ipsilateral trigeminal subnucleus caudalis on POD 1, 3, and 5 after inferior alveolar nerve injury compared with that in the sham group (P < 0.05, Figure 1B and C).
Effects of a Single Treatment with EphA4-Fc on Mechanical Allodynia
Figure 2 illustrates the anti-allodynic effects of a single treatment with EphA4-Fc, an EphA4 antagonist, on neuropathic mechanical allodynia on POD 3. Treatment with the vehicle did not affect mechanical allodynia induced by the malpositioned dental implant. Intracisternal administration of a low dose of EphA4-Fc (0.1 μg) did not affect the air-puff threshold; however, treatment with higher doses of EphA4-Fc (1 or 10 μg) produced significant anti-allodynic effects compared with vehicle treatment (F(3,20) = 514.1, P < 0.05). The anti-allodynic effects produced by a single treatment with EphA4-Fc appeared within 30 minutes and returned to the pretreated levels within 24 hours after injection. Although a high dose of EphA4-Fc (10 μg) provided effective pain relief, it caused motor dysfunction. Therefore, the high dose of EphA4-Fc was excluded from the following experiments.
Effects of Repeated Treatments with EphA4-Fc on Mechanical Allodynia
The present study investigated the anti-allodynic effects induced by daily treatment with EphA4-Fc for 3 days starting on POD 0 before the chronic neuropathic pain was established (Figure 3). The measurements of behavioral responses on POD 0 were omitted because the effects of drug administration could be masked as a result of anesthesia for surgery. Daily intracisternal treatments with both doses of EphA4-Fc (0.1 and 1 μg) produced significant anti-allodynic effects on POD 1 and 2 (P<0.05, Figure 3A). Anti-allodynic effects appeared within 1 hour after intracisternal administration of EphA4-Fc (1 μg) and persisted until 24 hours on both POD 1 and 2. Moreover, we measured air-puff thresholds once a day until POD 40 to investigate the long-term antinociceptive effects of EphA4-Fc. An early treatment protocol with 1 μg of EphA4-Fc for 3 days starting on POD 0 produced significantly prolonged anti-allodynic effects (F(2,15) = 41.1, P < 0.05, Figure 3B), which were sustained throughout the entire observation period until POD 36. Administration of vehicle or a low dose of EphA4-Fc (0.1 μg) did not produce prolonged anti-allodynic effects in rats with inferior alveolar nerve injury.
The present study also investigated the anti-allodynic effects after daily treatment with EphA4-Fc for 3 days starting on POD 3, when the chronic neuropathic pain was already established (Figure 4). Daily intracisternal administrations of a low dose of EphA4-Fc (0.1 μg) did not produce any anti-allodynic effects, except on POD 5 (3rd treatment). Daily treatment with 1 μg of EphA4-Fc produced significant anti-allodynic effects on POD 3, 4, and 5 (P < 0.05, Figure 4A). However, the anti-allodynic effects returned to the pretreatment levels by 24 hours after EphA4-Fc injection. In contrast to the early EphA4-Fc treatment protocol, late treatment with EphA4-Fc for 3 days starting on POD 3 did not produce any prolonged anti-allodynic effects when the chronic neuropathic pain was already established (Figure 4B).
Effects of Intracisternal Administration of EphA4 siRNA on Mechanical Allodynia
Figure 5 illustrates the effects of EphA4 siRNA on neuropathic mechanical allodynia following inferior alveolar nerve injury. Western blot analysis revealed that a single intracisternal administration of EphA4 siRNA, microinjected immediately after the malpositioned dental implantation, attenuated the upregulated EphA4 expression resulting from inferior alveolar nerve injury (P < 0.05, Figure 5A and B). However, the negative control did not affect the upregulated EphA4 expression. Furthermore, intracisternal injection of the negative control did not affect the air-puff threshold. However, all doses of single EphA4 siRNA produced an anti-allodynic effect in rats with trigeminal neuropathic pain (F(3,24) = 188.2, P < 0.05, Figure 5C). A high dose (1 nmol) of EphA4 siRNA produced a significantly prolonged anti-allodynic effect that persisted until 6 days after a single treatment.
Discussion
The present study is the first to demonstrate that central EphA4 activity is a key mechanism for the development of nerve injury-induced trigeminal neuropathic pain. Our current findings show that inferior alveolar nerve injury induced significant mechanical allodynia and up-regulated EphA4 expression in the ipsilateral trigeminal subnucleus caudalis. Although nerve injury-induced mechanical allodynia was unaffected by daily treatment with an EphA4 antagonist once the phenomenon had been established, this treatment effectively and persistently reduced mechanical allodynia when initiated shortly after the nerve-injury procedure. Moreover, the down-regulation of EphA4 by EphA4 siRNA produced significant prolonged anti-allodynic effects. These results suggest that the central EphA4 signaling pathway plays an important role in the development of trigeminal neuropathic pain after nerve injury.
