Back to Journals » Cancer Management and Research » Volume 13
Delayed Breast Cancer Detection in an Asian Country (Taiwan) with Low COVID-19 Incidence
Received 4 April 2021
Accepted for publication 15 July 2021
Published 28 July 2021 Volume 2021:13 Pages 5899—5906
DOI https://doi.org/10.2147/CMAR.S314282
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 5
Editor who approved publication: Dr Sanjeev K. Srivastava
Chen-Pin Chou,1– 3 Huey-Shyan Lin4
1Department of Radiology, Kaohsiung Veterans General Hospital, Kaohsiung, Taiwan; 2Department of Medical Laboratory Sciences and Biotechnology, Fooling University, Kaohsiung, 807, Taiwan; 3Department of Nursing, Shu-Zen Junior College of Medicine and Management, Kaohsiung, Taiwan; 4Department of Health-Business Administration, School of Nursing, Fooyin University, Kaohsiung, Taiwan
Correspondence: Chen-Pin Chou
Department of Radiology, Kaohsiung Veterans General Hospital, 386 Ta-Chung 1st Road, Kaohsiung, 813, Taiwan
Tel +886-7-3422121 ext 76254
Fax +886-7-3507581
Email [email protected]
Purpose: To assess whether the COVID-19 pandemic delayed breast cancer diagnosis in Taiwan, an Asian country with a low COVID-19 incidence.
Methods: The monthly volume of breast biopsies and breast cancers during the COVID-19 pandemic (during January 21 and July 31, 2020) was compared to the same period in 2019 (pre-COVID-19).
Results: Taiwan recorded a lower COVID-19 incidence rate (20.2 cases per million population) than other Asian countries. The screen-detected lesions accounted for 55% and 36% of 2019 and 2020 total biopsied lesions, respectively. Total breast biopsy, mammography-guided, and ultrasound-guided biopsies decreased by 17%, 23%, and 14%, respectively, from pre-COVID-19 to COVID-19. Monthly differences were significant in total biopsy (p=0.03), mammography-guided biopsy (p=0.04), and a benign pathology result after breast biopsy (p< 0.01). Nearly 46% decline was noted in the biopsy results of non-invasive breast cancer in 2020. The number of total breast cancers and early breast cancers (stages 0 and 1) decreased by 10% and 38%, respectively, during pandemic. Individuals with early breast cancer accounted for 71% and 49% of the total diagnosed breast cancer in the pre-COVID-19 and COVID-19 periods, respectively (p< 0.001).
Conclusion: The pandemic significantly delayed early breast cancer detection in Taiwan despite low COVID-19 incidence.
Clinical Trial Registration: ClinicalTrials.gov, NCT04750018.
Keywords: COVID-19, breast cancer, mammography, pandemic, breast biopsy
Introduction
COVID-19 (Coronavirus disease 2019) has spread rapidly and globally since December 31, 2019.1 Taiwan confirmed its first COVID-19 case on January 21, 2020. Based on their experience with the severe acute respiratory syndrome (SARS) epidemic of 2003, Taiwan’s medical teams were quick to recognize the crisis and effectively established rapid public health responses to the COVID-19 outbreak.2 However, based on their experience with hospital-acquired SARS infections in 2003,3 several Taiwanese hesitated to visit hospitals for routine breast screening or seek consult for their breast symptoms during the COVID-19 pandemic.
Breast cancer is the most common cancer, with the fourth highest mortality rate among Taiwanese women.4 Population-based biennial mammography screening for Taiwanese women aged 45–69 is associated with a 41% reduction in breast cancer mortality.5 The delay of breast cancer diagnosis may reduce the chance of early detection and intervention. Our previous study in Taiwan showed that the diagnostic and screening mammography exams decreased to 6% and 51%, respectively, during the COVID-19 pandemic.6 Delayed management could be an independent predictor of more invasive breast cancer.7 A previous study of breast cancer patients in China indicated that delays of more than 6 months resulted in a worse disease-free survival.8 Since the delay of breast cancer diagnosis is critical in clinical practice, we investigated the impact of the COVID-19 pandemic on breast cancer diagnosis and cancer staging at a medical center in Taiwan.
