Back to Journals » International Journal of General Medicine » Volume 15
Development of Risk Prediction Model for Muscular Calf Vein Thrombosis with Acute Exacerbation of Chronic Obstructive Pulmonary Disease
Authors Hu X , Li X, Xu H, Zheng W, Wang J, Wang W, Li S, Zhang N, Wang Y , Han K
Received 15 May 2022
Accepted for publication 25 July 2022
Published 10 August 2022 Volume 2022:15 Pages 6549—6560
DOI https://doi.org/10.2147/IJGM.S374777
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Scott Fraser
Xiaoman Hu, Xincheng Li, Huifen Xu, Weili Zheng, Jian Wang, Wenyu Wang, Senxu Li, Ning Zhang, Yunpeng Wang, Kaiyu Han
Department of Respiratory and Critical Medicine, The Second Affiliated Hospital of Harbin Medical University, Harbin, People’s Republic of China
Correspondence: Kaiyu Han, Email [email protected]
Purpose: This study aims to establish a risk prediction model for muscular calf vein thrombosis (MCVT) in patients with acute exacerbation of chronic obstructive pulmonary disease (AECOPD).
Methods: The research sample consisted of 248 patients with AECOPD and all of them underwent vascular ultrasounds of both lower limbs in this retrospective study. Univariate analysis and multivariate logistic regression analysis were conducted on factors with significant group differences to screen for the independent risk factors of MCVT. A nomogram to predict the risk of MCVT was constructed and validated with bootstrap resampling.
Results: According to the exclusion criteria, 240 patients were included for analysis, divided into the MCVT group (n = 81) and the non-MCVT group (n = 159). Multivariate logistic regression analyses showed that hypertension, elevated MPV, reduced albumin (ALB), elevated D-dimer and bed rest ≥ 3 days were independent risk factors for MCVT in AECOPD. A nomogram model for predicting AECOPD with MCVT was established based on them. The area under the curve (AUC) of receiver operating characteristic (ROC) curve for the prediction model and the simplified Wells score was 0.784 (95% CI: 0.722– 0.847) and 0.659 (95% CI: 0.583– 0.735), respectively. The cut-off value and Youden index of prediction model were 0.248 and 0.454, respectively. At the same time, the sensitivity, specificity, positive predictive value, and negative predictive value of the prediction model were 85.9%, 59.5%, 84.6%, and 77.4%, respectively. The sensitivity and specificity of the simplified Wells score were 67.9% and 56.3%, respectively. Validation by the use of bootstrap resampling revealed optimal discrimination and calibration, and the decision analysis curve (DAC) suggested that this prediction model involved high clinical practicability.
Conclusion: We developed a nomogram that can predict the risk of MCVT for AECOPD patients. This model has the potential to assist clinicians in making treatment recommendations and formulating corresponding prevention measures.
Keywords: acute exacerbation of chronic obstructive pulmonary disease, muscular calf vein thrombosis, risk factors, nomogram, model
Introduction
Chronic obstructive pulmonary disease (COPD) is a common respiratory disease characterized by persistent respiratory symptoms and airflow limitation due to airway and/or alveolar abnormalities.1 Nearly 3 million people die from COPD every year and COPD is currently the third-leading cause of death worldwide.2 Most patients with COPD have persistent systemic inflammation, chronic hypoxia and impaired pulmonary function over a long time. Therefore, COPD patients show decreased exercise tolerance and lower limb movement, often accompanied by lower limb venous endothelial injury and blood stasis,3,4 which create the pathophysiological conditions for forming deep vein thrombosis (DVT). Acute exacerbation of chronic obstructive pulmonary disease (AECOPD) refers to the deterioration of respiratory symptoms in a short period and requires additional treatment. It is reported that the risk of DVT in patients with AECOPD is up to 12.4% and the risk of pulmonary embolism (PE) is also increased accordingly, which can be highly life-threatening.5–8
Muscular calf vein thrombosis (MCVT) belonging to isolated distal deep vein thrombosis (IDDVT) refers to thrombosis in the calf muscle veins, most commonly found in halibut and fibula muscles. The incidence of MCVT accounts for 50% of IDDVT and 5.6–31.3% of DVT.9 The symptoms of lower limb swelling and pain are often inconspicuous because the muscular calf vein branch is thin and the inflammatory stimulation caused by thrombosis is light, which patients and clinicians do not easily detect. Thus, patients with MCVT may miss the best timing of treatment and develop PE.10,11
The nomogram has been accepted as a reliable tool for creating a simple, intuitive graph of a statistical predictive model that quantifies the risk of a clinical event.12 The nomogram risk prediction model is widely utilized in clinic. In contrast, research about the risk prediction model in AECOPD patients with MCVT is seldom reported, and there are discrepancies in the findings of studies. As a result, this study has constructed the risk prediction model of MCVT. It is designed to help clinicians identify patients at risk of MCVT and provide a theoretical and decision-making basis for early thromboprophylaxis.
