Back to Journals » Cancer Management and Research » Volume 9
Cytoreductive surgery and hyperthermic intraperitoneal chemotherapy for appendiceal goblet cell carcinomas with peritoneal carcinomatosis: results from a single specialized center
Authors Yu HH , Yonemura Y , Hsieh MC , Mizumoto A, Wakama S, Lu CY
Received 25 July 2017
Accepted for publication 20 September 2017
Published 19 October 2017 Volume 2017:9 Pages 513—523
DOI https://doi.org/10.2147/CMAR.S147227
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Antonella D'Anneo
Hsin-Hsien Yu,1,2 Yutaka Yonemura,3–5 Mao-Chih Hsieh,1,2 Akiyoshi Mizumoto,4 Satoshi Wakama,3 Chang-Yun Lu1,2
1Department of Surgery, School of Medicine, College of Medicine, Taipei Medical University, Taipei, Taiwan; 2Division of General Surgery, Department of Surgery, Wan Fang Hospital, Taipei Medical University, Taipei, Taiwan; 3Peritoneal Dissemination Center, Kishiwada Tokushukai Hospital, Kishiwada, Osaka, Japan; 4Department of Surgery, Kusatsu General Hospital, Kusatsu, Shiga, Japan; 5Nonprofit Organization to Support Peritoneal Surface Malignancy Treatment, Kyoto, Osaka, Japan
Background: Goblet cell carcinomas (GCCs) of the appendix are rare and aggressive malignancies with early peritoneal dissemination. The aim of the present article is to describe our experience in the management of GCCs with peritoneal carcinomatosis (PC) through cytoreductive surgery (CRS) and hyperthermic intraperitoneal chemotherapy (HIPEC) and to determine the impact of multiple clinical characteristics on the prognosis.
Methods: From a prospectively maintained database of patients receiving CRS and HIPEC for peritoneal surface malignancy, the data of 15 patients with GCC and PC were collected. Neoadjuvant laparoscopic HIPEC was performed if indicated. CRS and HIPEC with mitomycin-C or 5-fluorouracil plus oxaliplatin were performed. Adjuvant chemotherapy was also arranged if suitable for the patient’s condition.
Results: Nine males and six females with a mean age of 52.4 years were enrolled. The estimated median survival after the diagnosis of GCC with PC and after definitive CRS–HIPEC was 28 and 17 months, respectively. The 1-, 2-, 3-, 4-year survival rates were 86%, 69%, 57%, and 24%, respectively. Log-rank test revealed that the significant independent risk factors for more favorable outcomes were age >50 years, peritoneal cancer index (PCI) <27, postoperative PCI <20, administration of HIPEC, and adjuvant chemotherapy. Multivariate analyses confirmed that administration of HIPEC played a crucial role in providing prognostic benefit.
Conclusion: The management of GCC with PC remains challenging. We recommend CRS and HIPEC, followed by adjuvant systemic chemotherapy, as a promising strategy to improve survival, especially in selected patients with low PCI and possibility to achieve complete cytoreduction.
Keywords: cytoreductive surgery, hyperthermic intraperitoneal chemotherapy, appendiceal goblet cell carcinomas, peritoneal carcinomatosis
Introduction
Goblet cell carcinomas (GCCs) of the appendix are rare malignancies, accounting for only 5% of all primary appendiceal tumors.1 Worldwide, the annual incidence is 0.05 per 1,00,000 in the general population, with a relatively equal gender distribution. GCCs have been regarded as intermediates between neuroendocrine tumors and adenocarcinomas because of their pathologic composition and outcomes after management.2 GCCs are potentially malignant and develop from the pluripotent undifferentiated stem cells of the appendix, with divergent neuroendocrine and mucinous differentiation. GCC cells differ completely from the endocrine cells observed in typical carcinoid tumors. Because GCCs of the appendix have a mixed phenotype with partial neuroendocrine differentiation and intestinal-type goblet cell morphology, various terms are used for this condition: adenocarcinoid, goblet cell carcinoid, mucinous or mucin-producing carcinoid, intermediate-type carcinoid, crypt cell carcinoma and amphicrine neoplasia, and mixed adenoneuroendocrine carcinoma.3 GCCs are more malignant and aggressive than carcinoid tumors, and up to 10%–25% of GCC cases have already metastasized to the peritoneum at the time of diagnosis.
