Back to Journals » International Journal of Chronic Obstructive Pulmonary Disease » Volume 15
Critical Error Frequency and the Impact of Training with Inhalers Commonly used for Maintenance Treatment in Chronic Obstructive Pulmonary Disease
Authors Collier DJ , Wielders P, van der Palen J , Heyes L , Midwinter D , Collison K , Preece A , Barnes N , Sharma R
Received 23 July 2019
Accepted for publication 21 April 2020
Published 9 June 2020 Volume 2020:15 Pages 1301—1313
DOI https://doi.org/10.2147/COPD.S224209
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Richard Russell
David J Collier,1,2 Pascal Wielders,3 Job van der Palen,4,5 Logan Heyes,6 Dawn Midwinter,6 Kathryn Collison,7 Andy Preece,6 Neil Barnes,1,6 Raj Sharma6
1William Harvey Research Institute, Barts & The London School of Medicine & Dentistry, Queen Mary University of London, London, UK; 2Wolfson Institute of Preventive Medicine, Barts & The London School of Medicine & Dentistry, Queen Mary University of London, London, UK; 3Department of Pulmonary Diseases, Catharina Hospital, Eindhoven, Netherlands; 4Department of Pulmonology, Medisch Spectrum Twente, Enschede, Netherlands; 5Department of Research Methodology, Measurement, and Data Analysis, University of Twente, Enschede, Netherlands; 6Respiratory Therapy Area Unit, GlaxoSmithKline Plc., Stockley Park, Uxbridge, UK; 7Respiratory Medical Franchise, GlaxoSmithKline Plc., Research Triangle Park, Durham, NC, USA
Correspondence: David J Collier
William Harvey Research Institute Clinical Research Centre, Barts & The London School of Medicine & Dentistry, Queen Mary University of London, Charterhouse Square, London EC1M 6BQ, UK
Tel +44 7961 383925 Fax +44 207 253 1901 Email [email protected]
Introduction: Training in correct inhaler use, ideally in person or by video demonstration, can minimize errors but is rarely provided in clinics. This open-label, low-intervention study evaluated critical error rates with dry-powder inhalers (DPIs), before and after training, in patients with chronic obstructive pulmonary disease.
Methods: Patients prescribed an inhaled corticosteroid (ICS)/long-acting β2-agonist (LABA) (ELLIPTA, Turbuhaler, or DISKUS), long-acting muscarinic antagonist (LAMA)/LABA (ELLIPTA or Breezhaler), or LAMA-only DPI (ELLIPTA, HandiHaler, or Breezhaler) were enrolled. Critical errors were assessed before training (Visit 1 [V1]; primary endpoint) and 6 weeks thereafter (Visit 2 [V2]; secondary endpoint). Logistic regression models were used to calculate odds ratios (ORs) for between-group comparisons.
Results: The intent-to-treat population comprised 450 patients. At V1, fewer patients made ≥ 1 critical error with ELLIPTA (10%) versus other ICS/LABA DPIs (Turbuhaler: 40%, OR 4.66, P=0.005; DISKUS: 26%, OR 2.48, P=0.114) and other LAMA or LAMA/LABA DPIs (HandiHaler: 34%, OR 3.50, P=0.026; Breezhaler: 33%, OR 3.94, P=0.012). Critical error rates with the primary ICS/LABA DPI were not significantly different between ELLIPTA ICS/LABA (10%) and ICS/LABA plus LAMA groups (12– 25%). Critical errors with the primary ICS/LABA DPI occurred less frequently with ELLIPTA ICS/LABA with or without LAMA (11%) versus Turbuhaler ICS/LABA with or without LAMA (39%, OR 3.99, P< 0.001) and DISKUS ICS/LABA with or without LAMA (26%, OR 2.18, P=0.069). Simulating single-inhaler versus multiple-inhaler triple therapy, critical error rates were lower with ELLIPTA fluticasone furoate/vilanterol (FF/VI; 10%) versus ELLIPTA FF/VI plus LAMA (22%), considering errors with either DPI (OR 2.50, P=0.108). At V2, critical error rates decreased for all DPIs/groups, reaching zero only for ELLIPTA. Between-group comparisons were similar to V1.
Conclusion: Fewer patients made critical errors with ELLIPTA versus other ICS/LABA, and LAMA or LAMA/LABA DPIs. The effect of “verbal” training highlights its importance for reducing critical errors with common DPIs.
Keywords: inhaler technique, critical errors, ELLIPTA, inhaled corticosteroid, long-acting β2-agonist, long-acting muscarinic antagonist
Plain Language Summary
Chronic obstructive pulmonary disease (COPD) is a chronic lung disease that can have a significant burden on patients’ everyday lives. In order to treat COPD, patients are prescribed medications in one or more inhalers; however, many different inhalers are available, each one with its own unique mechanism of use, and errors in inhaler technique are common. So-called “critical” errors can reduce the amount of medication delivered to an extent which may lead to poorer disease management and outcomes. One way to reduce errors in inhaler technique is for healthcare professionals to explain to patients how to use their inhaler(s) properly and then to monitor their technique over time. In practice, this often does not occur. In this study, patients with COPD were receiving COPD treatments from one or two of five different inhalers (ELLIPTA, HandiHaler, DISKUS, Turbuhaler, or Breezhaler). Correct use of their inhaler(s) was assessed at two study visits (Visit 1 and Visit 2). Without receiving any training at Visit 1, fewer patients made critical errors with the ELLIPTA inhaler versus the other inhalers tested. At Visit 2 (6 weeks later), after training (if required), the number of patients making critical errors was much lower for all inhalers and no patients made critical errors using ELLIPTA alone. Overall, this study demonstrates the existence of poor inhaler technique and the importance of training to reduce errors with inhalers used for the treatment of COPD. Inhaler training may lead to improved treatment outcomes for patients with COPD.
