Back to Journals » Infection and Drug Resistance » Volume 13
Detection of Chromosomal and Plasmid-Mediated Quinolone Resistance Among Escherichia coli Isolated from Urinary Tract Infection Cases; Zagazig University Hospitals, Egypt
Authors Esmaeel NE , Gerges MA , Hosny TA , Ali AR, Gebriel MG
Received 25 November 2019
Accepted for publication 11 January 2020
Published 11 February 2020 Volume 2020:13 Pages 413—421
DOI https://doi.org/10.2147/IDR.S240013
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Suresh Antony
Noura E Esmaeel, 1 Marian A Gerges, 1 Thoraya A Hosny, 2 Ahmed R Ali, 3 Manar G Gebriel 1
1Medical Microbiology and Immunology Department, Faculty of Medicine, Zagazig University, Zagazig, Egypt; 2Clinical Pathology Department, Faculty of Medicine, Zagazig University, Zagazig, Egypt; 3Urology Department, Faculty of Medicine, Zagazig University, Zagazig, Egypt
Correspondence: Marian A Gerges
Medical Microbiology and Immunology Department, Faculty of Medicine, Zagazig University, 44519, Egypt
Tel +20 1003819530
Email [email protected]
Introduction: Resistance to fluoroquinolones (FQ) in uropathogenic Escherichia coli (UPEC) has emerged as a growing problem. Chromosomal mutations and plasmid-mediated quinolone resistance (PMQR) determinants have been implicated. Data concerning the prevalence of these determinants in UPEC in our hospital are quite limited.
Purpose: To investigate the occurrence and genetic determinants of FQ resistance in UPEC isolated from urinary tract infection (UTI) cases in Zagazig University Hospitals.
Patients and Methods: Following their isolation, the identification and susceptibility of UPEC isolates were performed by matrix-assisted laser desorption/ionization-time-of-flight mass spectrometer (MALDI-TOF MS). FQ resistance was detected by the disc diffusion method. Ciprofloxacin minimal inhibitory concentration (MIC) was determined using E-test. Chromosomal mutations in the gyrA gene were detected using polymerase chain reaction-restriction fragment length polymorphism (PCR-RFLP), and for detection of PMQR, a couple of multiplex PCR reactions were used.
Results: Among a total of 192 UPEC isolates, 46.9% (n=90) were FQ resistant. More than half of the isolates (57.8%) exhibited high-level ciprofloxacin resistance (MIC > 32 μg/mL). Mutations in gyrA were detected in 76.7% of isolates, with 34.4% having mutations at more than one site. PMQR determinants were detected in 80.1% of UPEC isolates, with aac(6ʹ)-Ib-cr gene being the most frequent found in 61.1% of isolates.
Conclusion: There is a high prevalence of both gyrA mutations and PMQR determinants among UPEC isolates in our hospital which contribute to high-level ciprofloxacin resistance, a finding that may require the revision of the antibiotics used for empirical treatment of UTI.
Keywords: gyrA mutations, qnr determinants, uropathogenic E. coli, ciprofloxacin resistance
Introduction
Escherichia coli (E. coli) is a common human pathogen that is frequently implicated in causing urinary tract infections (UTIs) such as cystitis and pyelonephritis.1 Fluoroquinolones (FQ) are broad-spectrum antibiotics that are commonly recommended for treatment of UTIs, in particular, those caused by genera and species of Enterobacteriaceae.2
Following their widespread use, resistance to FQ has emerged globally. This has increased the risk of treatment failure and furthermore, limited the selection of antibiotics used for empirical treatment of UTIs caused by Enterobacteriaceae.3,4
Fluoroquinolones tend to bind tightly to the bacterial enzymes (DNA gyrase and topoisomerase IV) inhibiting their activity in the control of DNA supercoiling and converting them into toxic enzymes that fragment the bacterial chromosome preventing DNA replication.5 E. coli traditionally resists FQ by the development of chromosomal mutations mainly in the quinolone resistance-determining regions (QRDRs) of the target genes; gyrA which encodes DNA gyrase and parC which encodes topoisomerase IV. In Gram-negative bacteria, DNA gyrase is more susceptible to inhibition by quinolones than is topoisomerase IV.6 Mutations at serine-83 (Ser-83) and asparagine-87 (Asp-87) in the gyrA gene are among the most often observed mutations in E. coli mutant strains.7
Although bacterial resistance to quinolones is principally mediated by chromosomal mutations, it can also be plasmid-mediated. Plasmid-mediated quinolone resistance has been reported first in 1998 from a Klebsiella pneumoniae strain in the University of Alabama at Birmingham Medical Center.8 Since then, it has been increasingly reported in most parts of the world and arisen as a significant concern.9,10
Generally, three mechanisms of plasmid-mediated quinolone resistance have been described: (i) qnr (qnrA, qnrB and qnrS) proteins that protect the target enzyme (DNA gyrase) against quinolone inhibition and encoded by qnr (quinolone resistance) determinants, (ii) aac(6ʹ)-Ib-cr gene, first discovered in 2003, which encodes a variant of aminoglycoside acetyltransferase enzyme that acetylates and inactivates ciprofloxacin and norfloxacin, and (iii) Efflux pumps associated with qepA gene which excretes hydrophobic fluoroquinolones (ciprofloxacin and norfloxacin).11
Plasmid-mediated resistance is usually associated with low-level resistance, yet it can confer high-level resistance by facilitating the selection of chromosomal mutation. In addition, it poses a major threat by allowing the rapid spread of resistance among different organisms.10
The aim of this study is to investigate the occurrence and genetic determinants of FQ resistance in E. coli isolated from urinary tract infection patients hospitalized in Zagazig University Hospitals which could help proper treatment choices.