Numerous studies have demonstrated significant improvements in survival and prognosis through the use of cytoreductive surgery (CRS) and hyperthermic intraperitoneal chemotherapy (HIPEC) in patients with peritoneal carcinomatosis (PC) from appendiceal cancer, colorectal cancer, malignant peritoneal mesothelioma, gastric cancer, pseudomyxoma peritonei (PMP), and ovarian cancer.4–8 Because of the rarity of GCC cases and the aggressive tumor behavior with early peritoneal dissemination, it is still difficult to establish a standard treatment protocol based on few reports. Butler et al reported a median survival of 7 months (range: 5–24 months) in four patients with GCC and PC, who underwent debulking surgery alone.9 Mahteme and Sugarbaker performed CRS and intraperitoneal chemotherapy on 20 GCC with PC patients (13 of whom underwent HIPEC; 17 patients treated with early postoperative intraperitoneal chemotherapy [EPIC]) and reported an improvement in overall median survival to 19.5 months (range: 3.2–95.1 months).2 Peter Cashin et al reported that after CRS–HIPEC for GCC, the survival was 36.6 months for patients with a completeness of cytoreduction (CC) score of ≤1, and 16.4 months with a CC score of >1.10 McConnell et al reported a favorable 3-year overall survival rate of 63.4% when CRS–HIPEC up to 71.1% was achieved for GCCs with PC.
In addition to conventional CRS and HIPEC, a more dedicated and comprehensive treatment protocol was developed for maximally eradicating invisible metastasis and malignant cells. The procedures included diagnostic laparoscopy, neoadjuvant intraperitoneal and systemic chemotherapy (NIPS) or bidirectional intraperitoneal and systemic induction chemotherapy (BISIC), CRS and HIPEC, extensive intraoperative peritoneal lavage (EIPL), EPIC, and postoperative adjuvant systemic/intraperitoneal chemotherapy. In our specialized peritoneal malignancy center, we have demonstrated the effectiveness of the comprehensive treatment for gastric cancer.6,11 The aim of the present study was to describe our experience in the management of GCCs with PC and to determine the impact of multiple clinical characteristics on the prognosis of GCC.
Materials and methods
From a prospectively maintained database of patients receiving CRS and HIPEC for peritoneal surface malignancy at the Peritoneal Dissemination Centers of Kishiwada Tokushukai Hospital and Kusatsu General Hospital, the data of 15 patients with GCC and PC between January 2006 and December 2016 were collected and reviewed retrospectively. The institutional review boards of the two participating hospitals approved this study. Written informed consent was obtained from all the patients enrolled in this study. The prior surgical score (PSS), which quantifies the extent of any previous surgery prior to definitive CRS–HIPEC, was also recorded.12 Meticulous medical examination and imaging studies were performed for a thorough investigation. Neoadjuvant chemotherapy was defined as the application of intravenous or oral chemotherapeutic agents after the diagnosis of PC and before definitive CRS.
Laparoscopic appendectomy or tissue sampling for diagnosis, with concurrent neoadjuvant (before CRS–HIPEC) laparoscopic HIPEC (L-HIPEC), was performed in patients who received diagnostic laparoscopy for tumor biopsy and pretreatment evaluation. Neoadjuvant L-HIPEC was useful in confirming the diagnosis, determining the appropriate treatment plan, and decreasing the tumor burden in some advanced PC cases such as in PMP and gastric cancer. The procedure was performed as previously described.13,14 Second-look laparotomy was performed as a subsequent intervention for CRS–HIPEC within 2 months after L-HIPEC.
