Back to Journals » Vascular Health and Risk Management » Volume 12
Detection of peripheral arterial disease with an improved automated device: comparison of a new oscillometric device and the standard Doppler method
Authors Span M, Gersak G, Millasseau S, Meza M, Kosir A
Received 16 February 2016
Accepted for publication 24 May 2016
Published 29 July 2016 Volume 2016:12 Pages 305—311
DOI https://doi.org/10.2147/VHRM.S106534
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Daniel Duprez
Matjaž Špan,1 Gregor Geršak,2 Sandrine C Millasseau,3 Marko Meža,4 Andrej Košir4
1Cardiovascular Department, Izola General Hospital, Izola, 2Laboratory of Metrology and Quality, Faculty of Electrical Engineering, University of Ljubljana, Ljubljana, Slovenia; 3Pulse Wave Consulting, Saint Leu La Foret, France; 4Faculty of Electrical Engineering, User-adapted Communication and Ambient Intelligence Lab, University of Ljubljana, Ljubljana, Slovenia
Abstract: In occidental countries, peripheral arterial disease (PAD) is an important health issue; however, most subjects are asymptomatic (~50%) and therefore undiagnosed and untreated. Current guidelines recommend screening for PAD in primary care setting using ankle brachial index (ABI) in all patients with cardiovascular risks. This is, however, not performed strictly because the standard Doppler method is cumbersome and time-consuming. Here, we evaluate the accuracy and reproducibility of ABI measurements obtained by an improved automated oscillometric device, the MESI ABPI MD® device, and the standard Doppler method. ABI was measured in random order in a general practice with Doppler probes by two operators separately (ABI_dop) and twice with the MESI ABPI MD device (ABI_mesi). ABI_dop was calculated dividing the highest systolic blood pressure from both tibial and dorsalis pedis arteries by the highest systolic blood pressure of both brachial arteries. ABI_mesi was obtained automatically with simultaneous measurements on three extremities. According to ABI_dop, PAD was present in 10% of the 136 screened subjects (68.2±7.4 years). Interoperator coefficient of variation was 5.5% for ABI_dop, while the intrasubject coefficient of variation for ABI_mesi was 3.0%. ABI_mesi was correlated with ABI_dop (R=0.61, P<0.0001). The difference between the two techniques was 0.06±0.14 with ABI_mesi providing slightly higher values (P<0.0001) and negligible bias across the range (R=0.19, P<0.0001). Therefore, ABI_mesi ≤1 had a sensitivity of 85% and specificity of 96% to detect ABI_dop ≤0.9 and hence PAD. Doppler measurements took seven times longer than MESI ABPI MD measurements to be performed. In conclusion, MESI improved automated oscillometric method and offered a faster and repeatable measurement of ABI with only a small, clinically irrelevant overestimation of ABI value. The tested MESI ABPI MD-improved oscillometric system can be used as a screening tool for patients in general practice and would enable family doctors to comply with current guidelines for PAD.
Keywords: ankle brachial index, peripheral arterial disease, automated oscillometric method, Doppler method, screening tool
Background
The incidence of the peripheral arterial disease (PAD) in lower extremities is increasing with age in the population. In Western European countries, more than 20% of people older than 65 years have PAD in lower extremities either in symptomatic or asymptomatic phase.1 More than 50% of patients with present PAD are asymptomatic2 and are therefore untreated.3 Progress of the PAD in the lower extremities can lead to onset of symptoms. First, leg pain during activity (intermittent claudication) occurs. If not treated, symptoms worsen and progress to critical limb ischemia, ulcer, or gangrene. Even more importantly, patients with PAD have four- to six-fold higher risk of cardiovascular or cerebrovascular events in comparison to the healthy population.4–6 Progression and severity of PAD can be altered by risk factor management and preventive medical treatment.7
The measurement of ankle brachial index (ABI) is a reliable noninvasive diagnostic tool with high sensitivity and specificity to diagnose symptomatic and asymptomatic PAD.5,8 Current guidelines for management of PAD advise to measure ABI in all subjects >65 years, >50 years if they have hypertension, diabetes, and/or are currently smokers, and in subjects with clinical symptoms of PAD.3,7
ABI obtained by Doppler method is usually regarded as the gold standard,9 where the operator detects the return of blood flow with Doppler probe during the deflation of a proximal cuff. But there are some drawbacks of this Doppler method: it requires an experienced operator, is time-consuming,10–13 and does not allow measurement of transient state such as after a strandness test during which it is important to measure ABI just after walking.
