Back to Journals » Drug Design, Development and Therapy » Volume 11
Does remifentanil attenuate renal ischemia–reperfusion injury better than dexmedetomidine in rat kidney?
Authors Erkılıç E, Kesimci E , Alaybeyoğlu F, Kılınç I, Tural R, Yazgan A, Gümüş T, Sepici Dinçel A, Dumlu EG, Kanbak O
Received 4 November 2016
Accepted for publication 9 February 2017
Published 8 March 2017 Volume 2017:11 Pages 677—683
DOI https://doi.org/10.2147/DDDT.S126701
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Qiongyu Guo
E Erkılıç,1 E Kesimci,1 F Alaybeyoğlu,1 I Kılınç,2 R Tural,3 A Yazgan,4 T Gümüş,1 A Sepici Dinçel,3 EG Dumlu,5 O Kanbak1
1Anesthesiology and Reanimation Department, 2General Surgery Department, Atatürk Training and Research Hospital, 3Biochemistry Department, Faculty of Medicine, Gazi University, 4Pathology Department, Atatürk Training and Research Hospital, 5General Surgery Clinic, Faculty of Medicine, Yıldırım Beyazit University, Ankara, Turkey
Background: Ischemia–reperfusion (I/R) injury is a common cause of patient morbidity and mortality in the perioperative period. Patients undergoing long-lasting, abdominal, and urogenital surgeries with risk factors such as advanced age, peripheral artery disease, diabetes mellitus, renovascular disease, and congestive heart failure are candidates for acute kidney injury (AKI) due to impaired renal perfusion and decreased functional renal reserve. Pharmacological agents with multiple functions and anti-oxidative and anti-inflammation properties may be promising preventative strategies for AKI. Recently, dexmedetomidine (dex) has been postulated to have renoprotective effects.
Objectives: We aimed to investigate the protective effects of an intravenous anesthetic remifentanil in renal I/R injury in the rat in comparison with dex.
Materials and methods: A total of 30 Sprague Dawley adult rats were randomly assigned into five groups: the control group (group C, n=6), the sham group (group Sh, n=6, saline-infused rats without I/R injury), the saline group (group S, n=6, saline-infused rats with I/R injury), the remifentanil-treated group (group REM, n=6), and the dexmedetomidine-treated group (group DEX, n=6). The infusions (saline, remifentanil, and dex) were started after anesthesia induction and right nephrectomy and continued until the end of the surgical procedure. In I/R injury groups, the left renal artery and vein were occluded together by a clamp for 30 minutes and reperfusion lasted for 30 minutes. The rats were sacrificed after reperfusion, and the left kidney tissue was harvested. Blood samples were drawn from all animals to evaluate plasma neutrophil gelatinase-associated lipocalin (NGAL) at the beginning, 15 minutes after ischemia, 15 minutes after reperfusion, and 6 hours after the surgical procedure (T0, T1, T2, and T3, respectively).
Results: The plasma NGAL levels exhibited increase at T1, T2, and T3 compared to the levels at T0 in group S (P<0.05). In group REM, there was a significant increase in plasma NGAL levels at T3 in comparison to those at T0, T1, and T2. The plasma NGAL levels at T2 in group S were significantly higher than those at T2 in group DEX (P<0.05). The groups S and REM showed significantly higher plasma NGAL levels at T3 compared to those at T0 (P<0.05). Upon histological examination, there was no difference among the study groups when left kidneys were evaluated (P>0.05).
Conclusion: The NGAL levels and histopathological findings reflected protection by dex against renal I/R injury. However, the same exact results could not be mentioned for remifentanil depending on our study results.
Keywords: ischemia–reperfusion, dexmedetomidine, remifentanil, neutrophil gelatinase-associated lipocalin
Introduction
Acute kidney injury (AKI) is a clinical event that is frequently observed in major surgical procedures due to modifications in renal perfusion pressure and mediators released as a result of surgical trauma.1 Temporarily reduced blood flow and the associated oxygen deprivation to the kidneys are suggested to generate oxygen-free radicals and reactive oxygen species, resulting in collection of inflammatory cells that also lead to a series of reactions. It is quite noteworthy to diagnose this tissue injury urgently so as to apply therapeutic measures as soon as possible.