CRS and HIPEC were performed according to the standard protocol of our institution. For the operation, a generous midline incision was made from the subxiphoid to the pubic symphysis. Then, ascitic fluid was aspirated for cytological examination, and frozen sections from peritoneal malignancy sampling were submitted for pathological examination. After profound lysis of adhesions resulting from previous surgical procedures, the dissemination status of the intra-abdominal malignancy was evaluated to determine the peritoneal cancer index (PCI) score, following the principles described by Sugarbaker.15 Before CRS and peritonectomy, EIPL was performed 10 times by using 1 L of saline each time (totally, 10 L). The EIPL using physiological saline delivered by a HIPEC machine was performed to wash away non-adherent peritoneal cancer cells, as recommended by Kuramoto et al.16 CRS combined with peritonectomy was intended to remove all the macroscopically visible tumors and all intra-abdominal organs affected by malignancy. For diffuse dissemination in the small bowel wall, segmental resection up to the maximal length possible without compromising the patient’s quality of life was performed. For small mesentery carcinomatosis, resection of the mesentery surface through electric cauterization or ablation was conducted using an argon beam coagulator (Japanese Medical Next Company, Osaka, Japan). After CRS and peritonectomy, EIPL was repeated 10 times by using 1 L of saline (totally 10 L). This was followed by the administration of HIPEC for 40 min by using the open technique. The procedure was initiated using 4 L of saline, with intraperitoneal chemotherapeutic drugs administered at 42.5°C–43.5°C during the whole procedure. The chemotherapeutic drugs used for HIPEC were mitomycin-C (12 mg/m2) or 5-fluorouracil (5-FU; 300 mg/m2) plus oxaliplatin (60 mg/m2).
The extent of CRS was determined using postoperative CC scores, following the criteria described by Sugarbaker: CC score=0, no residual tumor; 1, <2.5 mm of residual tumor; 2, residual tumor between 2.5 mm and 2.5 cm; and 3, >2.5 cm of residual tumor. After CRS–HIPEC, all patients were monitored closely in intensive care units. EPIC was performed in selected patients with condition allowed, from postoperative days 0 to 5 for 6 h, through the intraperitoneal administration of the chemotherapeutic agents 5-FU plus oxaliplatin or mitomycin-C. Postoperative morbidity and mortality were graded according to the Clavien–Dindo classification system. All patients were routinely followed up in outpatient clinics. Adjuvant chemotherapy was performed in selected patients with well performance status. Regular laboratory and imaging assessments for detecting cancer recurrence were conducted during the follow-up visits. Essential treatments were also provided for recurrent diseases including radiofrequency ablation (RFA) for liver metastasis and gastrointestinal bypass and resection surgery for bowel obstruction due to PC.
Statistical analysis
Survival after the diagnosis of GCC with PC and survival after CRS–HIPEC were recorded, with the endpoint being the latest follow-up or patient death. Survival times were estimated using the Kaplan–Meier method and compared among subgroups. The log-rank test was employed to identify significant differences. Cox proportional hazards regression was used to examine both univariate and multivariate associations between the predictors and overall survival after CRS–HIPEC. Data were analyzed using SPSS version 20.0 (SPSS; IBM Corporation, Armonk, NY, USA). P<0.05 was considered statistically significant.
Results
Patient characteristics
The clinical characteristics and perioperative parameters of the 15 patients with GCC and PC are listed in Table 1. Nine male and six female patients with a mean age of 52.4 years (range: 36–74 years) were assessed. After the diagnosis of PC and before CRS, nine patients (60%) received neoadjuvant chemotherapy with mFOLFOX-6 (n=2), Sandostatin-LAR (n=1), FOLFOX-6 (n=2; followed by FOFIRI plus bevacizumab and TS-1 in one patient), irinotecan plus TS-1 (n=1), docetaxel plus cisplatin and TS-1 (n=1), tegafur and uracil (n=1), and capecitabine plus bevacizumab (n=1). Four patients (26.7%) received neoadjuvant L-HIPEC before definitive CRS and HIPEC. These four patients all received laparoscopic examination, tissue sampling, and L-HIPEC as the “first” surgery after the diagnosis of PC. In addition, one patient (6.7%) underwent NIPS before CRS–HIPEC. Only two patients (13.3%) received CRS and HIPEC as an initial operation after the diagnosis of PC. Among the tumor markers, carcinoembryonic antigen (CEA) and CA-125 were elevated but not CA 19–9. PCI scores were generally high in patients with GCC and PC, with a median PCI score of 26; this indicated the aggressive behavior of this tumor entity.