Materials and Methods
A cross-sectional study was conducted over a period of 6 months (October 2018–March 2019) in Medical Microbiology and Immunology Department, Faculty of Medicine, Zagazig University and Clinical Pathology Department and the Urology Department, Zagazig University Hospitals.
Bacterial Isolates
Escherichia coli (E. coli) isolates were collected from urine specimens of hospitalized patients with suspected UTI from different wards of Zagazig University Hospitals, who had not yet received antibiotics, during the study period. To avoid testing multiple isolates from a single patient, E. coli was isolated in only one urinary culture from each patient. Urine specimens were collected by clean-catch midstream or from catheter in catheterized patients.
All urine specimens were immediately transported to the laboratory and the colony count semiquantitative method was performed according to surface streak procedure using calibrated loops onto the surface of MacConkey agar. The result of a single microorganism equal to or more than 105 CFU/mL was considered positive UTI. Identification up to species level and FQ resistance were done by matrix-assisted laser desorption/ionization-time-of-flight mass spectrometer (MALDI-TOF/MS) using the VITEK MS system (Biomérieux. Inc, Durham, USA). In brief, a fraction of a single colony of the freshly grown test isolates were smeared on the wells of disposable target slides to form a thin layer of the organism. Then, 1 µL of VITEK MS CHCA matrix solution (cyano-4-hydroxycinnamic acid) was applied over each sample and air-dried for 1–2 min at room temperature. The E. coli ATCC®8739TM strain (American Type Culture Collection Global Bioresource Center, Manassas, VA, USA) was used as a calibrator and internal identification control. It was inoculated on the calibration spots of each acquisition group. The target slide was then loaded into the VITEK MS system. Identification was done using a new database (v2.0) and MYLA software developed for in vitro diagnostic use. Identification results with confidence score values exceeding 90% were considered significant and displayed. E. coli isolates were maintained in glycerol broth at −20º C until use.
This study was conducted in accordance with the Declaration of Helsinki and was approved by the institutional review board (IRB) – Faculty of Medicine, Zagazig University. A written informed consent was obtained from each patient or the guardians of unconscious patients.
Phenotypic Detection of FQ Resistance
Collected E. coli isolates were subjected to phenotypic detection of FQ resistance by disc diffusion method using ciprofloxacin and levofloxacin antibiotic discs. Minimal inhibitory concentration (MIC) of ciprofloxacin was also determined by Epsillometer (E) test strips (Oxoid Limited, Basingstoke, Hants, England).
Disc Diffusion Method
Antimicrobial susceptibility testing was performed by the disc diffusion method (Modified Kirby–Bauer technique) using Muller Hinton agar following Clinical Laboratory Standards Institute (CLSI) guidelines.12 In addition to FQ discs (ciprofloxacin (CIP) 5 µg and levofloxacin (LVX) 5 µg), antimicrobial discs included that of carbapenems (Imipenem (IMP) 10 μg, Meropenem (MEM) 10 μg), aminoglycosides (Amikacin (AK) 30 μg, gentamicin (GEN) 10 μg), penicillin/β-lactamase inhibitor (amoxicillin/clavulanic acid (AMC) 20/10 μg), monobactam (Aztreonam (ATM) 30 μg), broad-spectrum cephalosporins (cefoxitin (FOX) 30 μg, ceftazidime (CAZ) 30 μg, cefepime (FEP) 30 μg), trimethoprim-sulfamethoxazole (SXT) 25 μg, and Nitrofurantoin (F) 300 μg, all were obtained from Oxoid Co. (Oxoid Limited, Basingstoke, Hampshire, England). E. coli ATCC®25922TM was used as a quality control strain (American Type Culture Collection Global Bioresource Center, Manassas, VA, USA).12 Multidrug resistance (MDR) phenotype was defined as the non-susceptibility to at least one agent in three or more antimicrobial categories, while extreme drug resistance phenotype (XDR) was defined as the non-susceptibility to at least one agent in all but two or fewer antimicrobial categories (ie, bacterial isolates remain susceptible to only one or two categories).13
Ciprofloxacin MIC Determination
For isolates found resistant to ciprofloxacin on primary antimicrobial susceptibility, E-test (Oxoid) strips were used to determine the MIC of isolates to ciprofloxacin (CIP 32 μg/mL - 0.002 μg/mL). Also, E. coli ATCC®25922TM was used as a quality control strain (American Type Culture Collection Global Bioresource Center, Manassas, VA, USA). Results were interpreted according to CLSI 2018 guidelines (susceptible, MIC ≤ 1 μg/mL, intermediate, MIC = 2 μg/mL, resistant, MIC ≥ 4 μg/mL).12
Genotypic Detection of FQ Resistance Determinants
E. coli isolates that were found resistant to FQ were subjected to polymerase chain reaction (PCR) amplification of the chromosomal QRDRs of gyrA, followed by restriction fragment length polymorphism (RFLP) to detect mutations in gyrA. Also, to multiplex PCR to detect plasmid-mediated quinolone resistance determinants (PMQR).
