Back to Journals » Infection and Drug Resistance » Volume 12
Differences in microbial etiology between hospital-acquired pneumonia and ventilator-associated pneumonia: a single-center retrospective study in Guang Zhou
Authors Feng DY , Zhou YQ, Zou XL , Zhou M , Zhu JX, Wang YH , Zhang TT
Received 9 February 2019
Accepted for publication 9 April 2019
Published 29 April 2019 Volume 2019:12 Pages 993—1000
DOI https://doi.org/10.2147/IDR.S204671
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Joachim Wink
Ding-Yun Feng,1,* Yu-Qi Zhou,1,* Xiao-Ling Zou,1 Mi Zhou,2 Jia-Xin Zhu,1 Yan-Hong Wang,1 Tian-Tuo Zhang1
1Department of Pulmonary and Critical Care Medicine, Third Affiliated Hospital of Sun Yat-sen University, Institute of Respiratory Diseases of Sun Yat-Sen University, Guangzhou, People’s Republic of China; 2Department of Surgery Intensive Care Unit, Third Affiliated Hospital of Sun Yat-sen University, Guangzhou, People’s Republic of China
*These authors contributed equally to this work
Purpose: Nosocomial pneumonia is a common nosocomial infection that includes hospital-acquired pneumonia (HAP) and ventilator-associated pneumonia(VAP). It is an important cause of morbidity and mortality in hospitalized patients. This study aimed to evaluate the differences in microbial etiology and outcomes between HAP and VAP, particularly in related risk factors of multidrug-resistant organism (MDRO) causing HAP and VAP.
Patients and methods: This single-center retrospective, observational study included patients with HAP/VAP. Clinical and epidemiological data of nosocomial pneumonia confirmed by microbial etiology that occurred in the Third Affiliated Hospital of Sun Yat-sen University, China, from January 2014 to December 2017 were obtained.
Results: A total of 313 HAP cases and 106 VAP cases were included. The leading pathogens of HAP and VAP were similar, including Acinetobacter baumannii, Pseudomonas aeruginosa, and Klebsiella pneumoniae. Antimicrobial susceptibility of the pathogens was low, and P. aeruginosa in VAP was less susceptible. In the multivariate logistic regression analysis, the risk factors associated with MDRO-HAP were chronic obstructive pulmonary disease, antibiotic therapy in the preceding 90 days, and prior endotracheal tracheostomy. The risk factor of MDRO-VAP was ≥5 days of hospitalization. The 30-day mortality rates of HAP and VAP were 18.5% and 42.5%.
Conclusion: The leading pathogens were similar in both HAP and VAP, and antimicrobial susceptibility of the pathogens was low. The risk factors associated with MDRO infection in HAP and VAP have significant variability; hence, attention should be paid to improve prognosis. VAP was associated with poorer outcomes compared with HAP.
Keywords: hospital-acquired pneumonia, ventilator-associated pneumonia, epidemiology, microbial etiology
Introduction
Nosocomial pneumonia (NP), including hospital-acquired pneumonia (HAP) and ventilator-associated pneumonia (VAP), is an important cause of morbidity and mortality in hospitalized patients despite advances in antimicrobial therapy and better supportive care modalities.1,2 Comparing the 2005 version guideline with the 2016 version guideline of the Infectious Diseases Society of America and the American Thoracic Society, HAP had different definitions; in 2016 version, it emphasized that HAP was not associated with mechanical ventilation (MV), but remained a confusing area.3,4 VAP is related to the duration of MV, intensive care unit (ICU) and hospital length of stay, and healthcare costs.2 This demands a focus on the difference between HAP and VAP characteristics and prognosis. Clinical practice guidelines developed by international societies need to be tailored according to local epidemiology.5 Meanwhile,bacterial infection is the main cause of NP, and multidrug-resistant organism(MDRO) has appeared in recent years;hence, the risk factors of MDRO must be identified to facilitate the accurate prescription of antibiotics and improve overall prognosis in cases of NP.6,7 This study aimed to evaluate the differences in microbial etiology between HAP and VAP, especially in related risk factors and prognosis caused by MDRO.