Methods
Study Design
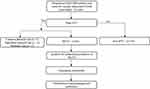
A total of 248 patients with AECOPD were hospitalized in the department of Respiratory Medicine at the Second Affiliated Hospital of Harbin Medical University from January 2017 to December 2020 with the simplified Wells score greater than or equal to 2. Then, all of them received vascular ultrasounds of both lower limbs and were screened for this study. Their data originated from the hospital information system (HIS) and the diagnostic criteria of the 2017 GOLD guidelines1 are used to confirm the diagnosis and severity grading of AECOPD. According to the 2016 American College of Chest Physicians (ACCP) Antithrombotic Therapy for VTE Disease13 and the vascular ultrasounds of both lower limbs, 89 patients were diagnosed DVT, among whom 81 with MCVT, 3 with common femoral vein thrombosis, 2 with superficial femoral vein thrombosis and 3 with popliteal vein thrombosis. A total of 240 patients were included in this research and divided into the MCVT group (81 cases) and the non-MCVT group (159 cases), see Figure 1 for details. The clinical data of these patients were retrospectively gathered for analysis and constructing a nomogram risk prediction model.
Inclusion criteria were as follows: (1) hospitalized patients with a confirmed diagnosis of AECOPD; (2) simplified Wells score greater than or equal to 2; (3) developed the vascular ultrasounds of both lower limbs.
Exclusion criteria were as follows: (1) patients with lying in bed in the last 1 month because of surgery (such as hip arthroplasties) and trauma (such as femoral shaft fracture); (2) patients with severe hepatic and renal dysfunction, severe hematological system diseases, immune deficiency and severe neurological disease; (3) patients having a recent history of anticoagulation treatment (such as warfarin, low-molecular-weight heparin, or rivaroxaban); (4) patients diagnosed DVT, but the thrombus located beyond the calf muscle veins; (5) patients diagnosed with chronic MCVT. This study was approved by the ethical committee of the Second Affiliated Hospital of Harbin Medical University. All procedures involving human participants complied with the Declaration of Helsinki, and informed consents were exempted from all patients.
Data Collection
The baseline characteristics include age, gender, underlying diseases, the history of venous thromboembolism (VTE), severity grading, FEV1 in percent of the predicted value (FEV1% pred), bed rest ≥3 days. Clinical manifestations such as lower limb swelling and fever ≥3 days. Laboratory examination such as blood routine, biochemical series, hemogram, blood gas analysis; other data such as simplified Wells score, vascular ultrasounds of both lower limbs and complications. Blood samples and relevant examinations were collected during admission.
Statistical Analysis
Data were analyzed using SPSS 25.0 and R 3.6.3 software. The categorical variables were described by the number of cases (rate) and were compared using the chi‐square test (χ2 test). Normally distributed variables were expressed as the mean ± standard deviation (mean ± SD), while non-normally distributed variables were shown as the median (M) and interquartile range (IQR). Univariate analysis and multivariate logistic regression analysis were performed on variables reaching statistical significance to determine the independent risk factors for MCVT. The independent risk factors were imported into R software for further analysis, and the “rms” program package in R software was used to establish the nomogram and correction diagram of MCVT occurrence. The performance of the nomogram was assessed by discrimination and calibration. The discriminative ability of the model was determined by the AUC of ROC analysis, which ranged from 0.5 (no discrimination) to 1 (perfect discrimination). The ROC analysis was also used to calculate the optimal cut-off values, which were determined by maximizing the Youden index (ie, sensitivity + specificity −1). The accuracy of the optimal cut-off value was assessed by sensitivity, specificity and predictive values. The calibration of the prediction model was performed by a visual calibration plot comparing the predicted and actual probability of MCVT. In addition, the nomogram was subjected to 1000 bootstrap resamples for internal validation to assess their predictive accuracies. The DAC was used to evaluate the nomogram’s net benefit and clinical practicability. A value of P < 0.05 was considered to be statistically significant.
Results
Baseline Characteristics
The proportion of patients who exhibited hypertension and VTE in MCVT group was significantly higher than that in the non-MCVT group (P < 0.05). Compared with the MCVT group, fewer patients in the non-MCVT group had lower swelling of lower limbs and bed rest ≥ 3 days. No statistically significant differences were found in age, gender, smoking history, complication, severity grading and FEV1% pred between the two sets (Table 1).
