Back to Journals » Cancer Management and Research » Volume 11
Cytoreductive surgery with hyperthermic intraperitoneal chemotherapy for peritoneal mesothelioma: patient selection and special considerations
Authors Enomoto LM , Shen P, Levine EA, Votanopoulos KI
Received 21 December 2018
Accepted for publication 26 March 2019
Published 7 May 2019 Volume 2019:11 Pages 4231—4241
DOI https://doi.org/10.2147/CMAR.S170300
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Lu-Zhe Sun
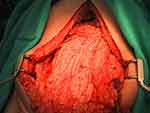
Laura M Enomoto, Perry Shen, Edward A Levine, Konstantinos I Votanopoulos
Wake Forest Baptist Medical Center, Medical Center Boulevard, Winston Salem, NC 27157, USA
Abstract: Malignant peritoneal mesothelioma (MPM) is a rare, aggressive malignancy that typically presents with vague symptoms, ascites, and/or diffuse peritoneal studding. Despite findings of advanced disease within the peritoneal cavity, spread beyond the abdomen is uncommon. Although advances in systemic chemotherapy have been made, cytoreductive surgery (CRS) and hyperthermic intraperitoneal chemotherapy (HIPEC) remain the mainstay of treatment. Median overall survival of approximately 50 months with CRS/HIPEC has been demonstrated, with age, gender, histologic subtype, peritoneal carcinomatosis index, comorbidities, nodal and extra-abdominal metastases, and completeness of cytoreduction all playing a role in prognosis. In patients with refractory malignant ascites and unresectable disease, complete resolution of ascites and improvement in quality of life have been demonstrated with palliative HIPEC. In appropriately selected patients, CRS/HIPEC plays a critical role in the treatment and palliation of MPM.
Keywords: malignant peritoneal mesothelioma, cytoreductive surgery, hyperthermic intraperitoneal chemotherapy, peritoneal surface disease
Introduction
Mesothelioma is an unusual malignancy of the serosal membrane, with potential involvement including the pleura, peritoneum, pericardium, and tunica vaginalis testes. First described over a century ago by Miller and Wynn,1 malignant peritoneal mesothelioma (MPM) is a rare lesion, with an estimated incidence of approximately 400 new cases per year in the United States.2 MPM typically presents with vague symptoms related to ascites and/or diffuse peritoneal studding with uncommon spread beyond the abdomen. Unlike its more common pleural counterpart, most research on MPM includes single-institution case series or multi-institutional cohort studies, with no randomized controlled trials. This review provides an overview of the epidemiology, diagnosis, treatment options, and prognosis of MPM.
Epidemiology
With an incidence in the United States of 1.94 and 0.41 cases per 100,000 for men and women, respectively, mesothelioma is a relatively uncommon disease.3,4 The vast majority of mesothelioma arises from the pleura, with only 7–30% of cases arising from the peritoneum.3–6 In contrast to pleural mesothelioma where there is a significant predominance of men diagnosed with the disease, there is an equal distribution of MPM among men and women.3,4 Mesothelioma has been linked to radiation,7 infection with simian virus 40,8 and mineral exposure, specifically erionite,9 but the most common and well-known carcinogen remains asbestos exposure.10,11 Unlike pleural mesothelioma where asbestos exposure accounts for approximately 80% of cases,10,12 MPM is less clearly associated with asbestos, if at all, and patients present at a younger age.13–15 Only 33–50% of patients with MPM report any prior asbestos exposure,10,11 and time and duration of exposure do not correlate with the development of the disease.16 It is noteworthy that at our institution, ferruginous, or asbestos bodies have never been found in any pathologic specimens from resections of MPM. Risk of developing MPM due to exposure to other minerals or pollutants has not been well quantified simply due to the rarity of the disease.
Diagnosis
Typically, MPM is diagnosed between 40 and 65 years of age17 and may present with vague, nonspecific symptoms that can be quite variable depending on the extent and distribution of disease throughout the peritoneum. Patients most commonly complain of increasing abdominal distension and abdominal pain. In the majority of patients, the increase in abdominal girth is due to ascites.18,19 Abdominal pain is generally diffuse and nonspecific, although occasionally a palpable mass or malignant bowel obstruction is discovered.19,20 Early satiety, weight loss, and nausea are also common complaints. Occasionally, MPM is discovered incidentally during laparoscopy for other indications.21 Because of the nonspecific presentation of MPM, diagnosis is often significantly delayed. Average time from onset of symptoms to diagnosis is 4–6 months.22 Not surprisingly, most patients have diffuse disease throughout the abdomen by the time of diagnosis; however, hematogenous and nodal metastases are rare occurrences.23
Diagnosis of MPM first begins with a thorough history and physical, with careful attention to asbestos and chemical exposures. Potential physical examination findings may include a protuberant abdomen with a fluid wave or palpable mass. Serum chemistry and tumor makers have a limited role. CA-125 may be elevated, but this is nonspecific for diagnosis and best used only as a marker for disease recurrence or progression.24,25
The most common modality used in detecting MPM is contrast-enhanced CT scan, although MRI with contrast can also be a useful alternative. MPM appears as a contrast enhancing, heterogeneous, solid, soft tissue mass in the peritoneum or omentum.26,27 It often lacks a distinct primary site as well as lymph node involvement or extra-abdominal metastasis, which may help to differentiate it from other malignancies.28 Peritoneal thickening, omental caking, and scalloping of solid organs indicative of tumor infiltration are often discovered,27 and ascites is present in over 60% of the patients (Figure 1).29,30 If the disease infiltrates the small bowel mesentery, the mesentery may have a pleated appearance while the mesenteric vessels have an uncharacteristically straight course.31 Late findings of MPM include small bowel obstruction and replacement of mesenteric fat by solid tumor (Figure 2).32
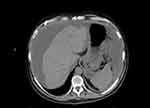 | Figure 1 CT scan demonstrating liver scalloping and ascites. |
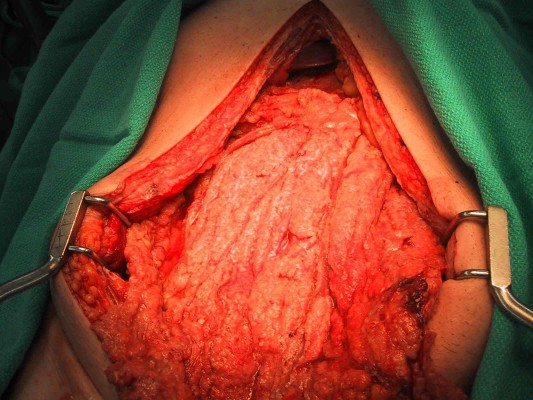
 | Figure 2 Extensive epithelioid malignant peritoneal mesothelioma involving the omentum. |
To definitively diagnose MPM, pathologic evaluation is required. As the majority of patients present with ascites, it is tempting to send this fluid for cytologic examination. However, due to the low number of malignant cells in the fluid, such analysis has a low diagnostic yield and is often inconclusive.10,19,33 Even if diagnostic paracentesis is suggestive of MPM, a pathologic specimen is still required for immunohistochemical staining to confirm a diagnosis. Fine-needle aspiration of peritoneal implants can confirm a diagnosis, but for improved accuracy, the preferred diagnostic modality is core-needle biopsy or direct tissue sampling by diagnostic laparoscopy.34 Diagnostic laparoscopy also offers the advantage of direct visualization of the abdominal cavity with an improved assessment of tumor burden, as CT scans often underestimate the volume of disease.28 If undertaken, it is incumbent upon the surgeon to define the extent of disease as well as to obtain tissue sufficient for accurate pathologic analysis.