MESI d.o.o. (Ljubljana, Slovenia) developed the MESI ABPI MD® system based on an improved oscillometric technology to provide an easy solution for PAD screening in clinical settings. The aim of this study was to evaluate the reproducibility and the accuracy of the MESI ABPI MD device from MESI compared with standard Doppler method to measure ABI in general practice population.
Methods
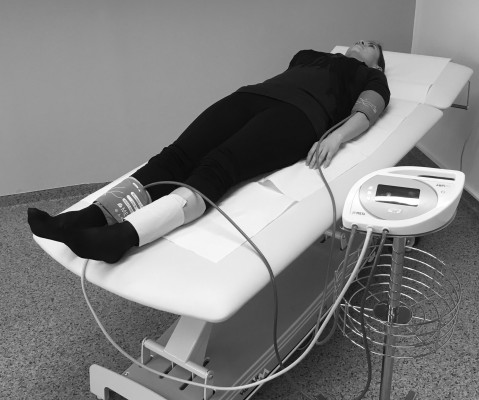
Subjects were approached at the general practitioner’s office during their regular visits. They were invited to participate in the study if they were 1) >65 years or 2) >50 years with smoking habit or diabetes or 3) had pain in the lower extremities during exercise. Subjects who fulfilled at least one of these criteria were included in the study, while subjects with known arrhythmia, upper extremity arteriovenous fistulas or symptomatic critical limb ischemia, ulcers or major lower leg amputations were excluded. All subjects provided written informed consent and the study protocol was approved by the Ljubljana University ethics committee and was in accordance with the Declaration of Helsinki. Permission was also obtained from the subject for the use of Figure 1.
  | Figure 1 Picture of the MESI ABPI MD® (MESI d.o.o., Ljubljana, Slovenia) device. |
Subject’s medical history was recorded by the general practitioner. After a few minutes of rest in supine position, four ABI measurements were obtained for each subject: one with the Doppler probe method and operator 1, one with the Doppler probe and operator 2, and two with the automatic oscillometric device (MESI ABPI MD). The order of the ABI method was chosen randomly, and the ABI results were hidden and not visible to the subjects or operators. There were two skilled operators who performed Doppler probe measurements on each subject separately and a third operator who performed both oscillometric measurements.
Measurement of ABI by means of Doppler probe method (ABI_dop) was performed according to the standard protocol9 with a calibrated sphygmomanometer and 8 MHz Doppler probes (Dopplex SD2, Huntleigh Healthcare Ltd, Cardiff, UK). ABI_dop was calculated as the ratio of the highest systolic blood pressure (SBP) obtained from both tibial and dorsalis pedis arteries at one ankle to the highest SBP of both brachial arteries. ABI_dop_L refers to ABI values obtained on the left leg, ABI_dop_R on the right leg. Measurement of ABI (ABI_mesi) with the improved automated oscillometric device (MESI ABPI MD; Figure 1) was performed according to the manufacturer’s instructions. In short, MESI ABPI MD system is based on simultaneous plethysmographic measurements through three cuffs, one conical cuff placed on the right or left arm, and two conical cuffs placed on each of the ankles. Precisely regulated system inflated and then deflated the three cuffs simultaneously at the same pressure level. Heart beats create oscillations on the plethysmographic signals, which are used to calculate blood pressure values from the three limbs using specific proprietary algorithms for the ankle blood pressure determination. The ankle cuffs measure oscillometric signals over the whole ankle and hence the highest SBP of all arteries within the ankle is detected.
Values of ABI_dop <0.90 were regarded as being a significant sign of PAD, whereas values >1.40 indicated incompressible ankle arteries and values in between were regarded as normal.
Time taken by operator 1, operator 2, and MESI ABPI MD to assess ABI was also measured.
Statistical analysis
Numerical data are presented as a mean ± standard deviation, while categorical data are expressed as percentage. Differences between measurements were tested with bilateral Student’s t-test. Coefficient of variation (CV) was defined as the mean standard deviation of measurements in a same subject divided by average of these measurements in a same patient. Relationship between variables was measured with the Pearson’s correlation coefficient and Bland–Altman plots were used to quantify the differences between measurements. Limits of agreement were determined by 95% confidence interval. Assuming that ABI values in a typical primary care should be between 0.6 and 1.6, we calculated that we needed at least 132 subjects to detect a difference of 0.04 with 95% confidence level and 90% power. All statistical analysis was performed using SPSS version 18 (SPSS Inc., Chicago, IL, USA).