DNA Extraction
Extraction of bacterial DNA was performed using QIAamp® DNA Mini kit (Qiagen GmbH, Hilden, Germany) according to manufacturer instructions.
PCR-RFLP to Detect gyrA Mutations
PCR amplification of QRDRs of gyrA was done using Taq PCR Master Mix (Qiagen GmbH, Hilden, Germany). Primers and thermocycling conditions were adjusted as described previously to detect mutations at Ser-83 and Asp-87 in the gyrA gene.14 This is achieved by the introduction of an artificial restriction enzyme cleavage site into PCR products by using a primer-specific restriction site modification method, followed by restriction site digestion of the PCR product to detect these mutations. The forward primer, EC-GYRA-A (5`-CGCGTACTTTACGCCATGAACGTA-3`), and the reverse primer, EC-GYRA-HinfI (5`-ATATAACGCAGCGAGAATGGCTGCGCCATGCGGACAATCGAG-3`) were used to amplify a 164-bp DNA product. The amplification consisted of 35 cycles of denaturation (94°C, 60 s), annealing (52°C, 50 s), and extension (72°C, 50 s).
PCR products were further analyzed by RFLP using HinfI (Thermo Fisher scientific Inc) to detect point mutations at positions Ser-83 and Asp-87 of gyrA.14 For RFLP, a 10-mL mixture containing 8 mL of the PCR product, 1 mL of HinfI, and 1 mL of 10 X digestion buffer, supplied by the manufacturer, was incubated at 37° C for 1–16 hrs. Ten microliters digested fragments were run in 3% agarose gel stained with ethidium bromide to visualize bands under UV transilluminator. DNA ladder (50 pb and 25 bp) was used as a molecular weight marker. Digestion products according to the mutation site are listed in Table 1.14
 |
Table 1 HinfI PCR-RFLP Patterns of gyrA |
When a wild type (no mutation) of the gyrA gene (164 bp DNA product) is digested with HinfI, there are two cleavage sites. This produces fragments of 109, 15, and 40 bp. The 15 bp fragment runs off the gel and is not observed. When a strain has mutations at both Ser-83 and Asp-87, both HinfI cleavage sites are destroyed and the 164 bp DNA fragment remains undigested. On the other hand, when a strain has a single mutation at Ser-83, HinfI fails to digest the PCR product at Ser-83 site and only digests at Asp-87 producing only two fragments of 124 bp and 40 bp. Similarly, when a strain has a single mutation at Asp-87, HinfI fails to digest the PCR product at Asp-87site and only digests at Ser-83 producing only two fragments of 109 bp and 55 bp.14
Multiplex PCR to Detect PMQR
Detection of five PMQR genes was performed using two multiplex PCR, the first for qnrA, qnrB and qnrS, while the second for aac(6ʹ)-Ib-cr and qepA. Amplification was done using Taq PCR Master Mix (Qiagen GmbH, Hilden, Germany) and the primers listed in Table 2.
 |
Table 2 Primer Sets Used for Detection of PMQR Genes |
For the first reaction, extracted DNA (2 ul) was subjected to multiplex PCR in a 50 ul reaction mixture containing PCR buffer with 20 pmol of each of the six primers (Table 2) and 2.5 U of Taq polymerase. The Amplification was carried out under the following conditions: 10 min at 95ºC followed by 35 cycles of amplification consisting of 1 min at 95ºC, 1 min at 54ºC and 1 min at 72ºC then 10 min at 72ºC for the final extension.15 DNA fragments were analyzed by electrophoresis in 2% agarose gel.
The second multiplex PCR was carried out for 35 cycles at 94ºC for 30 sec, 55ºC for 30 sec and 72ºC for 30 sec.16,17 The resulting amplicons were examined by electrophoresis with 2% agarose gel.
Results
During the study period, a total of 192 E. coli isolates were collected from hospitalized patients suspected to have UTIs from different wards of Zagazig University Hospitals including intensive care units (ICUs). Among them, 90 (46.8%) isolates were FQ-resistant (all isolates found resistant to ciprofloxacin were also resistant to levofloxacin) and were included in our study.