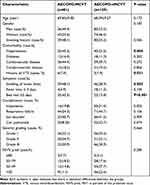 |
Table 1 Baseline Characteristics |
Comparison of Laboratory Parameters
There was no statistical difference between the MCVT group and the non-MCVT group in terms of WBC, HCT, MCV, RDW-CV, PDW, FIB, PaO2 and PaCO2 (Table 2). The concentration of D-dimer (Figure 2B) and MPV (Figure 2C) in the MCVT group was significantly higher than in the non-MCVT group, but the MCVT group had a remarkable level of PLT (Figure 2A) and ALB (Figure 2D) than in the non-MCVT group.
 |
Table 2 Laboratory Parameters |
 |
Figure 2 Laboratory examination results of the modeling group. The variables of (A) and (B) were presented as Log transformed values. The variables of (C) and (D) were presented as regime values. |
Logistic Multivariate Regression Analysis of the Modeling Group
The results of the univariate analysis indicated that VTE, hypertension, swelling of lower limbs, bed rest ≥3 days, RDW-SD, MPV, ALB and D-dimer were predictors for MCVT. The statistically significant factors (P < 0.05) shown in the univariate analysis were selected as independent variables of the multivariate logistic regression analysis. The results suggested that hypertension, elevated MPV, reduced ALB, elevated D-dimer and bed rest ≥3 days were independent risk factors for MCVT with AECOPD (Figure 3); however, VTE, swelling of lower limbs and RDW-SD were not significant.
 |
Figure 3 Univariate analysis and multi-factor analysis shows the risk factors of MCVT in AECOPD patients. |
Predictive Nomogram for the Probability of MCVT
Based on the final regression analysis, a nomogram was constructed that incorporated the 5 significant risk factors for predicting MCVT (Figure 4). A total score was calculated using hypertension, MPV, ALB, D-dimer, and bed rest ≥3 days. The value of each of these variables was given a score on the point scale axis. A total score could be easily calculated by adding each single score and, by projecting the total score to the lower total point scale, we were able to estimate the probability of MCVT.
Distinguishing Degree, Calibration and Clinical Effectiveness of the Predictive Model
The ROC curve of the nomogram model and simplified Wells score are shown in Figure 5. The prediction model and simplified Wells score had AUC value of 0.784 (95% CI: 0.722–0.847) and 0.659 (95% CI: 0.583–0.735), respectively. The sensitivity, specificity, positive predictive value, negative predictive value, cut-off value and Youden index of the model were 85.9%, 59.5%, 84.6%, 77.4%, 0.248, 0.454, accordingly. The sensitivity and specificity of the simplified Wells score were 67.9% and 56.3%, respectively. A calibration curve of the nomogram is presented in Figure 6, which shows that the MCVT probabilities predicted by the nomogram agreed well with the actual probabilities. In other words, this model had a high prediction accuracy. The performance of the nomogram was internally validated with Bootstrap resampling (1000 times) analysis, and it showed that the model had good predictive performances. Results of the DAC of the nomogram are shown in Figure 7. This model had good clinical utility, and it was feasible for making useful judgments because the decision curve was far from the 2 extreme curves.
 |
Figure 5 The ROC of the prediction model and simplified Wells score. |
Discussion
COPD is the most common chronic respiratory disease. The total number of patients with COPD is more than 100 million in China, causing substantial social and family economic burdens.14 Owing to the existence of systemic inflammation, platelet activation and oxidative stress in COPD pathogenesis, patients are prone to developing DVT and PE, and have a higher risk of DVT and PE in acute exacerbation.15–17 According to previous reports, the incidence of DVT and PE in AECOPD patients was 19–29%11,18 and 3.3–29.1%,19 respectively. DVT and PE have become the serious common comorbidity of AECOPD.
In the present study, we observed that the incidence of DVT with AECOPD who have perfected vascular ultrasounds of both lower limbs was 35.8% (89/240). This was higher than previous results. On the one hand, it might be due to having insufficient knowledge of the disease severity for AECOPD patients selected in the study, who had greater severity of illness at the time of hospital admission and prolonged bed time, with an increased risk of DVT, on the other hand, the difference possibly stemmed from the sample size included in other studies.