Neutrophil gelatinase-associated lipocalin (NGAL), which has been shown to be an early marker of AKI, is normally produced and secreted by kidney tubule cells at low levels. However, under ischemic, septic, or nephrotoxic conditions, the amount produced and secreted by the nephron increases dramatically.2–4 Besides, it is produced in the granules of activated neutrophils. Thus, it is immediately secreted into urine.5 Serum creatinine that has been accustomed to be used as an early biomarker for renal function, however, requires several days to show a detectable increase and is affected by parameters, including muscle mass, gender, ethnicity, and medication.6
In recent experimental and clinical studies, some authors have suggested that increased levels of NGAL in serum and/or urine are identified as an expression of AKI, even before increases in serum creatinine levels.7–10
There is continuing investigation in the field of anesthetics used for the prevention of perioperative injury of kidneys and treatment of renal function loss.11,12 dexmedetomidine (dex), which is widely used in clinical anesthesia, is a highly selective agonist of α2-adrenergic receptors.13 It is effective in sedation, analgesia, and hemodynamic stabilization during anesthesia.14 Moreover, it has been suggested to play an important role in ischemia–reperfusion (I/R) injury in several organs, inflammation, and sepsis, in both in vivo and in vitro models, apart from its use as an adjunct in anesthesia and analgesia.15,16
Remifentanil is an ultra-short-acting mu-opioid receptor agonist with very rapid onset and offset of clinical action, which makes it unique.17 As other opioid receptor agonists, it has also been shown to not only suppress the inflammatory response but also protect against I/R injury in some tissues.18,19 However, we have little information about remifentanil’s renoprotective effects.
The aims of the present study were to compare the possible preventive effects of two agents (dex vs remifentanil) in an experimental I/R model in rats by measuring NGAL levels and evaluate the relationship with histological examination in the first hours of AKI.
Materials and methods
Animals
Animals were housed and cared for in the Animal Resource Center and allowed free access to food and water. All procedures were reviewed and approved by the Institutional Animal Care and Use Committee of the Ankara Research and Training Hospital and were performed in accordance with the “Guiding Principles for Research Involving Animals and Human Beings”.
Experimental protocol
In the present study, 30 Sprague Dawley adult rats weighing 200–220 g were used. The study was approved by the ethical committee on animal research of Ankara Research and Training Hospital. The animals were housed under controlled conditions that included a 12-hour light/dark cycle (08:00–20:00 light; 20:00–08:00 dark), temperature of 23°C–25°C, humidity in the range of 55%–60%, and free access to standard food and drinking water. The rats were anesthetized by a single dose of Ketalar® (ketamine, ketamine HCl; Pfizer, Bristol, UK; 50 mg·kg−1) injection intraperitoneally (ip) before surgical operation.
Animals and experimental protocol
The preparation in the experiment was based on a previously described method for experimental renal I/R.20 The animals were randomly separated into five groups, each containing six rats. Following anesthesia induction, the abdomen of rats was dissected and right nephrectomy was performed. In the control group (group C, n=6), the rats were sacrificed after taking blood samples and removal of right kidneys without any infusion. In the saline group (group S, n =6), normal saline infusion was started after anesthesia induction and the right kidney was harvested without left renal ischemia and reperfusion. In the sham group (group Sh, n=6), following anesthesia induction and right nephrectomy, normal saline infusion was started as in group S but left renal ischemia was also performed by occlusion of the left renal artery for 30 minutes. Then, the arterial clamp was removed and reperfusion of the kidney was observed for 30 minutes. In dexmedetomidine-treated group (group DEX, n=6), right nephrectomy and left renal I/R were performed, but in addition to these, Precedex (dexmedetomidine hydrochloride 100 μg/2 mL; Abbott Laboratories, North Chicago, IL, USA) was infused at 3 μg·kg−1·h−1·following a bolus of 3 μg.kg−1 over 10 minutes simultaneously at the starting time of abdominal dissection until the end of the surgical procedure. In the remifentanil-treated group (group REM, n=6), remifentanil was infused at 2 μg·kg−1·min−1 starting simultaneously with abdominal dissection for right nephrectomy and left renal I/R until the end of the surgical procedure. Blood samples were drawn from all animals at predetermined time points, so as to evaluate serum levels of NGAL (T0: basal, T1: 15 minutes after ischemia, T2: 15 minutes after reperfusion, and T3: 6 hours after the end of the surgical procedure). Serum lipocalin-2/NGAL level was determined by Lipocalin-2/NGAL PicoKine™ ELISA kit (sandwich ELISA kit for quantitative detection; Boster Antibody and ELISA Experts, Boster Biological Technology, Pleasanton, CA, USA). Also, all through the procedure, the mean arterial pressure (MAP), heart rate (HR), and body temperature were monitored and recorded at the same predetermined time points. Then, all rats were sacrificed after 6 hours following reperfusion, and left kidney was harvested.