MPM is divided into three histopathological subtypes: epithelioid, sarcomatoid, and biphasic. Approximately 75% of MPM is epithelioid, 25% is biphasic, and sarcomatoid is rare and associated with very poor outcomes.35 Histologically, epithelioid MPM cells resemble normal mesothelial cells in a tubulopapillary or trabecular pattern with rare mitotic figures.31,36 Because of occasional signet ring cells and desmoplastic response, it can be difficult to distinguish from adenocarcinoma.37 In contrast, sarcomatoid MPM has tightly packed spindle cells with malignant osteoid, chondroid, or muscular elements. As the name suggests, the biphasic subtype contains both epithelioid and sarcomatoid cellular components, with each contributing to at least 10% of the overall histology.31,36 There are also indolent varieties of peritoneal mesothelioma, such as cystic and tubopapillary, which may not progress. The utility of expert pathologic consultation in these cases cannot be understated.
Because cellular histology is often similar to other tumors, immunohistochemical staining plays an important role in the diagnosis of MPM. No single marker is specific for MPM, but panels of antibodies are used to differentiate MPM from other tumors with similar cellular features, such as papillary serous carcinoma of the peritoneum, serous ovarian carcinoma, colorectal adenocarcinoma of the peritoneum, and borderline serous tumors.16 MPM stains positive for cytokeratin 5/6 (CK 5/6), calretinin, vimetin, epithelial membrane antigen, Wilms tumor 1 (WT-1), mesothelin, and anti-mesothelial cell antibody-1.38–40 Negative staining for CEA, Ber-EP4, thyroid transcription factor 1, PAX-2, LeuM1, Bg8, and B72.3 supports the diagnosis of MPM. Analysis for BAP1 mutation can also be helpful in assessing newly diagnosed patients. Current histopathologic recommendations include using two mesothelioma markers and two carcinoma markers for diagnosis.38–40
Pathologic characteristics of MPM are also prognostic even within the epithelioid group. We have found that histomorphologic features of the epithelioid subtype of MPM convey strong prognostic information.37 Specifically, using nuclear features and mitotic rate, the epithelioid MPM cases can be divided into low-risj and high-risk groups with significantly different 5-year survival rates after CRS/HIPEC of 57% versus 21% survival at 5 years.
Staging
The most widely used and accepted classification scheme to quantify the peritoneal disease burden is the peritoneal carcinomatosis index (PCI). In this system, the abdomen is divided into nine regions by two transverse planes and two sagittal planes (Figure 3). The regions are numbered in a clockwise fashion with 0 at the umbilicus and 1 encompassing the space beneath the right diaphragm. The small bowel is divided into four regions (upper and lower jejunum, upper and lower ileum) and numbered 9–12. Each region is assigned a lesion size (LS) score based on the amount of disease present. If no macroscopic tumor is present, the region is assigned LS 0. If the maximum diameter of the lesions is less than 5 mm, the region is given an LS 1. Tumor with a maximum diameter greater than 5 mm but less than 5.0 cm is assigned LS 2, and any tumor greater than 5.0 cm or confluence is given LS 3. The total score for all 13 regions is the PCI, with a minimum of 0 and maximum of 39.
 | Figure 3 Regions for calculating the peritoneal carcinomatosis index (PCI). |
Due to its diffuse spread throughout the abdomen and rare nodal or extra-abdominal metastatic spread, MPM does not logically fit into typical tumor-node-metastasis (TNM) staging systems. The 8th edition of the American Joint Committee on Cancer (AJCC) staging manual has a staging system for pleural mesothelioma, but does not have a staging system for MPM.41 To address this issue, a novel TNM staging system was proposed by Yan and colleagues.42 In this system, T was assigned based on the extent of disease burden quantified by intraoperative PCI and divided into four subgroups: T1 (PCI 1–10), T2 (PCI 11–20), T3 (PCI 21–30), and T4 (PCI 31–39). Node status (N) was assigned based on presence (N1) or absence (N0) of positive lymph nodes on histopathology of surgical specimens. Any extra-abdominal metastasis discovered on pre-operative imaging was assigned M1. Stage I disease included T1N0M0, stage II included T2-3N0M0, and stage III included T4N0M0 and N1 or M1 disease.42 Using this staging system, 5-year survival for stage I, II, and III disease was 87%, 53%, and 29%, respectively.42
Treatment
MPM is an aggressive disease and without treatment is uniformly fatal, with an estimated survival of 6–16 months from time of diagnosis.22,43 Due to its rarity, there are no randomized controlled trials evaluating the best treatment strategies. Recommendations for therapy are based on single institutional cohort studies and retrospective data from multi-institutional registries. Systemic chemotherapy, immunotherapy, and surgical resection are potential treatment strategies. Precision medicine for advanced disease is an evolving option. For patients who are not candidates for surgical resection but suffer from refractory ascites, HIPEC can, in selected patients, provide durable palliation. The use of bidirectional chemotherapy or pressurized intraperitoneal aerosol chemotherapy (PIPAC) to convert unresectable MPM to resectable disease has also recently been suggested.