Compared with other venous in lower limb, the muscular calf vein is more subjected to thrombus due to multiple branches, small vein valve and slow blood flow.20–22 However, it is worth noting that many patients may be missed because they are asymptomatic. In our study, MCVT made up 91% (81/89) of DVT, consistent with earlier reports. Previous studies suggested that MCVT might be the source of proximal DVT23,24 and the probability of MCVT progression towards proximal DVT was 10–33%,25 which could lead to increased incidence of PE.26 Furthermore, we also observed that IDDVT accounted for 33.7% (89/240) of DVT. IDDVT could cause a high VTE recurrence rate and high mortality rate and was correlated with post‐thrombotic syndrome (PTS).26–28 Hence, MCVT served as the most common type of IDDVT, and the early identification of MCVT in AECOPD patients can contribute to reduce the occurrence of adverse events and improve the quality of life.
The study has imposed statistical analysis on baseline characteristics and laboratory findings in MCVT group and non-MCVT group. Univariate analysis revealed that VTE, hypertension, swelling of lower limbs, bed rest ≥3 days, RDW-SD, MPV, ALB and D-dimer were predictors for MCVT. A further multivariate logistic regression analysis indicated that hypertension, elevated MPV, reduced ALB, elevated D-dimer and bed rest ≥3 days were independent risk factors for MCVT in AECOPD patients. The VTE history as a risk factor for VTE was generally recognised at home and abroad. However, it was only found to be a predictor, rather than an independent risk factor for MCVT in this study. We hypothesized that the depreciated significance could be resulted from the interplay between the factors included in the analysis. Therefore, it is still necessary to be vigilant about the possibility of MCVT for AECOPD patients with a history of VTE in the clinic.
Patients with COPD often reduce their physical activity and extend relatively bed rest due to cardiopulmonary insufficiency during exacerbation, which leads to venous stasis. It has been demonstrated that bed rest more than 3 days increases the incidence of venous thromboembolism and also is a risk factor for promoting DVT.29 Our results were in accordance with the findings. To remove this risk factor, physicians can guide them actively for rehabilitation exercise according to the current state of the organism, consequently decreasing the incidence of MCVT and improving the quality of life of patients.30
As the disease progresses, there are many comorbidities in AECOPD patients, among which hypertension is the most common. About 70.9% of AECOPD patients are accompanied by primary or secondary hypertension.31,32 During periods of acute COPD exacerbation, oxidative stress and inflammatory response presenting in the organism can induce airway epithelium injury and accelerate the progression of atherosclerosis, which leads to the occurrence of cardiovascular diseases such as hypertension.33 Chang et al34 found that MCVT existed easily in AECOPD patients complicated by hypertension. Long-term fluctuation of blood pressure easily causes vasodilator dysfunction, damaging the vascular endothelial cell, leading to the aggregation of platelet and fibrin, which in turn instigates venous thrombosis of the lower extremity.35 In parallel, the nomogram model in this study demonstrated that the AECOPD patients with hypertension were associated with higher risks of MCVT. Moreover, hypertension with MCVT increases the risk of adverse cardiovascular events and death.34 Consequently, well-controlled hypertension for managing the prognosis of AECOPD patients cannot be ignored.
As a multifunctional protein in the plasma, ALB is considered to be a negative acute-phase protein in inflammatory conditions.36 It possesses important antioxidant properties, plays an important role in defense against oxidative stress,37 and can serve as a biomarker for clinical evaluation of malnutrition.38 The majority of patients with AECOPD are elderly and usually associated with underlying diseases, easily suffering from malnutrition.39 Meanwhile, systemic inflammatory response and oxidative stress of them also result in a decline of ALB. The current study and Zinellu et al40 detected that the ALB level in AECOPD patients was lower than normal. A decrease in albumin level is indicated as a trigger of elevated platelet and fibrinogen aggregation, negatively correlating with the risk of venous thromboembolism.41 A prospective study42 suggested that reduced ALB level was the risk factor for DVT. Lung et al43 reported that lower ALB levels increased the prevalence of lower limb vein thrombosis dramatically. Consistent with these studies, our risk prediction model demonstrated that reduced ALB was the independent risk factor of AECOPD patients combined with MCVT. As a result, timely correcting the low level of ALB may play a positive role in preventing MCVT.
MPV represents the average size of platelets and correlates with platelet function and activation.44 Elevated MPV marks platelet activation and enlarged platelet volume.45 Large platelets with strong coagulation ability, fast aggregation and good mucoadhesion property can release more prothrombotic substances that are advantageous in venous thrombosis. They are also a predictor of venous thrombosis.45,46 AECOPD patients are usually accompanied by hypoxia and tachycardia, which can stimulate platelet activation and lead to an increase in MPV.47 Cui et al48 and Farah et al49 confirmed that elevated MPV was an independent predictor of DVT events. The model constructed in the current study revealed it was easy for AECOPD patients to develop MCVT when they present elevation of MPV. In addition, it was shown that elevated MPV could predict poor prognosis of AECOPD patients.50 Aydınyılmaz et al51 found that additional use of antiplatelet drug based on the above anticoagulant therapy reduced mortality for COVID-19 patients with MPV >10.45f/l. A systematic evaluation and meta-analysis study indicated that all-cause mortality in patients with AECOPD was decreased after taking antiplatelet therapy.52 Further prospective studies are still needed to confirm whether giving prophylactic antiplatelet medications will diminish the risk of death for AECOPD patients.
