Back to Journals » Infection and Drug Resistance » Volume 9
Diagnosing Clostridium difficile-associated diarrhea using enzyme immunoassay: the clinical significance of toxin negativity in glutamate dehydrogenase-positive patients
Authors Yuhashi K, Yagihara Y, Miasawa Y, Sato T, Saito R, Okugawa S , Moriya K
Received 30 January 2016
Accepted for publication 7 April 2016
Published 27 May 2016 Volume 2016:9 Pages 93—99
DOI https://doi.org/10.2147/IDR.S105429
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Professor Suresh Antony
Kazuhito Yuhashi,1 Yuka Yagihara,1 Yoshiki Misawa,1 Tomoaki Sato,1 Ryoichi Saito,2 Shu Okugawa,1 Kyoji Moriya1
1Department of Infection Control and Prevention, Faculty of Medicine, The University of Tokyo, 2Department of Microbiology and Immunity, Graduate School of Health Care Sciences, Tokyo Medical and Dental University, Bunkyo-ku, Tokyo, Japan
Purpose: The enzyme immunoassay (EIA) has lower sensitivity for Clostridium difficile toxins A and B than the polymerase chain reaction in the diagnosis of C. difficile-associated diarrhea (CDAD). Furthermore, toxin positivity with EIA performed on C. difficile isolates from stool cultures may be observed even in patients with EIA glutamate dehydrogenase (GDH)-positive and toxin-negative stool specimens. It is unclear whether such patients should be treated as having CDAD.
Methods: The present study retrospectively compared patient characteristics, treatment, and diarrhea duration among three groups of patients who underwent stool EIA testing for CDAD diagnosis: a toxin-positive stool group (positive stool group; n=39); a toxin-negative stool/toxin-positive isolate group (discrepant negative/positive group, n=14); and a dual toxin-negative stool and isolate group (dual negative group, n=15). All cases included were confirmed to be GDH positive on EIA test.
Results: Patients’ backgrounds and comorbidities were not significantly different among three groups. No difference was observed among the three groups with regard to antimicrobial drug use before diarrhea onset. Treatment was received by 82.1% of the positive stool group compared to 7.1% of the discrepant positive/negative group and 0% of the dual negative group, while mean diarrhea duration was 10.6 days compared to 7.9 days (P=0.6006) and 3.4 days (P=0.0312), respectively.
Conclusion: Even without treatment, patients with toxin-negative stool specimens had shorter diarrhea duration than those with toxin-positive stool specimens even with toxin-positive isolates. These findings may suggest a limited need for CDAD treatment for GDH-positive patients and toxin-negative stool specimens.
Keywords: Clostridium difficile toxin, diarrhea duration, treatment
Introduction
The anaerobic Gram-positive bacillus Clostridium difficile is a leading cause of antibiotic-associated diarrhea. Antimicrobial drugs disrupt the normal intestinal flora, allowing C. difficile to flourish and release C. difficile toxin A and toxin B that induce C. difficile-associated diarrhea (CDAD).1,2 Although CDAD is the common health-care-associated infectious disease, treatment or infection countermeasures are not applied to patients who do not present with diarrhea, including the tens of percents of asymptomatic adults who carry C. difficile in their intestines.3–5
CDAD diagnosis widely uses an enzyme immunoassay (EIA) kit to detect glutamate dehydrogenase (GDH) and toxins A and B (CD toxins) released by the C. difficile cells.6 However, the low detection sensitivity of EIA for the CD toxins has been reported.7,8 Compared to the ≥90% detection sensitivity of polymerase chain reaction (PCR) and cytotoxigenic culture, EIA detection sensitivity is usually thought to be ∼70% and has been reported to be as low as 50% or below.7,8
CDAD diagnosis is based on EIA results in Japan, because PCR has not been clinically available. If EIA testing is performed on C. difficile isolates cultured from the same specimen, an EIA GDH-positive and toxin-negative stool specimen may give a toxin-positive result. Bacterial content is higher in specimens containing colonies in solution compared to stool; therefore, toxin concentration may also be markedly higher.