Treatments and adverse events
The median postoperative PCI score was 18, and the CC score in most of these patients were 2 and 3 (73.3%), implying that it was difficult to achieve complete tumor clearance for this malignant tumor; therefore, a poor prognosis could be expected for GCCs with PC. Among the 15 patients, 11 received HIPEC with either mitomycin-C or 5-FU plus oxaliplatin after CRS. Among the four patients who did not receive HIPEC after CRS, one received EPIC instead; one received adjuvant paclitaxel chemotherapy; one was intolerant of adjuvant chemotherapy after a course of paclitaxel; and one died of intractable postoperative duodenal ulcer bleeding on postoperative day 5 after CRS. EPIC was performed in five patients. The median number of admission days for CRS–HIPEC was 28. One patient suffered from anastomotic leakage in the ileocolostomy, followed by intra-abdominal abscess and poor midline wound healing. The patient was managed through conservative wound care and abscess drainage and was discharged after 190 days of hospitalization. The morbidity rate after CRS–HIPEC was 46.7%, including complications of grades 1–2 in five patients and grades 3–5 in two patients. One patient experienced gastric perforation and received surgical repair on postoperative day 6. One patient died of intractable postoperative duodenal ulcer bleeding on postoperative day 5 after CRS.
Cytological and pathological examination
Intraoperative cytological examination of the ascitic fluid revealed malignant cells in seven patients (46.7%; Table 2). According to the subtypes of GCC, the papillary or tubular cells were observed in 5 patients and signet-ring (or signet-ring like) cells in 10 patients. In our study group, lymph node metastasis was observed in two patients, and lymphatic duct invasion (without lymph node metastasis) was noted in another two patients. A concurrent sigmoid colon adenocarcinoma was detected in one patient with GCC and PC. Immunohistochemical stains revealed frequent expression of cytokeratin (CK) 20 (positive vs negative=10:0), caudal-type homeobox transcription factor 2 (CDX2; positive vs negative=6:0), and equal expression of CK7 (positive vs negative=5:5; Figure 1). The mean and standard error of ki-67 (detected with MIB-1) for the proliferative activity of cancer was 31.9%±6.3% (range: 1%–46%).
  | Table 2 Cytological and pathological examination Abbreviation: LN, lymph node. |
Postoperative follow-up and survival
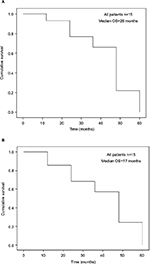
After recovery from CRS–HIPEC, adjuvant chemotherapy was deemed suitable for six patients (40%). The regimens included FOLFOX (n=3), FORFIRI (n=1), TS-1 (n=1), and paclitaxel (n=1). During regular follow-up, six patients died due to recurrent PC and two due to liver and lung metastasis. Two patients experienced colon and rectal recurrence and then received tumor resection at 31 and 27 months after CRS–HIPEC. One patient received bypass duodenocolostomy, and one patient received segmental resection of the small bowel for gastrointestinal obstruction caused by PC. The estimated median survival after PC diagnosis was 28 months (range: 6.5–56 months; Table 3); the 1-, 2-, 3-, 4-, 5-year survival rates were 93%, 77%, 66%, 22%, and 0%, respectively (Figure 2A). The estimated median survival after definitive CRS–HIPEC was 17 months (range: 0.5–51 months); the 1-, 2-, 3-, 4-, 5-year survival rates were 86%, 69%, 57%, 24%, and 0%, respectively (Figure 2B).