Introduction
Inhaled medications, namely licensed inhaled corticosteroid (ICS), long-acting β2-agonist (LABA), and long-acting muscarinic antagonist (LAMA) combinations, represent the cornerstone of maintenance therapy for chronic obstructive pulmonary disease (COPD).1 It is widely acknowledged, however, that patients frequently make errors when using their prescribed inhaler(s).2 These errors, particularly critical errors — those that result in no, or significantly reduced, medication being inhaled — are associated with a subsequent negative impact on treatment efficacy and also an increase in the economic burden associated with treatment.3
Both the type and number of inhalers required to deliver maintenance regimens can impact the frequency of errors (critical or otherwise) made by patients. COPD treatments can be delivered via multiple inhaler types, with different inhalers often requiring the user to learn different inhalation techniques.4 The ELLIPTA inhaler, a dry-powder inhaler (DPI) designed to deliver single agents or their combinations in a single inhalation, demonstrated the fewest critical errors when compared with other commonly used DPIs and metered-dose inhalers in clinical studies of COPD.5,6 While studies of errors in technique have been conducted in both the real world7 and clinical trial settings5,6 for many different inhalers, fewer data exist for more recently available inhalers such as ELLIPTA. Available data for ELLIPTA have largely been restricted to the trial setting thus far but the more widespread use of the ELLIPTA inhaler now provides an opportunity to study errors in technique in a routine clinical practice setting.
Optimal inhaler use should result in no (or very few) errors and successful delivery of medication. Training from healthcare professionals (HCPs) on correct inhaler use provides the best outcomes for patient technique in the clinic, and repetition of training or monitoring of ongoing inhaler technique reduces errors.1,8 Notably, a lack of instruction on correct inhaler use from HCPs is significantly (P<0.001) associated with risk of critical errors in patients with COPD or asthma.9 Nevertheless, training on correct inhaler technique is time-consuming to perform, while prior evidence shows that a significant proportion of HCPs have inadequate knowledge of and competency in correct inhaler use.10,11 In addition, assessment of inhaler technique over time is only conducted in a small percentage of patients8 and approximately half of patients who initially use their inhaler correctly do not maintain correct use over time;8 correct technique may decline within 1 month of receipt of training.12
Given the importance of training for correct inhaler technique, and the wider use of the ELLIPTA inhaler in routine clinical practice, we designed the current study to assess the rate of critical errors with ELLIPTA compared with a number of other DPIs. Due to the widespread use of multiple-inhaler triple therapies for COPD in practice, analyses were conducted for single DPIs and DPI combinations, both before and 6 weeks after refresher training (if required) on correct technique from an HCP.
Methods
Study Design and Patients
This open-label, low-intervention clinical study was carried out at 13 centers in the Netherlands (n=9; Catharina Ziekenhuis, Gelre Ziekenhuis Zutphen, Huisartsenmaatschap MCN, Huisartsenpraktijk Coenen, PreCare T&R, Q Clinical, Quality Care Research, Westfries Gasthuis, Ziekenhuisgroep Twente) and the United Kingdom (n=4; Medinova North London Clinical Studies Centre, Medinova South London Clinical Studies Centre, Saint George’s University of London, William Harvey Heart Clinical Research Centre) from June 2017 to March 2018. Eligible patients were identified from community pharmacies and clinical sites (hospitals, primary care sites, and contract research organizations) based on the patients’ regular inhaler(s) used to receive COPD maintenance medication. Expert training in inhaler technique was provided to staff at each of the study sites.
Enrolled patients were aged ≥40 years, with a physician’s diagnosis of COPD, and had received DPI treatment for ≥3 months prior to inclusion: the DPI groups included in this analysis are summarized in Table 1. Patients could have received ICS/LABA via ELLIPTA (fluticasone furoate/vilanterol [FF/VI]), Turbuhaler (budesonide/formoterol [BUD/FOR]), or DISKUS (fluticasone propionate/salmeterol [FP/SAL]), alone or in combination with a LAMA-only DPI (ELLIPTA umeclidinium [UMEC] or HandiHaler tiotropium [TIO]), or LAMA monotherapy via ELLIPTA (UMEC), HandiHaler (TIO), or Breezhaler (glycopyrronium [GLY]), or LAMA/LABA dual therapy via ELLIPTA (UMEC/VI) or Breezhaler (indacaterol/GLY [IND/GLY]). Exclusion criteria included a current diagnosis of asthma and participation in an inhaler study that involved training within 6 months of study entry. All patients provided written, informed consent. The study was conducted in accordance with the ethical principles outlined in the Declaration of Helsinki. At all participating sites, the study was approved by the relevant institutional review board or ethics committee, as required (East Midlands REC – Northampton, UK; Saint Antonius Ziekenhuis, the Netherlands).