If a specimen is GDH positive, follow-up testing with a nucleic acid amplification test, such as PCR, is recommended to compensate for the low sensitivity of EIA.9–11 PCR detection of tcdA or tcdB genes is the evidence for the presence of toxigenic C. difficile. While PCR findings have been reported to correlate well with clinical symptoms,12 bacterial toxin proteins are not detected; therefore, positive results may also include patients carrying C. difficile without CDAD.13 There is currently no definite method of determining whether diarrhea is caused by CD toxins present at concentrations below the detection sensitivity of EIA in patients with EIA toxin-negative stool or whether diarrhea is due to a cause other than CD toxins, despite the presence of C. difficile in the stool. Patients with toxin-negative stool may be dismissed as non-CDAD and thus overlooked in terms of treatment and infection countermeasures.
The present study investigated patients who had submitted stool specimens for CDAD diagnosis using EIA. Differences in patient characteristics, diarrhea duration, antibiotics usage, and severity were compared among patients with toxin-positive stool, toxin-negative stool but toxin-positive isolates, and toxin-negative stool and isolates.
The aim of the study is to investigate clinical history and need for the treatment of patients with GDH-positive and toxin-negative stool specimens.
Methods
Subjects
The present study retrospectively investigated patients (age ≥2 years) whose stool specimens were submitted to the microbiology laboratory at our institution for CD toxin testing between March 2013 and June 2014. Patients with C. difficile growth on stool culture and stool specimens confirmed to be GDH positive on EIA were included. Patients were divided into three groups based on the EIA CD toxin results in stool specimens and isolates obtained from bacterial culture: a toxin-positive stool group (positive stool group), a toxin-negative stool/toxin-positive isolate group (discrepant negative/positive group), and a dual toxin-negative stool and isolate group (dual negative group).
Bacteriological examination
C. Diff Quik Chek Complete (TechLab, Inc., Blacksburg, VA, USA) EIA was performed according to the manufacturer’s instructions to determine stool specimen positivity or negativity for GDH and CD toxins. Samples of toxin-negative stool specimens were inoculated onto cycloserine-cefoxitin mannitol agar (CCMA; Nissui Pharmaceutical Co., Ltd., Tokyo, Japan) and cultured for 48 hours under anaerobic conditions. EIA was performed on isolates from colonies using the same method as for the stool specimens to confirm the presence or absence of GDH and CD toxins. Isolates that were also GDH positive were determined to be C. difficile.
Clinical data collection
Bowel movement status was investigated using the electronic medical records (EMRs) of bowel movement frequency and characteristics. Patients with diarrhea or muddy stool passed ≥once per day or loose stool passed ≥four times per day were identified as symptomatic. Clinical data relating to white blood cell (WBC) count, C-reactive protein levels, serum albumin (Alb) levels, and body temperature on the day the specimens were submitted to the microbiology laboratory or, if measurements were unavailable for that day, on the day before or after, were also obtained from the EMR.
Statistical analysis
Analyses were performed using JMP Pro 11 (SAS Institute Inc., Cary, NC, USA). One-way analysis of variance was performed to compare the continuous variables among the three groups, and the Tukey–Kramer honest significant difference test was performed for multiple comparisons. Fisher’s exact test was used to analyze differences in proportions between the groups.
Ethics statement
The Ethics Committee of The University of Tokyo Graduate School of Medicine approved this study and waived the need for obtaining written informed consent from each patient.
Results
A flow chart of the study design and patient cohort is shown in Figure 1. During the study period, 195 stool specimens were included. Of these, 127 specimens were excluded for the following reasons: ≥second of multiple specimens submitted during the same diarrhea episode (n=54), no record of EIA results for isolates obtained from stool culture (n=47), patient age <2 years (n=4), and bowel movement status unclear from EMR or fever chart (n=22). The remaining 68 specimens were included in the analysis. All specimens underwent EIA, identifying 39 toxin-positive and 29 toxin-negative stool specimens. EIA performed on agar-cultured isolates from toxin-negative stool specimens identified 14 toxin-positive and 15 toxin-negative isolates. All stool specimens and isolates (n=68) were GDH positive.
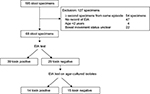  | Figure 1 Flow chart of the study. Notes: All patients and isolates included in this study were GDH positive on EIA test. Abbreviations: GDH, glutamate dehydrogenase; EIA, enzyme immunoassay. |
Table 1 shows the overall patient characteristics and clinical data for each group. No significant differences were observed in clinical characteristics such as the number of comorbidities, presence of malignant tumors, recent history of transplant, and use of tube feeding.
