Back to Journals » Infection and Drug Resistance » Volume 16
Diagnosis and Management of Drug-Induced Interstitial Lung Disease Associated with Amikacin Liposome Inhalation Suspension in Refractory Mycobacterium Avium Complex Pulmonary Disease: A Case Report
Authors Hashimoto K, Nii T, Sumitani H, Yokoyama M, Miyamoto S, Mihashi Y, Nagata Y, Matsuki T, Tsujino K, Miki K , Kida H
Received 26 June 2023
Accepted for publication 27 September 2023
Published 10 October 2023 Volume 2023:16 Pages 6629—6634
DOI https://doi.org/10.2147/IDR.S427544
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Professor Suresh Antony
Kazuki Hashimoto, Takuro Nii, Hitoshi Sumitani, Masashi Yokoyama, Satoshi Miyamoto, Yasuhiro Mihashi, Yuka Nagata, Takanori Matsuki, Kazuyuki Tsujino, Keisuke Miki, Hiroshi Kida
Department of Respiratory Medicine, National Hospital Organization Osaka Toneyama Medical Center, Osaka, Japan
Correspondence: Takuro Nii, Department of Respiratory Medicine, National Hospital Organization Osaka Toneyama Medical Center, 5-1-1 Toneyama, Toyonaka, Osaka, 560-8552, Japan, Tel +81-6-6853-2001, Fax +81-6-6853-3127, Email [email protected]
Abstract: Amikacin liposome inhalation suspension (ALIS) is a key drug for the treatment of refractory Mycobacterium avium complex pulmonary disease (MAC-PD). Although cases of drug-induced interstitial lung disease (DIILD) by ALIS have been reported, its diagnosis is challenging due to overlapping existing pulmonary shadows, airway bleeding, exacerbation of underlying conditions, and the potential for various concurrent infections. A 72-year-old woman started treatment with ALIS for refractory MAC-PD. Three weeks later, she had a fever, cough, and appetite loss. She was hospitalized because multiple infiltrative opacities were observed on chest X-ray and chest computed tomography. Because the opacities worsened after empiric antibiotic therapy with broad-spectrum antibiotics, we initiated corticosteroid therapy, suspecting DIILD caused by ALIS, although drug lymphocyte stimulation tests for ALIS and amikacin were negative. Three days later, we found signs of improvement and quickly tapered the corticosteroids. After obtaining informed consent, we performed a drug provocation test of ALIS. Seven days later, she exhibited fever, an increased peripheral white blood cell count, and elevated serum C-reactive protein level, all of which returned to baseline 4 days after stopping ALIS, leading to a diagnosis of DIILD caused by ALIS in this patient. DIILD caused by ALIS is rare but should be carefully diagnosed to ensure that patients with refractory MAC-PD do not miss the opportunity to receive ALIS treatment.
Keywords: amikacin liposome inhalation suspension, Mycobacterium avium complex pulmonary disease, drug-induced interstitial lung disease, drug provocation test, paradoxical response
Introduction
During the chronic course of refractory nontuberculous mycobacterial pulmonary disease (NTM-PD), it is sometimes exacerbated with the appearance of various opacities on chest radiography.1 A differential diagnosis in such cases is challenging because it includes various conditions such as expansion of the NTM-PD lesion, pneumonia caused by other bacteria or fungi,2 airway bleeding,3 and interstitial pneumonia, including idiopathic4 or drug-induced interstitial lung disease (DIILD).5–7
DIILD is caused by dose-dependent toxicity or immune-mediated inflammation. The diagnosis of DIILD is based on clinical findings consistent with ILD: a temporal relationship between the onset of symptoms and drug exposure; the exclusion of other possible causes such as infection, pulmonary edema, and radiation-induced lung injury; progression of the underlying disease; improvement upon withdrawal of the suspected causative agent with or without corticosteroid therapy; and, in some cases, deterioration upon re-challenge.8 DIILD caused by drugs constituting guideline-based therapy in the treatment of NTM-PD has been rarely reported.5–7
Amikacin liposome inhalation suspension (ALIS) was recently developed for the treatment of refractory Mycobacterium avium complex pulmonary disease (MAC-PD). Adding ALIS to the guideline-based therapy improved the rate of culture conversion among patients with refractory MAC-PD in a Phase III randomized controlled trial.9 In that trial, 3% of participants who received ALIS experienced hypersensitivity pneumonitis.9,10 However, descriptions of this adverse event have been limited, with only two case reports describing the clinical and imaging course of the event.11,12
In this case report, we describe ground-glass opacity in a patient with refractory MAC-PD that was discovered 3 weeks after starting ALIS, which improved upon the withdrawal of ALIS with corticosteroid therapy. ALIS therapy was re-started, and the patient exhibited a deterioration after 1 week of treatment.