 |
Table 1 Single and Combination DPI Groups Enrolled in the Study |
All patients were assessed only on the use of their own maintenance DPI (no change to a new DPI was investigated). The study comprised three visits: a screening (enrollment) visit (V0), a subsequent first assessment visit (Visit 1 [V1]; which could occur on the same day as V0), and a second assessment visit (Visit 2 [V2]) 6 weeks after V1. Patients’ baseline information (demographics, smoking history, COPD Assessment Test [CAT] score, medical history, time on each DPI, time since last trained, and educational information) was collected at V0 and errors made with their usual COPD maintenance DPI(s) were assessed at V1 without any instruction from their HCP. If the patient made errors at this stage, they received verbal instruction on correct use of their DPI(s) from the HCP. If the patient made no errors at this stage, they did not receive training and were simply informed of their correct use.
Patients returned for V2 6 weeks (±7 days) after V1; this time period reflects prior evidence showing a decline in correct inhaler technique within 1 month of training.12 DPI errors were assessed and noted, and any changes in health or prescriptions were captured. All patients were discharged from the study at V2. Patients were referred to their general practitioner for further training if they continued to demonstrate incorrect use of their prescribed DPI at V2.
Endpoints and Assessments
The primary endpoint was the percentage of patients making ≥1 critical error at V1. The key secondary endpoint was the percentage of patients making ≥1 critical error at V2. The primary comparisons of interest for this study were the percentage of patients making ≥1 critical error with each ICS/LABA DPI (ELLIPTA FF/VI vs other ICS/LABA DPIs) and with each LAMA or LAMA/LABA DPI (ELLIPTA UMEC or UMEC/VI vs other LAMA or LAMA/LABA DPIs). Additional comparisons examined the effect of adding a secondary LAMA DPI to the primary ICS/LABA inhaler, from the perspective of critical errors with the primary DPI and with both DPIs combined. Comparisons were also conducted to evaluate overall errors (ie, all errors made, whether classified as critical or non-critical). Furthermore, to simulate a comparison of single-inhaler triple therapy (SITT) versus multiple-inhaler triple therapy (MITT), critical error rates with ELLIPTA FF/VI alone versus ELLIPTA FF/VI plus LAMA were investigated.
Patients’ inhaler technique was assessed against a correct-use checklist for each relevant DPI by HCPs at each study site. All assessors purposefully received the same training in correct technique for each of the individual DPIs examined. No universally agreed checklists that define critical and non-critical errors for specific inhalers currently exist. Thus, checklists and critical errors for each individual DPI were developed based on the steps identified in the patient information leaflet for each inhaler, review of the available literature, and further review by external experts.5 These checklists were thus as objective as possible and were used by all study sites. Complete checklists are provided in the supplementary appendix (Tables S1–S5).
Critical errors, which were the focus of this study, were defined as those leading to no, or significantly reduced, medication being inhaled. Errors not meeting this definition were considered non-critical errors and were recorded for the purpose of also evaluating overall errors, ie, all errors made, whether classified as critical or not. The numbers of possible critical/overall errors for each DPI examined were: 4/9 for ELLIPTA; 6/12 for Turbuhaler; 5/9 for DISKUS; 7/13 for HandiHaler; and 7/12 for Breezhaler. Differences between the DPIs in the number of critical errors potentially reflect differences in complexity for correct inhaler use.
Statistical Analyses
A planned total of 50 patients in each DPI group was calculated to provide >94% power to demonstrate a statistically significant difference between the critical error rates of each of the paired DPI comparisons, assuming a true critical error rate of 5% for ELLIPTA and 30% for each of the other DPIs, based on previous studies.13,14 A two-sided 5% significance level was assumed to test the difference between critical error rates for each comparison of interest.
For the pre-specified analyses, the primary and key secondary endpoints were analyzed using a logistic regression model, with DPI group as a fixed effect and adjusting for the covariate of time on current primary DPI. Odds ratios (ORs), confidence intervals (CIs), and P-values were calculated for each comparison.
During development of this study, it became apparent that patients could have been using a particular DPI for less time than patients who were using other DPIs; for example, the majority of patients using ELLIPTA and Breezhaler DPIs had used these for 2 years or fewer, while most patients using Turbuhaler, DISKUS and HandiHaler DPIs had been using these for more than 3 years. As a result, there was a clear non-independent imbalance between time on DPI and treatment cohort across the different cohorts. Given the non-randomized design of the study and the potential confounding of the covariate, inclusion of the parameter of time on DPI resulted in the possibility that any treatment effect (in either direction) would be reduced due to this confounding by DPI type. We therefore also performed post-hoc sensitivity analyses in which the confounded covariate of time on DPI was removed from the logistic regression models to minimize the impact of this imbalance. All pre-specified and post-hoc sensitivity analyses, including those of safety, were conducted in the intent-to-treat (ITT) population, which comprised all patients who were enrolled and demonstrated use of their primary DPI. Data are presented for the pre-specified analyses (main text and tables) and for the post-hoc sensitivity analyses (tables only).