D-dimer is not only the smallest fibrin degradation product but also a specific degradation product of the fibrous matrix of venous thrombosis. It is abnormally elevated in the case of blood hypercoagulability or secondary hyperfibrinolysis and is a highly sensitive indicator of thrombosis and dissolution.53 Many studies have found that the level of D-dimer in MCVT with AECOPD patients was noticeably higher than those of the AECOPD without thrombosis, and that elevated D-dimer was the independent risk factor for MCVT.53,54 Like our study, these studies revealed that D-dimer levels had strong association with venous thrombosis. However, D-dimer can be affected by many factors, such as infection, tumor, severe hepatorenal insufficiency and pregnancy. As a result, D-dimer elevation at the time of hospitalization for AECOPD may indicate whether MCVT exists while we must preclude the possibility of other factors. We can observe the dynamic level of D-dimer and refine interrelated examinations for reducing the occurrence of adverse events.
This study combined hypertension, elevated MPV, reduced ALB, elevated D-dimer and bed rest ≥3 days to establish a nomogram model. When the calculated score increased in the nomogram model, the corresponding risk of MCVT also increased significantly. The AUC of ROC curve is calculated to verify the predictive performance. The simplified Wells score is a commonly used clinical likelihood assessment scale for VTE. In this study, we used the nomogram model and simplified Wells score to make the predictive diagnosis of MCVT in AECOPD patients, respectively. We evaluated the predictive diagnostic potential of the nomogram model against simplified Wells score, the most widely used method, in AECOPD patients. The AUC, sensitivity, and specificity of the prediction model ROC curve were found to be significantly higher than the simplified Wells score, suggesting that the model had good predictive value in estimating the probability of MCVT occurrence in AECOPD. Meanwhile, at the Youden index threshold value, the positive and negative predictive values were 84.6% and 77.4%, respectively, suggesting the effectiveness of the mode in predicting MCVT in AECOPD patients with certain high-risk factors. Furthermore, using the calibration curve and DAC to evaluate the model, it was found that the model had good calibration and clinical utility. These results indicate that the model may help clinicians recognize and prevent MCVT early in patients with AECOPD.
However, this study has some limitations. Firstly, this was a single-center retrospective study with a small sample size and a limited number of logistic multivariate regression analyses. Secondly, we did not conduct a follow-up of the patients after discharge. Thirdly, the model has not been verified in the external validation queue. Therefore, the sample size should be expanded, and the predictive value of the nomogram model should be verified by a multi-center study in future.
Conclusion
The present study found that hypertension, elevated MPV, reduced ALB, elevated D-dimer and bed rest≥3 days were independent risk factors of AECOPD patients with MCVT. Based on this, we have developed and internally validated a novel nomogram for predicting the risk of MCVT for patients with AECOPD. The nomogram is easy to use, is highly accurate, and exhibits excellent calibration. This nomogram might help clinicians to make individualized predictions of MCVT and to improve treatment recommendations for AECOPD patients with high-risk factors.
Acknowledgments
Study design was provided by Xiaoman Hu and Xincheng Li; Data collection by Huifen Xu, Weili Zheng, Jian Wang, Wenyu Wang, Senxu Li, Ning Zhang and Yunpeng Wang; Editorial support (in the form of writing assistance, collating author comments, assembling tables/figures, grammatical editing and referencing) was provided by Kaiyu Han, Professor.
Author Contributions
All authors made a significant contribution to the original text, whether that is in the conception, study design, execution, acquisition of data, analysis and interpretation, or in all these areas; took part in drafting, revising or critically reviewing the article; gave final approval of the version to be published; have agreed on the journal to which the article has been submitted; and agree to be accountable for all aspects of the work.
Funding
This study was supported by the funds from the National Natural Science Foundation of China (No.81700021).
Disclosure
The authors declare no conflicts of interest.
References
1. Global initiative for chronic obstructive lung disease. Global strategy for the diagnosis, management, and prevention of chronic obstructive pulmonary disease 2017 report [EB/OL]; 2016.