The results of antibiotic susceptibility among FQ-resistant E. coli isolates are summarized in Table 3. All of them except for one isolate (n=89) expressed the MDR phenotype. The highest rates of resistance were detected for broad-spectrum cephalosporin (100%), trimethoprim-sulfamethoxazole (92.2%) and aztreonam (80%). Meanwhile, isolates were found sensitive to meropenem (100%), imipenem (81.1%) followed by nitrofurantoin (51.1%).
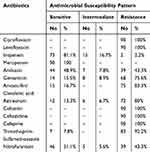 |
Table 3 Antibiotic Susceptibility Pattern of FQ-Resistant E. coli Isolates (No 90) |
Results of ciprofloxacin MIC are illustrated in Figure 1. More than half of the isolates exhibited high-level ciprofloxacin resistance with an MIC > 32 μg/mL being detected in 57.3% of FQ-resistant isolates.
 |
Figure 1 Ciprofloxacin MIC of FQ-resistant E. coli isolates. |
Mutations in the gyrA gene were observed in 69 (76.7%) FQ-resistant E. coli isolates, with 31 (34.4%) had mutations at both Ser-83 and Asp-87, 24 (26.7%) isolates had a mutation at Ser-83 and 14 (15.6%) had a mutation at Asp-87. Ciprofloxacin MIC levels of these isolates are summarized in Table 4.
 |
Table 4 Number of FQ-Resistant E. coli Isolates Harboring gyrA Gene Mutations and Their Corresponding Ciprofloxacin MIC (μg/mL) |
Out of the 90 FQ-resistant E. coli isolates, 73 (80.1%) isolates were positive for at least one PMQR gene. The most frequently occurred PMQR gene was aac(6ʹ)-Ib-cr, being found in 55/90 (61.1%) isolates, followed by qnrS 39/90 (43.3%), then by qnrB 20/90 (22.2%), and qepA 9/90 (10%). qnrA gene was not found in any of the isolates tested here. The aac(6ʹ)-Ib-cr gene was the most frequent either alone (14/90) or in combination with qnrS and/or qnrB (41/90). The qnrS and qnrB genes coexisted in 6 isolates.
Accordingly, FQ resistance was assumed to be due to gyrA mutations only in 16 (17.8%) isolates. Among them, 10 isolates (62.5%) were associated with high-level ciprofloxacin resistance (MIC > 32 μg/mL), and to be due to PMQR only in 20 (22.2%) isolates in which high-level ciprofloxacin resistance was detected in 3 isolates (15%). Both chromosomal mutations and plasmid determinants were present together in 53 (58.9%) isolates, of them 39 (73.6%) exhibited high-level ciprofloxacin resistance. Only one isolate was negative for both gyrA mutation and PMQR, although its ciprofloxacin MIC was 16 μg/mL (Table 5).
 |
Table 5 Number of FQ-Resistant E. coli Isolates with Resistance Determinants and Their Corresponding Ciprofloxacin MIC (μg/mL) |
Discussion
Uropathogenic E. coli (UPEC) infections and their treatment have a global burden in healthcare management. FQ constitutes an important line in the treatment of UPEC; however, their extensive use had led to a spread of quinolone-resistant strains worldwide, particularly in developing countries.18
In this study, the FQ resistance rate among UPEC was found to be 46.8%. This comes consistent with the previous published rate in Egypt (41.3%).19 On the global level, the rate varies from 35% to 57% in different geographical areas.20 A higher rate was reported in Iran (45.3–61.9%) and an extremely higher rate was documented in Pakistan (84.2%).21,22 These high rates could be explained by the wide and inappropriate use of quinolones as empirical treatment in UTIs. On the other hand, an obviously lower rate (5.3%) was reported by Sotto et al in France in E. coli isolates collected from hospitalized patients with acute UTI.23 This obvious variation has been documented previously where FQ resistance among UPEC has been shown to vary significantly in different regions and countries, recording the highest levels in developing countries and the lowest in North Europe.20
Nearly all FQ-resistant UPEC isolates in this study (89/90) were also resistant to at least one agent in at least three of the tested antimicrobial classes exhibiting an MDR profile or even an XDR one. This could be attributed to the coexistence of FQ resistance genes along with the genetic determinants of other antimicrobials, in particular, on mobile genetic elements. Similar findings were previously reported in different parts of the world.19,24,25 On the other hand, lower resistance rates of FQ-resistant UPEC to different antimicrobials were observed by Sotto et al in France.23 This variation could be due to the more adequate antibiotic prescription policy followed in developed countries as in France.