Systemic chemotherapy
Most of the data on systemic therapy for MPM is extrapolated from experience with pleural mesothelioma. Early trials of systemic chemotherapy for MPM used a doxorubicin-based regimen, but demonstrated a measurable response in only 43% of the patients.44 Of those who responded, median overall survival (OS) was 22 months; median OS for those with stable or progressive disease was 5 months.44 Since that time, a randomized clinical trial demonstrating longer median OS, longer disease-free progression, and higher rate of clinical response using pemetrexed plus cisplatin in treatment of pleural mesothelioma has prompted further study in MPM.45
Efficacy of pemetrexed alone or in combination with cisplatin on surgically unresectable MPM was reported by Janne et al.46 They found a median survival of 13.1 months for patients who received combination pemetrexed and cisplatin, compared to 8.7 months for those who received pemetrexed alone. Additionally, they showed the response rate by RECIST criteria for patients who received the combination was greater than for patients who received pemetrexed alone (30% versus 19%, respectively), and all patients with a complete response received the combined treatment. Pemetrexed was well-tolerated, with low rates of grade 3 or 4 toxicities. These results established pemetrexed in combination with cisplatin as first-line systemic chemotherapy for MPM.
Other chemotherapeutic drug combinations have also been investigated. Campbell and colleagues studied carboplatin instead of cisplatin in combination with pemetrexed and demonstrated a similar efficacy, with a 24% objective response rate and a 76% disease control rate.47 As carboplatin is often better tolerated than cisplatin, they proposed the use of carboplatin in older patients and for palliation. Gemcitabine in combination with pemetrexed was investigated as part of a larger study for pleural mesothelioma, but results were dismal.48 Due to toxicity, only 75% of the patients completed the planned treatment, and response rate and disease control rate were inferior to that of platinum-based regimens. As a result, pemetrexed in combination with a platinum agent remains first-line systemic treatment.
A trial reported at the European Society of Medical Oncology meeting from the Francophone trials group evaluated the utility of adding bevacizumab to pemetrexed and platinum for pleural mesothelioma.49 That study found that adding bevacizumab increased the median OS from 2.7 months to nearly 19 months. As a result, this three-drug regimen has become the current standard for MPM at many centers, including ours.
Immunotherapy
Immunotherapeutic approaches are being considered in MPM, similar to other malignancies. Tremelimumab, an anti-CTLA-4 agent, was studied as a second-line agent in patients with MPM who progressed on a platinum-based regimen.50 A modest benefit was shown, with a median OS of 10.7 months and median progression-free survival of 6.2 months. Investigations targeting epidermal growth factor receptor and phosphatidylinositol-3-kinase/mammalian target of rapamycin pathways are underway.51–54
Surgical resection
Despite advances in chemotherapy and immunotherapy, surgical resection provides the mainstay for treatment of MPM. A study of patients treated in the US and recorded in the National Cancer Database found that only 50% of MPM patients underwent CRS with or without HIPEC, and CRS/HIPEC offered the best survival when compared to CRS alone, systemic chemotherapy alone, or observation.55 Other studies have shown prolonged survival in well-selected patients, demonstrating a median survival of 34–92 months and 5-year survivals of 29–59%.17,19,21,55–63 There is a wide range of surgeon variability and CRS/HIPEC technique, although the overall goal of resecting all intra-abdominal disease is the same. Our techniques have been published in detail elsewhere, but are briefly outlined below.64
At our institution, prior to CRS/HIPEC, we first confirm a histologic diagnosis of MPM with a pathologic second opinion. Patients with the sarcomatoid variant of MPM are not candidates for resection; however, epithelioid and highly selected biphasic cases are.35 Exclusion criteria for CRS/HIPEC include comorbid conditions that significantly decrease functional status, extra-abdominal metastasis, or poor performance status (ECOG >2).
An estimation of residual tumor after CRS is important for prognosis and treatment and also determines eligibility for CRS/HIPEC. Two scoring systems are commonly used to quantify residual tumor after CRS: the completeness of cytoreduction (CC) score and the residual disease score (R score) from the AJCC staging manual.41 When no peritoneal disease is seen after CRS, a CC score of 0 is assigned. CC-1 indicates peritoneal nodules less than 2.5 mm in diameter remaining after CRS. A CC-2 is assigned if nodules are between 2.5 mm and 2.5 cm, and CC-3 indicates nodules greater than 2.5 cm or confluence of an unresectable tumor. CC-0 and CC-1 are considered a complete cytoreduction because CC-1 tumor size is thought to be penetrable by intraperitoneal chemotherapy.65 Alternatively, the R score quantifies residual disease by resection margin. After CRS, an R0 resection is considered complete macroscopic resection with negative margins on final pathology.41 Complete macroscopic resection with positive microscopic margins is classified as R1. An R2 resection is considered an incomplete macroscopic resection and is subdivided based on the maximal size of residual disease (R2a ≤5 mm, R2b 5 mm to 20 cm, R2c >2 cm). Since the peritoneal (radial) margins are positive by definition in MPM, we classify all complete cytoreductions as R0/R1. If a patient has a tumor burden so extensive on preoperative imaging or diagnostic laparoscopy as to preclude an R2a resection or better, at our institution they are not a candidate for CRS/HIPEC,57,64 although palliative HIPEC for refractory ascites may still be a consideration. Patients with voluminous ascites or high PCI are referred for upfront systemic chemotherapy aiming to downstage their volume of disease and increase the potential of complete cytoreduction in the future.
Anesthesia is secured with arterial line monitoring, and nasogastric and urinary catheters are routinely placed. If the patient has significant pelvic disease volume or a complex history of prior surgery, we arrange for temporary external ureteral stents to be placed at the outset of the case to facilitate retroperitoneal dissection. A wide prep including the lower chest is performed, and antibiotics and venous thrombosis prophylaxis are routine. We start with a midline laparotomy incision to thoroughly explore the abdomen and quantify the PCI. We perform a routine supracolic omentectomy and resection of all gross disease. Peritoneal stripping and resection of intra-abdominal organs are performed only as indicated by the presence of visible disease. Small tumor implants on the small bowel or mesentery are treated with resection, electrofulguration or ultrasonic surgical aspiration if they are too numerous or diffuse to be removed with small bowel resection.