Histopathological evaluation
The extracted kidneys were fixed in 10% formalin and embedded in paraffin. Sections of the tissue were cut at 5 μm and mounted on slides. The coded kidney specimens were stained with hematoxylin and eosin and periodic acid–Schiff (PAS) method and examined in a blinded manner under a light microscope.
Histological alterations were evaluated by quantitative measurement of acute tubular necrosis (ATN), which was assessed by counting the number of necrotic and apoptotic cells, loss of tubular brush border, tubular dilatation, and cast formation as follows: 0= none, 1=0–10%, 2=11%–25%, 3=26%–45%, 4=46%–75%, and 5=76%–100%. Histological changes because of ATN score were evaluated in the outer line of the outer medulla on each PAS-stained slide. As a minimum, 10 fields (×20) were evaluated for each slide. Apoptosis was described as cellular rounding and shrinkage and nuclear chromatin condensation. Apoptotic tubular cells were quantitatively evaluated per 10 high-power fields (×40) in the outer line of the outer medulla by the pathologist in a blinded manner.
Statistical analysis
Study data were entered into the computer and analyzed using the Statistical Package for the Social Sciences (SPSS) for Windows version 22.0 (SPSS Inc., Chicago, IL, USA). Descriptive statistics were presented as median (range). Normal distribution of the variables was tested by visual (histogram and probability graphs) and analytical methods (Shapiro–Wilk test). In order to assess the statistical significance between two dependent groups, Wilcoxon signed-rank test; between three dependent groups, Friedman test; and between three independent groups, Kruskal–Wallis test were used for variables with non-normal distribution. In case of a statistically significant difference between three dependent groups and between three independent groups, post hoc Bonferroni correction was used. Statistical significance level was set at P<0.05.
Results
All of the 30 animals successfully completed the experimental protocol. There was no statistically significant difference in regard to hemodynamic parameters (MAP, HR) and body temperatures of the animals.
Renal function changes
For in-group comparisons, the plasma NGAL levels (pg·mL−1) did not change at any time point (585.6 pg·mL−1) in group C. In group S, the plasma NGAL levels exhibited a considerable increase at T1 (567.3 pg·mL−1), T2 (670.6 pg·mL−1), and T3 (640.6 pg·mL−1) compared to the levels measured at T0 (530.6 g·mL−1; P=0.014; Figure 1). In group REM, there was a significant increase in plasma NGAL levels at T3 (624.0 pg·mL−1) in comparison to T0 (530.6 pg·mL−1), T1 (574.0 g·mL−1), and T2 (537.3 pg·mL−1) time points as shown in Figure 1 (P=0.019). In group DEX, plasma NGAL levels at three predetermined time points were not different from basal levels and each other (P>0.05).
When the groups were compared to each other, plasma NGAL levels measured at T2 (670.6 pg·mL−1) in group S were significantly higher than the levels measured at the same time point (520.6 pg·mL−1) in group DEX (P=0.031). The plasma NGAL levels (pg·mL−1) measured at T2 and T3 in groups S and REM varied significantly in comparison to other groups (P=0.014 and P=0.019, respectively). There was no significant difference between the NGAL levels of groups Sh, DEX, and REM at the mentioned time points (T2 and T3; Figure 1).
Renal histology changes
With respect to ATN, there was no difference between right and left kidneys in all groups (P>0.05). All the groups, except group C, showed statistically significant difference between right and left kidneys (P<0.05; Figure 2).