Materials and Methods
Ethics Statement
This retrospective study was approved by the hospital’s institutional review board, and the requirement for informed consent was waived. Medical records of patients receiving breast biopsy procedures were retrieved from a 1455-bed public academic medical center that provides screening and diagnostic breast exams in Taiwan.
Patients
The test period of the COVID-19 pandemic was between January 21 and July 31, 2020. Moreover, the control period of the pre-COVID-19 pandemic was between January 21 and July 31, 2019, before there was any report about the virus. All patients receiving a breast biopsy with the concordant histopathological results (benign breast disease or breast cancer) and complete medical records were included in the current study. Confirmed COVID-19 case numbers in Taiwan were acquired from Taiwan’s National Health Command Center.9 Anonymized records of breast biopsies and cancer registration in a single institution in southern Taiwan in the pre-COVID-19 and COVID-19 periods were reviewed.
Study Design
Patients receiving biopsies with a BI-RADS assessment of 4 or 5 from screening or diagnostic mammography or ultrasound were analyzed. We further categorized the biopsy data as ultrasound-guided or mammography-guided biopsy based on procedure coding. Ultrasound-guided core needle biopsy (CNB) was performed for breast lesions visible on ultrasound. Mammography-guided vacuum-assisted breast biopsy (VAAB) was performed for lesions identified only on mammography, such as microcalcification or architectural distortion. The biopsy results of our study were classified into benign breast disease and breast cancer (including stage zero). Benign breast disease is defined as any non-malignant breast condition and includes a wide range of pathological entities. Breast cancer staging of patients was according to the 8th edition of the American Joint Committee on Cancer (AJCC) system.10 Stage 0 or non-invasive breast cancer is used for ductal carcinoma in situ (DCIS). A pre-treatment clinical stage, assigned before neoadjuvant therapy, was determined by biopsy results of the primary breast cancer and axillary lymph nodes supplemented by imaging exams. Patients without neoadjuvant chemotherapy were staged based on surgical pathology and imaging examination. We further categorized stage 0–1 as early breast cancer and stage 2–4 as non-early breast cancer.
Statistical Analysis
The monthly differences of biopsy procedures and pathology results were calculated by subtracting the monthly pre-COVID-19 case number from the monthly number in the same months during the COVID-19 period. The normal distribution of quantitative data was tested using the Kolmogorov–Smirnov test. Categorical variables were analyzed using the chi-square test for the association between categorical variables, paired t-test for the monthly data, and unpaired t-test for the age difference. We calculated the monthly difference of breast cancers and monthly new COVID-19 cases with bivariate (Pearson’s R) correlations to evaluate the direction and strength.11 The Phi coefficient was used as a measure for the correlation of categorical results.12 We evaluated biopsy efficiency in detecting cancers by calculating positive biopsy rates (number of positive biopsies divided by total number of biopsies). Statistical analysis was performed using SPSS software (version 20.0; IBM Corp., Armonk, NY, USA). All statistical tests are based on two-tailed probability and a significance level set at α = 0.05. When the p-value is smaller than 0.05, the result is statistically significant.
Results
As of July 31, 2020, Taiwan had a total of 466 laboratory-confirmed cases and seven deaths from COVID-19 (Figure 1). Taiwan has reported a lower COVID-19 incidence rate (20.2 cases per million population) compared to other Asian countries (286 and 279 per million in Japan and Korea at the same period, respectively).