2. Rana JS, Khan SS, Lloyd-Jones DM, et al. Changes in Mortality in Top 10 Causes of Death from 2011 to 2018. J Gen Intern Med. 2021;36(8):2517-2518. doi:10.1007/s11606-020-06070-z
3. Vanfleteren LE, Spruit MA, Groenen M, et al. Clusters of comorbidities based on validated objective measurements and systemic inflammation in patients with chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 2013;187(7):728–735. doi:10.1164/rccm.201209-1665OC
4. Bakeer M, Funk GC, Valipour A. Chronic obstructive pulmonary disease phenotypes: imprint on pharmacological and non-pharmacological therapy. Ann Transl Med. 2020;8(21):1472. doi:10.21037/atm-20-2219
5. Dutt TS, Udwadia ZF. Prevalence of venous thromboembolism in acute exacerbations of chronic obstructive pulmonary disease: an Indian perspective. Indian J Chest Dis Allied Sci. 2011;53(4):207–210. doi:10.5005/ijcdas-53-4-207
6. Rizkallah J, Man SFP, Sin DD. Prevalence of pulmonary embolism in acute exacerbations of COPD: a systematic review and metaanalysis. Chest. 2009;135(3):786–793. doi:10.1378/chest.08-1516
7. Nishiwaki S, Morita Y, Yamashita Y, et al. Impact of no, distal, and proximal deep vein thrombosis on clinical outcomes in patients with acute pulmonary embolism: from the COMMAND VTE registry. J Cardiol. 2021;77(4):395–403. doi:10.1016/j.jjcc.2020.10.019
8. Rali PM, Criner GJ. Submassive Pulmonary Embolism. Am J Respir Crit Care Med. 2018;198(5):588–598. doi:10.1164/rccm.201711-2302CI
9. Bertoletti L, Couturaud F. COPD is not only one of the several VTE risk factors. Eur J Intern Med. 2021;84:14–15. doi:10.1016/j.ejim.2020.12.013
10. Garcia R, Probeck K, Elitharp DM, et al. Diverse management of isolated calf deep venous thrombosis in a university hospital. J Vasc Surg Venous Lymphat Disord. 2018;6(2):139–145. doi:10.1016/j.jvsv.2017.08.021
11. Su H, Liu H, Liu J, Wang X. Elderly patients with intertrochanteric fractures after intramedullary fixation: analysis of risk factors for calf muscular vein thrombosis. Orthopade. 2018;47(4):341–346. doi:10.1007/s00132-018-3552-5
12. Stark GF, Hart GR, Nartowt BJ, et al. Predicting breast cancer risk using personal health data and machine learning models. PLoS One. 2019;14:e0226765. doi:10.1371/journal.pone.0226765
13. Kearon C, Akl EA, Ornelas J, et al. Antithrombotic therapy for VTE disease: CHEST guideline and expert panel report [published correction appears in Chest. 2016 Oct;150(4):988]. Chest. 2016;149(2):315–352. doi:10.1016/j.chest.2015.11.026
14. Liao JP, Wang X, Liu F, et al. Serum surfactant protein D, lung function decline, and incident chronic obstructive pulmonary disease: a longitudinal study in Beijing. J Thorac Dis. 2021;13(1):92–100. doi:10.21037/jtd-20-1675
15. Polosa R, Malerba M, Cacciola RR, et al. Effect of acute exacerbations on circulating endothelial, clotting and fibrinolytic markers in COPD patients. Intern Emerg Med. 2013;8(7):567–574. doi:10.1007/s11739-011-0636-1
16. Koupenova M, Kehrel BE, Corkrey HA, et al. Thrombosis and platelets: an update. Eur Heart J. 2017;38(11):785–791. doi:10.1093/eurheartj/ehw550
17. Wang Q, Zennadi R. Oxidative stress and thrombosis during aging: the roles of oxidative stress in RBCs in venous thrombosis. Int J Mol Sci. 2020;21(12):4259. doi:10.3390/ijms21124259
18. Hassen MF, Tilouche N, Jaoued O, et al. Incidence and impact of pulmonary embolism during severe COPD exacerbation. Respir Care. 2019;64(12):1531–1536. doi:10.4187/respcare.06661
19. Pourmand A, Robinson H, Mazer-Amirshahi M, Pines JM. Pulmonary embolism among patients with acute exacerbation of chronic obstructive pulmonary disease: implications for emergency medicine. J Emerg Med. 2018;55(3):339–346. doi:10.1016/j.jemermed.2018.05.026
20. Ro A, Kageyama N, Mukai T. Pathophysiology of venous thromboembolism with respect to the anatomical features of the deep veins of lower limbs: a review. Ann Vasc Dis. 2017;10(2):99–106. doi:10.3400/avd.ra.17-00035