Acute tubular cell numbers were significantly higher in left kidneys of group C in comparison with the ones in group S and the ones in group REM (P<0.05; Figure 3). The median number of apoptotic cells in left kidneys was statistically significantly higher in groups C and S in comparison to groups Sh, REM, and DEX (P<0.05; Figures 2, 4, and 5).
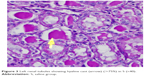  | Figure 3 Left renal tubules showing hyaline cast (arrow) (>75%) in S (×40). |
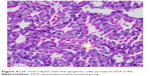  | Figure 4 Left renal tubules with few apoptotic cells (arrow) in DEX (×40). |
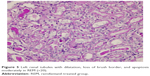  | Figure 5 Left renal tubules with dilatation, loss of brush border, and apoptosis moderately in REM (×20). |
On the other side, in groups Sh, S, REM, and DEX, apoptotic cell numbers showed an increase with relevance to time (P=0.042, P=0.043, P=0.043, and P=0.043, respectively; Figure 2). In these groups, apoptotic cell numbers of right kidneys were significantly higher than the ones of left kidneys (P>0.05; Figure 2).
Discussion
In the present study, we compared, for the first time to our knowledge, the probable protective effects of infusions of two agents, namely, dex and remifentanil. The right kidneys were the ones exposed to only intravenous anesthetic agents. They were not treated with any study infusions. Hence, higher ATN and apoptosis observed in right kidneys were attributed to the absence of the possible renoprotective effects of these agents.
Dex has been studied intensely as a sedative agent both in intensive care units and surgical settings, due to its superiority to benzodiazepines and propofol.21 As a result of the presence of adrenoceptors in human kidneys, dex has been reported to be significantly beneficial in perioperative renal functions.12,22,23 However, the underlying mechanism of this activity is still not clear. Most of the authors have supported the role of dex in reducing renin release with increasing water and sodium excretion and urinary output perioperatively.24–26 Additionally, dex has been shown to attenuate renal I/R injury, primarily in murine models.27,28 According to a study performed by Kocoglu et al,29 dex-treated rats had less apoptotic cells and histological lesions. Si et al30 also demonstrated improved tubular histology in renal ischemia model of rats when dex was infused. In our study, left kidneys in DEX had less necrotic cells and apoptotic tubular cells with regard to the left kidneys of saline-infused group. Besides, plasma NGAL levels were comparable between dex-treated and sham groups at all measurement times. This might be attributed to reduction in excessive free radicals indirectly by reduced noradrenalin secretion due to dex’s activity at presynaptic alpha-2 adrenoceptor.31 An increase in the concentration of antiapoptotic proteins and reduction in necrotic cell death caused as a result of decreased sympathetic tone by dex are the other possible underlying mechanisms.32 Our finding is similar to other authors’ reports in murine models,30,31 but there are also clinical studies32 reporting unfavorable notifications about dex. Most of these trials, which were performed in cardiac surgery patients, had indicated no influence of dex on renal functions.25,33 Kulka et al34 associated this protection provided by these agents with their adrenergic-mediated vasoconstriction. Indeed, these contradictory results might have resulted from different timings and dosings of dex infusion used in trials. Cakir et al35 pointed that 10–100 mg·kg−1 doses of dex had renoprotective effects in renal I/R injury, similar to the other studies in this subject. On the other hand, in a study, it is reported to be administered before ischemia for renal function improvement.12 We actually started infusions, as soon as anesthesia was induced. This effect was supported by the obvious reduction in blood urea and creatinine levels and significant changes in histopathology in another study.12 The low dose we applied was the same as had been tried in a recent study.34
Remifentanil, currently being one of the most used agents in clinical practice of anesthesia, has been reported to have protective effects against I/R injury.35 In a study, cardioprotective effect of remifentanil via alpha and beta receptors had been shown.36 However, there have been no controlled studies for comparison of remifentanil and dex regarding their protective effects. Yang et al37 demonstrated remifentanil’s activity in suppressing inflammatory response and reduction of hepatic I/R injury. As there are reports suggesting the effectiveness of continuous infusion of remifentanil as much as pre-/post-conditioning with it, we also preferred to infuse remifentanil, following anesthesia induction.38 In a clinical study performed in uterine I/R injury, the authors demonstrated remifentanil’s activity in reduction of histopathological lesions and oxidative stress markers.39 This is probably due to the opioid-binding sites in rat kidneys. We also observed that remifentanil decreased necrotic cell number and tubular cell damage when compared with non-treated groups. The effect of remifentanil on apoptotic cell number was not as effective as in the dex-treated group. Additionally, the plasma level of NGAL was as low as in the group not exposed to I/R injury, after both ischemia and reperfusion. However, the histopathological findings and NGAL levels were not as satisfactory as in the group DEX. In our current clinical practice, remifentanil is more commonly used than dex, so its favorable effects are also important for clinicians.