Following CRS, we use a closed abdomen HIPEC technique and perfuse with cisplatin according to the National Cancer Institute (NCI)–described protocol with sodium thiosulfate given intravenously.58 Due to a paucity of trials and comparison studies, there is no standardized HIPEC technique, and a variety of chemoperfusion regimens are currently used. In their series, Deraco et al found no statistically significant difference between cisplatin plus mitomycin versus cisplatin plus doxorubicin on OS or progression-free survival (PFS).60 In a study conducted at our institution, Blackham et al compared DFS, PFS, event-free survival (EFS), and OS in patients who underwent HIPEC with mitomycin versus cisplatin.57 Prior to 2004, we perfused with 30–40 mg/m2 of mitomycin; however, based on data from the NCI, we began using cisplatin in 2004. When comparing survival with cisplatin or mitomycin perfusion, we demonstrated a statistically significant OS benefit at 1, 2, and 3 years in patients perfused with cisplatin (80% vs 47%, 80% vs 47%, and 80% vs 42%, respectively).57 Median OS for cisplatin and mitomycin was 40.8 months and 10.8 months, respectively, although this difference did not reach statistical significance. DFS, PFS, and EFS showed a trend of better outcomes for those perfused with cisplatin, but likely due to the small number of patients in the study and shorter follow-up period of the cisplatin cohort, these differences were not significant.
Recent work by the RENAPE study group showed improved OS when a two-drug combination was used versus one chemotherapeutic agent.66 This retrospective cohort study used the RENAPE database and included 249 patients with MPM from 20 tertiary referral centers. Six commonly used chemoperfusion regimens were investigated: cisplatin alone, cisplatin + doxorubicin, cisplatin + mitomycin, mitomycin alone, oxaliplatin alone, and oxaliplatin + irtinotecan. There was no significant difference in OS among the groups; however, when comparing patients who received a single chemotherapeutic agent to patients who received two, OS was significantly better in patients who received a combined regimen (HR 0.54, 95% CI 0.31–0.95). There was no difference in major morbidity between patients who received one drug versus those who received two chemotherapeutic agents (29.5% vs 29.8%, p=0.16).
Precision medicine
Using tumor DNA to identify actionable genetic mutations for use in adjuvant treatment is an emerging strategy in precision oncology. At our institution, work is underway to develop micro-engineered 3D tumor organoids from fresh tissue specimens to provide patient-specific models with which treatment optimization can be performed in vitro prior to initiation of adjuvant therapy. Specifically, Mazzocchi and colleauges have demonstrated the viability of this organoid platform in tumor specimens resected from two patients with MPM.67 They showed the results of in vitro chemotherapy on the organoids mimicked the response to chemotherapy observed in the patients themselves. Moreover, they identified a specific genetic mutation in one patient which conferred susceptibility to a non-standard treatment and further confirmed its effectiveness in tumor regression. Although a limited study, the results are promising for a personalized treatment strategy in MPM, and potentially other diseases.
Refractory malignant ascites
Malignant ascites is a common comorbid condition accompanying peritoneal surface disease and can occur with a wide range of neoplasms, including MPM.68,69 Loss of proteins and electrolyte disorders can cause diffuse edema, and the accumulation of fluid can facilitate sepsis, all contributing to a significant loss in patients’ quality of life.70 Treatment options include diuretics, paracentesis, immunotherapy, and shunts, all with varying, but limited, degrees of efficacy and morbidity.
HIPEC is a potential treatment strategy for patients who have refractory malignant ascites who are not candidates for curative CRS.69,71 Our institution examined the efficacy of HIPEC in controlling malignant ascites in patients in whom complete CRS was attempted but was not completed as a result of the distribution or volume of disease.71 Malignant ascites was found in 299 patients that underwent 310 procedures. The majority of the patients had an appendiceal primary (46%), followed by colorectal (17%), and MPM (15%). Most patients underwent a 120-min perfusion (78%) with mitomycin C (83%). Complete resolution of malignant ascites occurred in 93% of the patients, and 84% of these still had a residual macroscopic disease (R2 resection). In the 7% of the patients where the ascites did not completely resolve, the majority (86%) were R2 resections. There was no statistically significant difference in resolution of ascites based on resection status, primary tumor, perfusion duration, or perfusion agent, suggesting that resolution of malignant ascites is more likely a function of HIPEC rather than CRS. We also suggest that if ascites is unexpectedly discovered at the time of a planned CRS/HIPEC in which only an R2b or R2c resection can be achieved, patients may still benefit from proceeding with the HIPEC portion of the procedure to control the ascites. In cases where a complete CRS is not possible, organ resections should be avoided.
Bidirectional chemotherapy and PIPAC
In patients who are deemed surgically unresectable due to high tumor burden, current treatment modalities are limited. Two groups in France, however, have proposed intraperitoneal chemotherapy strategies aimed at decreasing overall tumor burden to convert unresectable MPM to disease amenable to CRS/HIPEC. After diagnostic laparoscopy demonstrated unresectability, LeRoy and colleagues placed intraperitoneal catheters with subcutaneous ports through which they instilled multiple cycles of intraperitoneal pemetrexed combined with intravenous (IV) cisplatin/carboplatin or intraperitoneal oxaliplatin combined with IV gemcitabine.72 After a median of 5 cycles, 73.3% of the patients had a reduction of PCI and went on to CRS/HIPEC; in the remaining 26.7%, the peritoneal disease remained unresectable. In a study with similar aim, Alyami et al reported their results with PIPAC in decreasing intraperitoneal disease in a heterogenous group of unresectable tumor primaries, including MPM.73 After performing an exploratory laparoscopy, a pressurized aerosol containing cisplatin followed by doxorubicin, oxaliplatin alone, or mitomycin C alone, was applied intraperitoneally using a high-pressure injector inserted into the abdomen through a trocar. PIPAC was alternated with systemic chemotherapy every 6–8 weeks for multiple cycles. By the third PIPAC cycle, PCI decreased in 64.5% of the patients, but ultimately only 11.0% of patients went on to CRS/HIPEC. Clearly, more experience and long-term follow-up will be required for both modalities, but they may provide potential treatment options in patients with otherwise limited alternatives.