Results
Out of 150 screened subjects, six had atrial fibrillation and were hence excluded from the final analysis. In 14 (9.3%) subjects, it was not possible to assess ABI with Doppler probe and/or oscillometric device due to critical limb ischemia or incompressible arteries. Patients’ characteristics of the 136 remaining subjects are presented in Table 1.
Results from Doppler method
Both operators were experienced in measuring SBPs using Doppler probe. However, they had a strong tendency to round numbers as 90% of the pressure values reported ended by 0 or 5. Interoperator differences were small with highest interoperator difference for the right posterior tibial artery being 3.5±15.9 mmHg. There was no trend in the interoperator differences across patients confirming that both operators were experienced in measuring ABI_dop and that neither operator experienced a learning curve. ABI_dop values varied from 0.5 to 1.7 with a mean ABI_dop of 1.1±0.2 for both operators. The difference between operator was not significant (0.01±0.1, P=0.107) and the interoperator CV for ABI_dop was 5.9% for each leg.
Results from MESI device
Intrapatient differences between repeated measurements with the MESI ABPI MD device were small: 1.8±6.6, 3.0±8.8, and 2.8±8.0 mmHg for right arm, left ankle, and right ankle, respectively. ABI_mesi varied from 0.57 to 1.46 with mean values of 1.19±0.15 and 1.20±0.14 on left and right side, respectively. CV of repeated measurements was 3.5% on left side and 3.2% on the right side, showing excellent repeatability.
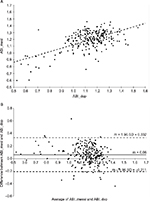
We compared the average of the two measurements of ABI_mesi_L with the average of ABI_dop_L from operators 1 and 2 as well as ABI_mesi_R with ABI_dop_R. As results were similar, they were pooled together, each subject having two data points (Figure 2A and B).
ABI_mesi was strongly correlated to ABI_dop (R=0.61, P<0.0001, Figure 2A). The error between the two methods exhibited a normal distribution and the Bland–Altman plot showed a mean difference of 0.06±0.14 (limits of agreement: −0.211 to 0.332, Figure 2B) representing a small overestimation (5.1%, P<0.0001) of ABI_mesi compared with ABI_dop. A small but significant bias across ABI range was observed with lower ABI value being slightly more overestimated than higher ABI value (R=0.19, P<0.0001).
The difference between Doppler and MESI ABPI MD device did not vary across recruitment (data not shown) confirming that MESI system does not drift with time.
To further evaluate the accuracy of the MESI ABPI MD device to screen for PAD, we looked at the number of patients with an ABI_dop ≤0.9 on at least one leg and found that 10% of the subjects had PAD on at least one leg according to the Doppler method. Using a threshold of 0.9 on ABI_mesi, MESI ABPI MD device diagnosed 6.8% of the subjects with a sensitivity of 57% and a specificity of 99%. However, several publications have advised to use ABI ≤1 for oscillometric techniques.14–17 In that case, MESI device reached 85% of sensitivity and 96% specificity to detect PAD in at least one leg.
Only 3.8% of the screened patients had an ABI_dop >1.4 (3% for ABI_mesi). This number is too small to calculate the specificity and sensitivity to detect arterial calcification.
Time spent to assess ABI
Time spent to measure ABI is presented in Figure 3. Confirming the experience of the operators, Doppler method took 14±1.54 minutes for operator 1 and 14±1.90 minutes for operator 2 (P=0.58) to measure arm and both left and right dorsalis pedis and tibial systolic pressures. Measurements with MESI ABPI MD took exactly 2 minutes whoever the patient and were hence complete seven times faster than the standard Doppler ABI measurement (P<0.001).
Discussion
This is the first study comparing the improved automated oscillometric MESI ABPI MD device from MESI d.o.o with the standard Doppler method. This device offers the opportunity to assess ABI easily from a proprietary method based on the analysis of oscillations from plethysmographic signals recorded through conical cuffs. Hence, there is no bias due to operator error. Even with experienced operators such as the ones we used in our study, there is a strong tendency to round up pressure values, which could lead to inaccuracy in ABI assessment. This is one of the various sources of errors which makes Doppler method having higher variability than oscillometric measurements.18 Our data confirm this as MESI variability was small (highest CV =3.5% on left ankle) compared with Doppler variability. Due to time constraints, we did not measure the intraoperator variability for Doppler measurements, but others19 have reported 8% for intraoperator and 9% for interoperator variability regardless of operator’s medical background.