Results
Study Population
Overall, 461 patients were enrolled and 450 comprised the ITT population. The mean (standard deviation [SD]) age was 67.2 (9.75) years and 54% were male (Table 2). At screening, the majority of patients (58%) reported a COPD duration of between 1 and 10 years. The mean (SD) CAT score at screening was 16.6 (7.23). Demographic characteristics were largely similar across DPI groups.
 |
Table 2 Patient Demographics and Clinical Characteristics (ITT Population and per Prescribed Inhaler at Study Entry) |
Inhaler Errors
Details of the specific errors (critical and non-critical) that were made by patients for each DPI are presented in the supplementary appendix (Figure S1). Almost all patients correctly opened the cover/removed the cap/removed the capsule of their DPI and formed a firm seal around the mouthpiece with their lips prior to inhalation. Among patients making any error (critical or non-critical), the most common critical error for the ELLIPTA, DISKUS and Breezhaler DPIs was exhaling directly into the mouthpiece (prior to medication inhalation) (18%, 37%, and 44% of patients, respectively). Not holding the inhaler upright (during dose preparation) was the most common critical error with the Turbuhaler DPI (41% of patients), while for the HandiHaler DPI it was the capsule not rattling, indicating failure to empty the capsule of medication (24% of patients).
Percentage of Patients Making ≥1 Critical Error at V1: ICS/LABA and LAMA or LAMA/LABA Primary DPIs
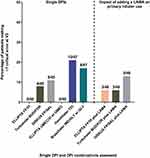
Patients using the ELLIPTA DPI demonstrated the lowest critical error rate across the single ICS/LABA DPIs assessed at V1 (Figure 1). The number of patients making ≥1 critical error with ELLIPTA FF/VI, Turbuhaler BUD/FOR, and DISKUS FP/SAL was 5/50 (10%), 20/50 (40%), and 13/50 (26%), respectively. The OR for making ≥1 critical error with Turbuhaler BUD/FOR versus ELLIPTA FF/VI was 4.66 (95% CI 1.58–13.69; P=0.005) (Table 3). For the DISKUS FP/SAL versus ELLIPTA FF/VI comparison, the OR for making ≥1 critical error was 2.48 (95% CI 0.80–7.63; P=0.114) (Table 3).
 |
Table 3 Percentage of Patients Making ≥1 Critical Error with their Prescribed DPI(s) at V1 |
For the LAMA or LAMA/LABA DPIs, the ELLIPTA DPI again showed the lowest critical error rate. The number of patients making ≥1 critical error was 5/51 (10%) with ELLIPTA UMEC/VI or UMEC, 17/50 (34%) with HandiHaler TIO, and 16/49 (33%) with Breezhaler IND/GLY or GLY (Figure 1). This corresponded to an OR for making ≥1 critical error with HandiHaler TIO versus ELLIPTA UMEC/VI or UMEC of 3.49 (95% CI 1.16–10.55; P=0.026), and an OR for making ≥1 critical error with Breezhaler IND/GLY or GLY versus ELLIPTA UMEC/VI or UMEC of 3.94 (95% CI 1.35–11.53; P=0.012) (Table 3).
Results of the post-hoc sensitivity analyses excluding the covariate of time on primary DPI (which was confounded with DPI type) mirrored the findings of the pre-specified analyses, but with slightly higher odds ratios for almost all comparators (vs ELLIPTA); this pattern was evident also for the comparisons described below.
Percentage of Patients Making ≥1 Critical Error at V1: ICS/LABA Plus LAMA Groups
Addition of a LAMA DPI had a minimal effect on critical error rates with the primary ICS/LABA DPI at V1. The number of patients making ≥1 critical error was 6/50 (12%) with ELLIPTA FF/VI, 19/50 (38%) with Turbuhaler BUD/FOR, and 13/50 (26%) with DISKUS FP/SAL (Figure 1).
Percentage of Patients Making ≥1 Critical Error at V1: ELLIPTA FF/VI Alone versus ICS/LABA DPIs Plus LAMA
The comparison of ELLIPTA FF/VI alone versus ICS/LABA DPIs plus LAMA DPIs effectively simulates the comparison of ELLIPTA SITT versus MITT (although in this investigation, ELLIPTA was not used by any patients to deliver a triple therapy regimen). When considering only errors with the primary ICS/LABA DPI, the number of patients making ≥1 critical error was 5/50 (10%) with ELLIPTA FF/VI and 6/50 (12%) with ELLIPTA FF/VI plus LAMA (Table 3). Compared with ELLIPTA FF/VI alone, the OR for patients making ≥1 critical error was 1.22 (95% CI 0.36–4.15; P=0.746) with ELLIPTA FF/VI plus LAMA, but when considering errors in either the primary or LAMA DPI, the number of patients making ≥1 critical error increased to 11/50 (22%) with ELLIPTA FF/VI plus LAMA, corresponding to an OR of 2.50 (95% CI 0.82–7.66; P=0.108) (Table 3).