In our hospitals, the most commonly used antibiotics for the treatment of UTIs are quinolones, trimethoprim-sulfamethoxazole and cephalosporins. This may explain our antibiotic susceptibility results, where the highest rates of resistance were found to broad-spectrum cephalosporin (100%) and trimethoprim-sulfamethoxazole (92.2%). Meanwhile, we found the isolates remain sensitive to meropenem (100%), imipenem (81.1%) followed by nitrofurantoin (51.1%). Similar findings were previously reported by El-Mahdy et al in Egypt,19 which may reflect the implementation of a similar antibiotic policy in managing UTI in different Egyptian hospitals. Our result comes also similar to the findings of Majlesi et al who reported that FQ-resistant Enterobacteriaceae isolates showed multidrug resistance to other antimicrobial agents like amoxicillin-clavulanic acid, cefoxitin, ceftazidime, cefotaxime, cefepime, aztreonam, tetracyclines, rifampicin, and trimethoprim-sulfamethoxazole, but remain susceptible to carbapenem antibiotics.26
Regarding the mutations in the gyrA gene, 76.7% of FQ-resistant UPEC isolates, in this study, had at least a single mutation, with more than one-third of the isolates (34.4%) harbored double mutations at both Ser-83 and Asp-87. Mutation at Ser-83 only was detected in 24 (26.7%) isolates, while 14 (15.6%) isolates had a mutation at Asp-87. High frequency of gyrA mutations has been reported previously. Shenagari et al reported that 55.2% of UPEC isolates were found to have a mutation at codon 83.21 An even higher ratio was recorded by El-Mahdy et al where 73.5% of ciprofloxacin-resistant E. coli were found to have double mutations at both Ser-83 and Asp-87, with 26.5% having a single mutation at Asp-87.19 In Brazil, Minarini et al reported that 47.7% of ciprofloxacin-resistant E. coli isolates from both community- and hospital-acquired infections showed double mutations at gyrA gene.27 This supports the high frequency found in this work. In contrast, a lower incidence was reported previously in Japan by Ozeki et al where only 18.1% of E. coli isolates derived from UTI had mutation in gyrA, with the incidence of mutation at Ser-83, at Asp-87, and at both codons have been found to be 10.4%, 4.4% and 3.3%, respectively.14 In the study of Ozeki et al, all urinary-derived E. coli isolates were included, in contrast, we targeted only FQ-resistant isolates, which explains the higher incidence of mutations in this study.
Strains with double mutations, in this study, were commonly associated with high-level ciprofloxacin resistance (23/31, 74.2%). This ratio was found to be lower in strains with a single mutation in gyrA (26/38, 68.4%). This is consistent with what has been declared by different authors where a correlation between FQ MIC and the number of mutations has been found and that strains with double mutations were significantly more quinolone-resistant than those with a single mutation.7,14,28
Nevertheless, a strain with a single gyrA mutation could have a greater opportunity to acquire additional resistant mutations during therapy which may predispose to failure of therapy, particularly upon repeated usage of the same drug.6,29
Although chromosomal mutations play an important role in conferring a high level of quinolone resistance, researchers believe that PMQR may contribute to an increase in quinolone resistance in clinical isolates of E. coli.30
In this study, the prevalence of PMQR genes was 80.1%, with aac(6ʹ)-Ib-cr gene being the most prevalent (61.1%), followed by qnrS (43.3%) and qnrB (22.2%); qepA gene had a low frequency (10%) and qnrA was not detected. The aac(6ʹ)-Ib-cr gene either existed alone or in combination with qnrS and/or qnrB. The qnrS and qnrB genes coexisted in 6 isolates.
The incidence and distribution of PMQR genes differ widely. A high prevalence of qnr genes was reported by Le et al, particularly qnrS gene, among antibiotic-resistant members of intestinal Enterobacteriaceae.16 High prevalence of PMQR determinants was also reported by El-sayed Marei et al among both extended-spectrum beta-lactamases (ESBLs) and non-ESBLs producing Enterobacteriaceae isolates (93.1% and 76%, respectively) from UTI.31 In another study carried out by Khalil et al, a prevalence of PMQR genes of 85.7% was found among ESBLs producing Enterobacteriaceae clinical isolates, with aac(6ʹ)-Ib-cr gene being the most prevalent (72.7%), followed by qnr genes (39.3%),32 which comes consistent with our result.
Lower prevalence of PMQR was reported in other studies and in contrast to our finding, very low incidences (<1%) were reported by other researchers.6,9,33,34
This obvious difference in the prevalence of different PMQR genes could be associated with the geographical distribution of these genes, differences in antibiotic resistance pattern, antibiotic uses and probably infection control policies. In spite of these differences, aac(6ʹ)-Ib-cr gene appears to be widely prevalent whether in this study or in others. Additionally, the aac(6ʹ)-Ib-cr gene was observed more frequently than qnr genes among Enterobacteriaceae clinical isolates in different previous studies.2,26,35,36 This comes to support our result.