Generally, Eph receptors have been classified into two subclasses, EphAs and EphBs, based on similarities in sequence and binding affinity.32 EphA receptors, identified by nine receptors, showed promiscuous binding activity with five ephrin-A ligands.3,33 The EphA pathway has been reported to regulate normal function and synaptic plasticity in the central nervous system.34,35 In particular, EphA4 is a synapse modulator involved in the formation of specific synapses and neuronal plasticity, such as long-term potentiation (LTP) in the adult hippocampus35,36 and the neuromuscular junction.37 In addition, blockade of EphA4 by EphA4-Fc, an EphA4 antagonist, delayed the development of experimental autoimmune encephalomyelitis (EAE) in mice,38 and attenuated the apoptotic cell death of hippocampal CA1 pyramidal neurons induced by ischemia/reperfusion in an experimental animal model of stroke.39 These findings suggest that EphA4 plays an important role in axonal reorganization and synaptic plasticity after various types of neuronal damage or injury. On the other hand, EphA4 promoted or enhanced the onset and severity of EAE and neuronal cell death in a stroke model.38,39 Therefore, EphA4 might play a role in the pathogenesis of neuronal damage or injury rather than axonal reorganization and synaptic plasticity.
The present data demonstrated that inferior alveolar nerve injury, which produces significant mechanical allodynia, increased EphA4 expression in the ipsilateral trigeminal subnucleus caudalis. Moreover, acute treatment with the antagonist EphA4-Fc attenuated mechanical allodynia induced by nerve injury in a dose-dependent and transient fashion, as compared with the vehicle-treated group. These results suggest that the upregulation of EphA4 expression plays a critical role in the development of mechanical allodynia in rats with trigeminal neuropathic pain after inferior alveolar nerve injury. The role of Eph receptors in central nociceptive processing, in particular the role of EphA4 in the development of trigeminal neuropathic pain, has not yet been addressed. In contrast, following spinal cord injury, another condition associated with an up-regulation of central EphA4 expression and persistent mechanical allodynia, the intensity of the latter was actually potentiated by suppression of EphA4 expression.40 These results indicate that the upregulation of EphA4 after spinal cord injury may limit the development of abnormal pain. The mechanisms underlying the conflicting findings observed in these different experimental animal models of nerve injury remain to be examined.
As a single treatment with EphA4-Fc produced significant but short-lasting anti-allodynic effects, we examined whether repeated daily treatment with the EphA4 antagonist would result in prolonged antinociceptive effects. Intracisternal daily microinjections of EphA4-Fc for 3 days starting on POD 3 (ie once allodynia was already established) failed to modify the intensity of allodynia during the treatment period or thereafter. However, when daily treatment with EphA4-Fc (at 1 μg) was initiated on POD 0 and repeated on the following 2 days, allodynia was markedly and persistently reduced over the entire 30-day observation period. These results suggest that the early blockade of the EphA4 pathway prior to the establishment of neuropathic pain effectively attenuated mechanical allodynia after inferior alveolar nerve injury. These early treatment findings were consistent with those of previous studies. Central hypersensitivity was established within POD 3 and was not modified by peripheral input after chronic pain was established.30 Early pain treatment can prevent the transition from acute to chronic pain.41,42 Moreover, early treatment with dexamethasone produces prolonged antinociceptive effects in rats with malpositioned dental implants, whereas delayed treatment has no effect.14 These results indicate that once neuropathic pain is established, it tends to become more difficult to treat successfully, requiring more aggressive treatments. Therefore, our findings indicate that early blockade of the EphA4 pathway can significantly limit the further development of neuropathic pain in response to nerve injury. On the other hand, it is noteworthy that EphA4-Fc is a glycosylated receptor decoy with a very short half-life in vivo, which is likely to have a negative effect on the duration of its antagonistic actions. In this regard, deletion of three of the N-linked glycosylation sites of the EphA4-Fc molecule by glycoengineering has been shown to dramatically improve the in vivo half-life and pharmacokinetic characteristics.43 Such a long-lasting EphA4 antagonist may prove to be more effective than EphA4-Fc in counteracting neuropathic pain induced by nerve injury.