Material and methods
This single-center retrospective, observational study included patients with HAP/VAP. Data on all episodes of NP that occurred in the Third Affiliated Hospital of Sun Yat-sen University, Guangdong, China, from January 2014 to December 2017 were collected. All patients were at least 18 years old. The criteria for a NPdiagnosis4 includedthe presence of a new pulmonary infiltrate acquired in the hospital with two or more of the following: temperature >38.3 °C or <36 °C, leukocyte count >10×109/L or <4×109/L, and the presence of purulent respiratory secretions. HAP was defined as pneumonia that occurred ≥48h after admission, with no association with MV. VAP was defined as pneumonia that occurred 48–72 h after the intubation and initiation of MV. All cases were confirmed by microbial etiology. Cases with acquired immunodeficiency syndrome and those missing key data were excluded.
The simplified version of the clinical pulmonary infection score (CPIS), which determined temperature, blood leukocytes, tracheal secretions, PaO2/FiO2, and chest radiograph to assess the severity of NP, was used.
A microbiological strategy was then followed for the establishment of diagnosis: lower respiratory tract samples were obtained by bronchoalveolar lavage, endotracheal aspiration,or adequate sputum culture with WBCs >25 cells/HPF and epithelial cells <10 cells on Gram stain. Pathogenic bacteria were further characterized by conventional biochemical tests to identify the specific strain using standard microbiological methods. Pathogenic organism susceptibility testing was performed using the microdilution method (Micro ScanSystem; Baxter Healthcare, West Sacramento, CA, USA), and the results were interpreted using the National Committee for Clinical Laboratory Standards guidelines published in 2012 (Clinical & Laboratory Standards Institute, 2012). Here, MDROs were defined as organisms that were resistant to at least one agent from each of the three or more antimicrobial categories in susceptibility tests of isolates from patients with NP.8 In the statistical analysis, the parametric data were reported as frequency, percentage, mean value, and standard deviation, while nonparametric data were reported as frequency and percentage. Simple logistic regression analysis was performed, and each independent variable was analyzed together with the dependent variable of NP. These were reported as the P-value and odds ratio (OR). The accepted level of significance was P<0.05. Statistical analysis was performed using IBM SPSS Statistics, version 20 (IBM Corp., Armonk, New York, America).
Results
A total of 1,158 patients had HAP, and only 313 cases were included because 556 patients did not undergo bacterial culture and 289 patients had a negative bacterial culture result. Among the 2,901 inpatients under MV, 106 patients had VAP, and they were included. By comparing the characteristics between HAP and VAP, we found that less VAP patients were aged >70years, and VAP patients had more antibiotic therapy in the preceding 90 days and prior endotracheal tracheostomy (Table 1). The leading pathogens were similar in both kinds of NP, including Acinetobacter baumannii, Pseudomonas aeruginosa, and Klebsiella pneumoniae, but A.baumanniiwas significantly abundant in VAP. K.pneumonia and Staphylococcus aureus were significantly predominant in HAP (Table 2). Of these, VAP had more MDROs (Table 1). Moreover, the antimicrobial susceptibility of A.baumannii, K. pneumonia and Staphylococcus aureus was similar in both groups (Table 3, Table 4). However, P. aeruginosa causing VAP was less susceptible than P. aeruginosa causing HAP (Table 3). A CPIS ≥6 was predominant in the VAP group. The 30-day mortality rates of HAP and VAP were 18.5% and 42.5%, respectively (Table 1).
 | Table 1 The characteristics difference between HAP and VAP |
 | Table 2 Distribution of pathogens between HAP and VAP |
 | Table 3 The antimicrobial susceptibility differences between HAP and VAP |
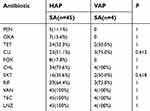 | Table 4 Antimicrobial susceptibility among Staphylococcus aureus of HAP and VAP |
To determine the distinction between HAP and VAP, this study analyzed the risk factors for MDRO-HAP and MDRO-VAP. The first logistic regression analysis identified the following risk factors for MDRO-HAP: chronic obstructive pulmonary disease (COPD), antibiotic therapy in the preceding 90 days, prior endotracheal tracheostomy, stomach tube intubation, and elevated blood urea nitrogen (BUN) level. Meanwhile, only ≥5 days of hospitalization and albumin level were considered the risk factors for MDRO-VAP. In the second multivariate analysis, COPD (P=0.023; OR, 3.006; 95% confidence interval [CI], 1.166–7.751), antibiotic therapy in the preceding 90 days (P=0.02; OR, 1.843; 95% CI, 1.102–3.081), and prior endotracheal tracheostomy (P=0.033; OR, 1.932; 95% CI, 1.055–3.539) were still associated with MDRO-HAP (Table 5). Furthermore, only≥5 days of hospitalization (P=0.01; OR, 0.021; 95% CI, 0.065–0.685) was associated with MDRO-VAP (Table 6).