In addition to its wide distribution, the findings of this study further demonstrated that among PMQR determinants, aac(6ʹ)-Ib-cr existed alone in 14 isolates, 7 of them (50%) exhibited ciprofloxacin MIC higher than 32µg/mL (Supplementary data). Though this high level of resistance could be partly explained by the co-existence of gyrA mutations in 6 isolates, it may yet reveal the possible importance of this gene.
Despite the arising debate about the significance of aac(6ʹ)-Ib-cr gene, due to its limited targets (ciprofloxacin and norfloxacin) and its presence in ciprofloxacin-susceptible isolates,9 researchers still believe that this enzyme plays an important role in FQ resistance for many reasons. First, aac(6ʹ)-Ib-cr is more prevalent in clinical isolates of Enterobacteriaceae than qnr determinants. Secondly, although it confers a low-level of resistance, it can become high-level resistance when both qnr and aac(6ʹ)-Ib-cr coexist in the same cell. Thirdly, the presence of aac(6ʹ)-Ib-cr can facilitate the selection of chromosomal mutants with a higher level of ciprofloxacin resistance upon exposure to ciprofloxacin.37
The results of the current work revealed that high-level ciprofloxacin resistance (MIC > 32 µg/mL) was detected in 3/20 isolates having PMQR genes, in the absence of gyrA mutations. This finding could be partly attributed to the existence of other chromosomal mutations, eg, parC mutations, which were not tested in this work. However, the contribution of other chromosomal mutations solely to high-level ciprofloxacin resistance in gram-negative bacteria is controversial and has been found to be secondary to gyrA mutations.38,39 Furthermore, UPEC isolates that had PMQR in combination, eg, aac(6ʹ)-Ib-cr combined with qnr genes, in this study, exhibited higher ciprofloxacin MIC than those with a single PMQR (Supplementary data). The possibility of qnrS and qnrB gene co-existence has been previously reported, but this combination did not have any additional effect on ciprofloxacin resistance.40 In contrast, strains carrying the qnrS, qnrB and aac(6ʹ)-Ib-cr, in the absence of mutation in the gyrA gene, were found to be fully resistant to ciprofloxacin.16 Moreover, the combination of plasmid determinants working in a different manner, as qnrS and qnrB, protecting DNA gyrase, and aac(6ʹ)-Ib-cr, modifying ciprofloxacin, was found to increase the resistance to ciprofloxacin.16 This supports our findings and further demonstrates that the effect of PMQR genes on FQ MIC appears to be reliant on the number and type of PMQR genes carried by the bacteria.
Only one isolate in this study was negative for both gyrA mutations and PMQR, though being resistant to quinolone. This may be attributed to the simultaneous presence of mutations in parC gene or another different mechanism as impermeability of the membrane that is not investigated in our study.
Still, there are some limitations in our study as the lack of sequencing to confirm the results of PCR-RFLP and to detect the exact status of base substitution among the mutant strains. Also, the lack of analysis of chromosomal mutations at genes other than the gyrA gene, which could give a more complete picture of FQ resistance determinants in UPEC isolates.
Conclusion
Chromosomal mutations in gyrA gene and PMQR determinants are widely prevalent among UPEC isolates and they contribute to high-level FQ resistance which necessitates periodic surveillance of the microbial population and probably, revision of the antibiotic policy used in UTI treatment in our hospital.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Alanazi MQ, Alqahtani FY, Aleanizy FS. An evaluation of E. coli in urinary tract infection in emergency department at KAMC in Riyadh, Saudi Arabia: retrospective study. Ann Clin Microbiol Antimicrob. 2018;17(1):3. doi:10.1186/s12941-018-0255-z
2. Azargun R, Sadeghi MR, Barhaghi MH, et al. The prevalence of plasmid-mediated quinolone resistance and ESBL-production in Enterobacteriaceae isolated from urinary tract infections. Infect Drug Resist. 2018;11:1007–1014. doi:10.2147/IDR
3. Gajdács M, Ábrók M, Lázár A, Burián K. Comparative epidemiology and resistance trends of common urinary pathogens in a tertiary-care hospital: a 10-year surveillance study. Medicina (Kaunas). 2019;55(7):356.
4. Yekani M, Baghi HB, Sefidan FY, Azargun R, Memar MY, Ghotaslou R. The rates of quinolone, trimethoprim/sulfamethoxazole and aminoglycoside resistance among Enterobacteriaceae isolated from urinary tract infections in Azerbaijan, Iran. GMS Hyg Infect Control. 2018;13:Doc07.