It is well known that bidirectional signaling between Eph receptors and ephrin ligands plays an important role in the cross-talk or communication between cells.44 The present study demonstrated that inferior alveolar nerve injury increased EphA4 expression in the superficial lamina of the trigeminal subnucleus caudalis. Upregulated EphA4 expression was observed in the astrocytes in the trigeminal subnucleus caudalis (unpublished data). Therefore, the upregulated EphA4 expression may participate in cross-talk or communication between the neuron and astrocytes after inferior alveolar nerve injury. However, further studies are needed to identify the underlying cellular mechanisms.
The present study also assessed the effects of EphA4 siRNA to confirm the participation of EphA4 in the development of trigeminal neuropathic pain. Intracisternal administration of EphA4 siRNA attenuated the upregulated EphA4 expression in the ipsilateral trigeminal subnucleus caudalis induced by inferior alveolar nerve injury, whereas the negative control did not affect EphA4 expression. Importantly, this same treatment also promoted long-lasting anti-allodynic effects. These results support our hypothesis that early EphA4 activity is a key mechanism for the development of trigeminal neuropathic pain after nerve injury.
In summary, early treatment with EphA4-Fc promoted persistent anti-nociceptive effects in rats with inferior alveolar nerve injury. Moreover, intracisternal administration of EphA4 siRNA produced anti-allodynic effects and limited the upregulation of EphA4 expression in the trigeminal subnucleus caudalis. These results suggest that early modulation of EphA4 signaling is a potentially important treatment strategy for trigeminal neuropathic pain.
Acknowledgments
This research was supported by the National Research Foundation of Korea (NRF) grant funded by the Korea government (NRF-2017R1A5A2015391 and NRF-2018R1D1A1B07049025). The authors thank Megagen (Gyeongsan, Republic of Korea) for providing miniature dental implants.
Author Contributions
All authors made substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data; took part in drafting the article or revising it critically for important intellectual content; gave final approval of the version to be published; and agree to be accountable for all aspects of the work.
Disclosure
The authors have no conflicts of interest to declare.
References
1. Klein R. Bidirectional modulation of synaptic functions by Eph/ephrin signaling. Nat Neurosci. 2009;12(1):15–20. doi:10.1038/nn.2231
2. Murai KK, Pasquale EB. Eph receptors and ephrins in neuron-astrocyte communication at synapses. Glia. 2011;59(11):1567–1578. doi:10.1002/glia.21226
3. Pasquale EB. Eph receptor signalling casts a wide net on cell behaviour. Nat Rev Mol Cell Biol. 2005;6(6):462–475. doi:10.1038/nrm1662
4. Du J, Fu C, Sretavan DW. Eph/ephrin signaling as a potential therapeutic target after central nervous system injury. Curr Pharm Des. 2007;13(24):2507–2518. doi:10.2174/138161207781368594
5. Coulthard MG, Morgan M, Woodruff TM, et al. Eph/Ephrin signaling in injury and inflammation. Am J Pathol. 2012;181(5):1493–1503. doi:10.1016/j.ajpath.2012.06.043
6. Goldshmit Y, Galea MP, Wise G, Bartlett PF, Turnley AM. Axonal regeneration and lack of astrocytic gliosis in EphA4-deficient mice. J Neurosci. 2004;24(45):10064–10073. doi:10.1523/JNEUROSCI.2981-04.2004
7. Frugier T, Conquest A, McLean C, Currie P, Moses D, Goldshmit Y. Expression and activation of EphA4 in the human brain after traumatic injury. J Neuropathol Exp Neurol. 2012;71(3):242–250. doi:10.1097/NEN.0b013e3182496149
8. Fabes J, Anderson P, Brennan C, Bolsover S. Regeneration-enhancing effects of EphA4 blocking peptide following corticospinal tract injury in adult rat spinal cord. Eur J Neurosci. 2007;26(9):2496–2505. doi:10.1111/j.1460-9568.2007.05859.x