21. Palareti G, Schellong S. Isolated distal deep vein thrombosis: what we know and what we are doing. J Thromb Haemost. 2012;10(1):11–19. doi:10.1111/j.1538-7836.2011.04564.x
22. Galanaud JP, Sevestre MA, Genty C, et al. Comparison of the clinical history of symptomatic isolated muscular calf vein thrombosis versus deep calf vein thrombosis. J Vasc Surg. 2010;52(4):932–938.e9382. doi:10.1016/j.jvs.2010.05.019
23. Elmi G, Rinaldi ER, Domanico A, et al. Calf deep vein thrombosis clinical relevance, diagnostic approaches and therapeutic options. Vasa. 2020;49(5):359–366. doi:10.1024/0301-1526/a000869
24. Gillet JL, Perrin MR, Allaert FA. Short-term and mid-term outcome of isolated symptomatic muscular calf vein thrombosis. J Vasc Surg. 2007;46(3):513–519. doi:10.1016/j.jvs.2007.04.040
25. Kret MR, Liem TK, Mitchell EL, et al. Isolated calf muscular vein thrombosis is associated with pulmonary embolism and a high incidence of additional ipsilateral and contralateral deep venous thrombosis. J Vasc Surg Venous Lymphat Disord. 2013;1(1):33–38. doi:10.1016/j.jvsv.2012.04.001
26. Wu AR, Garry J, Labropoulos N. Incidence of pulmonary embolism in patients with isolated calf deep vein thrombosis. J Vasc Surg Venous Lymphat Disord. 2017;5(2):274–279. doi:10.1016/j.jvsv.2016.09.005
27. Robert-Ebadi H, Righini M. Should we diagnose and treat distal deep vein thrombosis? Hematology Am Soc Hematol Educ Program. 2017;2017(1):231–236. doi:10.1182/asheducation-2017.1.231
28. Galanaud JP, Quenet S, Rivron-Guillot K, et al. Comparison of the clinical history of symptomatic isolated distal deep-vein thrombosis vs. proximal deep vein thrombosis in 11086 patients. J Thromb Haemost. 2009;7(12):2028–2034. doi:10.1111/j.1538-7836.2009.03629.x
29. Pang H, Wang L, Liu J, et al. The prevalence and risk factors of venous thromboembolism in hospitalized patients with acute exacerbation of chronic obstructive pulmonary disease. Clin Respir J. 2018;12(11):2573–2580. doi:10.1111/crj.12959
30. Spruit MA, Singh SJ, Garvey C, et al. An official American Thoracic Society/European Respiratory Society statement: key concepts and advances in pulmonary rehabilitation. Am J Respir Crit Care Med. 2013;188(8):e13–e64. doi:10.1164/rccm.201309-1634ST
31. Greulich T, Weist BJD, Koczulla AR, et al. Prevalence of comorbidities in COPD patients by disease severity in a German population. Respir Med. 2017;132:132–138. doi:10.1016/j.rmed.2017.10.007
32. Wang MT, Liou JT, Lin CW, et al. Association of cardiovascular risk with inhaled long-acting bronchodilators in patients with chronic obstructive pulmonary disease: a nested case-control study. JAMA Intern Med. 2018;178(2):229–238. doi:10.1001/jamainternmed.2017.7720
33. Choi BJ, Prasad A, Gulati R, et al. Coronary endothelial dysfunction in patients with early coronary artery disease is associated with the increase in intravascular lipid core plaque. Eur Heart J. 2013;34(27):2047–2054. doi:10.1093/eurheartj/eht132
34. Chang WT, Chang CL, Ho CH, et al. Long-term effects of unprovoked venous thromboembolism on mortality and major cardiovascular events. J Am Heart Assoc. 2017;6(5):e005466. doi:10.1161/JAHA.117.005466
35. Tural K, Kara F, Avcı S, et al. Can complete blood cell count parameters predict deep vein thrombosis? Acta Clin Croat. 2020;59(4):661–666. doi:10.20471/acc.2020.59.04.12
36. Gabay C, Kushner I. Acute-phase proteins and other systemic responses to inflammation [published correction appears in N Engl J Med 1999 Apr 29;340(17):1376]. N Engl J Med. 1999;340(6):448–454. doi:10.1056/NEJM199902113400607
37. Rossi R, Giustarini D, Milzani A, et al. Cysteinylation and homocysteinylation of plasma protein thiols during ageing of healthy human beings. J Cell Mol Med. 2009;13(9B):3131–3140. doi:10.1111/j.1582-4934.2008.00417.x
38. Zhang Z, Pereira SL, Luo M, et al. Evaluation of blood biomarkers associated with risk of malnutrition in older adults: a systematic review and meta-analysis. Nutrients. 2017;9(8):829. doi:10.3390/nu9080829