 | Table 5 Significant univariate and multivariate logistic regression analyses of risk factors associated with HAP |
 | Table 6 Significant univariate and multivariate logistic regression analyses of risk factors associated with VAP |
Discussion
In this study, we found that less VAP patients had age >70years. Although it was similar to HAP in that VAP incidence was lower in the elderly,9 the causes of these observations in the elderly are imperfectly understood. Survival bias might be a factor, that is, because of the elderly’s comorbid conditions, they died before or during VAP acquisition.10,11 VAP patients had to undergo more antibiotic therapy in the preceding 90 days, because this kind of patients had comorbidities requiring antibiotic therapy or preventive medication before MV. The patients who had prior endotracheal tracheostomy were more likely to have VAP in the current study; this is because they could easily be exposed to bacterial colonization,12 and changes to the airway structure are a further risk factor. As VAP often occurred in the ICU, it suggested that patients were in critical condition and frequently required stomach tube intubation and central venous catheterization. This was consistent with our research. We noted that the BUN level of VAP patients was higher than that of HAP patients. Because patients with pneumonia often had dehydrated status resulting in the increased reabsorption of urea by the kidneys, elevation of BUN level is frequently observed,13,14 especially with VAP.
In the current study, the leading pathogens were similar in both kinds of NP, with a predominance of A. baumannii, P. aeruginosa, and K. pneumonia. But A.baumannii was significantly abundant in VAP, and K. pneumonia and S. aureus were commonly predominant in HAP. This is similar to the findings reported previously.4,15,16 It suggested that Gram-negative bacteria were the major NP pathogens in our center. This may be attributed to the warmer climate in our city, increased prevalence of organisms in the environment, and the higher prevalence of Gram-negative bacterial infections, especially A.baumannii infection in VAP, in the developing world.17Although S. aureus is the most common Gram-positive pathogen of NP,18,19 it was still inferior to Gram-negative bacteria, especially in VAP. Our study demonstrated that VAP had more MDROs. Additionally, antimicrobial susceptibility of P. aeruginosa causing VAP was lower than that causing HAP, as in previous reports.20,21 Meanwhile, this study found a higher rate of A.baumannii resistant to meropenem in these populations (82.7% in the HAP group and 78% in the VAP group).Similar resistance rates(79.5%–92.5%) to carbapenems were observed in previous studies of other hospitals,22,23 indicating that MDRO infection remains a challenging aspect of NP etiology.
In this study, we assessed the risk factors associated with MDRO-NP. COPD was associated with MDRO-HAP because acute exacerbation of COPD usually requires antibiotic treatment.24,25 Moreover, studies have shown that the colonization rates of Gram-negative bacteria such as P. aeruginosa and A. baumannii were higher in patients with COPD; therefore, it is easier for them to be repeatedly infected.26,27 Antibiotics are crucial intreating infectious diseases including HAP. However, antibiotic resistance has increased, and the rise of MDR bacteria is threatening human health.28 Antibiotic therapy in the preceding 90 days was an important risk factor for MDRO-HAP in the present study. This finding is similar to that of the previous studies that suggested the impact of virulence and antibiotic tolerance.24,29 Therefore, among patients with HAP who have been exposed to antibiotics, MDRO should be targeted to control the condition effectively and improve prognosis. Tracheotomy was also likely to lead to MDRO infection. This is because patients who had required MV or undergone tracheotomy can easily be exposed to MDRO colonization.12 ICU admission or broad-spectrum antibiotic use also contributes to MDRO infection,30 and changes to the airway structure are a further risk factor. Meanwhile, in our hospital, the rehabilitation department is famous in Guang Zhou, and there are many patients with tracheotomy coming to that department for rehabilitation treatment. Hence, tracheotomy became a risk factor of MDRO-HAP. The risk factor for MDRO with HAP or VAP was ≥5 days of hospitalization.4,31 In this study, it was similar to VAP but not HAP, because VAP patients had long hospitalization, were easily exposed to antibiotics, and had induced bacterial resistance. In other words, the risk factors for MDRO in HAP and VAP were not the same.