9. Goldshmit Y, Spanevello MD, Tajouri S, et al. EphA4 blockers promote axonal regeneration and functional recovery following spinal cord injury in mice. PLoS One. 2011;6(9):e24636. doi:10.1371/journal.pone.0024636
10. Dong Y, Mao-Ying QL, Chen JW, Yang CJ, Wang YQ, Tan ZM. Involvement of EphB1 receptor/ephrinB1 ligand in bone cancer pain. Neurosci Lett. 2011;496(3):163–167. doi:10.1016/j.neulet.2011.04.008
11. Song XJ, Zheng JH, Cao JL, Liu WT, Song XS, Huang ZJ. EphrinB-EphB receptor signaling contributes to neuropathic pain by regulating neural excitability and spinal synaptic plasticity in rats. Pain. 2008;139(1):168–180. doi:10.1016/j.pain.2008.03.019
12. Ruan JP, Zhang HX, Lu XF, Liu YP, Cao JL. EphrinBs/EphBs signaling is involved in modulation of spinal nociceptive processing through a mitogen-activated protein kinases-dependent mechanism. Anesthesiology. 2010;112(5):1234–1249. doi:10.1097/ALN.0b013e3181d3e0df
13. Cao JL, Ruan JP, Ling DY, et al. Activation of peripheral ephrinBs/EphBs signaling induces hyperalgesia through a MAPKs-mediated mechanism in mice. Pain. 2008;139(3):617–631. doi:10.1016/j.pain.2008.06.023
14. Han SR, Yeo SP, Lee MK, Bae YC, Ahn DK. Early dexamethasone relieves trigeminal neuropathic pain. J Dent Res. 2010;89(9):915–920. doi:10.1177/0022034510374056
15. Won KA, Kim MJ, Yang KY, et al. The glial-neuronal GRK2 pathway participates in the development of trigeminal neuropathic pain in rats. J Pain. 2014;15(3):250–261. doi:10.1016/j.jpain.2013.10.013
16. Yaksh TL, Rudy TA. Chronic catheterization of the spinal subarachnoid space. Physiol Behav. 1976;17(6):1031–1036. doi:10.1016/0031-9384(76)90029-9
17. Kim HJ, Lee GW, Kim MJ, et al. Antinociceptive effects of transcytosed botulinum neurotoxin type A on trigeminal nociception in rats. Korean J Physiol Pharmacol. 2015;19(4):349–355. doi:10.4196/kjpp.2015.19.4.349
18. Yang GY, Lee MK, Bae YC, Ahn DK. Intracisternal administration of COX inhibitors attenuates mechanical allodynia following compression of the trigeminal ganglion in rats. Prog Neuropsychopharmacol Biol Psychiatry. 2009;33(4):589–595. doi:10.1016/j.pnpbp.2009.02.010
19. Yang KY, Mun JH, Park KD, et al. Blockade of spinal glutamate recycling produces paradoxical antinociception in rats with orofacial inflammatory pain. Prog Neuropsychopharmacol Biol Psychiatry. 2015;57:100–109. doi:10.1016/j.pnpbp.2014
20. Jeon HJ, Han SR, Lim KH, Won KA, Bae YC, Ahn DK. Intracisternal administration of NR2 subunit antagonists attenuates the nociceptive behavior and p-p38 MAPK expression produced by compression of the trigeminal nerve root. Mol Pain. 2011;7:46. doi:10.1186/1744-8069-7-46
21. Lee MK, Han SR, Park MK, et al. Behavioral evidence for the differential regulation of p-p38 MAPK and p-NF-κB in rats with trigeminal neuropathic pain. Mol Pain. 2011;7:57. doi:10.1186/1744-8069-7-57
22. Ahn DK, Lee SY, Han SR, et al. Intratrigeminal ganglionic injection of LPA causes neuropathic pain-like behavior and demyelination in rats. Pain. 2009;146(1–2):114–120. doi:10.1016/j.pain.2009.07.012
23. Ahn DK, Lim EJ, Kim BC, et al. Compression of the trigeminal ganglion produces prolonged nociceptive behavior in rats. Eur J Pain. 2009;13(6):568–575. doi:10.1016/j.ejpain.2008.07.008
24. Yang KY, Kim MJ, Ju JS, et al. Antinociceptive effects of botulinum toxin type A on trigeminal neuropathic pain. J Dent Res. 2016;95(10):1183–1190. doi:10.1177/0022034516659278
25. Lee GW, Son JY, Lee AR, Ju JS, Bae YC, Ahn DK. Central VEGF-A pathway plays a key role in the development of trigeminal neuropathic pain in rats. Mol Pain. 2019;15:1–14. doi:10.1177/1744806919872602
26. Han SR, Yang GY, Ahn MH, et al. Blockade of microglial activation reduces mechanical allodynia in rats with compression of the trigeminal ganglion. Prog Neuropsychopharmacol Biol Psychiatry. 2012;36(1):52–59. doi:10.1016/j.pnpbp.2011.10.007