Prognosis
Due to the rarity of the disease, the best data on MPM following CRS/HIPEC stem from large single-institution studies, multi-institutional registries, and national databases. The largest multi-institutional registry of patients with MPM treated with CRS/HIPEC included eight international institutions during a 10-year period and accrued 405 patients.23 The median OS was 53 months, and 1-, 3-, and 5-year survival rates were 81%, 60%, and 47%, respectively. Epithelioid subtype, absence of lymph node metastasis, CC0/CC1 resection, and HIPEC itself were independently associated with improved survival. The overall complication rate was 46%, of which 31% were grade 3 or 4 complications, and perioperative mortality was 2%. Another multi-institutional study with patients from three US institutions included 211 patients and demonstrated a median OS of 38.4 months with a 5-year survival rate of 41%.61 Independent predictors of survival included age, sex, histology, resection status, and chemoperfusate. A similar perioperative mortality rate (2.3%) and complication profile were found.
More recently, a meta-analysis that included 20 studies and 1,047 patients found a median OS ranging from 19 to 92 months, median PFS of 11–28 months, and median DFS from 7.2 to 40 months.62 OS at 1 and 5 years was 84% and 42%, respectively. There was a wide range of morbidity (8.3–90%) and mortality (0–20%), however, likely related to the steep learning curve in some reporting institutions.
Histologic subtype of MPM has remained one of the most consistent factors in predicting survival. Multiple studies have demonstrated that the epithelioid subtype confers a more favorable OS (51.5 months)74 compared to sarcomatoid and biphasic subtypes (10.5 months).23,74–76 The sarcomatoid subtype carries such a dismal prognosis that most centers, including ours, consider it a contraindication to CRS/HIPEC, as there is no proven survival benefit.74 Recent work at our institution, however, has demonstrated that for the biphasic subtype, long-term survival can be achieved in patients with a complete cytoreduction (CC-0) and HIPEC.35 The previously nihilistic view of the biphasic subtype likely stemmed from its rarity and traditional practice to group it with the sarcomatoid subtype. Median OS for biphasic subtypes with a CC-0 resection was 6.8 years but dropped off steeply with an incomplete resection (2.8 years for CC-1 resection). This study demonstrated that the biphasic subtype should not be considered an absolute contraindication to CRS/HIPEC as long-term survival can be attained with a complete cytoreduction.
The completeness of cytoreduction or resection status is also a well-established independent predictor of survival in patients with MPM who undergo CRS/HIPEC. In the largest analysis by Yan et al, the CC score had a statistically significant impact on survival, with a median OS of 94, 67, 40, and 12 months for CC 0, 1, 2, and 3, respectively.23 Alexander and colleagues had similar findings, showing that patients with a CC of 2 or 3 had nearly twice the risk of death compared to those with a CC of 0 or 1 (HR 1.81, p=0.02).61 In biphasic cohorts, survival depends so greatly on resection status that even a CC 1 resection has a shorter survival by 4 years.35 Thus, at many institutions, if a complete or near-complete cytoreduction cannot be obtained, CRS/HIPEC is considered contraindicated.
Staging systems, by definition, should include factors prognostic for survival. With the creation of a novel TNM staging system for MPM, Yan and colleagues identified seven prognostic factors previously shown to impact survival: age, gender, histologic subtype, CC score, PCI, and lymph node metastasis.42 As age, gender, and histologic subtype were intrinsic and not affected by disease progression, and CC score could only be determined postoperatively, only PCI, nodal status, and extra-abdominal metastasis were included in the staging system. Previous studies demonstrated a median OS of 119 months for patients with a PCI less than or equal to 20, but an only 39-month survival if the PCI was greater than 20.23 Likewise, Magge et al demonstrated that preoperative PCI was predictive of OS.74 Although a rare finding, patients with nodal metastasis had an OS of 20 months, as compared to 56 months in patients without nodal metastasis. The poor prognosis of involved lymph nodes was also confirmed by Baratti and colleagues, who found that pathologically negative nodes were independently correlated with increased OS.77 It is not surprising that extra-abdominal metastasis is a poor prognostic indicator, as this is true of all intra-abdominal malignancies. In their study, Yan et al included 12 patients with disease that penetrated the tendinous portion of the diaphragm. Despite resection of extra-abdominal disease in all cases, the median OS of 20 months was poor and significantly less than patients with no metastatic disease.23
Conclusion
MPM is a rare disease typically localized only to the abdominal cavity with low potential for lymphatic or extra-abdominal metastases. CRS/HIPEC has provided the mainstay of treatment, demonstrating long-term survival, especially in patients with a favorable subtype, low PCI, and complete cytoreduction. Treatment strategies will continue to be refined as more data emerge regarding intraperitoneal perfusion options and adjuvant systemic therapies. As targeted molecular therapies continue to evolve, a multimodal strategy is likely to involve both a surgical and systemic approach.
Disclosure
KIV reports a patent Organoid patent. The authors report no other conflicts of interest in this work.
References
1. Miller J, Wynn WH. Malignant tumor arising from endothelium of peritoneum, and producing mucoid ascitic fluid. J Pathol Bacteriol. 1908;12:267–278. doi:10.1002/path.1700120212
2. Yan TD, Brun EA, Cerruto CA, Haveric N, Chang D, Sugarbaker PH. Prognostic indicators for patients undergoing cytoreductive surgery and perioperative intraperitoneal chemotherapy for diffuse malignant peritoneal mesothelioma. Ann Surg Oncol. 2007;14(1):41–49. doi:10.1245/s10434-006-9169-7
3. Henley SJ, Larson TC, Wu M, et al. Mesothelioma incidence in 50 states and the District of Columbia, United States, 2003-2008. Int J Occup Environ Health. 2013;19(1):1–10. doi:10.1179/2049396712Y.0000000016
4. Moolgavkar SH, Meza R, Turim J. Pleural and peritoneal mesotheliomas in SEER: age effects and temporal trends, 1973-2005. Cancer Causes Control. 2009;20(6):935–944. doi:10.1007/s10552-009-9328-9
5. Price B. Analysis of current trends in United States mesothelioma incidence. Am J Epidemiol. 1997;145(3):211–218.
6. Price B, Ware A. Time trend of mesothelioma incidence in the United States and projection of future cases: an update based on SEER data for 1973 through 2005. Crit Rev Toxicol. 2009;39(7):576–588. doi:10.1080/10408440903044928
7. Gilks B, Hegedus C, Freeman H, Fratkin L, Churg A. Malignant peritoneal mesothelioma after remote abdominal radiation. Cancer. 1988;61(10):2019–2021.