The 30-day mortality rates of HAP and VAP were 18.5% and 42.5%, respectively, in this study. It was consistent with previous reports.32,33 The reason why VAP had higher mortality rate was that the patients often had severe infection34 or morbid state.35 CPIS is a vital tool that evaluates the severity of pulmonary infection, and this score is positively correlated with the severity of pulmonary infection and prognosis.36,37 The present study results support this finding, indicating an area for future research.
There are several limitations in our study. First, the sample size of this study was small, resulting in sparse data bias. Second, all clinical information related to the risk factors was collected retrospectively, which limits the generalization of our findings. Further prospective studies are warranted to reduce bias in the future.
Conclusion
HAP and VAP had similar pathogens, but antimicrobial susceptibility of VAP was lower, especially with P. aeruginosa. The risk factors associated with MDRO infection in HAP and VAP were different. VAP was associated with poorer outcomes compared with HAP.
Ethics approval and informed consent
The study has been approved by the institutional review board of the hospital and the ethics committee of the third affiliated hospital of Sun Yat-sen University. The patient consent was written informed consent and their information has been kept confidential.
Data availability
The data used to support the findings of this study are available from the corresponding author upon request.
Abbreviation list
NP, Nosocomial pneumonia; HAP, hospital-acquired pneumonia; VAP, ventilator-associated pneumonia; MV, mechanical ventilation; ICU, intensive care unit; MDRO, multidrug-resistant organism; OR, odds ratio; COPD, chronic obstructive pulmonary disease; BUN, blood urea nitrogen; CPIS, clinical pulmonary infection score; CI, 95% confidence interval.
Acknowledgment
We thank Dr He-Liang Li, Jia-Sheng He, Yi-Xiong Yuan for their help for data collection of this study.
Author contributions
All authors contributed toward data analysis, drafting and critically revising the paper, gave final approval of the version to be published, and agree to be accountable for all aspects of the work.
Disclosure
The authors have no conflicts of interest to disclose in this work.
References
1. Tao XB, Qian LH, Li Y, et al. Hospital-acquired infection rate in a tertiary care teaching hospital in China: a cross-sectional survey involving 2434 inpatients. Int J Infect Dis. 2014;27:7–9. doi:10.1016/j.ijid.2014.05.011
2. Vincent JL, de Souza Barros D, Cianferoni S. Diagnosis, management and prevention of ventilator-associated pneumonia: an update. Drugs. 2010;70:1927–1944. doi:10.2165/11538080?000000000?00000]
3.
4. Kalil AC, Metersky ML, Klompas M, et al. Management of adults with hospital-acquired and ventilator-associated pneumonia: 2016 clinical practice guidelines by the Infectious Diseases Society of America and the American Thoracic Society. Clin Infect Dis. 2016;63:e61–111. doi:10.1093/cid/ciw353
5. Awad LS, Abdallah DI, Mugharbil AM, et al. An antibiotic stewardship exercise in the ICU: building a treatment algorithm for the management of ventilator-associated pneumonia based on local epidemiology and the 2016 Infectious Diseases Society of America/American Thoracic Society guidelines. Infect Drug Resist. 2017;11:17–28. doi:10.2147/IDR.S145827 eCollection 2018.