There were 477 biopsy lesions in 430 women and 398 biopsy procedures in 355 women during the pre-COVID-19 and COVID-19 periods, respectively (Table 1). The age (mean ± SD) of breast biopsied cases between pre-COVID-19 and COVID-19 (51.1 ± 11.7 years and 51.2 ± 12.8 years) was not significantly different (p=0.97). The screen-detected lesions accounted for 55% and 36% of 2019 and 2020 total biopsied lesions, respectively. Total breast biopsy, mammography-guided, and ultrasound-guided biopsies decreased by 17%, 23%, and 14%, respectively, from the previous year. The monthly numbers of total biopsy and mammography biopsy were significantly lower in 2020 than in 2019 (p=0.03 and 0.04 respectively). Among the seven study months, only February had more total breast biopsy cases than the previous year because the Chinese Lunar New Year holiday 2019 fell in February (Table 2). A 43% decrease of total biopsy lesions in 2020 January could be due to the start of the COVID-19 outbreak coinciding with the Chinese Lunar New Year holiday. The monthly biopsy procedures showed a 35% decline in March 2020 when the pandemic peaked. As the incidence of COVID-19 declined, the monthly biopsy rate approached the previous year’s rate (−6%) in June 2020.
 |
Table 1 Clinical Characteristic of Breast Biopsy Lesions, Percentage Change and Significance of Monthly Difference During Pre-COVID-19 in 2019 (N=477) and COVID-19 in 2020 (N=398) |
 |
Table 2 Cross-Tabulation of Monthly Breast Biopsy Pathology and Monthly New COVID-19 Cases During 2019 (N=477) and 2020 (N=398) |
Pathology results revealed 128 and 115 breast cancers in pre-COVID-19 and COVID-19 period, respectively. There was no significant difference in the age (mean ± SD) of breast cancer women between the pre-COVID-19 and COVID-19 (55.6 ± 11.7 years and 58.7 ± 12.7 years, p=0.06). The positive biopsy rates were 27% and 29% for pre-COVID-19 and COVID-19 period, respectively. The monthly number of benign pathology result was significantly less in 2020 than in 2019 (p<0.01). The total breast cancers on biopsies showed a 10% decline in 2020, but the monthly number was significantly different in comparison with the previous year (p=0.52). Nearly 46% decline was noted in the biopsy results of non-invasive breast cancer in 2020. There was a moderate association between the proportion of cancers diagnosed and COVID-19 when the pandemic was slowing down in July (Phi=0.13). The monthly difference in breast cancer cases showed a highly significant negative linear correlation with monthly new COVID-19 cases (r=0.79, p<0.001) (Figure 2).
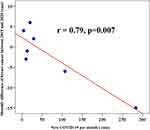 |
Figure 2 Correlation between new monthly COVID-19 cases (N=466) and monthly difference of confirmed breast cancer cases between 2019 (N=128) and 2020 (N=115). |
Ultimately, 123 cases in 2019 and 111 cases in 2020 were included for cancer staging. About 78% and 74% of the breast stagings were based on surgical pathology reports both in the pre-COVID-19 and COVID-19, while others were based on pre-treatment clinical evaluation before neoadjuvant chemotherapy. Stage 0 and stage 2 breast cancers in 2020 showed a 51% decrease and 84% increase, respectively (Figure 3). The proportion of cancer staging categories (stage 0 to 4) was significantly different between the pre-COVID-19 and COVID-19 era (p=0.01). Individuals with early breast cancer accounted for 71% and 49% of the total diagnosed breast cancer in the pre-COVID-19 and COVID-19 periods, respectively (p<0.001). There were a 30% decline and a 58% increase in the numbers of early and non-early breast cancer, respectively, in the pandemic period.
 |
Figure 3 Percentage change of the number in breast cancer staging categories (stage 0–4) from pre-COVID-19 to COVID-19. |
Discussion
In this single-institution study, a marked decline in breast biopsies occurred in March 2020 due to the peak of Taiwan’s pandemic. Taiwan’s national data on screening mammography and referral of abnormal screening also dropped rapidly in March 2020.13 The numbers of total breast biopsies, biopsy-proven breast cancer, and early breast cancer decreased by 17%, 10%, and 30%, respectively, from the pre-COVID-19 to the COVID-19 period. The decline of biopsy procedures clearly resulted in a delay in early breast cancer detection in the seven-month pandemic period. Monthly mammography-guided biopsy significantly decreased because fewer women underwent screening mammography during the pandemic.6 The monthly difference in confirmed breast cancers has a high negative correlation with monthly new COVID-19 cases. A 46% decline of non-invasive cancer during biopsy resulted in a 30% decrease of early breast cancer in 2020. The month number of total biopsies, mammography-guided biopsy and biopsy of benign breast disease were significantly lower in 2020 than the previous year.