Survival analysis
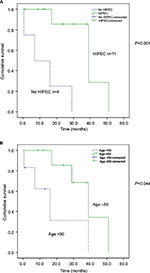
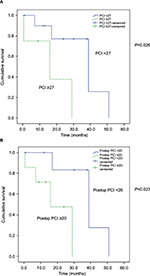
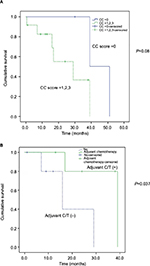
The Kaplan–Meier survival curves were examined through the log-rank test, and significantly improved overall survival was observed after surgery in patients who received HIPEC compared with those who could not receive HIPEC (39 vs 7 months, respectively, P=0.001; Figure 3A). Younger patients (age <50 years) with this aggressive cancer had poorer survival (16 vs 39 months, P=0.044; Figure 3B). Tumor dissemination severity was significantly associated with survival status (PCI score ≥27 [16 months] vs PCI <27 [39 months]; P=0.026; Figure 4A). After cytoreduction, the tumor clearance status represented by the postoperative remnant PCI score was also a significant factor for survival (postoperative PCI ≥20 [16 months] vs postoperative PCI <20 [39 months]; P=0.023; Figure 4B). A trend of improved survival was noted when CC=0, but this was not statistically significant (P=0.08; Figure 5A). Postoperative adjuvant chemotherapy also improved overall survival (16 vs 39 months, P=0.037), regardless of the type of chemotherapeutic regimen (Figure 5B). In the univariate Cox proportional hazards model, the significant characteristics for survival were intraoperative PCI scores and use of HIPEC (Table 4). We also tried to perform a multivariate analysis of significant variables from the univariate analysis. The result showed a potential role of HIPEC in prolonging survival (P=0.022; 95% CI: 0.001–0.574).
Discussion
To the best of our knowledge, only five dedicated papers with a total of <150 patients have focused on the impact of CRS and HIPEC on the effectiveness and prognosis of GCCs with PC. Randle et al reported a median survival of 18.4 months and identified the critical role of lymph node status in the prognosis of GCC.17 Michal Radomski et al demonstrated an estimated overall survival of 22 months for GCC patients, and further classified these patients into three subtypes by assessing the histopathologic features defined as per the Tang classification. The results indicated that the median overall survival after surgery among patients with Tang A, B, and C was 59, 22, and 13 months, respectively.18
Two papers have identified PCI scores as an indicator of outcomes after CRS–HIPEC.2,3 Accordingly, in the present study, poor survival was noted in patients with PCI >27. In four studies, CC for tumor clearance remained the most crucial factor for patient outcomes.2,3,10,18 Although we did not detect similar statistically significant results for the association of CC scores with survival, we did demonstrate that remnant PCI >20 after CRS was associated with poor survival. Therefore, we consider that postoperative remnant PCI, including the evaluation scores for all intra-abdominal regions, might be another more dedicated indicator for prognosis after CRS. Because patients with GCC and PC always present with aggressive intra-abdominal tumor dissemination with high PCI scores, improving the surgeon’s ability to achieve complete surgical cytoreduction of every intra-abdominal organ involved by the tumors remains critical for improved outcomes. The identification of resectability for cytoreduction is also critical for preoperative patient selection. In our multivariate Cox regression analysis, the only factor that predicted the prognosis was the administration of HIPEC (P=0.022, 95% CI: 0.001–0.574). Considering the unique tumor biology, including the non-mass-forming behavior, infiltrative growth patterns, and easy peritoneal dissemination, of GCCs, the effectiveness of HIPEC should be emphasized. However, the value of multivariate analysis was still questionable due to the statistics from small patient number.
In addition, we used mitomycin-C or 5-FU plus oxaliplatin for HIPEC; however, no difference in the impact on survival was observed between these two regimens. Randle et al also reported identical results in his study.17
L-HIPEC after diagnostic laparoscopy and before definitive CRS–HIPEC has also been reported for PC from gastric cancer.19 Yonemura has reported that L-HIPEC and neoadjuvant intraperitoneal/systemic chemotherapy could convert positive ascites cytology to negative in 70% of cases and could decrease PCI scores before CRS.11,14 Hirano and Yonemura also demonstrated that diagnostic laparoscopy following L-HIPEC is useful in determining the surgical treatment plan and reducing the tumor burden before definitive CRS–HIPEC in patients with PMP.13 We performed L-HIPEC in four patients (26.7%); the median survival was 51 months in the L-HIPEC group and 29 months in the non-L-HIPEC group, but the difference was nonsignificant (P=0.151). Further investigation of a large sample of patients might be necessary to establish the therapeutic role of L-HIPEC in GCCs with PC. In addition, we performed NIPS on one patient, who remained disease-free at 9 months after CRS–HIPEC.