6. Kim T, Chong YP, Park SY, et al. Risk factors for hospital-acquired pneumonia caused by carbapenem-resistant gram-negative bacteria in critically ill patients: A multicenter study in Korea. Diagn Microbiol Infect Dis. 2014;78(4):457–461. doi:10.1016/j.diagmicrobio.2013.08.011
7. Vallés J, Martin-Loeches I, Torres A, et al. Epidemiology, antibiotic therapy and clinical outcomes of healthcare-associated pneumonia in critically ill patients: a Spanish cohort study. Intensive Care Med. 2014;40:572–581. doi:10.1007/s00134-014-3239-2
8. Magiorakos AP, Srinivasan A, Carey RB, et al. Multidrug-resistant, extensively drug-resistant and pandrug-resistant bacteria: an international expert proposal for interim standard definitions for acquired resistance. Clin Microbiol Infect. 2012;18(3):268–281. doi:10.1111/j.1469-0691.2011.03570.x
9. Dananché C, Vanhems P, Machut A, et al. Healthcare-Associated Infections (HAIs) Surveillance Network of ICUs (Réseau REA-Raisin). Trends of incidence and risk factors of ventilator-associated pneumonia in elderly patients admitted to French ICUs between 2007 and 2014. Crit Care Med. 2018;46(6):869–877. doi:10.1097/CCM.0000000000003019
10. Dimopoulos G, Koulenti D, Blot S, et al. Extended prevalence of infection in intensive care study investigators: critically ill elderly adults with infection: analysis of the extended prevalence of infection in intensive care study. J Am Geriatr Soc. 2013;61:2065–2071. doi:10.1111/jgs.12544
11. Bagshaw SM, Webb SA, Delaney A, et al. Very old patients admitted to intensive care in Australia and New Zealand: A multi-centre cohort analysis. Crit Care. 2009;13:R45. doi:10.1186/cc7768
12. Thom KA, Rock C, Jackson SS, et al. Factors leading to transmission risk of Acinetobacter baumannii. Crit Care Med. 2017;45:e633–9. doi:10.1097/CCM.0000000000002318
13. Woodford-Williams E. Respiratory tract disease. Diagnosis and management of pneumonia in the aged. Br Med J. 1966;1:467–470. doi:10.1136/bmj.1.5485.467
14. Kasper DL, Fauci AS, Longo DL, editors. Harrison’s Principles of Internal Medicine.
15. Resende MM, Monteiro SG, Callegari B, Figueiredo PM, Monteiro CR, Monteiro-Neto V. Epidemiology and outcomes of ventilator-associated pneumonia in northern Brazil: an analytical descriptive prospective cohort study. BMC Infect Dis. 2013;13:119. doi:10.1186/1471-2334-13-119
16. Pássaro L, Harbarth S, Landelle C. Prevention of hospital-acquired pneumonia in non-ventilated adult patients: a narrative review. Antimicrob Resist Infect Control. 2016;5:43. eCollection 2016. doi:10.1186/s13756-016-0150-3
17. Reechaipichitkul W, Phondongnok S, Bourpoern J, Chaimanee P. Causative agents and resistance among hospital-acquired and ventilator-associated pneumonia patients at Srinagarind hospital, northeastern Thailand. Southeast Asian J Trop Med Public Health. 2013;44:490–502.