8. Rivera Z, Strianese O, Bertino P, Yang H, Pass H, Carbone M. The relationship between simian virus 40 and mesothelioma. Curr Opin Pulm Med. 2008;14(4):316–321. doi:10.1097/MCP.0b013e3283018220
9. Carbone M, Emri S, Dogan AU, et al. A mesothelioma epidemic in Cappadocia: scientific developments and unexpected social outcomes. Nat Rev Cancer. 2007;7(2):147–154. doi:10.1038/nrc2068
10. Bridda A, Padoan I, Mencarelli R, Frego M. Peritoneal mesothelioma: a review. MedGenMed. 2007;9(2):32.
11. Boffetta P. Epidemiology of peritoneal mesothelioma: a review. Ann Oncol. 2007;18(6):985–990. doi:10.1093/annonc/mdl345
12. Teta MJ, Mink PJ, Lau E, Sceurman BK, Foster ED. US mesothelioma patterns 1973-2002: indicators of change and insights into background rates. Eur J Cancer Prev. 2008;17(6):525–534. doi:10.1097/CEJ.0b013e3282f0c0a2
13. Rodriguez D, Cheung MC, Housri N, Koniaris LG. Malignant abdominal mesothelioma: defining the role of surgery. J Surg Oncol. 2009;99(1):51–57. doi:10.1002/jso.21167
14. Spirtas R, Heineman EF, Bernstein L, et al. Malignant mesothelioma: attributable risk of asbestos exposure. Occup Environ Med. 1994;51(12):804–811.
15. Faig J, Howard S, Levine EA, Casselman G, Hesdorffer M, Ohar JA. Changing pattern in malignant mesothelioma survival. Transl Oncol. 2015;8(1):35–39. doi:10.1016/j.tranon.2014.12.002
16. Kim J, Bhagwandin S, Labow DM. Malignant peritoneal mesothelioma: a review. Ann Transl Med. 2017;5(11):236. doi:10.21037/atm
17. Alexander HR, Hanna N, Pingpank JF. Clinical results of cytoreduction and HIPEC for malignant peritoneal mesothelioma. Cancer Treat Res. 2007;134:343–355.
18. Acherman YI, Welch LS, Bromley CM, Sugarbaker PH. Clinical presentation of peritoneal mesothelioma. Tumori. 2003;89(3):269–273.
19. Sugarbaker PH, Welch LS, Mohamed F, Glehen O. A review of peritoneal mesothelioma at the Washington Cancer Institute. Surg Oncol Clin N Am. 2003;12(3):605–621, xi.
20. Sethna K, Sugarbaker PH. Localized visceral invasion of peritoneal mesothelioma causing intestinal obstruction: a new clinical presentation. Hepato-gastroenterology. 2005;52(64):1087–1089.
21. Esquivel J, Averbach A. Combined laparoscopic cytoreductive surgery and hyperthermic intraperitoneal chemotherapy in a patient with peritoneal mesothelioma. J Laparoendosc Adv Surg Tech A. 2009;19(4):505–507. doi:10.1089/lap.2008.0392
22. Kaya H, Sezgi C, Tanrikulu AC, et al. Prognostic factors influencing survival in 35 patients with malignant peritoneal mesothelioma. Neoplasma. 2014;61(4):433–438. doi:10.4149/neo_2014_053
23. Yan TD, Deraco M, Baratti D, et al. Cytoreductive surgery and hyperthermic intraperitoneal chemotherapy for malignant peritoneal mesothelioma: multi-institutional experience. J Clin Oncol. 2009;27(36):6237–6242. doi:10.1200/JCO.2009.23.9640
24. Baratti D, Kusamura S, Martinetti A, et al. Prognostic value of circulating tumor markers in patients with pseudomyxoma peritonei treated with cytoreductive surgery and hyperthermic intraperitoneal chemotherapy. Ann Surg Oncol. 2007;14(8):2300–2308. doi:10.1245/s10434-007-9393-9
25. Baratti D, Kusamura S, Martinetti A, et al. Circulating CA125 in patients with peritoneal mesothelioma treated with cytoreductive surgery and intraperitoneal hyperthermic perfusion. Ann Surg Oncol. 2007;14(2):500–508. doi:10.1245/s10434-006-9192-8
26. Busch JM, Kruskal JB, Wu B. Best cases from the AFIP. Malignant peritoneal mesothelioma. Radiographics. 2002;22(6):1511–1515. doi:10.1148/rg.226025125
27. Park JY, Kim KW, Kwon HJ, et al. Peritoneal mesotheliomas: clinicopathologic features, CT findings, and differential diagnosis. AJR Am J Roentgenol. 2008;191(3):814–825. doi:10.2214/AJR.07.3628
28. Deraco M, Bartlett D, Kusamura S, Baratti D. Consensus statement on peritoneal mesothelioma. J Surg Oncol. 2008;98(4):268–272. doi:10.1002/jso.21055
29. Kebapci M, Vardareli E, Adapinar B, Acikalin M. CT findings and serum ca 125 levels in malignant peritoneal mesothelioma: report of 11 new cases and review of the literature. Eur Radiol. 2003;13(12):2620–2626. doi:10.1007/s00330-003-1851-6
30. Ros PR, Yuschok TJ, Buck JL, Shekitka KM, Kaude JV. Peritoneal mesothelioma. Radiologic appearances correlated with histology. Acta Radiol. 1991;32(5):355–358.