For many cancers, treatment delays of 2–6 months could lead to a considerable proportion of patients with early stage breast cancer progressing from curable to incurable.14 In a non-pandemic period, advanced-stage breast cancer could be related to multiple factors, such as delayed diagnosis, insurance status, facility distance, or pathology grade.7 A cross-sectional study in the United States showed a 46% decrease in the combined weekly numbers of 6 cancers during the pandemic, and breast cancer was the most affected, with a 51% reduction.15 The Netherlands Cancer Registry reported an average weekly decline of 27% in all national cancer incidences at the end of February 2020 due to delayed or postponed hospital visits.16 In this seven-month study, we did not find a significant decrease in the number of breast cancers in 2020 for Taiwan.
A meta-analysis including 38 studies of breast cancer found that delays of 3–6 months were significantly associated with worse survival.17 An average of 11 months of diagnostic delay of breast cancer resulted in a 1.3-fold larger breast cancer size and a 37% decrease in stage 0 breast cancer.18 We also found a 51% decrease in stage 0 in the seven-month COVID-19 period. As non-emergency mammography services and non-essential hospital visits were largely discouraged during the pandemic,19,20 suspension of this routine breast screening may predispose women at a higher risk of poor outcomes.21 Biennial screening mammography in Taiwan was substantially reduced during the pandemic, with a 45% decrease in the total volume during this period.6 In addition, the national diagnostic referral rate of abnormal screening mammography dropped to 7% in April 2020.5 A survey of the United States breast cancer survivors found that 44% of the participants also delayed clinical cancer care during the pandemic.22 Considerable increases in preventable breast cancer deaths are expected due to screening and diagnostic delays during the COVID-19 pandemic.23
Our study found a 51% decrease in early breast cancer during the seven-month pandemic period. There is also other evidence that the proportion of early breast cancers decreased during COVID-19.24,25 Such delayed diagnosis and biopsy may have late and long-term effects on cancer treatment.23 A national population-based modeling study from the UK estimated the impact of the diagnostic delay of breast cancer for the 12-month pandemic period and will cause a 1.0–1.1% decrease in cancer survival.23
There was no official recommendation about women with suspicious breast lesions from the Taiwan Breast Cancer Society during this period, and mammography screening and routine clinical breast care continued in Taiwan during the pandemic. However, several medical societies and breast cancer experts urged the medical staff to delay most breast imaging during the pandemic crisis. European breast specialists recommended that routine mammographic exams should be suspended except for symptomatic women.19 Biopsy of BI-RADS 4A lesions and short-term follow-up imaging examinations are considered “not immediately necessary”.7 To balance the risk of delayed treatment and risk of virus infection, the Breast Cancer Consortium developed a risk-stratified tool to generate a score based on clinical factors and recommendations during the pandemic.26
During this pandemic, tight surveillance can reduce suspected COVID-19 transmission, but it may also diminish women’s willingness for breast biopsy and further referral service after an abnormal screening mammogram. There are many potential barriers to breast cancer diagnosis. Barriers to early detection of breast cancer are particularly relevant due to the lack of medical information, fear of disease, decreased accessibility of the health care system, and other socio-cultural impediments. The pandemic could also be a temporary barrier to breast cancer biopsy and early cancer diagnosis. Government and health care providers should encourage women with suspicious symptoms to seek medical help and further work-ups during the pandemic.