Case Report
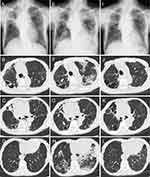
A 72-year-old woman (height 148 cm, body weight 33 kg) was referred to our hospital for the treatment of refractory Mycobacterium intracellulare pulmonary disease, which was diagnosed when she was 60 years old. She had been under observation without treatment until 4 years ago, when a cavitary lung nodule was detected. In the past 4 years, the patient has repeatedly received macrolide-containing multidrug therapies; however, they were not successful. Chest X-ray and chest computed tomography on the first visit to our hospital showed that the patient had fibrocavitary-type pulmonary disease (Figure 1A–D). She had a history of pulmonary tuberculosis. Although macrolide-resistant M. intracellulare was recently cultured from her sputum, the minimum inhibitory concentrations of clarithromycin and amikacin were >32 μg/mL and 8 μg/mL, respectively, clarithromycin was continued at an immunomodulatory dose (400 mg/day). Ethambutol (500 mg/day) and moxifloxacin (400 mg/day) were continued, and ALIS (590 mg/day) was added to these drugs.
Three weeks later, she had a fever, cough, and appetite loss. Chest X-ray and chest computed tomography revealed multiple infiltrative opacities in the left upper lobe and bilateral lower lobes (Figure 1E–H). ALIS was stopped, although the cavity wall and bronchial wall thicknesses decreased, suggesting that ALIS had a high likelihood of being effective against M. intracellulare-pulmonary disease in this patient (Figure 1F and G). A nose swab polymerase-chain reaction test for coronavirus disease 2019 (COVID-19) was negative. Serum (1,3)-beta-
ALIS was the key drug affecting the long-term outcome for this patient; therefore, we performed a drug provocation test of ALIS after obtaining informed consent. Before starting the drug provocation test, the patient’s body temperature was normal and her white blood cell count in peripheral blood was 5990/μL. Her serum C-reactive protein (CRP) concentration was 0.76 mg/dL. Seven days after ALIS re-challenge, the patient’s fever returned. The white blood cell count and CRP concentration increased to 7910/μL (normal range: 4500–11,000 cells/μL) and 5.08 mg/dL (normal range: 0–0.14mg/dL), respectively. Furthermore, her body temperature, white blood cell count, and CRP concentration returned to baseline 4 days after stopping ALIS. All these findings supported a diagnosis of DIILD induced by ALIS in this patient.
Two years later she had experienced four hospitalizations. One was due to COVID-19 infection and the other three were related to fevers requiring intravenous antibiotic therapy. Currently, the patient exhibits shortness of breath attributed to a decrease in lower limb muscle strength and restrictive ventilatory impairment. Nevertheless, the patient continues to attend outpatient appointments to maintain the multi-drug chemotherapy for her refractory MAC-PD.
Discussion
During the disease course of refractory NTM-PD, patients experience several clinical and radiographic exacerbations. Sometimes, the exacerbation is caused by NTM-PD itself; however, it is frequently caused by other complications, including viral, fungal, and bacterial pneumonia; airway bleeding; and DIILD. For physicians treating NTM-PD, DIILD has not been a significant concern, because the frequency of DIILD caused by antimicrobial agents, including anti-tuberculous drugs, was quite low.13 However, differential diagnosis has become more challenging since ALIS became available for use in clinical settings. In this case, it required 8 days to start steroid therapy after carefully excluding COVID-19 and bacterial and fungal pneumonia (Figure 2).