31. Levy AD, Arnaiz J, Shaw JC, Sobin LH. From the archives of the AFIP: primary peritoneal tumors: imaging features with pathologic correlation. Radiographics. 2008;28(2):
32. Yan TD, Haveric N, Carmignani CP, Chang D, Sugarbaker PH. Abdominal computed tomography scans in the selection of patients with malignant peritoneal mesothelioma for comprehensive treatment with cytoreductive surgery and perioperative intraperitoneal chemotherapy. Cancer. 2005;103(4):839–849. doi:10.1002/cncr.20836
33. Patel NP, Taylor CA, Levine EA, Trupiano JK, Geisinger KR. Cytomorphologic features of primary peritoneal mesothelioma in effusion, washing, and fine-needle aspiration biopsy specimens: examination of 49 cases at one institution, including post-intraperitoneal hyperthermic chemotherapy findings. Am J Clin Pathol. 2007;128(3):414–422. doi:10.1309/DV1JYBL8LLYYT4J5
34. Attanoos RL, Webb R, Dojcinov SD, Gibbs AR. Malignant epithelioid mesothelioma: anti-mesothelial marker expression correlates with histological pattern. Histopathology. 2001;39(6):584–588.
35. Votanopoulos KI, Sugarbaker P, Deraco M, et al. Is cytoreductive surgery with hyperthermic intraperitoneal chemotherapy justified for biphasic variants of peritoneal mesothelioma? Outcomes from the peritoneal surface oncology group international registry. Ann Surg Oncol. 2018;25(3):667–673. doi:10.1245/s10434-017-6293-5
36. Liu S, Staats P, Lee M, Alexander HR, Burke AP. Diffuse mesothelioma of the peritoneum: correlation between histological and clinical parameters and survival in 73 patients. Pathology. 2014;46(7):604–609. doi:10.1097/PAT.0000000000000181
37. Valente K, Blackham AU, Levine E, et al. A histomorphologic grading system that predicts overall survival in diffuse malignant peritoneal mesothelioma with epithelioid subtype. Am J Surg Pathol. 2016;40(9):1243–1248. doi:10.1097/PAS.0000000000000696
38. Lee M, Alexander HR, Burke A. Diffuse mesothelioma of the peritoneum: a pathological study of 64 tumours treated with cytoreductive therapy. Pathology. 2013;45(5):464–473. doi:10.1097/PAT.0b013e3283631cce
39. Robinson BW, Lake RA. Advances in malignant mesothelioma. N Engl J Med. 2005;353(15):1591–1603. doi:10.1056/NEJMra050152
40. Husain AN, Colby T, Ordonez N, et al. Guidelines for pathologic diagnosis of malignant mesothelioma: 2012 update of the consensus statement from the International Mesothelioma Interest Group. Arch Pathol Lab Med. 2013;137(5):647–667. doi:10.5858/arpa.2012-0214-OA
41. AJCC Cancer Staging Manual.
42. Yan TD, Deraco M, Elias D, et al. A novel tumor-node-metastasis (TNM) staging system of diffuse malignant peritoneal mesothelioma using outcome analysis of a multi-institutional database*. Cancer. 2011;117(9):1855–1863. doi:10.1002/cncr.25640
43. Eltabbakh GH, Piver MS, Hempling RE, Recio FO, Intengen ME. Clinical picture, response to therapy, and survival of women with diffuse malignant peritoneal mesothelioma. J Surg Oncol. 1999;70(1):6–12.
44. Antman KH, Pomfret EA, Aisner J, MacIntyre J, Osteen RT, Greenberger JS. Peritoneal mesothelioma: natural history and response to chemotherapy. J Clin Oncol. 1983;1(6):386–391. doi:10.1200/JCO.1983.1.6.386
45. Vogelzang NJ, Rusthoven JJ, Symanowski J, et al. Phase III study of pemetrexed in combination with cisplatin versus cisplatin alone in patients with malignant pleural mesothelioma. J Clin Oncol. 2003;21(14):2636–2644. doi:10.1200/JCO.2003.11.136
46. Janne PA, Wozniak AJ, Belani CP, et al. Open-label study of pemetrexed alone or in combination with cisplatin for the treatment of patients with peritoneal mesothelioma: outcomes of an expanded access program. Clin Lung Cancer. 2005;7(1):40–46. doi:10.3816/CLC.2005.n.020
47. Campbell NP, Kindler HL. Update on malignant pleural mesothelioma. Semin Respir Crit Care Med. 2011;32(1):102–110. doi:10.1055/s-0031-1272874
48. Simon GR, Verschraegen CF, Janne PA, et al. Pemetrexed plus gemcitabine as first-line chemotherapy for patients with peritoneal mesothelioma: final report of a phase II trial. J Clin Oncol. 2008;26(21):3567–3572. doi:10.1200/JCO.2007.15.2868
49. Zalcman G, Mazieres J, Margery J, et al. Bevacizumab for newly diagnosed pleural mesothelioma in the Mesothelioma Avastin Cisplatin Pemetrexed Study (MAPS): a randomised, controlled, open-label, phase 3 trial. Lancet. 2016;387(10026):1405–1414. doi:10.1016/S0140-6736(15)01238-6
50. Calabro L, Morra A, Fonsatti E, et al. Tremelimumab for patients with chemotherapy-resistant advanced malignant mesothelioma: an open-label, single-arm, phase 2 trial. Lancet Oncol. 2013;14(11):1104–1111. doi:10.1016/S1470-2045(13)70381-4
51. Foster JM, Radhakrishna U, Govindarajan V, et al. Clinical implications of novel activating EGFR mutations in malignant peritoneal mesothelioma. World J Surg Oncol. 2010;8:88. doi:10.1186/1477-7819-8-88
52. Govindan R, Kratzke RA, Herndon JE
53. Kanteti R, Dhanasingh I, Kawada I, et al. MET and PI3K/mTOR as a potential combinatorial therapeutic target in malignant pleural mesothelioma. PLoS One. 2014;9(9):e105919. doi:10.1371/journal.pone.0105919
54. Varghese S, Chen Z, Bartlett DL, et al. Activation of the phosphoinositide-3-kinase and mammalian target of rapamycin signaling pathways are associated with shortened survival in patients with malignant peritoneal mesothelioma. Cancer. 2011;117(2):361–371. doi:10.1002/cncr.25555