This study has several limitations. First, this retrospective study was conducted from a single large institution in Taiwan, and Taiwan’s prevalence of COVID-19 is much lower than that in other countries,1 these results may have limited generalizability to women in other affected countries. The clinical impact of COVID-19 on breast cancer diagnoses could be unprecedented and long-lasting in countries with high prevalence. Second, multiple causes may have led to delayed breast biopsy. However, within the current cohort, we were unable to accurately discriminate the causes of delay. As hospital resources and medical staffs were not limited during this pandemic period in Taiwan, psychological fear and anxiety of patients could directly impact the delayed diagnosis. Finally, the long-term effects of delayed breast cancer diagnosis during the COVID-19 pandemic or indications for subsequent waves and future disasters require data that is not currently available and is an important topic of future research.
Conclusions
Despite the lower prevalence of COVID-19 in Taiwan, the pandemic over a seven-month has significantly reduced the number of early breast cancer.
Abbreviations
COVID-19, Coronavirus disease 2019; SARS, severe acute respiratory syndrome; CNB, core needle biopsy; VAAB, vacuum assisted breast biopsy.
Data Sharing Statement
The data that support the findings of this study are available on reasonable request from the corresponding author. Some data are not publicly available due to their containing information that could compromise the privacy of research participants.
Ethical Approval
The study was approved by the institutional review board (IRB) of Kaohsiung Veterans General Hospital (KSVGH20-CT7-26). Written informed consent was waived.
Author Contributions
All authors made substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data; took part in drafting the article or revising it critically for important intellectual content; agreed to submit to the current journal; gave final approval of the version to be published; and agree to be accountable for all aspects of the work.
Funding
This work was supported by grants from Kaohsiung Veterans Hospital Research Found (VGHKS108-158, VGHKS109-146).
Disclosure
Chen-Pin Chou and Huey-Shyan Lin have no conflict of interest.
References
1. Cheng HY, Jian S-W, Liu D-P, et al. Contact tracing assessment of COVID-19 transmission dynamics in taiwan and risk at different exposure periods before and after symptom onset. JAMA Intern Med. 2020;180(9):1156–1163. doi:10.1001/jamainternmed.2020.2020
2. Wang CJ, Ng CY, Brook RH. Response to COVID-19 in Taiwan: big data analytics, new technology and Proactive Testing. JAMA. 2020;323(14):1341–1342.
3. Chang HJ, Huang N, Lee C-H, et al. The impact of the SARS epidemic on the utilization of medical services: SARS and the fear of SARS. Am J Public Health. 2004;94(4):562–564. doi:10.2105/AJPH.94.4.562
4. Chou CP, Pan H-B, Hsu G-C, et al. Assessing the first 3 years of Taiwan’s nationwide population-based mammography screening program. Breast J. 2012;18(5):498–499. doi:10.1111/j.1524-4741.2012.01289.x
5. Yen AM, Tsau HS, Fann JC, et al. Population-based breast cancer screening with risk-based and universal mammography screening compared with clinical breast examination: a propensity score analysis of 1429890 Taiwanese women. JAMA Oncol. 2016;2(7):915–921. doi:10.1001/jamaoncol.2016.0447
6. Chou CP, Pan HB, Yang TL, et al. Impact of the COVID-19 pandemic on the volume of mammography examinations in Southern Taiwan. Breast J. 2020. doi:10.1111/tbj.14019 [Epub ahead of print]
7. Ward WH, DeMora L, Handorf E, et al. Preoperative delays in the treatment of DCIS and the associated incidence of invasive breast cancer. Ann Surg Oncol. 2020;27(2):386–396. doi:10.1245/s10434-019-07844-4
8. Huo Q, Cai C, Zhang Y, et al. Delay in diagnosis and treatment of symptomatic breast cancer in China. Ann Surg Oncol. 2015;22(3):883–888. doi:10.1245/s10434-014-4076-9