No significant differences were observed among the three groups with regard to number and respective administration frequencies of antimicrobial drugs received by patients within the 30 days prior to diarrhea onset (Table 2).
A significant difference was observed in mean diarrhea duration between the 39 patients in the positive stool group and the 29 patients with toxin-negative stool specimens (10.6 days vs 5.6 days; P=0.0179). Among the 29 patients with toxin-negative stool specimens, mean diarrhea durations were 7.9 days and 3.4 days for the discrepant negative/positive group and the dual-negative group, respectively. A significant difference was observed in diarrhea duration among the three groups (P=0.0395; Figure 2). Comparison between each group found a significant difference in diarrhea duration between the positive stool group and the dual-negative group (P=0.0312). No significant difference was observed between the positive stool group and the discrepant negative/positive group (P=0.6006); however, diarrhea duration tended to be longer in the positive stool group. No difference was observed in diarrhea duration between either of the groups with toxin-negative stool specimens (P=0.3952).
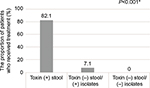
A significant difference was observed among the groups in the proportion of patients who received vancomycin or metronidazole treatment for CDAD: positive stool group, 82.1% (n=32); discrepant negative/positive group, 7.1% (n=1; vancomycin 500 mg three times daily for 13 days); and dual-negative group, 0% (n=0); P<0.001 (Figure 3). In positive stool group, 27 patients received oral vancomycin, four received oral metronidazole, and one received oral metronidazole (250 mg four times daily for 11 days) followed by oral vancomycin (125 mg four times daily for 7 days). Of the patients receiving oral vancomycin alone in positive stool group, 70.4% received 125 mg four times daily, 18.5% received 500 mg four times daily, 7.4% received 500 mg three times daily, or 3.7% received 250 mg four times daily. The median duration of oral vancomycin was 11 days (range 1–25). Of the patients receiving oral metronidazole alone in positive stool group, 25% received 250 mg four times daily and 75% received 500 mg three times daily. The median duration of oral metronidazole was 14 days (range 10–15).
The present findings demonstrated that, although significantly fewer patients in the discrepant negative/positive group received treatment compared to the positive stool group, diarrhea duration tended to be shorter.
No significant differences were observed among the groups with regard to inflammation findings, specifically WBC count, C-reactive protein levels, Alb levels, and body temperature, on the day stool specimens were submitted (Table 3).
Discussion
Of the 68 patients investigated in the present study, 39 (57.4%) had CD toxin-positive stool specimens, 14 (20.6%) had toxin-negative stool specimens but toxin-positive isolates, and 15 (22.1%) had both toxin-negative stool specimens and isolates.
Quinn et al14 investigated 174 stool specimens and found that 133 (76.4%) were negative for both GDH and CD toxins, 18 (10.3%) were positive for both GDH and toxins, and 23 (13.2%) were positive for GDH but negative for toxins. In the present study, similar rates of GDH positivity were found in both toxin-positive and toxin-negative patients with regard to stool specimens. Similar results were obtained. These GDH-positive but toxin-negative patients represent the proportion of cases in which diagnosis and treatment requirements are unclear.
Symptom duration was significantly shorter in patients with toxin-negative rather than toxin-positive stool specimens, despite the lack of CDAD drug treatment in all but one of the 29 toxin-negative stool specimen patients. Furthermore, diarrhea duration also tended to be shorter in patients with toxin-negative stool specimens but toxin-positive isolates compared to those with toxin-positive stool specimens, despite significantly fewer discrepant negative/positive group patients receiving treatment. This suggests that patients who tested positive for C. difficile on culture but negative for toxins in their stool had milder symptoms, despite having toxin-positive isolates. Polage et al15 found that EIA toxin-negative patients had significantly milder diarrhea, shorter diarrhea duration, and a lower mortality rate than toxin-positive patients. Patel et al16 also reported that an infection with an EIA toxin-negative strain of C. difficile presents less severe clinical course, such as lower white blood cell counts and shorter length of hospital stay, than infection with EIA-positive C. difficile. Meanwhile, Baker et al17 demonstrated that EIA toxin-negative patients present with milder clinical symptoms than toxin-positive patients. Conversely, other studies have reported no difference in clinical symptoms between toxin-negative and toxin-positive patients,18 and a consensus has yet to be reached. The present analysis supported the reports of Polage et al,15 Patel et al,16 and Baker et al,17 finding that mean diarrhea duration was significantly shorter in patients with toxin-negative (5.6 days) than toxin-positive (10.6 days) stool specimens, even with similar GDH positivity.