55. Verma V, Sleightholm RL, Rusthoven CG, et al. Malignant peritoneal mesothelioma: national practice patterns, outcomes, and predictors of survival. Ann Surg Oncol. 2018;25(7):2018–2026. doi:10.1245/s10434-018-6499-1
56. Yan TD, Welch L, Black D, Sugarbaker PH. A systematic review on the efficacy of cytoreductive surgery combined with perioperative intraperitoneal chemotherapy for diffuse malignancy peritoneal mesothelioma. Ann Oncol. 2007;18(5):827–834. doi:10.1093/annonc/mdl428
57. Blackham AU, Shen P, Stewart JH, Russell GB, Levine EA. Cytoreductive surgery with intraperitoneal hyperthermic chemotherapy for malignant peritoneal mesothelioma: mitomycin versus cisplatin. Ann Surg Oncol. 2010;17(10):2720–2727. doi:10.1245/s10434-010-1080-6
58. Feldman AL, Libutti SK, Pingpank JF, et al. Analysis of factors associated with outcome in patients with malignant peritoneal mesothelioma undergoing surgical debulking and intraperitoneal chemotherapy. J Clin Oncol. 2003;21(24):4560–4567. doi:10.1200/JCO.2003.04.150
59. Brigand C, Monneuse O, Mohamed F, et al. Peritoneal mesothelioma treated by cytoreductive surgery and intraperitoneal hyperthermic chemotherapy: results of a prospective study. Ann Surg Oncol. 2006;13(3):405–412. doi:10.1245/ASO.2006.05.041
60. Deraco M, Nonaka D, Baratti D, et al. Prognostic analysis of clinicopathologic factors in 49 patients with diffuse malignant peritoneal mesothelioma treated with cytoreductive surgery and intraperitoneal hyperthermic perfusion. Ann Surg Oncol. 2006;13(2):229–237. doi:10.1245/ASO.2006.03.045
61. Alexander HR
62. Helm JH, Miura JT, Glenn JA, et al. Cytoreductive surgery and hyperthermic intraperitoneal chemotherapy for malignant peritoneal mesothelioma: a systematic review and meta-analysis. Ann Surg Oncol. 2015;22(5):1686–1693. doi:10.1245/s10434-014-3978-x
63. Levine EA, Stewart J, Shen P, Russell GB, Loggie BL, Votanopoulos KI. Intraperitoneal chemotherapy for peritoneal surface malignancy: experience with 1,000 patients. J Am Coll Surg. 2014;218(4):573–585. doi:10.1016/j.jamcollsurg.2013.12.013
64. Dodson RM, Kuncewitch M, Votanopoulos KI, Shen P, Levine EA. Techniques for cytoreductive surgery with hyperthermic intraperitoneal chemotherapy. Ann Surg Oncol. 2018;25(8):2152–2158. doi:10.1245/s10434-018-6336-6
65. Harmon RL, Sugarbaker PH. Prognostic indicators in peritoneal carcinomatosis from gastrointestinal cancer. Int Semin Surg Oncol. 2005;2(1):3. doi:10.1186/1477-7800-2-3
66. Malgras B, Gayat E, Aoun O, et al. Impact of combination chemotherapy in peritoneal mesothelioma Hyperthermic Intraperitoneal Chemotherapy (HIPEC): the RENAPE Study. Ann Surg Oncol. 2018;25(11):3271–3279. doi:10.1245/s10434-018-6631-2
67. Mazzocchi AR, Rajan SAP, Votanopoulos KI, Hall AR, Skardal A. In vitro patient-derived 3D mesothelioma tumor organoids facilitate patient-centric therapeutic screening. Sci Rep. 2018;8(1):2886. doi:10.1038/s41598-018-21200-8
68. Facchiano E, Risio D, Kianmanesh R, Msika S. Laparoscopic hyperthermic intraperitoneal chemotherapy: indications, aims, and results: a systematic review of the literature. Ann Surg Oncol. 2012;19(9):2946–2950. doi:10.1245/s10434-012-2360-0
69. Valle M, Van der Speeten K, Garofalo A. Laparoscopic hyperthermic intraperitoneal peroperative chemotherapy (HIPEC) in the management of refractory malignant ascites: A multi-institutional retrospective analysis in 52 patients. J Surg Oncol. 2009;100(4):331–334. doi:10.1002/jso.21321
70. Cavazzoni E, Bugiantella W, Graziosi L, Franceschini MS, Donini A. Malignant ascites: pathophysiology and treatment. Int J Clin Oncol. 2013;18(1):1–9. doi:10.1007/s10147-012-0396-6
71. Randle RW, Swett KR, Swords DS, et al. Efficacy of cytoreductive surgery with hyperthermic intraperitoneal chemotherapy in the management of malignant ascites. Ann Surg Oncol. 2014;21(5):1474–1479. doi:10.1245/s10434-013-3224-y
72. Le Roy F, Gelli M, Hollebecque A, et al. Conversion to complete cytoreductive surgery and hyperthermic intraperitoneal chemotherapy for malignant peritoneal mesothelioma after bidirectional chemotherapy. Ann Surg Oncol. 2017;24(12):3640–3646. doi:10.1245/s10434-017-6033-x
73. Alyami M, Gagniere J, Sgarbura O, et al. Multicentric initial experience with the use of the pressurized intraperitoneal aerosol chemotherapy (PIPAC) in the management of unresectable peritoneal carcinomatosis. Eur J Surg Oncol. 2017;43(11):2178–2183. doi:10.1016/j.ejso.2017.09.010
74. Magge D, Zenati MS, Austin F, et al. Malignant peritoneal mesothelioma: prognostic factors and oncologic outcome analysis. Ann Surg Oncol. 2014;21(4):1159–1165. doi:10.1245/s10434-013-3358-y
75. Chua TC, Yan TD, Morris DL. Outcomes of cytoreductive surgery and hyperthermic intraperitoneal chemotherapy for peritoneal mesothelioma: the Australian experience. J Surg Oncol. 2009;99(2):109–113. doi:10.1002/jso.21177
76. Schaub NP, Alimchandani M, Quezado M, et al. A novel nomogram for peritoneal mesothelioma predicts survival. Ann Surg Oncol. 2013;20(2):555–561. doi:10.1245/s10434-012-2651-5
77. Baratti D, Kusamura S, Cabras AD, Laterza B, Balestra MR, Deraco M. Lymph node metastases in diffuse malignant peritoneal mesothelioma. Ann Surg Oncol. 2010;17(1):45–53. doi:10.1245/s10434-009-0756-2
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