18. Chastre J, Fagon JY. Ventilator-associated pneumonia. Am J Respir Crit Care Med. 2002;165:867–903. doi:10.1164/ajrccm.165.8.2106104
19. Naidus EL, Lasalvia MT, Marcantonio ER, Herzig SJ. The diagnostic yield of noninvasive microbiologic sputum sampling in a cohort of patients with clinically diagnosed hospital-acquired pneumonia. J Hosp Med. 2018;13(1):34–37. doi:10.12788/jhm.2868
20. Djordjevic ZM, Folic MM, Jankovic SM. Distribution and antibiotic susceptibility of pathogens isolated from adults with hospital-acquired and ventilator-associated pneumonia in intensive care unit. J Infect Public Health. 2017;10(6):740–744. doi:10.1016/j.jiph.2016.11.016
21. Zhao T, Liu Y, Cao B, et al. Prospective multicenter study of pathogen distributions in early-onset and late-onset hospital-acquired pneumonia in China. Antimicrob Agents Chemother. 2013;57:6404–6405. doi:10.1128/AAC.01539-13
22. Biedenbach DJ, Giao PT, Hung Van P, et al. Antimicrobial-resistant pseudomonas aeruginosa and acinetobacter baumannii from patients with hospital-acquired or ventilator-associated pneumonia in Vietnam. Clin Ther. 2016;38(9):2098–2105. doi:10.1016/j.clinthera.2016.07.172
23. Ju M, Hou D, Chen S, et al. Risk factors for mortality in ICU patients with Acinetobacter baumannii ventilator-associated pneumonia: impact of bacterial cytotoxicity. J Thorac Dis. 2018;10(5):2608–2617. doi:10.21037/jtd.2018.04.86
24. Nseir S, Ader F. Prevalence and outcome of severe chronic obstructive pulmonary disease exacerbations caused by multidrug-resistant bacteria. Curr Opin Pulm Med. 2008;14:95–100. doi:10.1097/MCP.0b013e3282f37a11
25. Seligman R
26. Gallego M, Pomares X, Espasa M, et al. Pseudomonas aeruginosa isolates in severe chronic obstructive pulmonary disease: characterization and risk factors. BMC Pulm Med. 2014;14:103. doi:10.1186/1471-2466-14-103
27. Grochowalska A, Kozioł-Montewka M, Sobieszczańska A. Analysis of Acinetobacter baumannii resistance patterns in patients with chronic obstructive pulmonary disease (COPD) in terms of choice of effective empiric antibiotic therapy. Ann Agric Environ Med. 2017;24:307–311. doi:10.26444/aaem/74710
28. Zaman SB, Hussain MA, Nye R, Mehta V, Mamun KT, Hossain N. A review on antibiotic resistance: alarm bells are ringing. Cureus. 2017;9(6):e1403. doi:10.7759/cureus.1403
29. Raman G, Avendano E, Berger S, Menon V. Appropriate initial antibiotic therapy in hospitalized patients with gram-negative infections: systematic review and meta-analysis. BMC Infect Dis. 2015;15:395. doi:10.1186/s12879-015-1123-5
30. Ang H, Sun X. Risk factors for multidrug-resistant Gram-negative bacteria infection in intensive care units: A meta-analysis. Int J Nurs Pract. 2018;e12644. doi:http://dx.doi.org/10.1111/ijn.12644
31. Ekren PK, Ranzani OT, Ceccato A, et al. Evaluation of the 2016 infectious diseases society of america/american thoracic society guideline criteria for risk of multidrug-resistant pathogens in patients with hospital-acquired and ventilator-associated pneumonia in the ICU. Am J Respir Crit Care Med. 2018;197(6):826–830. doi:10.1164/rccm.201708-1717LE
32. Ewan VC, Sails AD, Walls AW, Rushton S, Newton JL. Dental and microbiological risk factors for hospital-acquired pneumonia in non-ventilated older patients. PLoS One. 2015;10:e0123622. doi:10.1371/journal.pone.0123622
33. Ding C, Zhang Y, Yang Z, et al. Incidence, temporal trend and factors associated with ventilator-associated pneumonia in mainland China: a systematic review and meta-analysis. BMC Infect Dis. 2017;17(1):468. doi:10.1186/s12879-017-2566-7
34. Da Silveira F, Nedel WL, Cassol R, Pereira PR, Deutschendorf C, Lisboa T. Acinetobacter etiology respiratory tract infections associated with mechanical ventilation: what impacts on the prognosis? A retrospective cohort study. J Crit Care. 2019;49:124–128. doi:10.1016/j.jcrc.2018.10.034
35. Siniscalchi A, Aurini L, Benini B, et al. Ventilator associated pneumonia following liver transplantation: etiology, risk factors and outcome. World J Transplant. 2016;6(2):389–395. doi:10.5500/wjt.v6.i2.389
36. Esperatti M, Ferrer M, Giunta V, et al. Validation of predictors of adverse outcomes in hospital-acquired pneumonia in the ICU. Crit Care Med. 2013;41:2151–2161. doi:10.1097/CCM.0b013e31829133d6
37. Özvatan T, Akalın H, Sınırtaş M, et al. Nosocomial Acinetobacter pneumonia: treatment and prognostic factors in 356 cases. Respirology. 2016;21(2):363–369. doi:10.1111/resp.12698
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