Unlike for validation of brachial blood pressure electronic devices, there is no recognized protocol to test the accuracy of ABI devices. However, most publications reports: 1) the coefficient of correlation between the two techniques, 2) the difference between the two techniques, and 3) in a smaller proportion of studies, the sensitivity and specificity values of the oscillometric method in diagnosing PAD. In 2012, Verberk et al found 25 studies comparing oscillometric ABI determination with Doppler determination.18 Sixteen of them reported the coefficient of correlation which was on average 0.71±0.05, which is relatively similar to our coefficient of correlation of 0.61 (P<0.001). Eighteen studies reported the difference in ABI values assessed by the two methods with an average error of 0.02 indicating that in general, oscillometric determination of ABI gave slightly higher values than the Doppler method. Our results are also in agreement with this result as we observed a small overestimation of 0.06±0.14. This small overestimation of ABI with the MESI ABPI MD device is due to higher ankle systolic pressure values recorded on the ankles (5.8±14.8 mmHg on average compared with Doppler tibial or dorsalis pedis artery pressure, P<0.001). This could be explained by observer error: the time for the operator between hearing the Doppler signal and recording the pressure value from the sphygmomanometer might induce a lower systolic pressure value;18 however, we did not observe any differences between brachial SBP recorded with the MESI ABPI MD device and the Doppler method on the same arm.
The average sensitivity and specificity found by the meta-analysis by Verbeck et al were 69%±6% and 96%±0.8%, respectively.18 However, several authors have reported that using a cutoff value closer to 1 was better to diagnose PAD surely because of the systematic small overestimation of oscillometric method.14–17 When using 1 as a cutoff value, the MESI ABPI MD device sensitivity was 85% and 96% for specificity showing excellent detection of PAD. In our study, PAD diagnosis was solely based on Doppler ABI results. To truly evaluate sensitivity and specificity of MESI ABPI MD system, ABI results should be compared with PAD diagnosis from imagery techniques.
Both operators of our study took approximately 14 minutes to complete full measurement of ABI. Indeed, they had to successively inflate the cuff at least once on the arm and twice per leg for the tibial and dorsalis pedis arteries. In comparison, MESI ABPI MD took 2 minutes to perform measurement on the three cuffs simultaneously. This important discrepancy is intuitive and has been reported previously.18 What is more important is that our measurement did not take into account the required resting time. Indeed, with successive Doppler measurements, it is important to make sure all hemodynamic variables are stable before assessing ABI. While with MESI ABPI MD, systemic variations of blood pressure do not matter as both legs and arms are measured simultaneously.
There are some controversies regarding the use of oscillometric devices for ABI measurements; however, several dedicated ABI oscillometric devices have shown comparable results to the Doppler method.18 Some complain that if ankle pressure is particularly low, oscillometric might not be able to calculate ABI. This is, however, by itself, an important clinical diagnosis. It is also crucial to use specific ankle oscillometry technique and not a standard blood pressure designed for brachial pressure. Indeed, as lower leg geometry and anatomy are different from the upper arm, specific cuffs and specific pressure detection algorithms are necessary at the ankle level. Simultaneous measurements are also a plus in order to reduce measurements variability and “white coat” effect.18 MESI ABPI MD device has been designed in that respect and offers an accurate, easy, and rapid solution to avoid drawbacks of the Doppler method. In addition, MESI ABPI MD offers a comprehensive detection of critical limb ischemia (blood pressure <70 mmHg) by displaying a specific message (message E1) particularly relevant for patients with severe PAD.
Limitations
The main limitation of our study resided in the studied ABI range. Subjects recruitment was performed in general practice settings and we only diagnosed 10% of subjects with PAD. The distribution of ABI values is hence centered around normal ABI values with fewer data available <0.9 and >1.4. While further studies are required to verify MESI’s accuracy <0.9, our cohort represents typical primary care population and hence shows that the MESI device is suitable for such settings.
Conclusion
MESI ABPI MD device provides fast, accurate, and precise measurements of ABI in general practice. As ABI values are slightly overestimated and sensitivity compared with Doppler value is slightly lower, patients with borderline oscillometric results between 0.9 and 1 should be considered at very high risk of PAD. MESI ABPI MD can be accepted as an alternative to the Doppler method and be used as a large-scale screening tool for family doctors in order to comply with current guidelines for the diagnosis of PAD.
Acknowledgments
Part of this paper will be presented as a poster presentation with interim findings at the European Society of Hypertension congress on June 11, 2016. Organizers of the congress intend to publish the poster abstract in the August supplement issue of Journal of Hypertension.