When considering errors with the primary DPI for patients using any ICS/LABA plus LAMA combination (ie, ELLIPTA FF/VI, Turbuhaler BUD/FOR, or DISKUS FP/SAL, respectively, plus Handihaler TIO or ELLIPTA UMEC, respectively), ≥1 critical error was made by 38/150 (25%) patients. This corresponded to an OR of 2.29 (95% CI 0.85–6.16; P=0.100) versus ELLIPTA FF/VI alone. Including errors in either the primary or LAMA DPI, the number of patients making ≥1 critical error increased to 61/150 (41%). This corresponded to an OR of 4.89 (95% CI 1.85–12.90; P=0.001) versus ELLIPTA FF/VI alone.
A further post-hoc analysis comparing ELLIPTA FF/VI alone (n=50) with ICS/LABA plus LAMA combinations not using ELLIPTA (ie, Turbuhaler BUD/FOR and DISKUS FP/SAL each plus Handihaler TIO; n=99) found that significantly more patients using ICS/LABA plus LAMA made ≥1 critical error with the primary ICS/LABA DPI (10% vs 32%, OR 3.19, 95% CI 1.13–9.00; P=0.028) or with either DPI (10% vs 51%, OR 7.01, 95% CI 2.51–19.54; P<0.001).
Percentage of Patients Making ≥1 Critical Error at V1: ELLIPTA FF/VI Alone or Plus LAMA versus Other ICS/LABA DPIs Alone or Plus LAMA
For patients prescribed an ICS/LABA DPI, considering both those using it alone and those using it together with a LAMA, the number making ≥1 critical error with the primary ICS/LABA DPI was 11/100 (11%) with ELLIPTA FF/VI, 39/100 (39%) with Turbuhaler BUD/FOR, and 26/100 (26%) with DISKUS FP/SAL (Table 3). Compared with ELLIPTA FF/VI, the OR for patients making ≥1 critical error with the primary DPI was 3.99 (95% CI 1.81–8.83; P<0.001) with Turbuhaler BUD/FOR, and 2.18 (95% CI 0.94–5.06; P=0.069) with DISKUS FP/SAL (Table 3). When these comparisons considered critical errors made with either the primary or LAMA DPI, the number of patients making ≥1 critical error increased and was 16/100 (16%) for ELLIPTA FF/VI plus LAMA, 47/100 (47%) for Turbuhaler BUD/FOR plus LAMA, and 36/100 (36%) for DISKUS FP/SAL plus LAMA. Compared with ELLIPTA FF/VI plus LAMA, the OR for patients making ≥1 critical error with the primary ICS/LABA DPI and/or LAMA DPI was 3.94 (95% CI 1.86–8.34; P<0.001) with Turbuhaler BUD/FOR plus LAMA, and 2.39 (95% CI 1.09–5.26; P=0.030) with DISKUS FP/SAL plus LAMA (Table 3).
Percentage of Patients Making ≥1 Critical Error with their Prescribed DPI at V2
At V2, the percentage of patients making ≥1 critical error was reduced compared to V1 across all single DPIs and combinations tested (Figure 2; Table 4).
 |
Table 4 Percentage of Patients Making ≥1 Critical Error with their Prescribed DPI(s) at V2 |
It was again notable that the ELLIPTA DPI, as either a single ICS/LABA DPI or single LAMA/LABA DPI, had the lowest critical error rate. The number of patients making ≥1 critical error with ELLIPTA FF/VI at V2 was 0/50 (0%), compared with 4/48 (8%) with Turbuhaler BUD/FOR (OR 7.35, 95% CI 0.41–132.14; P=0.176) and 5/45 (11%) with DISKUS FP/SAL (OR 9.30, 95% CI 0.53–164.47; P=0.128).
For LAMA or LAMA/LABA single-inhaler comparisons, 0% of patients (0/50) made ≥1 critical error with ELLIPTA UMEC/VI or UMEC, compared with 10/47 (21%) with HandiHaler TIO (OR 20.45, 95% CI 1.19–351.61; P=0.038) and 8/47 (17%) with Breezhaler IND/GLY or GLY (OR 18.78, 95% CI 1.13–311.67; P=0.041) (Figure 2; Table 4).
Percentage of Patients Making ≥1 Overall Error with their Prescribed DPI
The correct-use checklists recorded all errors, referred to as overall errors (critical errors and non-critical errors). This enabled additional planned analyses of overall errors to be conducted. Comparisons were analogous to those reported above for critical errors.
Consistent with the data for critical errors, overall errors were made least often with the ELLIPTA DPI, as either a single ICS/LABA DPI or single LAMA/LABA DPI. The number of patients making ≥1 overall error with ELLIPTA FF/VI at V1 was 17/50 (34%), compared with 32/50 (64%) with Turbuhaler BUD/FOR (OR 2.40, 95% CI 1.02–5.68; P=0.045) and 30/50 (60%) with DISKUS FP/SAL (OR 1.82, 95% CI 0.77–4.32; P=0.172) (Table S6). For LAMA or LAMA/LABA single-inhaler comparisons, 19/51 (37%) patients made ≥1 overall error with ELLIPTA UMEC/VI or UMEC, compared with 37/50 (74%) with HandiHaler TIO (OR 2.99, 95% CI 1.23–7.27; P=0.016) and 27/49 (55%) with Breezhaler IND/GLY or GLY (OR 1.75, 95% CI 0.77–3.94; P=0.179) (Table S6).