Furthermore, 13 of 14 patients with toxin-negative stool specimens but toxin-positive isolates did not receive CDAD treatment, and diarrhea duration in this group did not differ significantly from that in the dual-negative group. The group with toxin-negative isolates may have been infected with nontoxigenic C. difficile, while the toxin concentration in the stool and intestines of the group with toxin-positive isolates but toxin-negative stool specimens was likely too low to be detected by EIA, despite C. difficile toxigenicity. Akerlund et al19 found that toxin concentrations in vitro from cultured C. difficile did not correlate with the toxin concentration in stool, which correlated with diarrhea frequency and abdominal pain. In addition to low toxin concentration, various intestinal factors, such as primary bile salt and human immunity, may contribute to lack of toxin detection in stool specimens in patients with discrepant negative/positive findings,20–26 because primary bile salts inhibit the growth of vegetative cells of C. difficile and human alpha-defensins cause loss of cytotoxicity of C. difficile toxin B. However, whatever the cause, diarrhea is comparatively mild in patients with toxin-negative stool, suggesting that severe symptoms are unlikely to develop at toxin concentrations below detection sensitivity. In this study, all cases were GDH positive. Toxin-negative patients with GDH positivity may be simply colonized with another cause of diarrhea, even if toxigenic C. difficile is isolated from stool.
Some classes of antibiotics, such as clindamycin, broad-spectrum cephalosporins, vancomycin, carbapenems, β-lactamase inhibitors combination antibiotics, and fluoroquinolones, are widely known risk factors for CDAD.5,27–29 On the other hand, linezolid has been reported to have a potential protective role against the development of C. difficile infection30 and a good in vitro activity against C. difficile isolates in a human gut model of C. difficile infection.31 In this study, 48% patients received β-lactamase inhibitors combination antibiotics and 25% patients received carbapenems. Linezolid was not used in the present study. Although cephamycin and fourth-generation cephalosporin tend to be more frequently used in toxin (+) stool group than other groups, there were no significant differences among the groups in antimicrobial drug use within the 30 days prior to toxin testing. Furthermore, no clear correlation was observed between type of antimicrobial drug administered prior to CDAD onset and the presence of toxins.
In the majority of patients, stool testing was conducted within several days of diarrhea onset, but no significant differences were observed among the groups with regard to clinical inflammation during the same period, suggesting no strong correlation between toxin detection and inflammation at the time of symptom onset. These findings indicate that it is difficult to predict toxin negativity or positivity prior to toxin testing based on drug use status or other test results.
The sensitivity of EIA for toxin detection is far inferior to that of PCR, which amplifies the toxin coding regions.14,32 However, PCR detects the presence of bacteria with toxin genes rather than toxin protein production; therefore, patients who are identified as toxin-positive on PCR may include carriers with diarrhea due to a cause other than toxin.13 EIA directly measures the toxin proteins in stool and is thus more likely to reflect the patient’s actual disease status. While EIA is often associated with low sensitivity, it has a high positive predictive value,14 making it an important test in determining treatment approach.
Limitations of the present study include the retrospective nature of the study, the small sample size, and the absence of molecular biological testing. Potential differences in the gene sequences related to toxin production between patients with toxin-positive stool and those with toxin-positive isolates were also not investigated. Hypervirulent C. difficile strain, such as BI/NAP-1/027 strain, has emerged and causes severe and recurrent diseases. In Japan, the NAP-1 strain is low prevalence.33 Recently, Polage et al34 reported that patients with a positive molecular test result and a negative toxin immunoassay test result had outcomes that were comparable to patients without C. difficile by either method and that the prevalence of hypervirulent strain was 107 of 293 isolates. Thus, the strains may not affect the results of the study. However, further large number analysis is required to determine whether the patient groupings in the present study reflect bacterial genetic characteristics.