Disclosure
SCM received financial compensation for her work on the analysis, the interpretation of the data, as well as editing this manuscript. MŠ has some shares in MESI d.o.o. The other authors report no conflicts of interest in this work.
References
Diehm C, Allenberg JR, Pittrow D, et al. Mortality and vascular morbidity in older adults with asymptomatic versus symptomatic peripheral artery disease. Circulation. 2009;120(21):2053–2061. | ||
Abramson BL, Huckell V, Anand S, et al. Canadian Cardiovascular Society Consensus Conference: peripheral arterial disease – executive summary. Can J Cardiol. 2005;21(12):997–1006. | ||
Norgren L, Hiatt WR, Dormandy JA, Nehler MR, Harris KA, Fowkes FG; TASC II Working Group. Inter-society consensus for the management of peripheral arterial disease (TASC II). J Vasc Surg. 2007;45(Suppl S):S5–67. | ||
Criqui MH, Langer RD, Fronek A, et al. Mortality over a period of 10 years in patients with peripheral arterial disease. N Engl J Med. 1992;326(6):381–386. | ||
Heald CL, Fowkes FG, Murray GD, Price JF; Ankle Brachial Index Collaboration. Risk of mortality and cardiovascular disease associated with the ankle-brachial index: systematic review. Atherosclerosis. 2006;189(1):61–69. | ||
Fowkes FG, Murray GD, Butcher I, et al; Ankle Brachial Index Collaboration. Ankle brachial index combined with Framingham Risk Score to predict cardiovascular events and mortality: a meta-analysis. JAMA. 2008;300(2):197–208. | ||
Rooke TW, Hirsch AT, Misra S, et al. 2011 ACCF/AHA focused update of the guideline for the management of patients with peripheral artery disease (updating the 2005 guideline): a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines. J Am Coll Cardiol. 2011;58(19):2020–2045. | ||
Khan NA, Rahim SA, Anand SS, Simel DL, Panju A. Does the clinical examination predict lower extremity peripheral arterial disease? JAMA. 2006;295(5):536–546. | ||
Lovelace T, Moneta G. Peripheral vascular diagnostic methods. In: Lanzer P, Topol E, editors. Pan Vascular Medicine SE - 27. Berlin/Heidelberg: Springer; 2002:398–419. | ||
Beckman JA, Higgins CO, Gerhard-Herman M. Automated oscillometric determination of the ankle-brachial index provides accuracy necessary for office practice. Hypertension. 2006;47(1):35–38. | ||
Meyer D, Bureau JM, Vu TD. [Ankle brachial index: motivations, training, and practices among 165 general practitioners in Île-de-France]. J Mal Vasc. 2014;39(1):18–25. | ||
Mohler ER 3rd, Treat-Jacobson D, Reilly MP, et al. Utility and barriers to performance of the ankle-brachial index in primary care practice. Vasc Med. 2004;9(4):253–260. | ||
Bendermacher BL, Teijink JA, Willigendael EM, et al. Applicability of the ankle-brachial-index measurement as screening device for high cardiovascular risk: an observational study. BMC Cardiovasc Disord. 2012;12:59. | ||
Guo X, Li J, Pang W, et al. Sensitivity and specificity of ankle-brachial index for detecting angiographic stenosis of peripheral arteries. Circ J. 2008;72(4):605–610. | ||
Clairotte C, Retout S, Potier L, Roussel R, Escoubet B. Automated ankle-brachial pressure index measurement by clinical staff for peripheral arterial disease diagnosis in nondiabetic and diabetic patients. Diabetes Care. 2009;32(7):1231–1236. | ||
Kollias A, Xilomenos A, Protogerou A, Dimakakos E, Stergiou GS. Automated determination of the ankle-brachial index using an oscillometric blood pressure monitor: validation vs. Doppler measurement and cardiovascular risk factor profile. Hypertens Res. 2011;34(7):825–830. | ||
Ichihashi S, Hashimoto T, Iwakoshi S, Kichikawa K. Validation study of automated oscillometric measurement of the ankle-brachial index for lower arterial occlusive disease by comparison with computed tomography angiography. Hypertens Res. 2014;37(6):591–594. | ||
Verberk WJ, Kollias A, Stergiou GS. Automated oscillometric determination of the ankle-brachial index: a systematic review and meta-analysis. Hypertens Res. 2012;35(9):883–891. | ||
Holland-Letz T, Endres HG, Biedermann S, et al. Reproducibility and reliability of the ankle-brachial index as assessed by vascular experts, family physicians and nurses. Vasc Med. 2007;12(2):105–112. |
 © 2016 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2016 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.