In comparisons of ELLIPTA FF/VI alone versus ICS/LABA DPIs plus LAMA, addition of a LAMA DPI to ELLIPTA FF/VI had no effect on the number of patients making ≥1 overall error with the primary DPI (17/50, 34%); however, the number increased when errors with either DPI were considered (23/50, 46%; OR 1.93, 95% CI 0.84–4.43; P=0.120). Similarly, more patients made ≥1 overall error with any ICS/LABA plus LAMA compared with ELLIPTA FF/VI alone, and with any ICS/LABA alone or plus a LAMA compared with ELLIPTA FF/VI alone or plus a LAMA; this was true when considering errors with the primary DPI, or errors with either DPI (Table S6).
Similar patterns were observed at V2 (Table S7), where again the overall error rate was reduced compared to V1 across all single DPIs and combinations tested. The ELLIPTA DPI demonstrated the lowest overall error rate (FF/VI: 6/50, 12%; UMEC or UMEC/VI: 4/50, 8%) compared with equivalent ICS/LABA DPIs (Turbuhaler BUD/FOR: 17/48, 35%, OR 3.48, 95% CI 1.22–9.96; P=0.020; DISKUS FP/SAL: 12/45, 27%, OR 2.28, 95% CI 0.76–6.90; P=0.143) and LAMA or LAMA/LABA DPIs (HandiHaler TIO: 17/47, 36%, OR 5.27, 95% CI 1.62–17.19; P=0.006; Breezhaler IND/GLY or GLY: 15/47, 32%, OR 4.66, 95% CI 1.48–14.67; P=0.009).
Discussion
Errors in inhaler technique remain an ever-present issue in COPD maintenance therapy, and the association between these errors, particularly critical errors, and reduced treatment efficacy is widely acknowledged.3 As such, the optimization of inhaler technique through training represents an important route by which the efficacy of maintenance therapy may be improved.15
This study was designed to assess the ability of patients with COPD to correctly use their prescribed DPI in a low-intervention setting. Here, the notion that inhaler training reduces the percentage of patients making critical (and overall) errors is clearly demonstrated by the comparison of results between V1 and V2, despite an interval of 6 weeks. Our findings also reveal differences in ease of use for each inhaler, exemplified by large differences in critical and overall error rates in the absence of training.
For the single DPIs (ICS/LABA, and LAMA or LAMA/LABA), the ELLIPTA DPI was associated with the lowest percentage of patients making ≥1 critical error at V1, providing agreement with previous, similar studies.6,14 This difference was statistically significant for all but one of the single-DPI comparisons at V1; only the comparison between ELLIPTA FF/VI and DISKUS FP/SAL did not reach statistical significance (P=0.114). Findings from the pre-specified analyses were confirmed in the additional post-hoc sensitivity analyses, in which the covariate of time on primary DPI was excluded due to confounding with type of DPI used.
Recent findings from a randomized, open-label, placebo-inhaler crossover study, including ELLIPTA, indicated that patients with COPD make fewer critical errors with single-inhaler regimens compared with multiple-inhaler regimens.5 This idea of improved technique when using fewer inhalers is also supported here by comparing ELLIPTA FF/VI with ICS/LABA DPIs plus LAMA, which demonstrated relatively high ORs for ≥1 critical error in favor of ELLIPTA at both V1 and V2. The smaller, non-significant differences in critical errors observed between ELLIPTA FF/VI and ELLIPTA FF/VI plus LAMA likely reflect the proportion of patients (23/50) already using an ELLIPTA DPI for the secondary LAMA.
The comparison of ELLIPTA FF/VI versus the ICS/LABA plus LAMA combination group is essentially equivalent to simulating SITT with ELLIPTA versus MITT with any DPIs. In additional post-hoc analyses, patients using ELLIPTA ICS/LABA (simulated SITT) demonstrated significantly fewer critical errors at V1 compared with an ICS/LABA plus LAMA comparator group that excluded the ELLIPTA DPIs (ie, non-ELLIPTA MITT); a similar but non-significant trend was observed at V2.
Incorrect inhaler technique is common and has been well documented in the literature.2,7,16 A systematic literature review of studies evaluating incorrect use of DPIs by patients with asthma or COPD found that 4–94% of patients (depending on the type of DPI and method of assessment) did not use their inhalers correctly, while up to 25% had never received verbal instruction on inhaler technique.16 Physical demonstration and verbal instruction have been shown to markedly reduce critical errors with DPIs, including ELLIPTA and Breezhaler.17 Our findings also clearly show that verbal instruction substantially reduces the number of critical and overall inhaler errors with these, and other, DPIs. Considering five different DPIs, we found that ≥1 critical error occurred in 10–40% of patients at V1, least often with the ELLIPTA DPI and most often with the Turbuhaler DPI. Error rates were reduced after training for all five DPIs at V2 (0–21%); this is comparable with previous studies and meta-analyses demonstrating a reduction in errors after visual demonstration or additional training.6,17,18 In this study, exhaling into the inhaler mouthpiece prior to inhalation was the most common critical error with the ELLIPTA FF/VI, DISKUS FP/SAL and Breezhaler IND/GLY or GLY DPIs. This may introduce moisture into the mouthpiece, causing clumping of the medication.19
The enrollment of patients on their prescribed DPIs is a notable strength of this study, adding an element of real-world design to the protocol and meaning results more accurately reflect error rates in the clinic. The incorporation of minimal study visits also mimics real-world scenarios, allowing patients to feel less constrained by the schedule of clinical trial protocol visits.