Conclusion
In this study, around half with toxin-negative stool specimens among the patients with EIA GDH-positive stool specimens had toxin-positive isolates. Since toxigenic C. difficile was thus present in their stool, these patients are at risk of transferring the bacteria to other patients and require care from the perspective of preventing hospital infection. However, from the perspective of treatment for individual patients, diarrhea duration is shorter in patients with toxin-negative stool specimens compared to those with toxin-positive stool specimens even without treatment. Nucleic acid amplification tests are recommended to detect toxinogenic C. difficile in stool for a diagnosis of CDAD; however, toxin production is not evaluated. These results may suggest a limited need for CDAD treatment for patients with GDH-positive and toxin-negative stool specimens.
Author contributions
All authors contributed toward data analysis, drafting and critically revising the paper and agree to be accountable for all aspects of the work.
Disclosure
The authors report no conflicts of interest in this study.
References
Lyerly DM, Krivan HC, Wilkins TD. Clostridium difficile: its disease and toxins. Clin Microbiol Rev. 1988;1(1):1–18. | ||
Kelly CP, Pothoulakis C, LaMont JT. Clostridium difficile colitis. N Engl J Med. 1994;330(4):257–262. | ||
Kelly CP, LaMont JT. Clostridium difficile infection. Annu Rev Med. 1998;49:375–390. | ||
Bartlett JG. Clinical practice. Antibiotic-associated diarrhea. N Engl J Med. 2002;346(5):334–339. | ||
Rupnik M, Wilcox MH, Gerding DN. Clostridium difficile infection: new developments in epidemiology and pathogenesis. Nat Rev Microbiol. 2009;7(7):526–536. | ||
Sharp SE, Ruden LO, Pohl JC, Hatcher PA, Jayne LM, Ivie WM. Evaluation of the C. diff quik chek complete assay, a new glutamate dehydrogenase and A/B toxin combination lateral flow assay for use in rapid, simple diagnosis of Clostridium difficile disease. J Clin Microbiol. 2010;48(6):2082–2086. | ||
Peterson LR, Mehta MS, Patel PA, et al. Laboratory testing for Clostridium difficile infection: light at the end of the tunnel. Am J Clin Pathol. 2011;136(3):372–380. | ||
Humphries RM, Uslan DZ, Rubin Z. Performance of Clostridium difficile toxin enzyme immunoassay and nucleic acid amplification tests stratified by patient disease severity. J Clin Microbiol. 2013;51(3):869–873. | ||
Goldenberg SD, Cliff PR, Smith S, Milner M, French GL. Two-step glutamate dehydrogenase antigen real-time polymerase chain reaction assay for detection of toxigenic Clostridium difficile. J Hosp Infect. 2010;74(1):48–54. | ||
Larson AM, Fung AM, Fang FC. Evaluation of tcdB real-time PCR in a three-step diagnostic algorithm for detection of toxigenic Clostridium difficile. J Clin Microbiol. 2010;48(1):124–130. | ||
Surawicz CM, Brandt LJ, Binion DG, et al. Guidelines for diagnosis, treatment, and prevention of Clostridium difficile infections. Am J Gastroenterol. 2013;108(4):478–498. quiz 499. | ||
Berry N, Sewell B, Jafri S, et al. Real-time polymerase chain reaction correlates well with clinical diagnosis of Clostridium difficile infection. J Hosp Infect. 2014;87(2):109–114. | ||
Koo HL, Van JN, Zhao M, et al. Real-time polymerase chain reaction detection of asymptomatic Clostridium difficile colonization and rising C. difficile-associated disease rates. Infect Control Hosp Epidemiol. 2014;35(6):667–673. | ||
Quinn CD, Sefers SE, Babiker W, et al. C. diff quik chek complete enzyme immunoassay provides a reliable first-line method for detection of Clostridium difficile in stool specimens. J Clin Microbiol. 2010;48(2):603–605. | ||
Polage CR, Chin DL, Leslie JL, Tang J, Cohen SH, Solnick JV. Outcomes in patients tested for Clostridium difficile toxins. Diagn Microbiol Infect Dis. 2012;74(4):369–373. | ||