In this article, we also report elevated CEA and CA-125 levels, but not CA 19–9 levels, in patients with GCC and PC. We noted that the abnormal tumor markers were applicable only for diagnostic use but were not correlated with the disease survival. Furthermore, the GCC with PC specimens from CRS exhibited inconsistent immunoreactivity for neuroendocrine and cancer markers. The common positive immunohistochemical markers identified were MUC2, CEA, CDX2, CK7, and CK20.20 In our series, we also obtained similar results, with particularly frequent expression of CK20, CDX2, and MUC2.
In our study of patients with GCC and PC, the mean age was 52.4 years (range: 36–74 years). Although previous studies have reported no influence of age on survival, our data surprisingly revealed a shorter survival period after CRS–HIPEC in younger patients (<50 years of age). There are debates that younger cancer patients exhibit more aggressive disease, poorer histologic subtypes, and more genetic defects than do older patients, such as in breast or rectal cancer. However, the small sample of patients may have introduced bias for this conclusion. Because the administration of HIPEC was a crucial factor for survival, we noted that more HIPEC procedures were performed for patients >50 years of age (88.9% vs 50%) in our series.
Due to the rarity of GCC, no randomized control trial about postoperative adjuvant systemic chemotherapy is reported in the literature. Because GCCs exhibit a clinical and histological resemblance to colorectal adenocarcinoma (rather than to metastatic carcinoids), the choice of adjuvant therapy for GCCs should be similar to that for colorectal adenocarcinoma. 5-FU and leucovorin-based FOLFOX (5-FU, leucovorin, and oxaliplatin) and FOLFIRI (5-FU, folic acid, and irinotecan) chemotherapies are the recommended standard regimens. In our analysis, patients who received adjuvant systemic chemotherapy seemed to achieve better outcomes in survival curve by log-rank test. But in our univariate analysis, adjuvant chemotherapy reached a P value of only 0.073. Further, a study with a large number patients might be necessary to confirm the role of adjuvant systemic chemotherapy for patients with GCC and PC. In our study, six patients (40%) also received EPIC. EPIC begins just after CRS–HIPEC, because the invisible cancer cells or tumor volume is assumed to be the least or smallest at this time. The benefit of EPIC for improving survival has been reported in gastric cancer and colorectal cancer, although it remains controversial.21,22 However, in our study of patients with GCC and PC, the administration of EPIC after CRS–HIPEC did not result in a significant improvement in overall survival.
This study has several limitations. First, the results are unavoidably limited by the small sample size. Second, the follow-up period was inadequate to determine the 5-year survival rate after CRS–HIPEC. Despite these limitations, we contribute our experience to the literature to consolidate the importance of CRS–HIPEC in improving survival in patients with GCC and PC. This is also the first report of applying CRS–HIPEC on Asian patients having GCC with PC.
Conclusion
The management of GCC with PC remains challenging. We recommend CRS and HIPEC, followed by adjuvant systemic chemotherapy, as a promising strategy to improve survival, especially in selected patients with low PCI and possibility to achieve complete cytoreduction.
Disclosure
The authors report no conflicts of interest in this work.