39. Collins PF, Yang IA, Chang YC, et al. Nutritional support in chronic obstructive pulmonary disease (COPD): an evidence update. J Thorac Dis. 2019;11(Suppl 17):S2230–S2237. doi:10.21037/jtd.2019.10.41
40. Zinellu E, Zinellu A, Fois AG, et al. Reliability and usefulness of different biomarkers of oxidative stress in chronic obstructive pulmonary disease. Oxid Med Cell Longev. 2020;2020:4982324. doi:10.1155/2020/4982324
41. Chi G, Gibson CM, Liu Y, et al. Inverse relationship of serum albumin to the risk of venous thromboembolism among acutely ill hospitalized patients: analysis from the APEX trial. Am J Hematol. 2019;94(1):21–28. doi:10.1002/ajh.25296
42. Zhu Y, Chen W, Li J, et al. Incidence and locations of preoperative deep venous thrombosis (DVT) of lower extremity following tibial plateau fractures: a prospective cohort study. J Orthop Surg Res. 2021;16(1):113. doi:10.1186/s13018-021-02259-y
43. Lung BE, Kanjiya S, Bisogno M, et al. Risk factors for venous thromboembolism in total shoulder arthroplasty. JSES Open Access. 2019;3(3):183–188. doi:10.1016/j.jses.2019.07.003
44. Onder HI, Kilic AC, Kaya M, et al. Relation between platelet indices and branch retinal vein occlusion in hypertensive patients. Indian J Ophthalmol. 2013;61(4):160–162. doi:10.4103/0301-4738.111063
45. Braekkan SK, Mathiesen EB, Njølstad I, et al. Mean platelet volume is a risk factor for venous thromboembolism: the Tromsø study. J Thromb Haemost. 2010;8(1):157–162. doi:10.1111/j.1538-7836.2009.03498.x
46. Djordjevic D, Rondovic G, Surbatovic M, et al. Neutrophil-to-lymphocyte ratio, monocyte-to-lymphocyte ratio, platelet-to-lymphocyte ratio, and mean platelet volume-to-platelet count ratio as biomarkers in critically ill and injured patients: which ratio to choose to predict outcome and nature of bacteremia? Mediators Inflamm. 2018;2018:3758068. doi:10.1155/2018/3758068
47. Colkesen Y, Muderrisoglu H. The role of mean platelet volume in predicting thrombotic events[J]. Clin Chem Lab Med. 2012;50(4):631–634. doi:10.1515/cclm.2011.806
48. Cui LN, Li N, Fu S, et al. Combination of preoperative D-dimer and mean platelet volume predicts postoperative deep venous thrombosis in breast cancer patients. Cancer Biomark. 2018;21(4):909–913. doi:10.3233/CBM-170975
49. Farah R, Nseir W, Kagansky D, et al. The role of neutrophil-lymphocyte ratio, and mean platelet volume in detecting patients with acute venous thromboembolism. J Clin Lab Anal. 2020;34(1):e23010. doi:10.1002/jcla.23010
50. Zhang H, Wu D, Ma D, et al. The clinical value of the ratio of average platelet volume to platelet count in predicting the short-term prognosis of elderly patients with chronic obstructive pulmonary disease in the acute exacerbation period. Chin J Geriatr. 2020;39(06):627–631.
51. Aydınyılmaz F, Aksakal E, Pamukcu HE, et al. Significance of MPV, RDW and PDW with the severity and mortality of COVID-19 and effects of acetylsalicylic acid use. Clin Appl Thromb Hemost. 2021;27:10760296211048808. doi:10.1177/10760296211048808
52. Pavasini R, Biscaglia S, D’Ascenzo F, et al. Antiplatelet treatment reduces all-cause mortality in COPD patients: a systematic review and meta-analysis. COPD. 2016;13(4):509–510. doi:10.3109/15412555.2015.1099620
53. He J, Sun J, Zhang T, Liu G. Construction and validation of risk prediction model for deep vein thrombosis in acute exacerbations of chronic obstructive pulmonary disease based on serum angiopoietin 2 levels. Ann Palliat Med. 2021;10(7):7905–7918. doi:10.21037/apm-21-1448
54. Chen C, Liu Y, Wu H, Feng J, Li A, Wu Z. The outcome of isolated calf muscle vein thrombosis after open reduction and internal fixation for closed intra-articular distal femur fractures: a prospective cohort study. BMC Musculoskelet Disord. 2021;22(1):397. doi:10.1186/s12891-021-04289-3
 © 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.