However, the results of this study may not be widely comparable with those of similar, existing studies because patients were enrolled on their currently prescribed DPI. Previous studies have tended to use inhaler-naïve patients and, as such, the critical error rates reported in this study may be lower than generally expected based on existing data. Moreover, this study was open-label and non-randomized, though baseline and demographic characteristics were broadly similar across all treatment groups and much of this potential bias was thus reduced. Additionally, participating sites did not recruit an equal number of participants using each of the DPIs tested but all site staff assessing participants for errors in correct use received the same training on all DPIs, to limit the effect of this.
A further limitation of this study is that conclusions regarding the relationship between inhaler errors and treatment outcomes cannot be drawn, as clinical outcomes were not assessed. Previous studies and reviews have, however, highlighted a positive correlation between inhaler technique and treatment outcomes7,15 and critical errors may have an adverse impact on patient outcomes.20
In this study, the retention of inhaler training knowledge was assessed over 6 weeks. Longer-term studies are needed to further understand the retention of inhaler training over time periods when patients would not typically receive further guidance in clinical settings. Future studies in this area may also make use of additional inhalers not considered here to provide a more comprehensive overview of inhalers available in clinical practice, and should also consider patient compliance and satisfaction, to better understand how these factors may impact clinical outcomes.21
The observed differences between visits in this low-intervention study clearly show the value and importance of verbal instruction on correct inhaler use by HCPs. In a recent review of 39 studies of inhalation technique education for both asthma and COPD, 89% involved a physical or video demonstration of inhaler technique, and 90% of the same studies reported a significant improvement in inhaler technique, indicating a strong relationship between training and improved technique.15 Physical or video demonstration of correct inhaler technique may be ideal in most cases, the latter being an approach that might easily be adopted by most practices (eg, during patient waiting times).
Conclusion
Patients with COPD can make critical inhaler errors frequently, though rates differ according to inhaler type. Critical error rates were lower after verbal refresher training, demonstrating the importance of HCP instruction on inhaler technique for commonly used DPIs. In this study, the ELLIPTA DPI resulted in fewer critical errors compared with other ICS/LABA, and LAMA or LAMA/LABA DPIs assessed in a low-intervention clinical setting in patients with COPD. Our findings will help HCPs and patients make informed decisions regarding the choice of, training with and use of DPIs as an integral part of COPD maintenance therapy regimens.
Abbreviations
BMI, body mass index; BUD, budesonide; CAT, COPD Assessment Test; CI, confidence interval; COPD, chronic obstructive pulmonary disease; DPI, dry-powder inhaler; FF, fluticasone furoate; FOR, formoterol; FP, fluticasone propionate; GLY, glycopyrronium; HCP, healthcare professional; ICS, inhaled corticosteroid; IND, indacaterol; ITT, intent-to-treat; LABA, long acting β2-agonist; LAMA, long-acting muscarinic antagonist; MITT, multiple-inhaler triple therapy; OR, odds ratio; SAL, salmeterol; SD, standard deviation; SITT, single-inhaler triple therapy; TIO, tiotropium; UMEC, umeclidinium; VI, vilanterol; V0, Visit 0; V1, Visit 1; V2, Visit 2.
Data Sharing Statement
Anonymized individual patient data and study documents can be requested for further research from www.clinicalstudydatarequest.com.
Acknowledgments
Trademarks are owned by or licensed to their respective owners (the GlaxoSmithKline group of companies, and AstraZeneca, Boehringer Ingelheim, and Novartis). Editorial support (in the form of writing assistance, collating author comments, assembling tables and figures, grammatical editing, fact checking, and referencing) was provided by Matthew Hallam MSc(Res), and Molly Macpherson, BSc, of Gardiner-Caldwell Communications (Macclesfield, UK) and was funded by GlaxoSmithKline plc.
Author Contributions
All authors contributed to data analysis, drafting or revising the article, gave final approval of the version to be published, and agree to be accountable for all aspects of the work.
Funding
This study was funded by GlaxoSmithKline plc. (ClinicalTrials.gov number NCT02982187; GlaxoSmithKline plc. study 204981).
Disclosure
David J Collier was supported in part by the NIHR Barts Biomedical Research Centre. Job van der Palen reports personal fees from AstraZeneca, Boehringer Ingelheim, and GlaxoSmithKline plc., and grants from Chiesi, outside the submitted work. Logan Heyes was an employee of GlaxoSmithKline plc. at the time of the study, and is currently employed by Pharmaceutical Management Agency (PHARMAC). Dawn Midwinter, Kathryn Collison, Andy Preece, Neil Barnes, and Raj Sharma are employees of, and have shares in, GlaxoSmithKline plc. The authors report no other conflicts of interest in this work.
References
1. GOLD, Global Initiative for Chronic Obstructive Lung Disease. Global strategy for the diagnosis, management and prevention of chronic obstructive pulmonary disease. 2019. Report, 2019.