27. Jeon HJ, Han SR, Park MK, Yang KY, Bae YC, Ahn DK. A novel trigeminal neuropathic pain model: compression of the trigeminal nerve root produces prolonged nociception in rats. Prog Neuropsychopharmacol Biol Psychiatry. 2012;38(2):149–158. doi:10.1016/j.pnpbp.2012.03.002
28. Kim MJ, Shin HJ, Won KA, et al. Progesterone produces antinociceptive and neuroprotective effects in rats with microinjected lysophosphatidic acid in the trigeminal nerve root. Mol Pain. 2012;8:16. doi:10.1186/1744-8069-8-16
29. Sessle BJ, Hu JW, Amano N, Zhong G. Convergence of cutaneous, tooth pulp, visceral, neck and muscle afferents onto nociceptive and non-nociceptive neurones in trigeminal subnucleus caudalis (medullary dorsal horn) and its implications for referred pain. Pain. 1986;27(2):219–235. doi:10.1016/0304-3959(86)90213-7
30. Brantley DM, Cheng N, Thompson EJ, et al. Soluble Eph A receptors inhibit tumor angiogenesis and progression in vivo. Oncogene. 2002;21(46):7011–7026. doi:10.1038/sj.onc.1205679
31. Curatolo M, Petersen-Felix S, Arendt-Nielsen L, Giani C, Zbinden AM, Radanov BP. Central hypersensitivity in chronic pain after whiplash injury. Clin J Pain. 2001;17(4):306–315. doi:10.1097/00002508-200112000-00004
32. Eph Nomenclature Committee. Unified nomenclature for Eph family receptors and their ligands the ephrins. Cell. 1997;90(3):403–404. doi:10.1016/S0092-8674(00)80500-0
33. Pasquale EB. Eph-ephrin promiscuity is now crystal clear. Nat Neurosci. 2004;7(5):417–418. doi:10.1038/nn0504-417
34. Goldshmit Y, McLenachan S, Turnley A. Roles of Eph receptors and ephrins in the normal and damaged adult CNS. Brain Res Rev. 2006;52(2):327–345. doi:10.1016/j.brainresrev.2006.04.006
35. Filosa A, Paixão S, Honsek SD, et al. Neuron-glia communication via EphA4/ephrin-A3 modulates LTP through glial glutamate transport. Nat Neurosci. 2009;12(10):1285–1292. doi:10.1038/nn.2394
36. Murai KK, Nguyen LN, Irie F, Yamaguchi Y, Pasquale EB. Control of hippocampal dendritic spine morphology through ephrin-A3/EphA4 signaling. Nat Neurosci. 2003;6(2):153–160. doi:10.1038/nn994
37. Lai KO, Ip FC, Cheung J, Fu AK, Ip NY. Expression of Eph receptors in skeletal muscle and their localization at the neuromuscular junction. Mol Cell Neurosci. 2001;17(6):1034–1047. doi:10.1006/mcne.2001.0997
38. Munro KM, Dixon KJ, Gresle MM, et al. EphA4 receptor tyrosine kinase is a modulator of onset and disease severity of experimental autoimmune encephalomyelitis (EAE). PLoS One. 2013;8(2):e55948. doi:10.1371/journal.pone.0055948
39. Li J, Liu N, Wang Y, Wang R, Guo D, Zhang C. Inhibition of EphA4 signaling after ischemia-reperfusion reduces apoptosis of CA1 pyramidal neurons. Neurosci Lett. 2012;518(2):92–95. doi:10.1016/j.neulet.2012.04.060
40. Cruz-Orengo L, Figueroa JD, Velázquez I, et al. Blocking EphA4 upregulation after spinal cord injury results in enhanced chronic pain. Exp Neurol. 2006;202(2):421–433. doi:10.1016/j.expneurol.2006.07.005
41. Hefferan MP, O’Rielly DD, Loomis CW. Inhibition of spinal prostaglandin synthesis early after L5/L6 nerve ligation prevents the development of prostaglandin-dependent and prostaglandin-independent allodynia in the rat. Anesthesiology. 2003;99(5):1180–1188. doi:10.1097/00000542-200311000-00027
42. Verdi J, Jafari-Sabet M, Mokhtari R, Mesdaghinia A, Banafshe HR. The effect of progesterone on expression and development of neuropathic pain in a rat model of peripheral neuropathy. Eur J Pharmacol. 2013;699(1–3):207–212. doi:10.1016/j.ejphar.2012.11.052
43. Pegg CL, Cooper LT, Zhao J, et al. Glycoengineering of EphA4 Fc leads to a unique, long-acting and broad spectrum, Eph receptor therapeutic antagonist. Sci Rep. 2017;7(1):6519. doi:10.1038/s41598-017-06685-z
44. Pasquale EB. Eph-ephrin bidirectional signaling in physiology and disease. Cell. 2008;133(1):38–52. doi:10.1016/j.cell.2008.03.011
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.