In this study, our last check of the parameters was at the sixth hour after reperfusion, due to high cost of the laboratory tests; thus, we could not follow whether the renoprotective effect continued until the 24th hour or not. However, we believe that our findings might be informative.
Either ischemic or toxic kidney injury has been reported to damage endothelial and tubular epithelial cells of kidneys, leading to interstitial fibrosis and glomerulosclerosis. Therefore, recognizing new effective therapies and the preventive effects of drugs to suppress organ damage during I/R is clinically important.40
We tried to associate the plasma levels of NGAL with the severity of renal I/R injury and renoprotective effects of two agents. Recently, NGAL has been demonstrated as an early biomarker of rapid prediction of renal insufficiency.41 In the perioperative period, this is important, especially in debilitated patients with comorbidities, since early detection and management of AKI treatment deserve particular importance. Sprenkle et al42 reported that NGAL might be a useful marker for detecting renal injury with poorer preoperative renal function and susceptibility to AKI.
In this study, we had an ischemic injury period of 30 minutes, followed by a reperfusion period of 30 minutes. Although there are variabilities in this subject from one study to another, we believe we were in the range reported in the other experimental study.43 Our results demonstrated that remifentanil might reduce the effects of renal I/R injury, but it was not as effective as dex. We used remifentanil at a very low and constant dose in this study, so we could not make an interpretation for differentiation of the response due to different opioid receptors. Remifentanil, used mainly as a continuous infusion agent, may protect against ischemic injury during preconditioning and post-conditioning. We experienced this effect in this study. Thus, this study leads a way toward understanding the effect of remifentanil as a safe opioid. We believe we need new experimental models that will be similar to clinical situations due to the limited studies in this aspect.
Conclusion
Dexmedetomidine has been demonstrated to play an important role in protection against I/R injury in rat kidneys, supported by histological changes and a biochemical marker NGAL. However, other intravenous anesthetic agents that are commonly used should also be investigated for comparison in other studies so that these results might guide the preference for the appropriate anesthetic agent.
Disclosure
The authors report no conflicts of interest in this work.
References
Kanbay M, Kasapoglu B, Perazella MA. Acute tubular necrosis and pre-renal acute kidney injury: utility of urine microscopy in their evaluation- a systematic review. Int Urol Nephrol. 2010;42(2):425–433. | ||
Mishra J, Mori K, Ma Q, Kelly C, Barasch J, Devarajan P. Neutrophil gelatinase-associated lipocalin: a novel early urinary biomarker for cisplatin nephrotoxicity. Am J Nephrol. 2004;24(3):307–315. | ||
Mori K, Nakao K. Neutrophil gelatinase-associated lipocalin as the real-time indicator of active kidney damage. Kidney Int. 2007;71(10):967–970. | ||
Mishra J, Dent C, Tarabishi R, et al. Neutrophil gelatinase-associated lipocalin (NGAL) as a biomarker for acute renal injury after cardiac surgery. Lancet. 2005;365(9466):1231–1238. | ||
Gonzalez F, Vincent F. Biomarkers for acute kidney injury in critically ill patients. Minerva Anestesiol. 2012;78(12):1394–1403. | ||
Soni SS, Pophale R, Ronco C. New biomarkers for acute renal injury. Clin Chem Lab Med. 2011;49(8):1257–1263. | ||