2. Chrystyn H, van der Palen J, Sharma R, et al. Device errors in asthma and COPD: systematic literature review and meta-analysis. NPJ Prim Care Respir Med. 2017;27(1):22. doi:10.1038/s41533-017-0016-z
3. Usmani OS, Lavorini F, Marshall J, et al. Critical inhaler errors in asthma and COPD: a systematic review of impact on health outcomes. Respir Res. 2018;19(1):10. doi:10.1186/s12931-017-0710-y
4. Usmani OS. Choosing the right inhaler for your asthma or COPD patient. Ther Clin Risk Manag. 2019;15:461–472. doi:10.2147/TCRM.S160365
5. van der Palen J, Moeskops-van Beurden W, Dawson CM, et al. A randomized, open-label, single-visit, crossover study simulating triple-drug delivery with Ellipta compared with dual inhaler combinations in patients with COPD. Int J Chron Obstruct Pulmon Dis. 2018;13:2515–2523. doi:10.2147/COPD.S169060
6. van der Palen J, Thomas M, Chrystyn H, et al. A randomised open-label cross-over study of inhaler errors, preference and time to achieve correct inhaler use in patients with COPD or asthma: comparison of ELLIPTA with other inhaler devices. NPJ Prim Care Respir Med. 2016;26:16079. doi:10.1038/npjpcrm.2016.79
7. Molimard M, Raherison C, Lignot S, et al. Chronic obstructive pulmonary disease exacerbation and inhaler device handling: real-life assessment of 2935 patients. Eur Respir J. 2017;49(2):1601794. doi:10.1183/13993003.01794-2016
8. Price D, Bosnic-Anticevich S, Briggs A, et al. Inhaler competence in asthma: common errors, barriers to use and recommended solutions. Respir Med. 2013;107(1):37–46. doi:10.1016/j.rmed.2012.09.017
9. Melani AS, Bonavia M, Cilenti V, et al. Inhaler mishandling remains common in real life and is associated with reduced disease control. Respir Med. 2011;105(6):930–938. doi:10.1016/j.rmed.2011.01.005
10. Plaza V, Giner J, Rodrigo GJ, et al. Errors in the use of inhalers by health care professionals: a systematic review. J Allergy Clin Immunol Pract. 2018;6(3):987–995. doi:10.1016/j.jaip.2017.12.032
11. Self TH, Arnold LB, Czosnowski LM, et al. Inadequate skill of healthcare professionals in using asthma inhalation devices. J Asthma. 2007;44(8):593–598. doi:10.1080/02770900701554334
12. Ovchinikova L, Smith L, Bosnic-Anticevich S. Inhaler technique maintenance: gaining an understanding from the patient’s perspective. J Asthma. 2011;48(6):616–624. doi:10.3109/02770903.2011.580032
13. Molimard M, Raherison C, Lignot S, et al. Assessment of handling of inhaler devices in real life: an observational study in 3811 patients in primary care. J Aerosol Med. 2003;16(3):249–254. doi:10.1089/089426803769017613
14. van der Palen J, Thomas M, Chrystyn H, et al. Inhaler errors after reading the patient information leaflet in patients with COPD: a comparison of Ellipta® with five inhaler devices. Am J Respir Crit Care Med. 2016;193:A6811.
15. Klijn SL, Hiligsmann M, Evers SMAA, et al. Effectiveness and success factors of educational inhaler technique interventions in asthma & COPD patients: a systematic review. NPJ Prim Care Respir Med. 2017;27(1):24. doi:10.1038/s41533-017-0022-1
16. Lavorini F, Magnan A, Christophe Dubus J, et al. Effect of incorrect use of dry powder inhalers on management of patients with asthma and COPD. Respir Med. 2008;102(4):593–604. doi:10.1016/j.rmed.2007.11.003
17. Man KN, Tian Z, Lam DC-L, et al. Satisfaction, preference and error occurrence of three dry powder inhalers as assessed by a cohort naïve to inhaler operation. Int J Chron Obstruct Pulmon Dis. 2018;13:1949–1963. doi:10.2147/COPD.S152285
18. Komase Y, Asako A, Kobayashi A, et al. Ease-of-use preference for the ELLIPTA® dry powder inhaler over a commonly used single-dose capsule dry powder inhaler by inhalation device-naïve Japanese volunteers aged 40 years or older. Int J Chron Obstruct Pulmon Dis. 2014;9:1365–1375. doi:10.2147/COPD.S72762
19. Sulaiman I, Seheult J, Sadasivuni N, et al. The impact of common inhaler errors on drug delivery: investigating critical errors with a dry powder inhaler. J Aerosol Med Pulm Drug Deliv. 2017;30(4):247–255. doi:10.1089/jamp.2016.1334
20. Price DB, Román-Rodríguez M, McQueen RB, et al. Inhaler errors in the CRITIKAL study: type, frequency, and association with asthma outcomes. J Allergy Clin Immunol Pract. 2017;5(4):1071–1081.e9. doi:10.1016/j.jaip.2017.01.004
21. Pascual S, Feimer J, De Soyza A, et al. Preference, satisfaction and critical errors with Genuair and Breezhaler inhalers in patients with COPD: a randomised, cross-over, multicentre study. NPJ Prim Care Respir Med. 2015;25(1):15018. doi:10.1038/npjpcrm.2015.18
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.