Patel H, Randhawa J, Nanavati S, Marton LR, Baddoura WJ, DeBari VA. Laboratory and clinical features of EIA toxin-positive and EIA toxin-negative community-acquired Clostridium difficile infection. Ann Clin Lab Sci. 2015;45(3):333–339. | ||
Baker I, Leeming JP, Reynolds R, Ibrahim I, Darley E. Clinical relevance of a positive molecular test in the diagnosis of Clostridium difficile infection. J Hosp Infect. 2013;84(4):311–315. | ||
Guerrero DM, Chou C, Jury LA, Nerandzic MM, Cadnum JC, Donskey CJ. Clinical and infection control implications of Clostridium difficile infection with negative enzyme immunoassay for toxin. Clin Infect Dis. 2011;53(3):287–290. | ||
Akerlund T, Svenungsson B, Lagergren A, Burman LG. Correlation of disease severity with fecal toxin levels in patients with Clostridium difficile-associated diarrhea and distribution of PCR ribotypes and toxin yields in vitro of corresponding isolates. J Clin Microbiol. 2006;44(2):353–358. | ||
Sorg JA, Sonenshein AL. Bile salts and glycine as cogerminants for Clostridium difficile spores. J Bacteriol. 2008;190(7):2505–2512. | ||
Sorg JA, Sonenshein AL. Inhibiting the initiation of Clostridium difficile spore germination using analogs of chenodeoxycholic acid, a bile acid. J Bacteriol. 2010;192(19):4983–4990. | ||
Taur Y, Pamer EG. Harnessing microbiota to kill a pathogen: fixing the microbiota to treat Clostridium difficile infections. Nat Med. 2014;20(3):246–247. | ||
Giesemann T, Guttenberg G, Aktories K. Human alpha-defensins inhibit Clostridium difficile toxin B. Gastroenterology. 2008;134(7):2049–2058. | ||
Furci L, Baldan R, Bianchini V, et al. New role for human alpha-defensin 5 in the fight against hypervirulent Clostridium difficile strains. Infect Immun. 2015;83(3):986–995. | ||
Goldberg E, Amir I, Zafran M, et al. The correlation between Clostridium-difficile infection and human gut concentrations of Bacteroidetes phylum and clostridial species. Eur J Clin Microbiol Infect Dis. 2014;33(3):377–383. | ||
Kyne L, Warny M, Qamar A, Kelly CP. Asymptomatic carriage of Clostridium difficile and serum levels of IgG antibody against toxin A. N Engl J Med. 2000;342(6):390–397. | ||
Loo VG, Poirier L, Miller MA, et al. A predominantly clonal multi-institutional outbreak of Clostridium difficile-associated diarrhea with high morbidity and mortality. N Engl J Med. 2005;353(23):2442–2449. | ||
Johnson S, Samore MH, Farrow KA, et al. Epidemics of diarrhea caused by a clindamycin-resistant strain of Clostridium difficile in four hospitals. N Engl J Med. 1999;341(22):1645–1651. | ||
Gerding DN. Clindamycin, cephalosporins, fluoroquinolones, and Clostridium difficile-associated diarrhea: this is an antimicrobial resistance problem. Clin Infect Dis. 2004;38(5):646–648. | ||
Valerio M, Pedromingo M, Munoz P, et al. Potential protective role of linezolid against Clostridium difficile infection. Int J Antimicrob Agents. 2012;39(5):414–419. | ||
Baines SD, Noel AR, Huscroft GS, et al. Evaluation of linezolid for the treatment of Clostridium difficile infection caused by epidemic strains using an in vitro human gut model. J Antimicrob Chemother. 2011;66(7):1537–1546. | ||
Swindells J, Brenwald N, Reading N, Oppenheim B. Evaluation of diagnostic tests for Clostridium difficile infection. J Clin Microbiol. 2010;48(2):606–608. | ||
Senoh M, Kato H, Fukuda T, et al. Predominance of PCR-ribotypes, 018 (smz) and 369 (trf) of Clostridium difficile in Japan: a potential relationship with other global circulating strains? J Med Microbiol. 2015;64(10):1226–1236. | ||
Polage CR, Gyorke CE, Kennedy MA, et al. Overdiagnosis of Clostridium difficile infection in the molecular test era. JAMA Intern Med. 2015;175(11):1792–1801. |
 © 2016 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2016 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.