Schmidt-Ott KM, Mori K, Kalandadze A, et al. Neutrophil gelatinase associated lipocalin-mediated iron traffic in kidney epithelia. Curr Opin Nephrol Hypertens. 2006;15(4):442–449. | ||
Kümpers P, Hafer C, Lukasz A, et al. Serum neutrophil gelatinase-associated lipocalin at inception of renal replacement therapy predicts survival in critically ill patients with acute kidney injury. Crit Care. 2010;14(1):R9. | ||
Hall IE, Yarlagadda SG, Coca SG, et al. IL-18 and urinary NGAL predict dialysis and graft recovery after kidney transplantation. J Am Soc Nephrol. 2010;21(1):189–197. | ||
Haase M, Devarajan P, Haase-Fielitz A, et al. The outcome of neutrophil gelatinase-associated lipocalin-positive subclinical acute kidney injury: a multicenter pooled analysis of prospective studies. J Am Coll Cardiol. 2011;57(17):1752–1761. | ||
Moore E, Bellomo R, Nichol A. Biomarkers of acute kidney injury in anesthesia, intensive care and major surgery: from the bench to clinical research to clinical practice. Minerva Anesthesiol. 2010;76(6):425–440. | ||
Gu J, Sun P, Zhao H, et al. Dexmedetomidine provides renoprotection against ischemia-reperfusion injury in mice. Crit Care. 2011;15(3):153. | ||
Afonso J, Reis F. Dexmedetomidine: current role in anesthesia and intensive care. Rev Bras Anestesiol. 2012;62(1):118–133. | ||
Sanders RD, Maze M. Alpha2-adrenoceptor agonists. Curr Opin Investig Drugs. 2007;8(1):25–33. | ||
Taniguchi T, Kidani Y, Kanakura H, Takemoto Y, Yamamoto K. Effects of dexmedetomidine on mortality rate and inflammatory responses to endotoxin-induced shock in rats. Crit Care Med. 2004;32(6):1322–1326. | ||
Memis D, Hekimoglu S, Vatan I, Yandim T, Yüksel M, Süt N. Effects of midazolam and dexmedetomidine on inflammatory responses and gastric intramucosal pH to sepsis, in critically ill patients. Br J Anaesth. 2007;98(4):550–552. | ||
Kim HS, Cho JE, Hong SW, Kim SO, Shim JK, Kwak YL. Remifentanil protects myocardium through activation of anti-apoptotic pathways of survival in ischemia-reperfused rat heart. Physiol Res. 2010;59(3):347–356. | ||
Minguet G, Brichant JF, Joris J. Opioids and protection against ischemia-reperfusion injury: from experimental data to potential clinical applications. Acta Anaesthesiol Belg. 2012;63(1):23–34. | ||
Vianna PT, Castiglia YM, Braz JR, et al. Remifentanil, isoflurane, and preconditioning attenuate renal ischemia/reperfusion injury in rats. Transplant Proc. 2009;41(10):4080–4082. | ||
Azevedo VL, Santos PS, Oliveira GS Jr, et al. The effect of 6% hydroxyethyl starch vs. Ringer’s lactate on acute kidney injury after renal ischemia in rats. Acta Cir Bras. 2013;28(1):5–9. | ||
Devabhakthuni S, Pajoumand M, Williams C, Kufera JA, Watson K, Stein DM. Evaluation of dexmedetomidine: safety and clinical outcomes in critically ill trauma patients. J Trauma. 2011;71(5):1164–1171. | ||
Billings FT 4th, Chen SW, Kim M, et al. alpha2-Adrenergic agonists protect against radio contrast-induced nephropathy in mice. Am J Physiol Renal Physiol. 2008;295(3):741–748. | ||
Kiliç K, Hanci V, Selek S, et al. The effects of dexmedetomidine on mesenteric arterial occlusion-associated gut ischemia and reperfusion-induced gut and kidney injury in rabbits. J Surg Res. 2012;178(1):223–232. | ||
Villela NR, do Nascimento Junior P, de Carvalho LR, Teixeira A. Effects of dexmedetomidine on renal system and on vasopressin plasma levels. Experimental study in dogs. Rev Bras Anestesiol. 2005;55(4):429–440. | ||
Leino K, Hynynen M, Jalonen J, et al; Dexmedetomidine in Cardiac Surgery Study Group. Renal effects of dexmedetomidine during coronary artery bypass surgery: a randomized placebo-controlled study. BMC Anesthesiol. 2011;11:9. | ||