5. Aldred KJ, Kerns RJ, Osheroff N. Mechanism of quinolone action and resistance. Biochemistry. 2014;53(10):1565–1574. doi:10.1021/bi5000564
6. Jacoby GA. Mechanisms of resistance to quinolones. Clin Infect Dis. 2005;41(Suppl 2):S120–S126. doi:10.1086/428052
7. Varughese LR, Rajpoot M, Goyal S, Mehra R, Chhokar V, Beniwal V. Analytical profiling of mutations in quinolone resistance determining region of gyrA gene among UPEC. PLoS One. 2018;13(1):e0190729. doi:10.1371/journal.pone.0190729
8. Martínez-Martínez L, Pascual A, Jacoby GA. Quinolone resistance from a transferable plasmid. Lancet. 1998;351(9105):797–799. doi:10.1016/S0140-6736(97)07322-4
9. Xue G, Li J, Feng Y, et al. High prevalence of plasmid-mediated quinolone resistance determinants in Escherichia coli and Klebsiella pneumoniae isolates from pediatric patients in China. Microb Drug Resist. 2017;23(1):107–114. doi:10.1089/mdr.2016.0004
10. Nazik H, Bektöre B, Öngen B, et al. Plasmid-mediated quinolone resistance genes in Escherichia coli urinary isolates from two teaching hospitals in Turkey: coexistence of TEM, SHV, CTX-M and VEB-1 type -lactamases. Trop J Pharm Res. 2011;10(3):325–333. doi:10.4314/tjpr.v10i3.9
11. Jacoby GA, Strahilevitz J, Hooper DC. Plasmid-mediated quinolone resistance. Microbiol Spectr. 2014;2(5). doi:10.1128/microbiolspec.PLAS-0006-2013
12. Clinical and Laboratory Standards Institute. M100 Performance Standards for Antimicrobial Susceptibility Testing,
13. Magiorakos AP, Srinivasan A, Carey RB, et al. Multidrug-resistant, extensively drug-resistant and pandrug-resistant bacteria: an international expert proposal for interim standard definitions for acquired resistance. Clin Microbiol Infect. 2012;18(3):268–281. doi:10.1111/j.1469-0691.2011.03570.x
14. Ozeki S, Deguchi T, Yasuda M, et al. Development of a rapid assay for detecting gyrA mutations in Escherichia coli and determination of incidence of gyrA mutations in clinical strains isolated from patients with complicated urinary tract infections. J Clin Microbiol. 1997;35(9):2315–2319. doi:10.1128/JCM.35.9.2315-2319.1997
15. Cattoir V, Poirel L, Rotimi V, Soussy CJ, Nordmann P. Multiplex PCR for detection of plasmid-mediated quinolone resistance qnr genes in ESBL-producing enterobacterial isolates. J Antimicrob Chemother. 2007;60(2):394–397. doi:10.1093/jac/dkm204
16. Le TM, Baker S, Le TP, et al. High prevalence of plasmid-mediated quinolone resistance determinants in commensal members of the Enterobacteriaceae in Ho Chi Minh City, Vietnam. J Med Microbiol. 2009;58(pt12):1585–1592. doi:10.1099/jmm.0.010033-0
17. Wareham DW, Umoren I, Khanna P, Gordon NC. Allele-specific polymerase chain reaction (PCR) for rapid detection of the aac (6′)-Ib-cr quinolone resistance gene. Int J Antimicrob Agents. 2010;36(5):476–477. doi:10.1016/j.ijantimicag.2010.07.012
18. Fasugba O, Gardner A, Mitchell BG, Mnatzaganian G. Ciprofloxacin resistance in community- and hospital-acquired Escherichia coli urinary tract infections: a systematic review and meta-analysis of observational studies. BMC Infect Dis. 2015;15:545. doi:10.1186/s12879-015-1282-4
19. El-Mahdy RH, Saleh MA, Aboelnour A. GyrA mutations in nosocomial ciprofloxacin-resistant Escherichia coli isolates associated with urinary tract infections. Int J Curr Microbiol App Sci. 2017;6(2):1902–1907. doi:10.20546/ijcmas.2017.602.215
20. Tandogdu Z, Cek M, Wagenlehner F, et al. Resistance patterns of nosocomial urinary tract infections in urology departments: 8-year results of the global prevalence of infections in urology study. World J Urol. 2014;32(3):791–801. doi:10.1007/s00345-013-1154-8
21. Shenagari M, Bakhtiari M, Mojtahedi A, Roushan ZA. High frequency of mutations in gyrA gene associated with quinolones resistance in uropathogenic Escherichia coli isolates from the north of Iran. Iran J Basic Med Sci. 2018;21(12):1226–1231. doi:10.22038/ijbms.2018.31285.7539
22. Muhammad I, Uzma M, Yasmin B, Mehmood Q, Habib B. Prevalence of antimicrobial resistance and integrons in Escherichia coli from Punjab, Pakistan. Braz J Microbiol. 2011;42(2):462–466. doi:10.1590/S1517-83822011000200008
23. Sotto A, De Boever CM, Fabbro-Peray P, Gouby A, Sirot D, Jourdan J. Risk factors for antibiotic-resistant Escherichia coli isolated from hospitalized patients with urinary tract infections: a prospective study. J Clin Microbiol. 2001;39(2):438–444. doi:10.1128/JCM.39.2.438-444.2001
24. Karlowsky JA, Hoban DJ, DeCorby MR, Laing NM, Zhanel GG. Fluoroquinolone-resistant urinary isolates of Escherichia coli from outpatients are frequently multidrug resistant: results from the North American Urinary Tract Infection Collaborative Alliance-Quinolone Resistance study. Antimicrob Agents Chemother. 2006;50(6):2251–2254. doi:10.1128/AAC.00123-06
25. Niranjan V, Malini A. Antimicrobial resistance pattern in Escherichia coli causing urinary tract infection among inpatients. Indian J Med Res. 2014;139(6):945–948.