For a diagnosis of DIILD caused by ALIS, it is important to avoid ambiguity as much as possible because patients have very limited other treatment options and may lose the benefit of ALIS administration in the future, once diagnosed with DIILD by ALIS. Recently, a case of DIILD caused by ALIS was reported, in which a transbronchial lung biopsy sample showed findings of organizing pneumonia.12 The accumulation of such studies may reveal histological findings specific to DIILD caused by ALIS. However, there is a concern regarding the spread of pathogens through the airways during bronchoscopy, especially in patients with conditions such as cavitary lesions, where a high bacterial burden is anticipated. In our case, the patient presented with cavitary lesions and was additionally undergoing oxygen therapy via a nasal cannula. Therefore, bronchoscopy was deemed risky and not performed.
The possibility of a paradoxical response, which has been defined among patients with tuberculosis but never reported among patients with NTM-PD, could not be completely excluded in this case. Paradoxical response describes the transient worsening of tuberculosis following anti-tuberculosis treatment. The median onset time of paradoxical response was reported as 26 days after the initiation of anti-tuberculosis treatment.14 In our case, a drug-induced lymphocyte stimulation test for amikacin and ALIS, which were performed before steroid administration, were negative, suggesting the immune response was not directly targeted by amikacin or ALIS. It was speculated that necrotic tissue, which sloughed off the thickened cavitary or bronchial wall, might have been aspirated to other parts of the lungs, where it could have stimulated an immune reaction causing infiltrative opacity in this patient.
Since the approval of ALIS by the Japanese government in July 2021, we have started to prescribe ALIS to patients with refractory MAC-PD and experienced seven cases of chest radiographic deterioration during ALIS administration (Table 1). The median age of the patients was 79 years (interquartile range [IQR], 73–80), and the median disease duration was 12 years (IQR, 10.5–14.0). The deterioration was caused by the exacerbation of MAC-PD itself in four patients, pneumonia complications caused by other bacteria in two patients, and DIILD caused by ALIS in one patient. In this study, we demonstrated that patients with refractory MAC-PD for whom ALIS was prescribed may experience radiographic exacerbations for a variety of reasons. Because ALIS is a key drug for patients with refractory MAC-PD, an incorrect diagnosis of DIILD can have negative consequences for patients. Therefore, it is important to make a careful and accurate differential diagnosis. The participants of a phase III randomized controlled trial had a younger age (median, 65 years ; IQR, 40–87) and shorter disease duration (median, 4.6 years ; IQR, 0.8–32.4) than our cases with chest radiographic deterioration during ALIS administration.9 Because the importance of achieving early culture conversion was recently emphasized,15,16 ALIS should be prescribed to patients with early-stage NTM-PD.
 |
Table 1 Cases with Radiographic Deterioration During Treatment with Amikacin Liposome Inhalation Suspension |
Conclusion
Patients with refractory NTM-PD who are treated with ALIS may experience radiographic exacerbations for various reasons, including exacerbation of NTM-PD itself, pneumonia caused by other bacteria, and DIILD caused by ALIS. It is crucial to accurately differentiate between these conditions to ensure appropriate management and prevent potential harm to patients. It is important to perform a drug provocation test after obtaining sufficient informed consent.
Abbreviations
ALIS, Amikacin liposome inhalation suspension; MAC-PD, Mycobacterium avium complex pulmonary disease; DIILD, drug-induced interstitial lung disease; NTM, Nontuberculous mycobacterium; CRP: C-reactive protein; IQR: Interquartile range.
Data Sharing Statement
The dataset generated during the current study is not publicly available in consideration of patient privacy but is available from the corresponding author upon reasonable request.
Ethics Approval
National Hospital Organization Osaka Toneyama Medical Center approve to publish the case detail.
Consent for Publication
The patient provided written informed consent for her personal or clinical details along with any identifying images to be published in this study.
Acknowledgments
We thank J. Ludovic Croxford, PhD, from Edanz for editing a draft of this manuscript.
Funding
This work was supported in part by grants from AMED (number 23fk0108673h0701) and JSPS KAKENHI (number 21K08194).
Disclosure
Dr Keisuke Miki reports patents (6798747 and PCT/JP2019/037405) for expiratory pressure load training management device and system for reducing shortness of breath of a COPD patient and improving exercise capability and physical activity. The present invention is a training management device for a user who has COPD. The authors report no other conflicts of interest in this work.