References
Roy P, Chetty R. Goblet cell carcinoid tumors of the appendix: an overview. World J Gastrointest Oncol. 2010;2(6):251–258. | ||
Mahteme H, Sugarbaker PH. Treatment of peritoneal carcinomatosis from adenocarcinoid of appendiceal origin. Br J Surg. 2004;91(9):1168–1173. | ||
McConnell YJ, Mack LA, Gui X, et al. Cytoreductive surgery with hyperthermic intraperitoneal chemotherapy: an emerging treatment option for advanced goblet cell tumors of the appendix. Ann Surg Oncol. 2014;21(6):1975–1982. | ||
Glehen O, Kwiatkowski F, Sugarbaker PH. Cytoreductive surgery combined with perioperative intraperitoneal chemotherapy for the management of peritoneal carcinomatosis from colorectal cancer: A Multi-Institutional Study. J Clin Oncol. 2004;22(16):3284–3292. | ||
Yan TD, Deraco M, Baratti D, et al. Cytoreductive surgery and hyperthermic intraperitoneal chemotherapy for malignant peritoneal mesothelioma: multi-institutional experience. J Clin Oncol. 2009;27(36):6237–6242. | ||
Canbay E, Mizumoto A, Ichinose M, et al. Outcome data of patients with peritoneal carcinomatosis from gastric origin treated by a strategy of bidirectional chemotherapy prior to cytoreductive surgery and hyperthermic intraperitoneal chemotherapy in a single specialized center in Japan. Ann Surg Oncol. 2014;21(4):1147–1152. | ||
Chua TC, Moran BJ, Sugarbaker PH, et al. Early- and long-term outcome data of patients with pseudomyxoma peritonei from appendiceal origin treated by a strategy of cytoreductive surgery and hyperthermic intraperitoneal chemotherapy. J Clin Oncol. 2012;30(20):2449–2456. | ||
Bakrin N, Cotte E, Golfier F, et al. Cytoreductive surgery and hyperthermic intraperitoneal chemotherapy (HIPEC) for persistent and recurrent advanced ovarian carcinoma: a multicenter, prospective study of 246 patients. Ann Surg Oncol. 2012;19(13):4052–4058. | ||
Butler JA, Houshiar A, Lin F, Wilson SE. Goblet cell carcinoid of the appendix. Am J Surg. 1994;168(6):685–687. | ||
Cashin P, Nygren P, Hellman P, Granberg D, Andréasson H, Mahteme H. Appendiceal adenocarcinoids with peritoneal carcinomatosis treated with cytoreductive surgery and intraperitoneal chemotherapy: a retrospective study of in vitro drug sensitivity and survival. Clin Colorectal Cancer. 2011;10(2):108–112. | ||
Yonemura Y, Canbay E, Li Y, et al. A comprehensive treatment for peritoneal metastases from gastric cancer with curative intent. Eur J Surg Oncol. 2016;42(8):1123–1131. | ||
Harmon RL, Sugarbaker PH. Prognostic indicators in peritoneal carcinomatosis from gastrointestinal cancer. Int Semin Surg Oncol. 2005;2(1):3. | ||
Hirano M, Yonemura Y, Canbay E, et al. Laparoscopic diagnosis and laparoscopic hyperthermic intraoperative intraperitoneal chemotherapy for pseudomyxoma peritonei detected by CT examination. Gastroenterol Res Pract. 2012;2012:741202. | ||
Yonemura Y, Ishibashi H, Hirano M, et al. Effects of neoadjuvant laparoscopic hyperthermic intraperitoneal chemotherapy and neoadjuvant intraperitoneal/systemic chemotherapy on peritoneal metastases from gastric cancer. Ann Surg Oncol. 2017;24(2):478–485. | ||
Sugarbaker PH. Peritonectomy procedures. Ann Surg. 1995;221(1):29–42. | ||
Kuramoto M, Shimada S, Ikeshima S, et al. Extensive intraoperative peritoneal lavage as a standard prophylactic strategy for peritoneal recurrence in patients with gastric carcinoma. Ann Surg. 2009;250(2):242–246. | ||
Randle RW, Griffith KF, Fino NF, et al. Appendiceal goblet cell carcinomatosis treated with cytoreductive surgery and hyperthermic intraperitoneal chemotherapy. J Surg Res. 2015;196(2):229–234. | ||
Radomski M, Pai RK, Shuai Y, et al. Curative surgical resection as a component of multimodality therapy for peritoneal metastases from goblet cell carcinoids. Ann Surg Oncol. 2016;23(13):4338–4343. | ||
Yonemura Y, Endou Y, Sasaki T, et al. Surgical treatment for peritoneal carcinomatosis from gastric cancer. Eur J Surg Oncol. 2010;36(12):1131–1138. | ||
Shenoy S. Goblet cell carcinoids of the appendix: tumor biology, mutations and management strategies. World J Gastrointest Surg. 2016;8(10):660–669. | ||
Yu WS, Sugarbaker PH. Early postoperative intraperitoneal chemotherapy for gastric cancer. Cancer Treat Res. 1991;55:265–275. | ||
Mahteme H, Hansson J, Berglund A, et al. Improved survival in patients with peritoneal metastases from colorectal cancer: a preliminary study. Br J Cancer. 2004;90(2):403–407. |
 © 2017 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2017 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.