26. Majlesi A, Kakhki RK, Nejad ASM, et al. Detection of plasmid-mediated quinolone resistance in clinical isolates of Enterobacteriaceae strains in Hamadan, West of Iran. Saudi J Biol Sci. 2018;25(3):426–430. doi:10.1016/j.sjbs.2016.11.019
27. Minarini LA, Darini AL. Mutations in the quinolone resistance-determining regions of gyrA and parC in Enterobacteriaceae isolates from Brazil. Braz J Microbiol. 2012;43(4):1309–1314. doi:10.1590/S1517-83822012000400010
28. Lindgren PK, Karlsson Å, Hughes D. Mutation rate and evolution of fluoroquinolone resistance in Escherichia coli isolates from patients with urinary tract infections. Antimicrob Agents Chemother. 2003;47(10):3222–3232. doi:10.1128/AAC.47.10.3222-3232.2003
29. van der Starre WE, van Nieuwkoop C, Paltansing S, et al. Risk factors for fluoroquinolone-resistant Escherichia coli in adults with community-onset febrile urinary tract infection. J Antimicrob Chemother. 2011;66(3):650–656. doi:10.1093/jac/dkq465
30. Tran JH, Jacoby GA, Hooper DC. Interaction of the plasmid-encoded quinolone resistance protein Qnr with Escherichia coli DNA gyrase. Antimicrob Agents Chemother. 2005;49(1):118–125. doi:10.1128/AAC.49.1.118-125.2005
31. El-sayed Marei Y, Mohamed Aboul-Ola O, Zakaria El-Azab S, Mohammad Kishk R, Mohammad Fouad M. Detection of plasmid-mediated quinolone resistance determinants in ESBLs and non-ESBLs producing Enterobacteriaceae from urinary tract infections. Ann Clin Immunol Microbiol. 2019;1(2):1008.
32. Khalil M, Elsherif R, Ghaith D, et al. Quinolone resistance detection by PCR-RFLP and multiplex-PCR among extended- spectrum β- lactamase producing Enterobacteriaceae. Int J Clin Med Microbiol. 2017;2:119. doi:10.15344/2456-4028/2017/119
33. Wang M, Tran JH, Jacoby GA, Zhang Y, Wang F, Hooper DC. Plasmid-mediated quinolone resistance in clinical isolates of Escherichia coli from Shanghai, China. Antimicrob Agents Chemother. 2003;47(7):2242–2248. doi:10.1128/AAC.47.7.2242-2248.2003
34. Jeong J-Y, Yoon HJ, Kim ES, et al. Detection of qnr in clinical isolates of Escherichia coli from Korea. Antimicrob Agents Chemother. 2005;49(6):2522–2524. doi:10.1128/AAC.49.6.2522-2524.2005
35. Kao CY, Wu HM, Lin WH, et al. Plasmid-mediated quinolone resistance determinants in quinolone-resistant Escherichia coli isolated from patients with bacteremia in a university hospital in Taiwan, 2001–2015. Sci Rep. 2016;6:32281. doi:10.1038/srep32281
36. Akya A, Lorestani RC, Elahi A, Ghadiri K. The impact of mutations in topoisomerase genes and the plasmid-mediated quinolone resistance (PMQR) determinants on the resistance to fluoroquinolones in Klebsiella pneumoniae. Arch Clin Infect Dis. 2017;14(4):e57290.
37. Robicsek A, Strahilevitz J, Jacoby GA, et al. Fluoroquinolone-modifying enzyme: a new adaptation of a common aminoglycoside acetyltransferase. Nat Med. 2006;12(1):83. doi:10.1038/nm1347
38. Heisig P. Genetic evidence for a role of parC mutations in development of high-level fluoroquinolone resistance in Escherichia coli. Antimicrob Agents Chemother. 1996;40(4):879–885. doi:10.1128/AAC.40.4.879
39. Lindbäck E, Rahman M, Jalal S, Wretlind B. Mutations in gyrA, gyrB, parC, and parE in quinolone-resistant strains of Neisseria gonorrhoeae. APMIS. 2002;110(9):651–657. doi:10.1034/j.1600-0463.2002.1100909.x
40. Hu FP, Xu XG, Zhu DM, Wang MG. Coexistence of qnrB4 and qnrS1 in a clinical strain of Klebsiella pneumoniae. Acta Pharmacol Sin. 2008;29(3):320–324. doi:10.1111/j.1745-7254.2008.00757.x
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