References
1. Fukushima K, Kitada S, Abe Y, et al. Long-term treatment outcome of progressive Mycobacterium avium complex pulmonary disease. J Clin Med. 2020;9(5):1315. doi:10.3390/jcm9051315
2. Fukushima K, Kida H. New/different look at the presence of Aspergillus in mycobacterial pulmonary diseases. Long-term retrospective cohort study. Microorganisms. 2021;9(2):270. doi:10.3390/microorganisms9020270
3. Lee SH, Lee JH, Chang JH, et al. Hemoptysis requiring bronchial artery embolization in patients with nontuberculous mycobacterial lung disease. BMC Pulm Med. 2019;19(1):117. doi:10.1186/s12890-019-0881-z
4. Yamamoto Y, Tsujino K, Kuge T, et al. Pleuroparenchymal fibroelastosis in Mycobacterium avium complex pulmonary disease: clinical characteristics and prognostic impact. ERJ Open Res. 2021;7(1):00765–2020. doi:10.1183/23120541.00765-2020
5. Terzano C, Petroianni A. Clarithromycin and pulmonary infiltration with eosinophilia. BMJ. 2003;326(7403):1377–1378. doi:10.1136/bmj.326.7403.1377
6. Saha K, Bandyopadhyay A, Sengupta A, Jash D. A rare case of ethambutol induced pulmonary eosinophilia. J Pharmacol Pharmacother. 2013;4(4):300–302. doi:10.4103/0976-500X.119724
7. Nishio C, Sato A, Tsuboi T, Sumi K, Kurasawa T. Pneumonitis induced by rifampicin. Kekkaku. 2011;86(4):473–476.
8. Matsuno O. Drug-induced interstitial lung disease: mechanisms and best diagnostic approaches. Respir Res. 2012;13(1):39. doi:10.1186/1465-9921-13-39
9. Griffith DE, Eagle G, Thomson R, et al. Amikacin liposome inhalation suspension for treatment-refractory lung disease caused by Mycobacterium avium Complex (CONVERT). A prospective, open-label, randomized study. Am J Respir Crit Care Med. 2018;198(12):1559–1569. doi:10.1164/rccm.201807-1318OC
10. Winthrop KL, Flume PA, Thomson R, et al. Amikacin liposome inhalation suspension for Mycobacterium avium complex lung disease: a 12-month open-label extension clinical trial. Ann Am Thorac Soc. 2021;18(7):1147–1157. doi:10.1513/AnnalsATS.202008-925OC
11. Kidogawa M, Yamasaki K, Nemoto K, Yatera K. Liposomal amikacin inhalation suspension-induced pneumonitis. Intern Med. 2022;61(16):2547–2549. doi:10.2169/internalmedicine.8796-21
12. Takao D, Takeda K, Takazono T, et al. A case of drug-induced organizing pneumonia caused by amikacin liposome inhalation suspension. J Infect Chemother. 2023;29(8):806–808. doi:10.1016/j.jiac.2023.04.013
13. Matsumoto K, Nakao S, Hasegawa S, et al. Analysis of drug-induced interstitial lung disease using the Japanese adverse drug event report database. SAGE Open Med. 2020;8:1–13. doi:10.1177/2050312120918264
14. Cheng SL, Wang HC, Yang PC. Paradoxical response during anti-tuberculosis treatment in HIV-negative patients with pulmonary tuberculosis. Int J Tuberc Lung Dis. 2007;11(12):1290–1295.
15. Im Y, Hwang NY, Kim K, Kim H, Kwon OJ, Jhun BW. Impact of time between diagnosis and treatment for nontuberculous mycobacterial pulmonary disease on culture conversion and all-cause mortality. Chest. 2022;161(5):1192–1200. doi:10.1016/j.chest.2021.10.048
16. Hasegawa N, Nishimura T, Ohtani S, et al. Therapeutic effects of various initial combinations of chemotherapy including clarithromycin against Mycobacterium avium complex pulmonary disease. Chest. 2009;136(6):1569–1575. doi:10.1378/chest.08-2567
 © 2023 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2023 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.