Bayram A, Esmaoglu A, Akin A, et al. The effects of intraoperative infusion of dexmedetomidine on early renal function after percutaneous nephrolithotomy. Acta Anaesthesiol Scand. 2011;55(5):539–544. | ||
Gu J, Chen J, Xia P, Tao G, Zhao H, Ma D. Dexmedetomidine attenuates remote lung injury induced by renal ischemia-reperfusion in mice. Acta Anaesthesiol Scand. 2011;55(10):1272–1278. | ||
Sugita S, Okabe T, Sakamoto A. Continuous infusion of dexmedetomidine improves renal ischemia-reperfusion injury in rat kidney. J Nippon Med Sch. 2013;80(2):131–139. | ||
Kocoglu H, Ozturk H, Ozturk H, Yilmaz F, Gulcu N. Effect of dexmedetomidine on ischemia-reperfusion injury in rat kidney: a histopathologic study. Ren Fail. 2009;31(1):70–74. | ||
Si Y, Bao H, Han L, et al. Dexmedetomidine protects against renal ischemia and reperfusion injury by inhibiting the JAK/STAT signaling activation. J Transl Med. 2013;11:141. | ||
Gonullu E, Ozkardesler S, Kume T, et al. Comparison of the effects of dexmedetomidine administered at two different times on renal ischemia/reperfusion injury in rats. Braz J Anesthesiol. 2014;64(3):152–158. | ||
Engelhard K, Werner C, Eberspächer E, et al. The effect of the alpha 2-agonist dexmedetomidine and the N-methyl-D-aspartate antagonist S(+)-ketamine on the expression of apoptosis regulating proteins after incomplete cerebral ischemia and reperfusion in rats. Anesth Analg. 2003;96(2):524–531. | ||
Ji F, Li Z, Nguyen H, et al. Perioperative dexmedetomidine improves outcomes of cardiac surgery. Circulation. 2013;127(15):1576–1584. | ||
Kulka PJ, Tryba M, Zenz M. Preoperative alpha2-adrenergic receptor agonists prevent the deterioration of renal function after cardiac surgery: results of a randomized, controlled trial. Crit Care Med. 1996;24(6):947–952. | ||
Cakir M, Polat A, Tekin S, et al. The effect of dexmedetomidine against oxidative and tubular damage induced by renal ischemia reperfusion in rats. Ren Fail. 2015;37(4):704–708. | ||
de Carvalho AL, Vital RB, Kakuda CM, et al. Dexmedetomidine on renal ischemia-reperfusion injury in rats: assessment by means of NGAL and histology. Ren Fail. 2015;37(3):526–530. | ||
Yang LQ, Tao KM, Liu YT, et al. Remifentanil preconditioning reduces hepatic ischemia-reperfusion injury in rats via inducible nitric oxide synthase expression. Anesthesiology. 2011;114(5):1036–1047. | ||
Chun KJ, Park YH, Kim JS, et al. Comparison of 5 different remifentanil strategies against myocardial ischemia-reperfusion injury. J Cardiothorac Vasc Anesth. 2011;25(6):926–930. | ||
Atalay YO, Aktas S, Sahin S, Kucukodaci Z, Ozakpinar OB. Remifentanil protects uterus against ischemia-reperfusion injury in rats. Acta Cir Bras. 2015;30(11):756–761. | ||
Ranganathan P, Jayakumar C, Ramesh G. Proximal tubule-specific overexpression of netrin-1 suppresses acute kidney injury-induced interstitial fibrosis and glomerulosclerosis through suppression of IL-6/STAT3 signaling. Am J Physiol Renal Physiol. 2013;304(8):F1054–F1065. | ||
Medic B, Rovcanin B, Vujovic KS, Obradovic D, Duric D, Prostran M. Evaluation of novel biomarkers of acute kidney injury: the possibilities and limitations. Curr Med Chem. 2016;23(19):1981–1997. | ||
Sprenkle PC, Wren J, Maschino AC, et al. Urine neutrophil gelatinase-associated lipocalin as a marker of acute kidney injury after kidney surgery. J Urol. 2013;190(1):159–164. | ||
Punuru P, Sujatha D, Kumari BP, Charisma VV. Evaluation of aqueous extract of Murraya koenigii in unilateral renal ischemia reperfusion injury in rats. Indian J Pharmacol. 2014;46(2):171–175. |
 © 2017 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2017 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.