9. Taiwan Centers for Disease Control Daily press release of COVID-19. Available from: https://www.cdc.gov.tw/En.
10. Giuliano AE, Edge SB, Hortobagyi GN. Eighth Edition of the AJCC cancer staging manual: breast cancer. Ann Surg Oncol. 2018;25(7):1783–1785. doi:10.1245/s10434-018-6486-6
11. Cohen J. Statistical Power Analysis for the Behavioral Sciences.
12. Akoglu H. User’s guide to correlation coefficients. Turk J Emerg Med. 2018;18(3):91–93. doi:10.1016/j.tjem.2018.08.001
13. Peng S-M, Yang K-C, Chan WP, et al. Impact of the COVID-19 pandemic on a population-based breast cancer screening program. Cancer. 2020;126(24):5202–5205. doi:10.1002/cncr.33180
14. Sud A, Torr B, Jones ME, et al. Effect of delays in the 2-week-wait cancer referral pathway during the COVID-19 pandemic on cancer survival in the UK: a modelling study. Lancet Oncol. 2020;21(8):1035–1044. doi:10.1016/S1470-2045(20)30392-2
15. Kaufman HW, Chen Z, Niles J, et al. Changes in the number of US patients with newly identified cancer before and during the coronavirus disease 2019 (COVID-19) pandemic. JAMA Netw Open. 2020;3(8):e2017267. doi:10.1001/jamanetworkopen.2020.17267
16. Dinmohamed AG, Visser O, Verhoeven RHA, et al. Fewer cancer diagnoses during the COVID-19 epidemic in the Netherlands. Lancet Oncol. 2020;21(6):750–751. doi:10.1016/S1470-2045(20)30265-5
17. Richards MA, Westcombe AM, Love SB, et al. Influence of delay on survival in patients with breast cancer: a systematic review. Lancet. 1999;353(9159):1119–1126. doi:10.1016/S0140-6736(99)02143-1
18. Tartter PI, Pace D, Frost M, et al. Delay in diagnosis of breast cancer. Ann Surg. 1999;229(1):91–96. doi:10.1097/00000658-199901000-00012
19. Curigliano G, Cardoso MJ, Poortmans P, et al. Recommendations for triage, prioritization and treatment of breast cancer patients during the COVID-19 pandemic. Breast. 2020;52:8–16. doi:10.1016/j.breast.2020.04.006
20. Dietz JR, Moran MS, Isakoff SJ, et al. Recommendations for prioritization, treatment, and triage of breast cancer patients during the COVID-19 pandemic. the COVID-19 pandemic breast cancer consortium. Breast Cancer Res Treat. 2020;181(3):487–497. doi:10.1007/s10549-020-05644-z
21. Moy L, Toth HK, Newell MS, et al. Response to COVID-19 in breast imaging. J Breast Imaging. 2020;2(3):180–185. doi:10.1093/jbi/wbaa025
22. Papautsky EL, Hamlish T. Patient-reported treatment delays in breast cancer care during the COVID-19 pandemic. Breast Cancer Res Treat. 2020;184(1):249–254. doi:10.1007/s10549-020-05828-7
23. Maringe C, Spicer J, Morris M, et al. The impact of the COVID-19 pandemic on cancer deaths due to delays in diagnosis in England, UK: a national, population-based modelling study. Lancet Oncol. 2020;21(8):1023–1034. doi:10.1016/S1470-2045(20)30388-0
24. The American Society of Breast Surgeons. ASBrS and ACR joint statement on breast screening exams during the COVID-19 pandemic; 2020. Available from: https://www.breastsurgeons.org/news/?id=45.
25. Shim E, Tariq A, Choi W, et al. Transmission potential and severity of COVID-19 in South Korea. Int J Infect Dis. 2020;93:339–344. doi:10.1016/j.ijid.2020.03.031
26. Smith BL, Nguyen A, Korotkin JE, et al. A system for risk stratification and prioritization of breast cancer surgeries delayed by the COVID-19 pandemic: preparing for re-entry. Breast Cancer Res Treat. 2020;183(3):515–524. doi:10.1007/s10549-020-05792-2
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.

